Abstract
Oral mucosal lesions are a common pathology that impact the quality of life due to pain, inappetence, weight loss, and low productivity. The purpose of this study is to evaluate the effects of Tarantula cubensis extract on wound healing in rats with buccal mucosal lesions. A total of 40 male Wistar albino rats weighing 250–300 g were used in the study. The rats were divided into four equal groups. A 3 mm diameter mucosal defect was created in the buccal mucosa of each rat. Groups one and three (controls) assessed spontaneous healing at 3 and 6 days post trauma, respectively. Groups two and four (treatment) received 0.2 ml T. cubensis extract subcutaneously. Group two was treated for 2 days and assessed at 3 days and group four was treated for 5 days and assessed at 6 days. All rats were euthanatized prior to tissue sample collection. Tissue samples were compared between control and treatment groups using histopathology and immunohistochemistry. Tissue samples taken from the defect areas were immunohistochemically evaluated for cytokeratin and collagen expressions. In both the 3 and 6 day treatment groups, the improvements were statistically different compared to the control groups. T. cubensis extract was found to increase cytokeratin and collagen in both epithelial and connective tissue and to have a significant healing effect on mucosa on both gross and microscopic findings.
Introduction
The structural and functional properties of the oral mucosa are to provide protection and defense; however, exposure to the external environment can lead to trauma and infection, thereby, serving as a gateway for infection to spread both locally and systemically. The oral epithelium is essential for defense and normal homeostasis, its structure and function are important indicators of health and disease. However, it is comprised of soft tissue and can easily be damaged by trauma and or disease. Stomatitis is often used as a diagnosis of extensive oral ulceration and inflammation; however, “stomatitis” is a general term to describe any inflammation of the oral mucosa. 1
Oral mucosal wounds and stomatitis are painful lesions that can develop in both humans and animals. One of the most common causes is trauma, however, mouth lesions can also be attributed from microorganisms or disease. In humans, recurrent aphthous stomatitis (RAS), a disease that affects the oral mucosa is characterized by recurrent ulcers 2 and is painful. Although the precise etiology of RAS is unknown, it is thought to be a combination of factors, including environmental, immunological, genetic, systemic, nutritional, and microbial. 3 These factors are also relevant in the development of oral lesions in animals. Oral lesions may occur more frequently in animals than humans, acquired from eating hard foods, lack of oral hygiene, and thereby become chronic due to a delayed diagnosis. As a result, treatment can be prolonged and challenging due to the severity of the disease. Mouth lesions develop during the course of many diseases and therefore must be diagnosed and treated promptly. 4 Stomatitis is a problem that clinicians frequently encounter in both human and veterinary medicine, and treatment is often challenging. This is particularly relevant in dentistry, where clinicians are seeking medications and methods to hasten the healing of mouth lesions and recovery of their patients to alleviate pain, discomfort, and difficulty to eat.
A naturopathic alcohol extract medicinea obtained from the Tarantula cubensis spider has recently been used by veterinarians with great success. 5 Several studies have reported successful results in the treatment of skin papillomatosis in cattle and breast adenocarcinomas in dogs, as well as many traumatic and inflammatory lesions and injuries.6–9 In addition, the extract has been shown to have demarcation, regeneration, antiphlogistic, and resorptive properties.6–9 Despite this success in the treatment of various diseases, its mechanisms of action remain unknown. Another study reported anti-inflammatory and inhibitory effects on catabolic conditions. 10
Despite these anti-inflammatory effects, and the treatment of systemic diseases and use in conjunction with other treatment methods, with reported healing effects, there have been few studies on its use in oral lesions. A number of publications based on clinical evaluations on the use of T. cubensis extract in mouth sores have been published,11–13 however, there were no pathologically examined experimental studies on oral inflammation. There is also no information available on how the extract achieves its cellular effects. The purpose of this study is to assess the effects of T. cubensis extract on the healing of a buccal mucosal defect in rats using histopathological and immunohistochemical methods.
Materials and Methods
The study was performed in accordance with the ARRIVE (Animal Research: Reporting in vivo Experiments) guidelines 2.0 and approved by the Local Ethical Committee on Animal Research of Burdur Mehmet Akif Ersoy University, Turkey (MAKU-HADYEK-608, 16.01.2020). Rats were provided food and water ad lib, kept at room temperature, and housed under a 12-h light/dark cycle.
Experimental Procedure
Forty Wistar albino male rats weighing between 250 and 300 g were used in the study. Rats were divided into four equal groups. Group one and two comprised a control and an experimental group, respectively, and was conducted over 3 days. Group three and four comprised a control and an experiment group, respectively, and was conducted over 6 days.
A 3 mm diameter mucosal defect was created using a 3 mm biopsy punchb on Day 0 in all rats. The defect in the control groups was left to heal spontaneously. The rats in groups two and four had 0.2 ml of T. cubensis extract injected subcutaneously (SQ) once daily starting on Day 0. Group two rats received further doses once daily for two days, while the rats in group four received further doses once daily for five days. Rats were euthanized on either day 3 or day 6 as per the study protocol using ketaminec (90 mg/kg) and xylazined (10 mg/kg) by intraperitoneal (IP) injection.
After euthanasia, tissue samples were collected from the oral mucosa lesion area and examined grossly, histopathologically, and immunohistochemically for cytokeratin and collagen expressions.
Histopathological Method
Post-necropsy, the wound-induced right cheek areas of rats were examined macroscopically. Tissue samples were collected from the lesion-induced areas and fixed in 10% neutral formalin solutione. To prevent the tissues from curling, samples were immersed in the formalin solution before being placed on a flat surface for five minutes to harden. Tissue samples were taken to a fully automated tissue processing devicef for routine tissue processing. The samples were embedded in paraffin, and serial sections of 5-micron thickness were obtained using a Leica 2155 rotary microtomeg. The sections were then stained with hematoxylinh and eosini and Picrosirius redj for connective tissue.
Immunohistochemistry Method
After deparaffinizing and rehydrating sections on polylysine-coated slidesk for immunohistochemical examinations, the “streptavidin-biotin complex peroxidase method” was used. Immunohistochemical analysis was performed using commercially available ready-to-use kitsl. Sections were stained by collagen-4m and cytokeratin-1n immunohistochemically according to the manufacturer instructions. The ready-to-use kits and 3,3′-diaminobenzidine (DAB)o was used as the chromogen. Instead of primary antibodies, slides were incubated with antibody dilution solutionsp as negative controls. For counterstaining, they were stained with hematoxylin and examined under a light microscopeq. For immunohistochemical staining, semiquantitative analysis was used, with scores ranging from 0 to 3, where 0 indicates no staining, 1—light staining, 2—moderate staining, and 3—heavy staining. The scores obtained were statistically analyzed. Morphometric evaluations and microphotography were evaluated using an imaging software systemr.
Statistical Analysis
The immunohistochemical scores were statistically analyzed using a one-way ANOVA tests. The Duncan test was used to compare group differences. Values of P < .05 were considered statistically significant.
Results
Clinical Findings
There were no deaths or healing complications observed in any of the rats. Body weight measurements were measured before and after the study. While weight loss was observed in the control groups, weight gain was observed in both the 3-day and 6-day periods in the treated groups. However, the difference between the groups was statistically insignificant (Table 1).
Rat's Weights At the Beginning and At the End of the Study (Grams).
Gross Findings
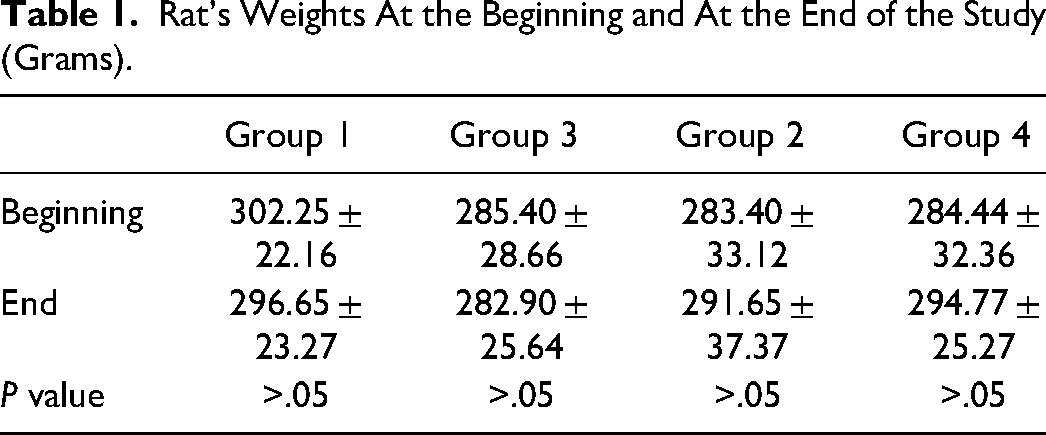
During necropsy, the mucosal defects were examined. The control and treatment groups were compared in both the 3-day and 6-day periods. The treatment groups exhibited greater healing in both periods. During the 3-day study period, many rats in the treatment group (Group two) had epithelization and marked healing, while ulcers and necrotic masses were clearly visible in the control group (Group one). Despite the fact that wounds were closed in many rats during the 6-day study period, incomplete areas were observed in some of the defects in the control group (Group three). All rats in the 6-day study period treatment group (Group four) had macroscopically complete recovery (Figure 1A–D).
Macroscopic view of the defect area (arrows). (A) Day 3—control group. (B) Day 3—treatment group. (C) Day 6—control group. (D) Day 6—treatment group.
Histopathological Findings
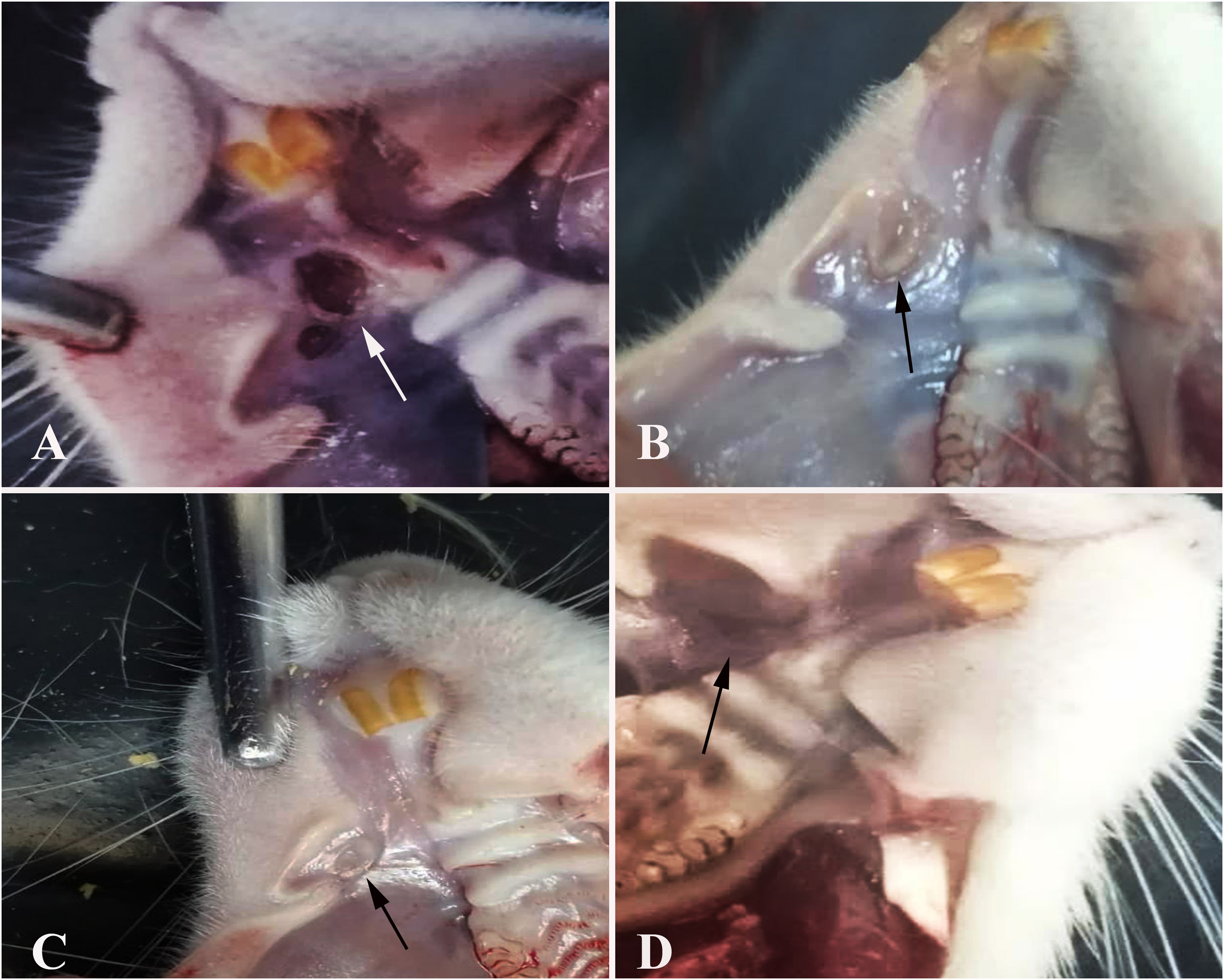
During the 3-day study period, the control group (Group one) had crust formation on the upper surfaces of the defects created in the oral mucosa with no epithelization. Propria mucosa had extensive hemorrhagic foci and severe inflammatory cell infiltration. The treatment group (Group two) had begun epithelization in the defect area. Hemorrhage and infiltration were much less severe than in the control group.
During the 6-day study period, the control group (Group three) defects formed in the oral mucosa were very small and nearly closed. In the experimental treatment group (Group four), the defect area had completely healed (Figure 2A–D).
Histopathological appearance of the defect area (arrows) between the groups. (A) Day 3—necrotic crust (arrowhead) on the lesioned area of control group. (B) Day 3—treatment group. (C) Day 6—control group. (D) Day 6—treatment group. HE, bars = 200 μm.
Picrosirius Red Findings
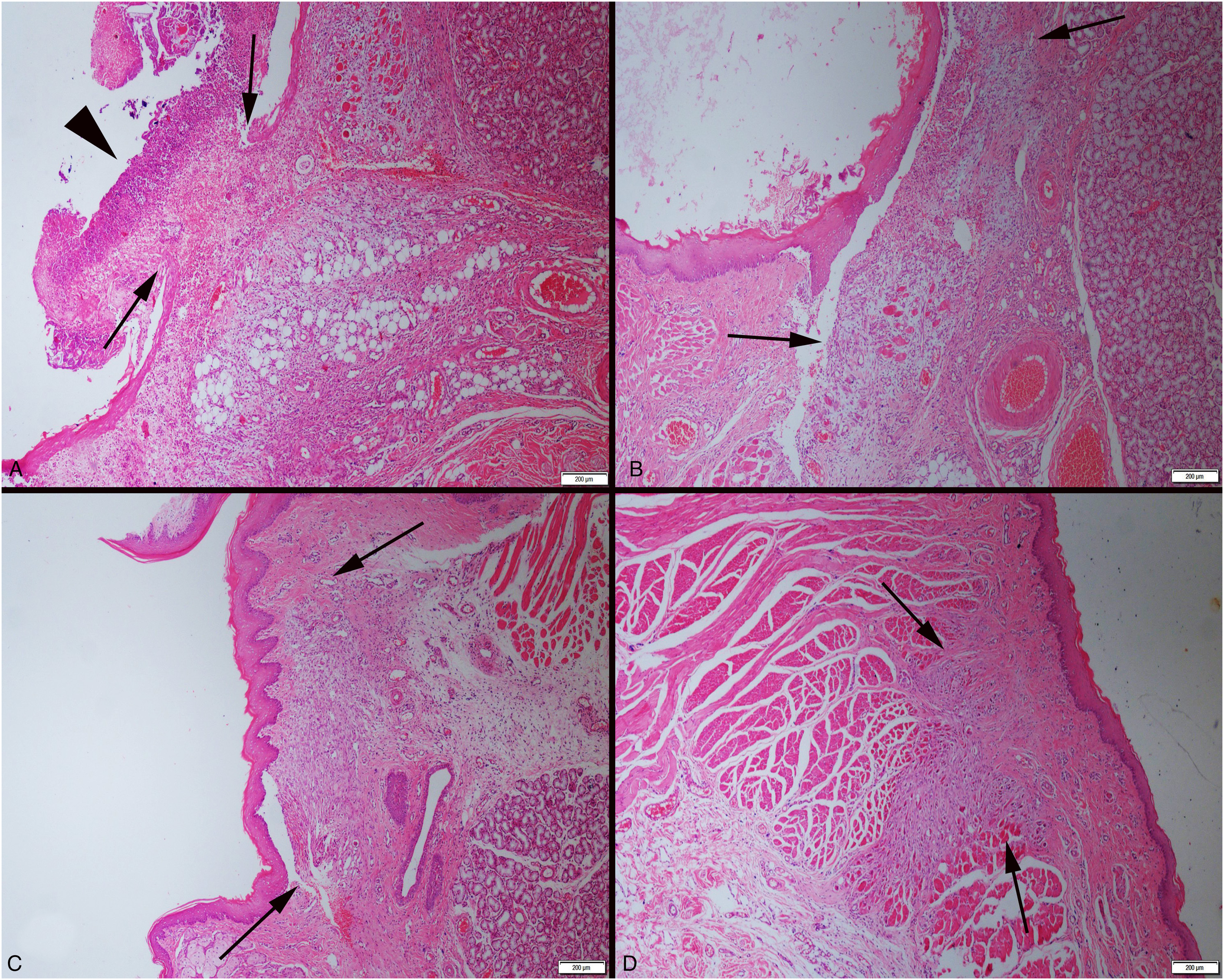
Picrosirius red staining revealed that collagen formations started within 3 days of evaluating connective tissue development in the defect area. The 3-day control group (Group one) showed the lowest collagen reaction, whereas the 6-day treatment group (Group four) showed the highest amount of mature collagen (Figure 3A–D).
Connective tissue maturation (arrows) between the groups. (A) Day 3—slight mature collagen in control group. (B) Day 3—increased amount of mature collagen in treatment group. (C) Day 6—moderate increase in mature collagen in control group. (D) Day 6—marked increase in mature collagen in treatment group. Picrosirius red method, bars = 100 μm.
Immunohistochemical Findings
As epithelization had not occurred, there was no expression of cytokeratin immunoreactions in the defect areas of the 3-day control group (Group one). The 3-day treatment group (Group two) had slight expressions, in contrast of remarkable expressions observed in the 6-day control (Group three) and treatment groups (Group four) (Figure 4A–D). When the collagen immunoreactions of the defect areas were examined, mild collagen expression was observed in the 3-day control group (Group one), with more pronounced expressions noted in the treatment group (Group two). Over the course of the 6-day study, marked expressions were observed in the control and treatment groups (Groups three and four) (Figure 5A–D). Table 2 shows the statistical analysis results of cytokeratin and collagen immunohistochemical scores.
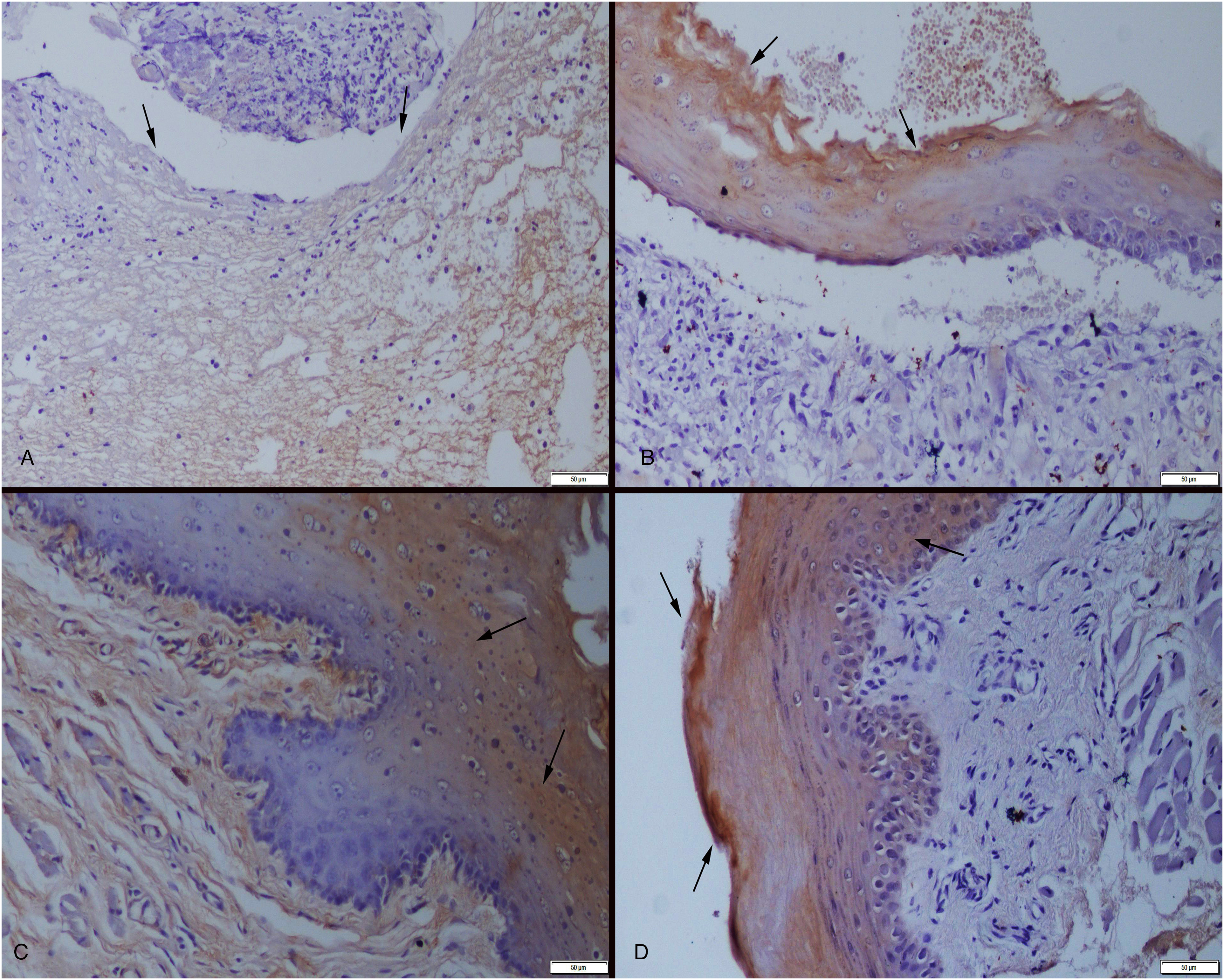
Cytokeratin expressions (arrows) between the groups. (A) Day 3—negative expression due to lack of the epithelial layer in control group. (B) Day 3—increased expression in regenerated epithelial cells in treatment group. (C) Day 6—moderate expression in newly regenerated epithelial layer in control group. (D) Day 6—marked increase in epithelial layer in treatment group. Streptavidin-biotin-peroxidase method, bars = 50 μm.
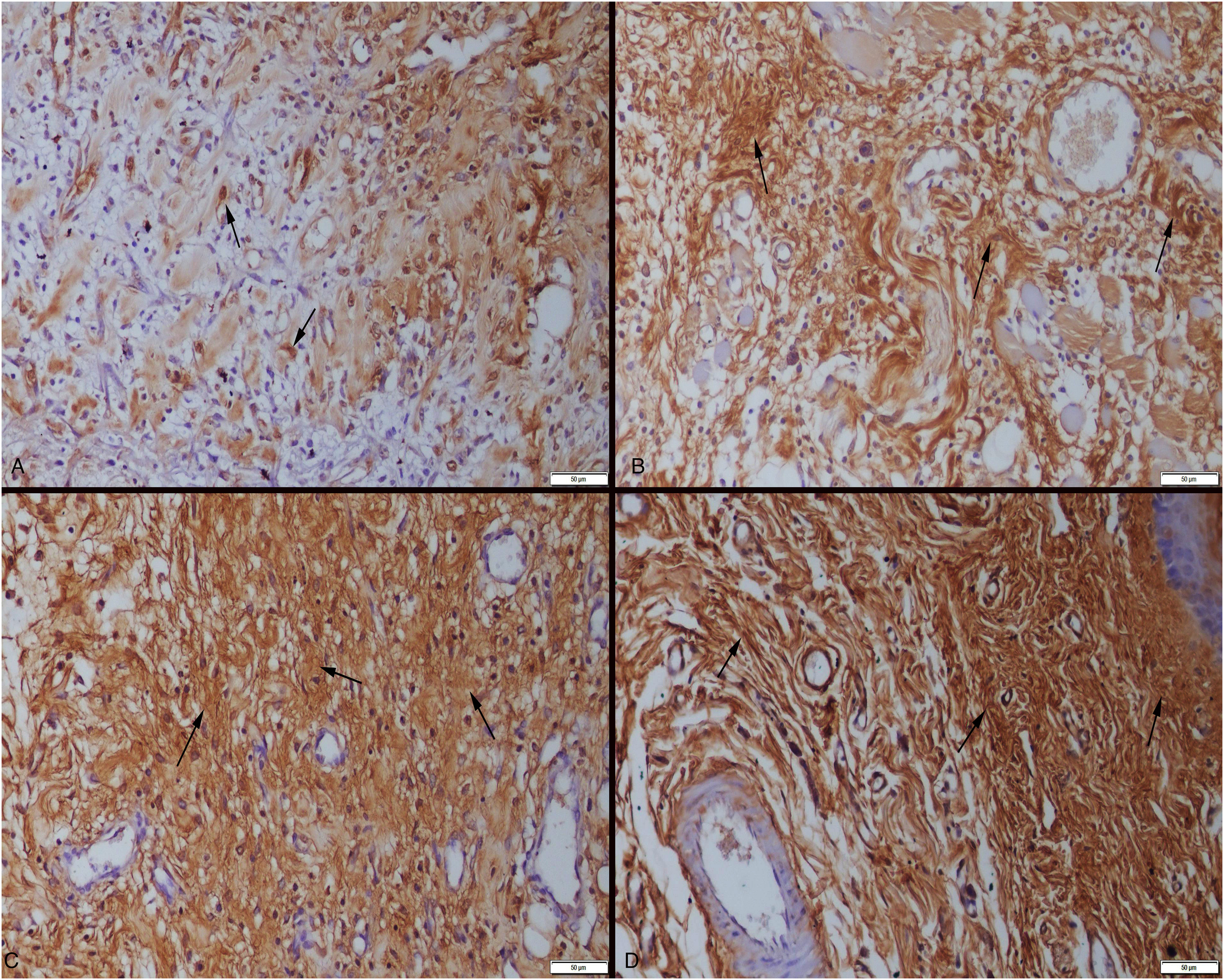
Collagen-1 expressions (arrows) among the groups. (A) Day 3—slight expression in control group. (B) Day 3—increased expression in treatment group. (C) Day 6—moderate expression in control group. (D) Day 6—marked increase in treatment group. Streptavidin-biotin-peroxidase method, bars = 50 μm.
Statistical Analysis Results of Immunohistochemical Scores.
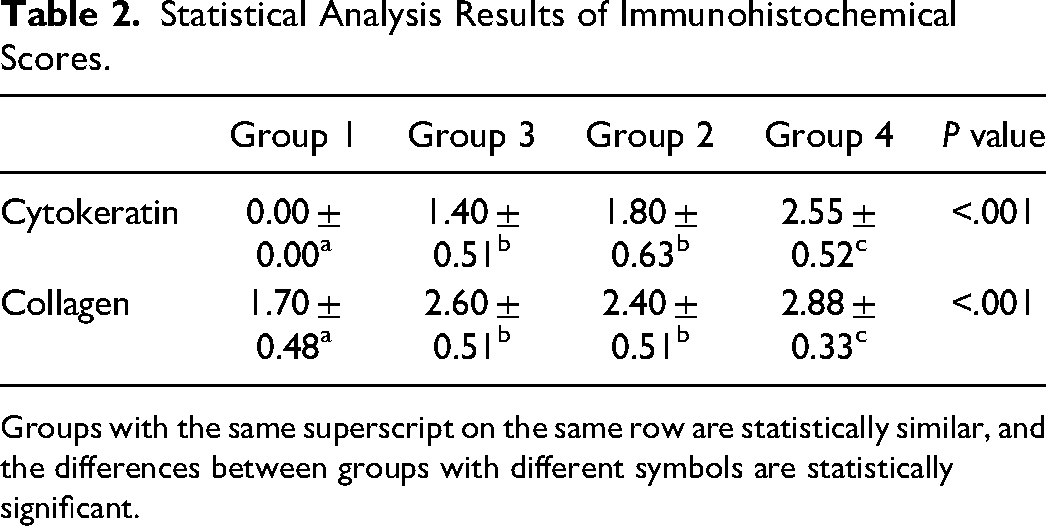
Groups with the same superscript on the same row are statistically similar, and the differences between groups with different symbols are statistically significant.
Discussion
The oral mucosa is located at the beginning of the digestive system and is constantly exposed to traumatic factors making it susceptible to oral lesions and stomatitis. There are numerous diseases that cause specific lesions in the oral cavity which can greatly impact the quality of life for both humans and animals.2–4 The most common causes are trauma, food debris, microorganisms, chemicals, burns, autoimmune deficiencies, and other physical factors.
Reducing the healing time of oral lesions will increase the quality of life for humans and animals, a shorter recovery time prevents a long and painful treatment process, particularly when combined with tooth and gum disease. Reduced healing time of oral lesions will reduce and/or prevent economic losses in animal production stock and herds. Mouth lesions are painful and often lead to the inability to eat, which in turn can cause immunosuppression. Anorexia and immunosuppression leave these animals vulnerable to disease which can rapidly spread throughout the herd. Reducing the healing process time of oral lesions can possibly inhibit and/or prevent the rapid spread of disease, maintain healthy herd condition, and sustain economic output.
The use of the alcoholic extract of T. cubensis, a licensed naturopathic medicine in animals, has escalated in recent years for treatment in domestic animals. The extract has been reported to have a healing effect for many infectious, toxic, and neoplastic diseases, and studies on these effects have increased.6–9,14,15 The successful results in animals have led to the suggestion that this drug could be used in humans. For this purpose, research using this drug with experimental animals has begun. This study investigated the effect of T. cubensis extract on mucosal defects created in rats, findings concluded that mucosal defects closed in a shorter period of time and with no reported healing complications.
Although T. cubensis extract has successfully treated many diseases, its mechanism of action is not fully understood. The healing properties of the drug are thought to be due to its regenerative, resorptive, antiphlogistic, anti-inflammatory, demarcation-enhancing, and catabolic effects.10,14 Currently there are no existing studies that examine the pathological effects or pathogenetic mechanisms. This study found a wound-healing effect by providing epithelization and an increase in connective tissue.
The extract is licensed and sold as a veterinary “homeopathic” medicine. Horse, cattle, dog, cat, pig, sheep, and goat are specified as target species. In these animals, the drug has been reported to have varying degrees of healing effects in septic or toxic diseases, dermatitis, inflammatory nail diseases (panaricium, foot rot), phlegmon, ulcers, abscesses, and purulent necrotic cases.6,8,14–16 When used in addition to the standard treatment protocol in a cat with upper lip ulcer and tongue erosion, lesions in the tongue were reported to completely disappear after 14 days, and re-epithelization was observed in the ulcerated regions of the lips. 15 Another study used regeneration, resorption, antiphlogistic, and demarcation effects of cows in the postpartum period to investigate the effect on retentio secundarium, vaginal discharge, and uterus involution. 16 As a result, it has been reported for use in the postpartum period to prevent retentio secundarium, reduce pathological vaginal discharge, and accelerate uterus involution. 16
There is limited information on the effects of the commercially available product on healing diseases, lesions, oral inflammation, and mouth lesions. According to one study, cattle with blue tongue disease recovery faster. 11 It has also been reported that this drug can help manage foot and mouth disease.12,13 In the current study, the effect on healing of oral mucosa lesions was improved. Further, epithelial and connective tissue healing of oral inflammation was improved. This feature is thought to reduce recovery time by assisting in the healing of mouth lesions in a shorter period of time. Oral lesions can make food intake difficult and cause a variety of problems ranging from weight loss to the systemic spread of disease. The results of this study support use in the treatment of oral lesions in rats.
Oral injuries are problems that all animals suffer on a regular basis throughout life and it is important that lesions heal as soon as possible. Wound healing is a complicated process. In general, injuries and repair stages are divided into three phases: exudative, proliferative, and regenerative. In each of these phases, a series of cellular, physiological, and biochemical events take place, and healing occurs as a result of their compatibility. Many endogenous and exogenous factors influence the healing process. The pathogenesis of wound healing should be thoroughly evaluated in order to develop effective and successful approaches to wound treatment. 17 Although experimentally induced stomatitis is not the same disease as immune-mediated stomatitis seen in dogs and cats, the pathological examination results of this study suggest that T. cubensis extract may promote healing of stomatitis in these species. The findings of this study show that T. cubensis extract has regulating and accelerating effects in all phases of wound healing. It also revealed that weight loss did not occur during the treatment process, which was regarded as an important contribution to the treatment process in both humans and animals.
Human studies are currently being performed on tissue culture cells, where it has been reported to destroy tumor cells by increasing apoptosis in tumor cells. 18 It has also been reported in experimental studies to be effective in tumor treatment. 19 In addition, a study found no side effects in horses. 20 In the current study, findings confirmed no toxic effects in rats, and no deaths of any test subjects.
As previously stated, stomatitis and oral inflammation can develop into a serious health issues for humans and animals. It can occur at all stages of life in both humans and animals placing a negative impact on their quality of life. Due to the increased prevalence of stomatitis and oral inflammation, researchers are investigating therapeutic agents to treat this condition. In this study, T. cubensis extract was found to be effective in the treatment of oral lesions induced experimentally in rats. This is thought to be attributed to the ability to extract, demarcate and regenerate tissues. In this context, it is considered to be a highly effective treatment. The results showed that T. cubensis extract can be used to treat oral wounds and potentially oral inflammation and stomatitis. This study serves as a model for future research on this subject. It is thought that clinicians may benefit from considering the use of the T. cubensis extract in the management of stomatitis, though additional studies are warranted.
Materials
Theranekron, Richter Pharma AG, Austria
Kruuse, Denmark
Ketasol, Richter Pharma AG, Austria
Brasilazin, BaVet, Türkiye
Formaldehyde Sol. Merck, Germany
Leica ASP300S; Leica Microsystems, Nussloch, Germany
Leica Microsystems, Nussloch, Germany
Harris Hematoxylin, Surgipath, USA
Eosin (Aqueous), Surgipath, USA
Picro Sirrius Red Staining Kit, ab150681, Abcam, UK
Polysine Slides, Thermo Scientific, Germany
UltraVision Detection System Large Volume Anti-Polyvalent, HRP (RTU) (TA-060-HL), Thermo Shandon Limited, Cheshire, England
Collagen 4 polyclonal antibody (bs-4595R), Bioss Antibodies Inc., Massachusetts, USA
Cytokeratin-1 [cytokeratin 1 (4D12B3): sc-65999], Santa cruz, USA
UltraVision detection System Large Volume DAB Substrate System (RTU) (TA-060-HD), Thermo Shandon Limited, Cheshire, England
Antibody Diluent, Abcam, UK
Olympus BX51, Tokyo, Japan
Database Manual CellSens Life Science Imaging Software System, Olympus Corporation, Tokyo, Japan
SPSS 22.00 package program, IL, USA
Footnotes
Acknowledgments
The authors would like to express their gratitude to the staff of the Burdur Mehmet Akif Ersoy University Experimental Animal Production and Experimental Research Center for their valuable support throughout the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Scientific Projects Commission of University of Burdur Mehmet Akif Ersoy (Project number: 0641-YL-20).
