Abstract
The aim of this study was to evaluate the reliability of electric pulp test (EPT), cold pulp test (CPT) and tooth transillumination (TTI) in the assessment of pulpal health in dog teeth. Forty-five client-owned dogs requiring tooth extraction or pulpectomy were included. For each patient, one affected and two control healthy teeth were evaluated with EPT, followed by CPT and TTI. Direct pulp inspection was used as a gold standard. The real pulpal health (vital or necrotic) was determined by the presence or absence of bleeding after creating access to the pulp chamber. Sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) and accuracy of EPT, CPT and TTI were obtained for each pulp test using the binomial Clopper-Pearson exact method to establish confidence intervals. Forty-five affected teeth were tested. Forty-three were tested with EPT, CPT, and TTI, and two were tested solely with EPT and CPT. All dogs tested with EPT and TTI were included in the study whereas 21 out of 45 (47%) dogs tested with CPT were excluded. The sensitivity, specificity, PPV, NPV and accuracy were respectively 0.96, 1.00, 1.00, 0.96 and 0.98 for EPT; 1.00, 0.89, 0.92, 1.00 and 0.95 for CPT; and 0.59, 0.95, 0.94, 0.67 and 0.76 for TTI. This study concluded that EPT is a highly reliable diagnostic test to evaluate pulpal health in dogs. The high accuracy of CPT is conditional on the patient's responsiveness to stimulation applied to its control healthy teeth. TTI was the least reliable test in the study.
Keywords
Introduction
The diagnosis of pulpal health is essential to determine the appropriate treatment of several dental pathologies. Accurate assessment of pulpal status is a challenge in veterinary dentistry as pulp is encased in a hard shell of enamel and dentin, and patients do not self-report their pain. Unless there is direct pulp exposure, veterinarians must rely almost exclusively on radiographic features of pulp necrosis (wide root canal consistent with arrested dentinogenesis, tooth resorption or periapical lucency consistent with periapical lesions) to assess pulpal health. However, these features may take months to years to develop and dogs may live with pulpal necrosis and associated pain for a long time before a diagnosis is made.1‐4 Indirect diagnostic methods of pulpal health are now standard adjunct tools in human dentistry,5–8 and these techniques could help the diagnosis of pulp disease in veterinary medicine.
Sensibility tests assess whether there is a neuronal response to tooth stimulation using thermal (cold or heat) or electrical conduction (electric pulp tester). The integrity of the A-delta fibres in the dentin-pulp complex is tested by applying a brief stimulus to the outer surface of the tooth and evaluating behaviour responses to the painful stimuli. These two sensibility tests are currently the most widely used tests to assess pulpal health in human dentistry.7–10 Assessment of tooth transillumination is another non-invasive test to evaluate pulpal health status. The latter is occasionally used in veterinary dentistry11–14 and considered to be a helpful adjunct to conventional pulp tests in human dentistry.7,8,15 In this test, a source of light is placed behind the tooth. The light passes through a vital pulp because of its translucency, resulting in a transilluminated tooth. A necrotic pulp appears dull and dark because of its compromised blood supply and the degeneration of the pigments inside its dentinal tubules.9,11
Current diagnostic methods for pulpal health assessment have shortcomings in terms of reliability and reproducibility. Many studies in human dentistry5,6 have compared the effectiveness and reliability of pulp tests but, to the authors’ knowledge, the reliability of sensibility tests and tooth transillumination have not been evaluated in veterinary dentistry.1,11–14
The aim of this study was to evaluate the reliability (ability of a test to discriminate the presence or absence of a disease in any given population based on its global accuracy) of electric pulp test (EPT), cold pulp test (CPT) and tooth transillumination (TTI) in the assessment of pulpal health in permanent dog teeth. Based on the results of similar studies in human dentistry and the authors’ clinical experience in veterinary dentistry, we hypothesized that EPT and TTI would have very good to excellent reliability while the CPT would show only good reliability.
Materials and Methods
This study was approved by the local animal care committee of the Faculty of Veterinary Medicine, Université de Montréal (protocol number 17-Rech-1906).
Study Population
The study involved 45 client-owned dogs (mean ± standard deviation; 5.5 ± 2.1 years old; 23.4 ± 15.3 kg body weight), after owners’ informed consent. Dogs presented for a dental consultation at the Dentistry Service of the Centre hospitalier universitaire vétérinaire, Faculty of Veterinary Medicine, Université de Montréal, between February 2017 and October 2018 and requiring a tooth extraction or a pulpectomy were considered for this study.
Inclusion Criteria
Irrespective of breeds, dogs between 2 and 8 years of age requiring one or more tooth extraction or pulpectomy were included. The specific age group was selected to minimize bias related to individual responses unrelated to stimulation such as age-induced anatomic changes (ie, incomplete root development in young dogs and decreasing pulpal innervation and dentinal tubules/fluids in older dogs).4,16–22 To assure inclusion of vital and nonvital teeth in the study, all teeth requiring extraction or pulpectomy (irrespective of the reason) were included. Only one affected tooth per animal was included in this study.
Exclusion Criteria
Patients that were reluctant to permit oral examination even after sedation and patients necessitating a different anesthesia protocol were excluded. Affected teeth with previous restoration, crown or pulpectomy were also excluded. The ability of each patient to show a reaction to pulp stimulation was evaluated. All the patients that did not show pain reaction when two healthy teeth were tested were excluded from that test.
Study Design
All patients without a previous clinical diagnosis of pulpal health status were examined. The affected tooth was evaluated with EPT, CPT, and TTI. The two first tests were performed in the conscious patient in the examination room before the dental procedure. The EPT was applied first followed by the CPT with a minimum of 2 minute intervals between the two tests. If sedation was required for oral examination, acepromazinea (0.02-0.05 mg/kg) and hydromorphoneb (0.1 mg/kg) were administered intramuscularly at least 15 min prior to the tests. The TTI was performed under general anesthesia after induction with propofolc to effect and maintenance with isofluraned in 100% oxygen after teeth cleaning. All pulp tests were evaluated by the same investigator (CP). Direct pulp inspection was used as a gold standard for comparisons with EPT, CPT and TTI results. Direct pulpal status (vital or necrotic) was determined by creating access to the pulp chamber with a round carbide bure on a highspeed handpiece. The lack of bleeding confirmed a necrotic pulp (nonvital tooth), while bleeding confirmed a vital pulp and tooth. When there was no bleeding at pulp access, the pulp was explored with a Pathfinderf stainless steel endodontic hand file. If bleeding was present in the more apical part of the canal (partially necrotic pulp),7,14,16,23 pulpal health was still considered necrotic (nonvital tooth) (Figure 1).
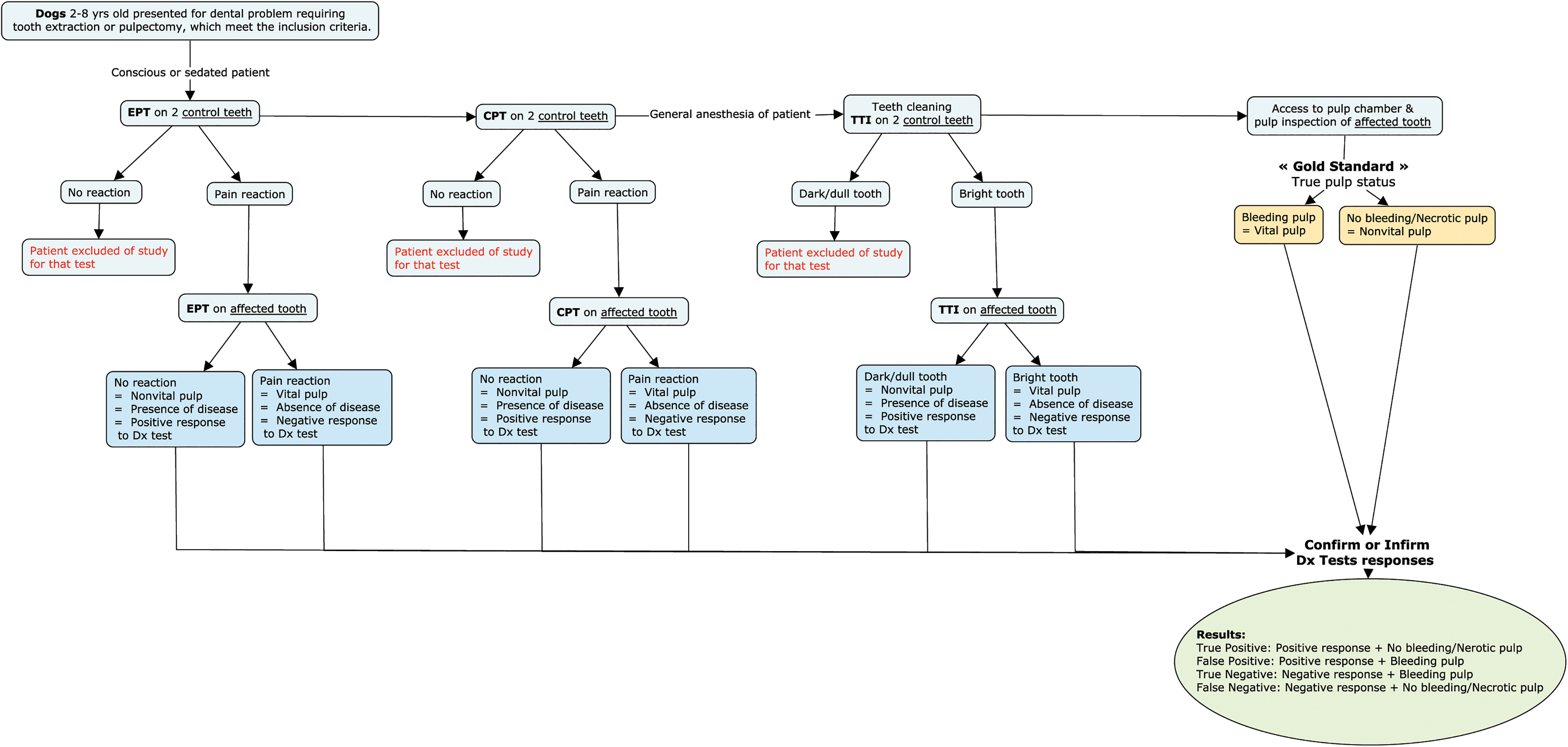
Study Design.
Electric Pulp Test
The EPT was performed by using an analog electric pulp testerg (Figure 2a). The tooth surface was dried with a cotton gauze. An interface conducting mediumh was applied to the electrode tip (1 mm diameter), and the electrode was placed on the middle third of the buccal surface of the tooth. A gradual increase of the current (90 mA; DC 0-10 V) every 2 to 5 seconds allowed the patient to react to the painful stimulus. Any mouth reaction (eg, involuntary jaw movement) was considered a negative response (compatible with a vital tooth) whereas no pain reaction at the maximum current was recorded as a positive response (compatible with a nonvital tooth) (Videos 1, 2).
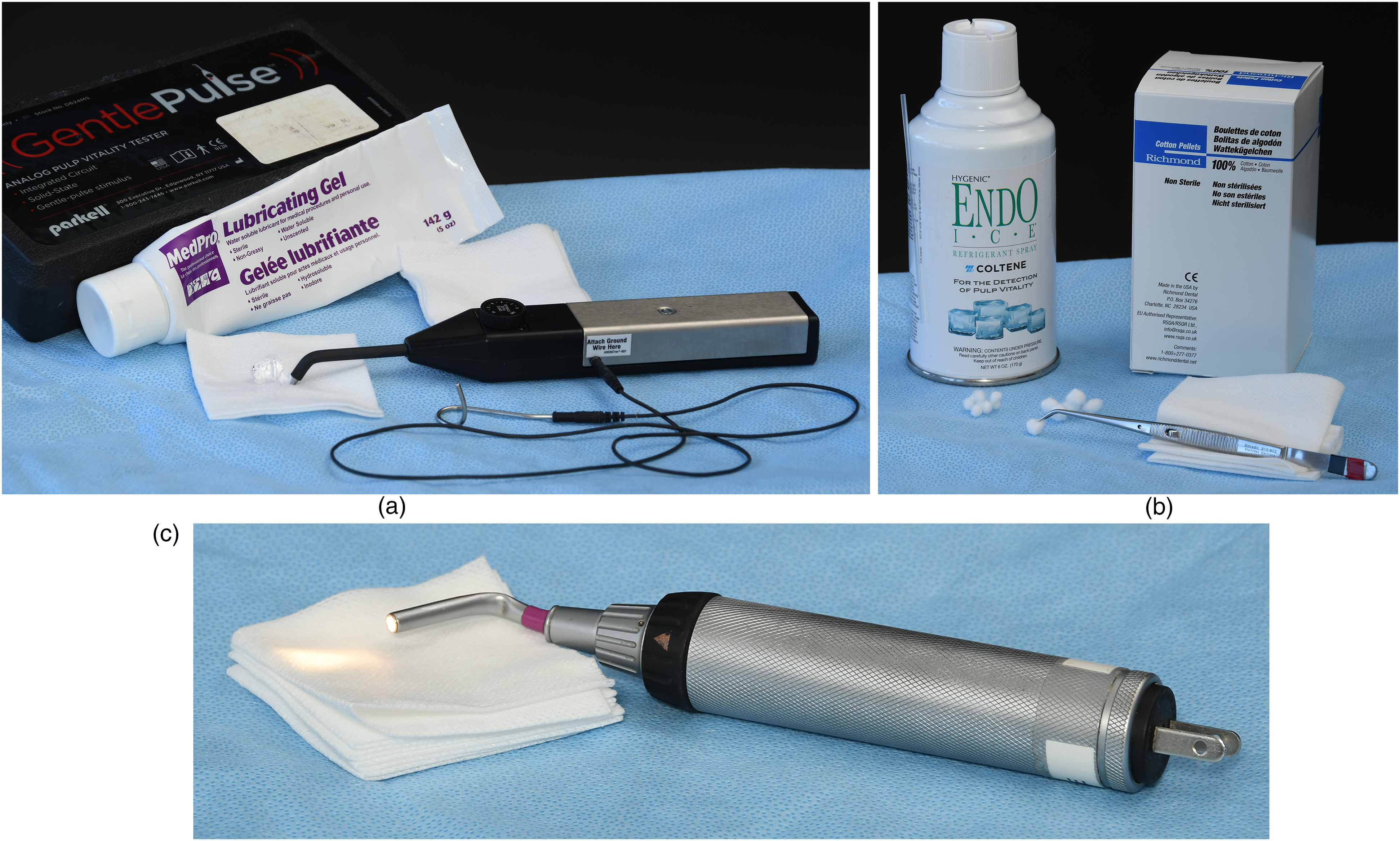
(a) Analog electric pulp tester, cotton gauze, interface conducting medium for electric pulp test (EPT). (b) Refrigerant spray (1,1,1,2-tetrafluoroethane, Endo Ice), cotton pellets and cotton pliers for cold pulp test (CPT). (c) Transilluminator for tooth transillumination (TTI).
Cold Pulp Test
The cold test was performed by using a refrigerant spray (1,1,1,2-tetrafluoroethane, Endo Icei) (Figure 2b), which is released at −26 °C. 7 A #2 cotton pelletj, for medium and large breed dogs, or #3 cotton pelletj, for small breed dogs, 7 was soaked with Endo Ice, carried by cotton pliersk and placed on the buccal surface of the tooth until the patient responded or for a maximum of 10 s (cut-off). If there was no reaction, the response to the test was considered positive (compatible with a nonvital tooth). Any mouth or head reaction (eg, jaw or tongue movement, turning head away from cold pellet) was considered a negative response (compatible with a vital tooth) (Videos 3, 4).
Transillumination
A strong narrow beam of lightl (Figure 2c) was placed on and behind the tooth, and the passage of the light through the tooth was evaluated. If the light passed through the tooth (ie, transillumination), it was considered vital (Figure 3a). If the tooth appeared dark and dull, especially in the central portion, it was considered nonvital (Figure 3b).

(a) Tooth 404 transilluminates which corresponds to a negative response compatible with a vital tooth. (b) Tooth 204 does not transilluminate which corresponds to a positive response compatible with a nonvital tooth.
For the three tests, the affected tooth, its contralateral and another healthy tooth were tested. When the contralateral tooth was affected or absent, another healthy tooth in the opposite arcade was tested.
Statistical Analysis
The sample size was determined using the EpiTools Epidemiological Calculator. 24 Power analysis was based on EPT and CPT sensitivity range of 0.71 to 0.76 and 0.81 to 0.92 found in human dentistry, and a power of 0.90 with the desired precision of 0.10. Sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) and accuracy of EPT, CPT and TTI were obtained using the total number of teeth included in the study for each pulp test. Ninety-five percent (95%) confidence intervals (CI) were calculated for each parameter using the Binomial (Clopper-Pearson) exact method.
Sensitivity, Specificity, PPV, NPV and Accuracy
Sensitivity denotes the ability of a test to detect disease among a population of patients with the disease. Conversely, specificity denotes the ability of a test to detect the absence of disease among a population of patients without the disease. Sensitivity and specificity are good indicators of a test's discriminative ability; they evaluate the performance of a test in relation to patients with known disease states.5,25,26 However, in clinical practice, there is an interest in evaluating a diagnostic test to determine its probability to provide the correct diagnosis in patients with unknown disease states.5,26 The measurement of PPV and NPV may indicate how good the test is at predicting illness or health in patients with unknown disease states. 5 Finally, the accuracy is the proportion of test responses that agreed with the gold standard. 16 In the context of this study, the accuracy is the proportion of pulp test responses that agreed with direct visualization of the pulp.
The criteria used to define sensitivity, specificity, PPV, and NPV should be similar across studies, so results are comparable. Additionally, only adjusted predictive values and accuracy to a common prevalence should be compared since the disease prevalence in the studied population can influence predictive values and accuracy.25–29
Results
A total of 45 dogs from 25 different breeds and mixed breeds among which Labrador retriever (7), Beagle (4) and Chihuahua (4) were the most common breeds. Forty-three affected teeth were tested with EPT, CPT, and TTI, and two were tested solely with EPT and CPT since 2 teeth were not tested for TTI. Table 1 shows the type of teeth tested in the study. Of 45 teeth, 25 had tooth fracture, 12 had periodontal disease, 4 had tooth dyschromia, 2 were involved in malocclusion, 1 had severe attrition, and 1 was affected by an oral mass.
Type of Teeth Included in the Study.
The EPT identified 96% of the nonvital teeth and 100% of the vital teeth since no pain reaction was observed in 24 out of 25 nonvital teeth tested with EPT whereas all the 20 vital teeth elicited a pain reaction to EPT stimulation.
Forty-five teeth were tested with CPT and 21 (47%) teeth had to be excluded due to absence of pain reaction when testing the control healthy teeth of the patients. Therefore, only 24 teeth were eligible to be included in the study for that test. For those 24 teeth, the CPT identified 100% of the nonvital teeth and 89% of the vital teeth since all fifteen teeth (100%) with necrotic pulp elicited no pain reaction to CPT stimulation whereas 8 out of 9 teeth with vital pulp did.
The TTI identified 59% of the nonvital teeth and 95% of the vital teeth since only 13 out of 22 teeth with necrotic pulps did not transilluminate whereas 20 out of 21 teeth with vital pulp let the light pass through and appeared bright at TTI (Tables 2, 3).
Results of the Electric Pulp Test (EPT), Cold Pulp Test (CPT) and Tooth Transillumination (TTI) in Comparison with the Gold Standard (Direct Visualization of the Pulp) and Calculated Sensitivity, Specificity, Positive Predictive Value, Negative Predictive Value and Accuracy for Each Diagnostic Test. (Adapted from Oakden-Rayner, 2018). 28
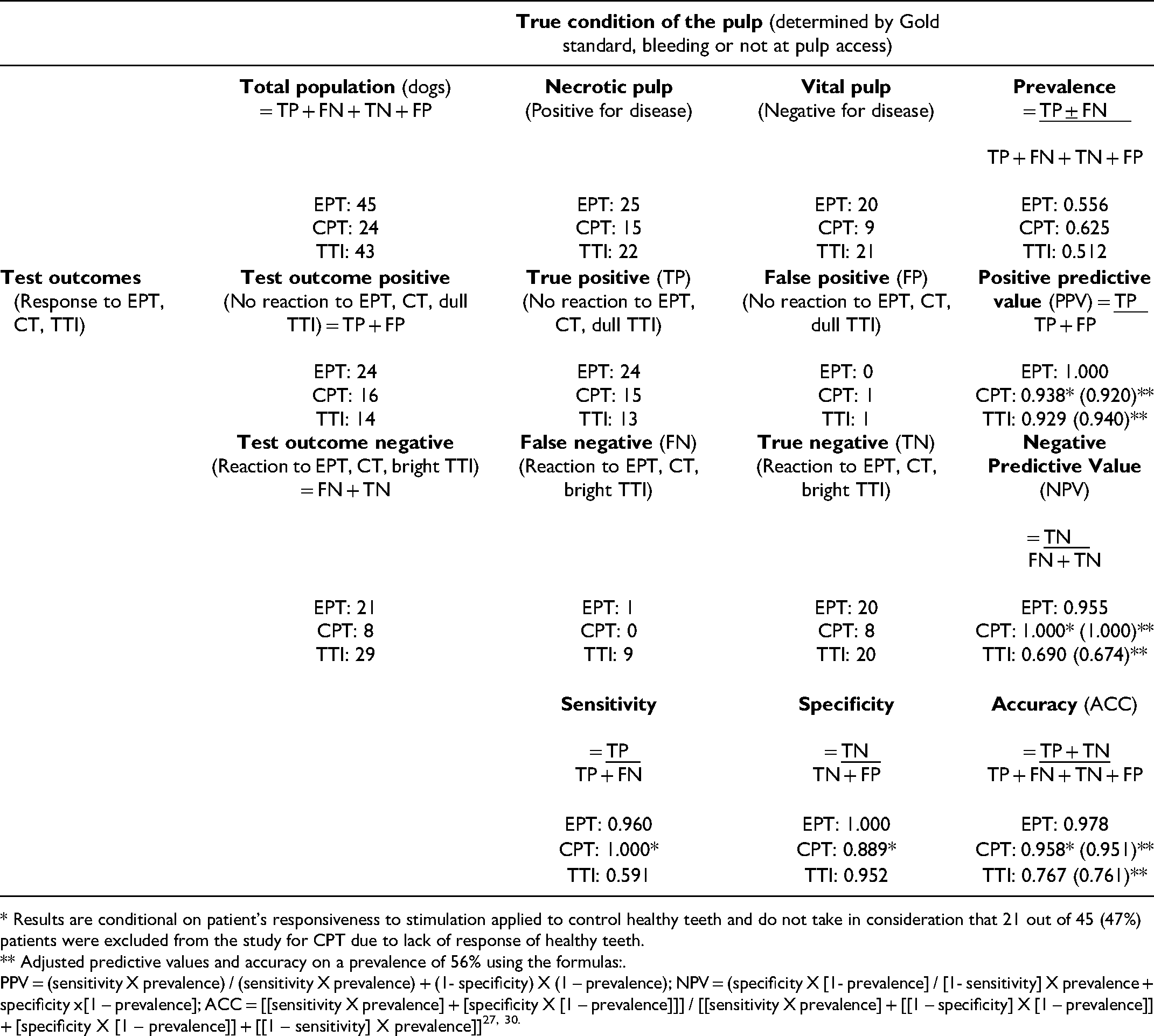
* Results are conditional on patient’s responsiveness to stimulation applied to control healthy teeth and do not take in consideration that 21 out of 45 (47%) patients were excluded from the study for CPT due to lack of response of healthy teeth.
** Adjusted predictive values and accuracy on a prevalence of 56% using the formulas:.
PPV = (sensitivity X prevalence) / (sensitivity X prevalence) + (1- specificity) X (1 – prevalence); NPV = (specificity X [1- prevalence] / [1- sensitivity] X prevalence + specificity x[1 – prevalence]; ACC = [[sensitivity X prevalence] + [specificity X [1 – prevalence]]] / [[sensitivity X prevalence] + [[1 – specificity] X [1 – prevalence]] + [specificity X [1 – prevalence]] + [[1 – sensitivity] X prevalence]]27, 30.
Sens., sensibility; Spec., specificity; PPV, positive predictive value; NPV, negative predictive value; ACC, accuracy, CI, confidence interval; N/E, not evaluated.
* * Results are conditional on patient’s responsiveness to stimulation applied to control healthy teeth and do not take in consideration that 21 out of 45 (47%) patients were excluded from the study for CPT due to lack of response of healthy teeth.
** Adjusted predictive values and accuracy on a prevalence of 56% using the same formulas as in Table 2.
Discussion
In this clinical study, the EPT was found to be the most accurate (98%) technique to evaluate pulpal health status in dogs. This test correctly identified 100% of the nonvital teeth and 96% of the vital teeth. The CPT also showed good accuracy (95%) but only when excluding patients with absence of pain reaction on their control healthy teeth (47%). TTI was found to be the least accurate test (75%) with correct identification of vital teeth in only 67% of the cases. Therefore, both EPT and CPT may aid in the diagnosis of pulp disease in conscious dogs and provide guidance in the treatment of these teeth in the clinical setting. However, veterinarians should keep in mind that there is a high prevalence of absence of pain reaction to CPT stimulation in control healthy teeth which limits the usefulness of this test.
The EPT relies on an ionic shift in the dentinal fluid within the dentinal tubules. By applying an electric current to the tooth surface, the ionic movement in the dentinal fluid causes a local depolarization and impulse propagation through the A-delta fibres creating the sudden, sharp pain sensation.8–10 This pain sensation was exhibited by a sudden involuntary jaw movement in all dogs with vital pulp and was easily recognized by the observer. Only one of the 25 dogs with a nonvital pulp had a false-negative response (ie, pain reaction) to EPT stimulation. False-negative results obtained with sensitive pulp tests may be due to stimulation through adjacent periodontal attachment, partial pulp necrosis (eg, vital nerve fibers after coronal necrosis, multi-rooted teeth, infected pulp), patient's anxiety, ineffective tooth isolation, drugs that decrease patient's threshold for pain and contact with restorations.7,16,31
Thermal pulp testing relies on the outward (cold) or inward (heat) movement of the dentinal fluid within the dentinal tubules. A rapid temperature change of the tooth causes a sudden fluid flow within the tubules which stimulates the A-delta fiber nociceptors in the dentin-pulp complex, leading to a sudden, sharp pain sensation lasting for the duration of the stimulus. A gradual change in the tooth temperature, however, would rather excite C fibers and elicit a delayed, dull and diffused aching.7–10 As previously mentioned, 47% of the control healthy teeth (21 dogs) tested elicited an absence of pain reaction to CPT stimulation and were excluded from the study. For the remaining 24 dogs, the response to CPT was, for most of them, a more delayed and subtle response (eg, jaw or tongue movement, turning head away from cold pellet). Two studies showed that the thickness of the enamel and dentin, and the number of nerve fibres in the underlying pulp affect the response to pulp testing21,32 with CPT being more affected than EPT.16,21 Alternatively, large breed and older dogs with thicker dentin tissues could be less prone to CPT. Further studies are warranted to investigate the lack of responsiveness of dogs to CPT stimulation. On the other hand, CPT was highly accurate for the patients that elicited a pain reaction in their control healthy teeth. In these cases, CPT identified all teeth with necrotic pulp, while only one affected tooth with vital pulp elicited a false positive response (ie, no pain reaction) to CPT stimulation. False positive results may be obtained with calcific obliterations in the root canals, recently traumatized teeth, immature apex, drugs that increase the patient's threshold for pain, and inadequate application of refrigerant.7,16,31
The TTI showed the least accuracy (76%) due to its low NPV (0.67), which indicates difficulty in detecting necrotic pulp (sensitivity 0.59). The TTI appears to be useful when the nonvital tooth presents dyschromia; however, it may fail to diagnose necrotic pulp when there is no tooth dyschromia such as in cases of false negative responses (ie, transillumination of nonvital teeth). Additionally, TTI may give false positive results (ie, no transillumination of vital teeth) in discoloured teeth caused by other pathology that did not compromise pulp health (eg, enamel defects, drug-induced stained teeth, uncomplicated slab fracture).
Our results provide specific information on sensitivity, specificity, PPV, NPV, and accuracy for three different modalities for pulpal health assessment in dogs. It is also possible to make comparisons with similar human studies. In humans as in dogs, EPT showed higher specificity and PPV and lower sensitivity and NPV whereas CPT presented higher sensitivity and NPV and lower specificity and PPV. Therefore, in both species, the EPT can be considered more reliable to detect vital teeth than nonvital teeth. However, the accuracy for EPT in dogs was higher than in both human studies due to higher sensitivity and NPV suggesting better discrimination to detect nonvital teeth in dogs than in humans. This variation may be due to differences in EPT threshold to consider the tooth nonvital, to the higher incidence of restored teeth and closer proximity of teeth in humans than in dogs, which may conduct electrical current to adjacent teeth.
Converse to the EPT, the CPT can be considered more reliable to detect nonvital teeth than vital teeth. Additionally, the accuracy of CPT in the present study lies within the confidence interval provided by one study 27 and slightly outside another. 25 This difference can be attributed to the higher sensitivity and NPV, suggesting a better ability for CPT to detect nonvital teeth in dogs than in humans. However, the CPT has several limitations as discussed above in terms of absence of pain reaction in control healthy teeth in dogs.
In addition to the aforementioned differences between dog and human studies, inter-species differences could be due to the ability of humans to self-report pain, the subjective nature of pain assessment in dogs by a proxy and the inclusion and exclusion criteria that could bias the results of these studies.
As in similar human dentistry studies,5,6 direct inspection of the dental pulp was the gold standard used in the present study. Histological examination of the dental pulp would be the ideal gold standard for determining the stage of pulpal heath. However, the implementation of this gold standard would have required the extraction of the tooth, which would have required the exclusion of the pulpectomy treated teeth. It would have also been difficult to compare our results to human dentistry studies. The ultimate goal of vitality tests is to extrapolate the histologic status of the pulp from the results. More research is needed to evaluate the performance of vitality tests in the veterinary clinical setting, to examine the effect of age, analgesic use, tooth type, correlation with radiographic findings and correlation with pathology of affected teeth. 16
Conclusion
The EPT was shown to be a highly reliable diagnostic test to assess pulpal health of permanent teeth in dogs whereas the accuracy of CPT was conditional on the responsiveness of the patient to testing of control healthy teeth with a high prevalence of absence of pain reaction on control healthy teeth. The TTI is the least effective of the three pulp tests evaluated. The electric pulp tester is an inexpensive, safe and easy to use adjunct tool for the early diagnosis of pulpal necrosis and associated pain. The instrument could be easily integrated into veterinary dental practice for the diagnosis of pulp disease in dogs. To the authors’ knowledge, this study is the first to evaluate the reliability of EPT, CPT and TTI in veterinary dentistry and more research with the inclusion of histologic data as well as more detailed information regarding patient responses in large-scale pulp vitality testing studies would provide the most accurate and clinically applicable data.
Footnotes
Acknowledgments
The authors gratefully acknowledge the assistance of Mr Marco Langlois, Faculty of Veterinary Medicine, Université de Montréal, in the production of photographs and videos presented in this document.
Funding
The authors disclosed receipt of financial support for the research of this article: Fonds du Centenaire (RH000669), Faculty of Veterinary Medicine, Université de Montréal.
Materials
Atravet, acepromazine maleate; Boehringer Ingelheim, 5158 South Service Road, Burlington, ON, Canada. Hydromorphone HP; Sandoz, Canada Inc., 145 Jules Léger, Boucherville, QC, Canada. PropoFlo 28; Zoetis Canada Inc., Kirkland, QC, Canada. Isoflurane USP; Fresenius Kabi Canada Ltd, Richmond Hill, ON, Canada. Round carbide burs (#1/2, #2; standard, friction grip); Henry Schein, 45 Townline Road, Niagara-on-the-Lake, ON, Canada. Pathfinder stainless steel endodontic hand file (25 mm); Henry Schein, 45 Townline Road, Niagara-on-the-Lake, ON, Canada. Gentle Pulse™ Analog Pulp Vitality Tester; Parkell Inc., Edgewood, NY, USA. MedPro Lubricating Gel; AMG Medical Inc., Montreal, QC, Canada. Endo-Ice; Coltene Whaledent, Cuyahoga Falls, OH, USA. Cotton Pellets size 2 (7/32 in, 2550/Bx), size 3 (5/32, 2500/Bx); Richmond Dental Company, Charlotte, NC, USA. Cotton Pliers, Self-Locking; Henry Schein Animal Health, 3403 Rue Griffith, Saint-Laurent, QC, Canada. Finnoff Ocular Transilluminator (41100, 3.5 V); Welch Allyn, 160 Matheson Blvd E, Mississauga, ON Canada.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fonds du Centenaire, Faculty of Veterinary Medicine, Université de Montréal. (grant number RH000669).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

