Abstract
Background
Dementia prevalence and incidence rise sharply among the oldest-old (≥85 years), yet whether age-at-diagnosis shapes functional trajectories remains unclear. Understanding age-specific patterns is critical for tailoring care strategies.
Objectives
To determine whether age at dementia diagnosis influences patterns of functional decline and recovery, and to identify the functional domains driving age-specific trajectories.
Methods
Using Manitoba Follow-up Study data, participants were grouped by age at dementia diagnosis (<85, 85–89, ≥90) or no dementia. We tracked Basic Activities of Daily Living (BADLs), Instrucmental Activities of Daily Living (IADLs), and SF-36 Physical/Mental scores. Generalized additive mixed models and mixed-effects random forest analyses assessed trajectories, dependence, and recovery.
Results
Age at diagnosis showed differences in trajectories. The ≥85 group showed steep post-diagnosis decline; the <85 group declined more steadily. Complex tasks (e.g., cutting toenail) were most impaired. Recovery occurred, particularly in IADLs and in the ≥90 group.
Conclusions
Age at dementia diagnosis showed meaningful differences in functional decline and recovery, supporting age-specific care strategies.
Keywords
Introduction
Dementia is a progressive, incurable syndrome affecting more than 55 million people worldwide and constitutes a leading cause of death, disability, and healthcare burden in later life (World Health Organization, 2023). Its incidence increases exponentially with age (Corrada et al., 2010; Jorm, 1998; Wolters et al., 2020), from about 4 per 1,000 person-years at age 65–69 to roughly 65 per 1,000 person-years at age 85–89 (Wolters et al., 2020), with higher rates beyond age 90 (Corrada et al., 2010; St John et al., 2025).
The oldest-old (≥85 years) (Lund & Wang, 2020) is an important subgroup in dementia research because advancing age is strongly associated with increased dementia incidence and prevalence (Corrada et al., 2010; Jorm, 1998; Plassman et al., 2007; Wolters et al., 2020). This group is rapidly expanding, projected to triple globally from 126.5 million in 2015 to 446.6 million by 2050 (He et al., 2016), and dementia affects over 30% of those aged 90+ in the community (Plassman et al., 2007). Importantly, the oldest-old are not a homogeneous group: those reaching 90+ without dementia represent a selectively resilient subset who have delayed major diseases (Kravitz et al., 2012). When dementia does occur in this group, its functional trajectories may differ from younger-onset cases, but previous studies have been limited by small sample sizes (Corrada et al., 2010).
Neuropathological evidence supports age-specific heterogeneity in the oldest-old. Dementia in this group is associated with fewer neuritic plaques and neurofibrillary tangles than in younger individuals with dementia (Middleton et al., 2011; Savva et al., 2009; Yang et al., 2013), suggesting differences in pathology or neurocompensation that may shape clinical manifestations. Functional decline is central to both the clinical definition of dementia and its disease burden. Diagnostic criteria require evidence that cognitive deficits interfere with independence in everyday activities, reflecting impairments in memory, executive function, or attention that compromise daily functioning (American Psychiatric Association, 2013; World Health Organization, 2022). In parallel, functional status is a key determinant of independence, care needs, quality of life, and survival (Bozkurt et al., 2016; Gao et al., 2023; Sharma, 2020).
Despite extensive work on cognitive decline and neuropathology in the oldest-old, several gaps remain in understanding functional trajectories. First, most research treats the oldest-old as a homogeneous group, largely due to the small samples (Corrada et al., 2010), ignoring heterogeneity that may exist within this population. Second, studies typically focus on total BADL or IADL scores rather than specific sub-items, obscuring which functional abilities drive overall decline and which may show recovery. Limited research has identified that IADL functions like financial management and shopping deteriorate earliest (Edjolo et al., 2016; Njegovan et al., 2001), but whether the sequence and timing of specific functional losses vary by diagnosis age remains unknown. Third, while some evidence suggests functional recovery can occur even with dementia (Chiu et al., 2004), age-specific patterns of recovery have not been systematically examined.
Understanding age-specific functional trajectories has important clinical and policy implications. The trajectory of functional decline—and the order in which IADLs and BADLs are lost—may be informative for prognosis and could prompt clinical and cognitive assessment. Functional decline may herald the onset of dementia, and prompt clinical and cognitive assessment. It may also guide homecare supports for functions lost earlier and adapting as later losses emerge. Understanding the disabling process of dementia would help caregivers and older adults anticipate disease progression.
This study addresses these gaps by examining functional and quality-of-life trajectories surrounding the dementia diagnosis in individuals aged 85 and older, stratified by age at dementia diagnosis (<85, 85-89, ≥90 years), and compared them to individuals without a dementia diagnosis. We used both chronological age-based and diagnosis-timeline approaches to: (1) characterize whether trajectory patterns of BADL, IADL, and quality of life (Physical and Mental Component Scores) differ by age at diagnosis; (2) identify which specific functional items contribute to overall decline in each age group; and (3) assess whether recovery potential in BADL and IADL sub-items differs across age-at-diagnosis groups. These findings have important implications for understanding the natural history of functional change in the oldest-old with dementia and for developing targeted interventions for this rapidly growing but understudied population.
Method
Data Source
The Manitoba Follow-Up Study (MFUS) serves as the data source. MFUS is a prospective cohort study initiated in 1948 (Tate et al., 2015). It consisted of 3983 Royal Canadian Air Force aircrew male recruits (mean age 31). Only 42 participants have been lost to follow-up over the entire study period from 1948 to the present (St John et al., 2016). Initially focused on cardiovascular epidemiology, the study expanded its scope in 1996 to successful aging, as population aging became a policy concern and most MFUS participants were ≥75 years old.
MFUS introduced the Successful Ageing Questionnaire (SAQ) in 1996, and it was administered in 2000, 2002, and annually since 2004. Response rates have consistently exceeded 70% among living participants at each assessment wave (St John et al., 2016). The SAQ includes BADL, IADL, and SF-36 to assess functioning and quality-of-life (Ware & Sherbourne, 1992). For this analysis, we included participants with at least two SAQ measurements to enable trajectory modeling and excluded those who could not fully understand or answer questions. The MFUS dataset is a valuable resource for investigating functional decline trajectories associated with dementia through its long-term follow-up, large cohort, and detailed functional status information.
Study Variables
Health-related quality of life was assessed using the SF-36, which captures the Physical Component Score (PCS) for physical functioning and the Mental Component Score (MCS) for mental health functioning (Cooper, 2007). Both scores range from 0 to 100, with higher scores indicating better health status (von der Heyde & Droege, 2007).
Daily functioning was measured using BADL and IADL scales. IADLs involve more complicated thinking abilities, such as shopping and managing finances, while BADLs are fundamental self-care activities like walking and feeding oneself (Edemekong et al., 2023). The BADL scale includes 15 items, and one sub-item (“nursing care”) was only administered once and excluded from analysis due to inconsistent interpretation among participants. The IADL scale includes 9 items. Higher scores indicate greater functional limitations (Wallace et al., 2007).
Follow-up measurements were collected over 27 years (1996–2023). Dementia status and date of diagnosis were determined by MFUS study physicians through review of participants’ medical records. Formal cognitive testing data were not abstracted for the cohort because cognitive assessments and testing practices varied over time and place and were not uniformly available for all participants. Consequently, we cannot report the proportion of participants who underwent formal cognitive testing.
Statistical Analysis
We modeled non-linear functional trajectories of BADL, IADL, MCS, and PCS scores across three age-at-diagnosis groups (<85, 85–89, ≥90 years) using Generalized Additive Mixed Models (GAMMs). GAMMs use flexible smooth functions to capture complex, non-linear trends, and accommodate unbalanced, irregularly spaced longitudinal data (Gueorguieva & Krystal, 2004; Lin & Zhang, 1999). The GAMM model (Lin & Zhang, 1999; Sørensen et al., 2021) can be written as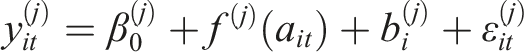
We fitted the models with
We implemented two modeling approaches: (1) chronological age-based trajectories examining functional and quality-of-life changes as a function of age; and (2) diagnosis relative time trajectories aligning outcomes relative to dementia diagnosis (negative values representing pre-diagnosis periods).
For item-level analysis, we calculated functional dependency prevalence for each IADL and BADL sub-items at multiple time points relative to dementia diagnosis. Dependency was defined as any difficulty or need for assistance in performing a given IADL or BADL task. We used this inclusive definition to capture the full spectrum of functional limitation, as even mild difficulty represents meaningful functional decline in this population. Recovery was defined as transition from any level of dependency (difficulty or requiring assistance) to complete independence (no difficulty and no assistance needed). Proportion of participants with dependency at each time point was calculated separately for the three age-at-diagnosis groups and a non-dementia group, and visualized with line plots.
We used mixed-effects random forest (MERF) to identify the relative importance of specific functional items contributing to overall functional decline at dementia diagnosis. MERF allows flexible nonparametric estimation of fixed effects and random effects, effectively handling non-linear trends and within-person correlation (Hajjem et al., 2014). It shows superior prediction accuracy for high-dimensional longitudinal data compared to alternative methods (Yiğit & Ahmed, 2024). Variable importance was quantified using the Mean Decrease in Impurity (MDI) metric (also known as Gini Importance), defined as the total drop in Gini impurity attributable to a feature across all splits in which it appears. This metric reflects each item’s relative contribution to the model’s predictive performance for functional decline. Higher MDI values indicate greater importance. Variable importance was compared across age groups.
Finally, we assessed functional state transitions by calculating the percentage of participants moving from dependent to independent status on each BADL and IADL item between assessments across the entire follow-up period (1996–2023). Transitions were compared between diagnosis groups and the non-dementia group to address whether recovery potential differs by age at dementia diagnosis.
Analyses were conducted using R, version 4.2.3 (R Core Team, Vienna, Austria).
Result
Of the 3,983 men originally enrolled in the MFUS, SAQ-based functional status assessment was not implemented until 1996, when 1,963 participants remained alive. From this group, we excluded 154 participants who were unable to fully understand or answer the questions and 419 participants with only a single SAQ measurement, leaving a final analytic sample of 1,390 participants (Supplemental Figure S1).
Supplemental Table S1 summarizes baseline characteristics by diagnosis group. All groups showed minimal BADL impairment at baseline (median = 0.0 across all groups). Some differences were observed across groups, with the ≥90 group being slightly older and having higher PCS scores, while the <85 group had higher IADL impairment and lower MCS scores at baseline. Supplemental Table S2 compares included versus excluded participants. Excluded participants were older and had greater functional impairment (higher BADL and IADL scores) and lower quality of life scores (lower MCS and PCS).
Age at Dementia Diagnosis and Functional Trajectories
All trajectory models showed statistically significant age- or time-related changes within each diagnosis group (p < 0.001 for all smooth terms). Between-group differences described below represent substantive comparisons of trajectory shapes, timing, and slopes and magnitude of functional change.
Stratification by age at dementia diagnosis showed differences in functional trajectories across both BADL and IADL domains (Figure 1). Differences were more apparent when trajectories were examined by chronological age, whereas trajectories aligned to the time of diagnosis showed more comparable overall shapes across older diagnosis groups. • Individuals diagnosed before age 85 experienced steady, linear functional decline beginning years before diagnosis and continuing post-diagnosis. BADL scores declined progressively from near zero at age 70 to approximately 2–2.5 points by age 95 (Figure 1a). IADL showed similar linear deterioration but with steeper slopes, rising to approximately 5–6 points by age 95 (Figure 1c). • Those diagnosed at 85–89 years maintained functional stability similar to non-dementia controls until approximately age 85, after which their function deteriorated dramatically. BADL scores rose sharply from near zero to approximately 6 to 6.5 points by ages 95–100. IADL scores followed the same deterioration pattern, reaching an even greater magnitude of approximately 8 to 9 points by age 100 (Figure 1c). • Individuals diagnosed at age 90 or older showed the mildest functional decline. They maintained functional scores comparable to the non-dementia group until roughly age 90, followed by moderate increases in impairment. BADL scores reached approximately 2.5–3 points by age 100 (Figure 1a), and IADL scores reached 6–7 points (Figure 1c). Functional trajectories across dementia diagnosis groups by age and diagnosis timeline
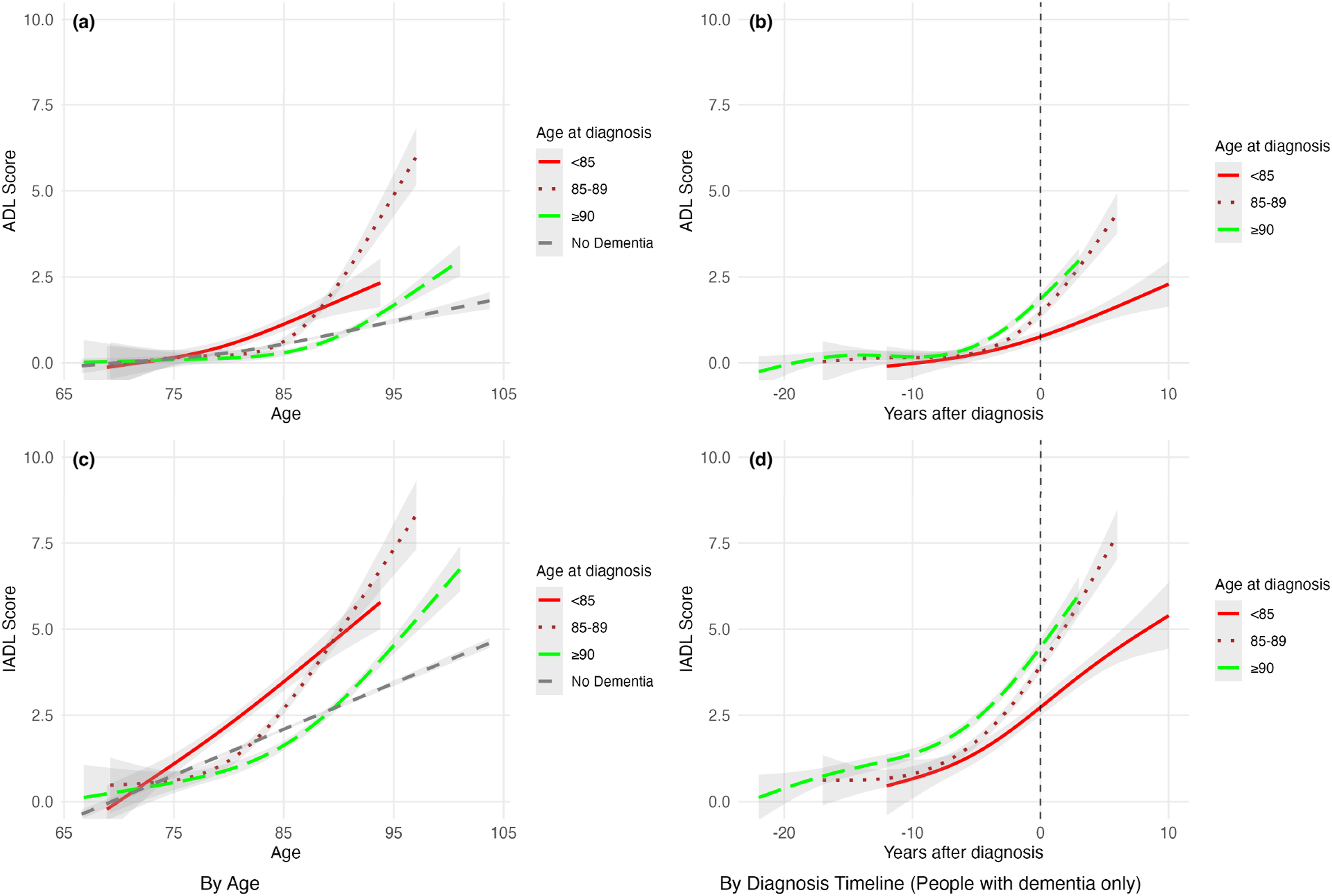
The non-dementia reference group maintained low BADL scores (approximately 0-1.5 points) and IADL scores (approximately 1-4 points) across the entire age spectrum from 65 to 105 years (Figures 1a, 1c).
When trajectories were examined relative to diagnosis timing (Figures 1b, 1d; vertical dashed line at year 0), all dementia groups showed relatively stable BADL and IADL scores during the pre-diagnosis period, extending up to 15–20 years before diagnosis. Following diagnosis, the <85 group continued its steady decline. Both the 85–89 and ≥90 groups showed acceleration in functional decline immediately after diagnosis, with broadly similar trajectory shapes on the diagnosis timeline, although differences remained in the overall magnitude of impairment.
Item-Level Functional Decline Patterns
To understand what drives these age-specific trajectory patterns, we examined which functional tasks showed the greatest impairment and contributed most to overall decline.
BADL item-level analysis found that complex motor tasks requiring dexterity, balance, and strength showed the earliest and greatest functional limitations across all age groups (Figures 2). “Cutting toenails” showed the highest increases in dependency, particularly in the ≥90 group where the proportion of participants with limitations increased from under 5% at 20 years before diagnosis to 50% at diagnosis. “Going up and down stairs” showed similar patterns, increasing from <5% to approximately 30% at diagnosis in the ≥90 group. In contrast, core self-care activities—such as “eating,” “using toilet,” “washing or bathing,” and “getting in and out of bed”—remained dependent in less than 10% of individuals across all age groups, even at the time of diagnosis. Proportion of limitations in 15 BADL items across the dementia diagnosis timeline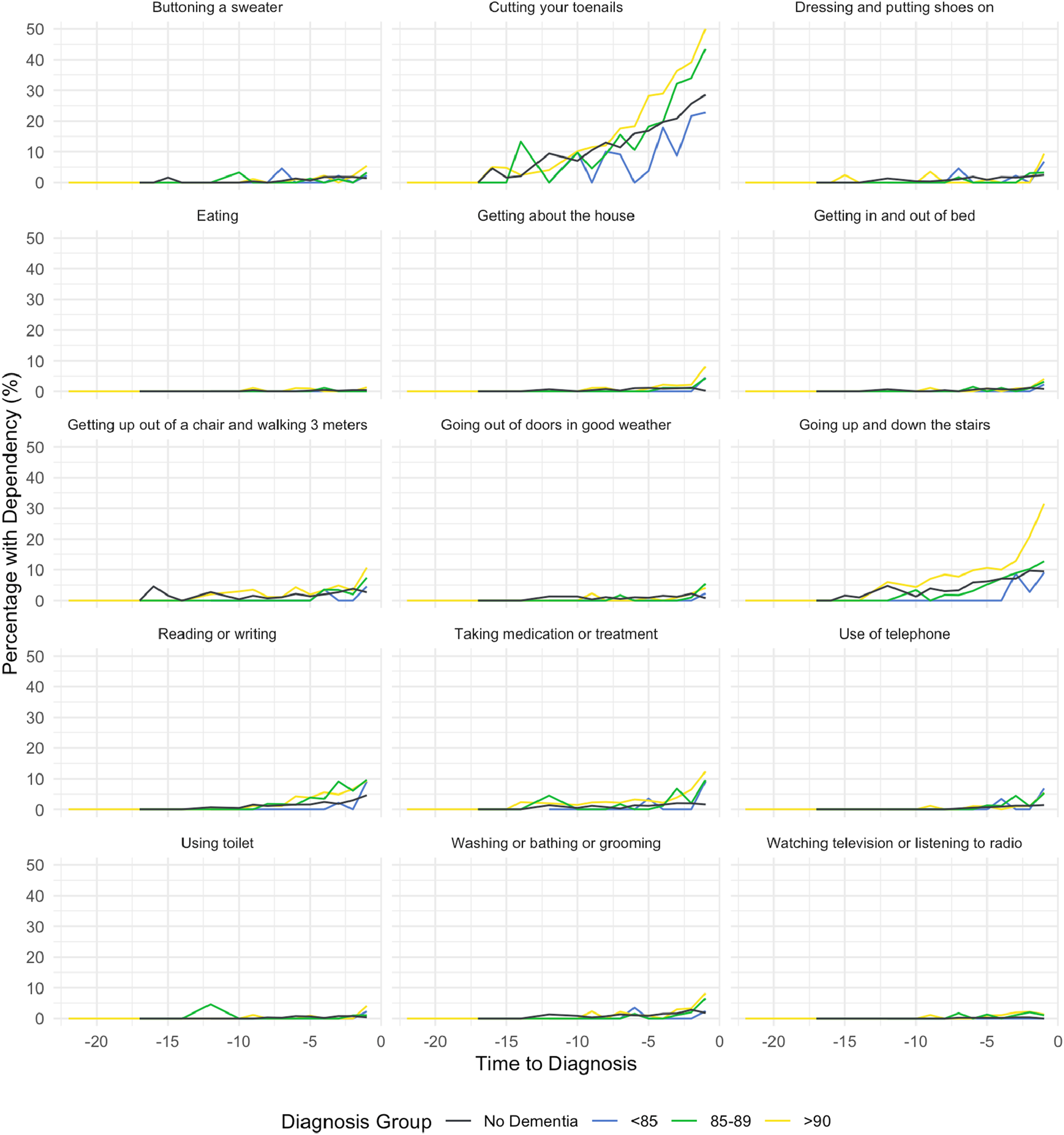
IADL item-level patterns showed that physically demanding tasks demonstrated the most rapid functional decline (Figure 3). The items “Major house or household repairs,” “doing heavy housework,” and “shovelling and yardwork” showed steep increases, with the ≥90 group reaching approximately 75–90% dependence by diagnosis. In contrast, less physically demanding tasks such as “making a cup of tea,” “shopping,” and “managing financial affairs” showed earlier onset of limitations but remained below 30% dependence at diagnosis across most groups. Proportion of limitations in 9 IADL items across the dementia diagnosis timeline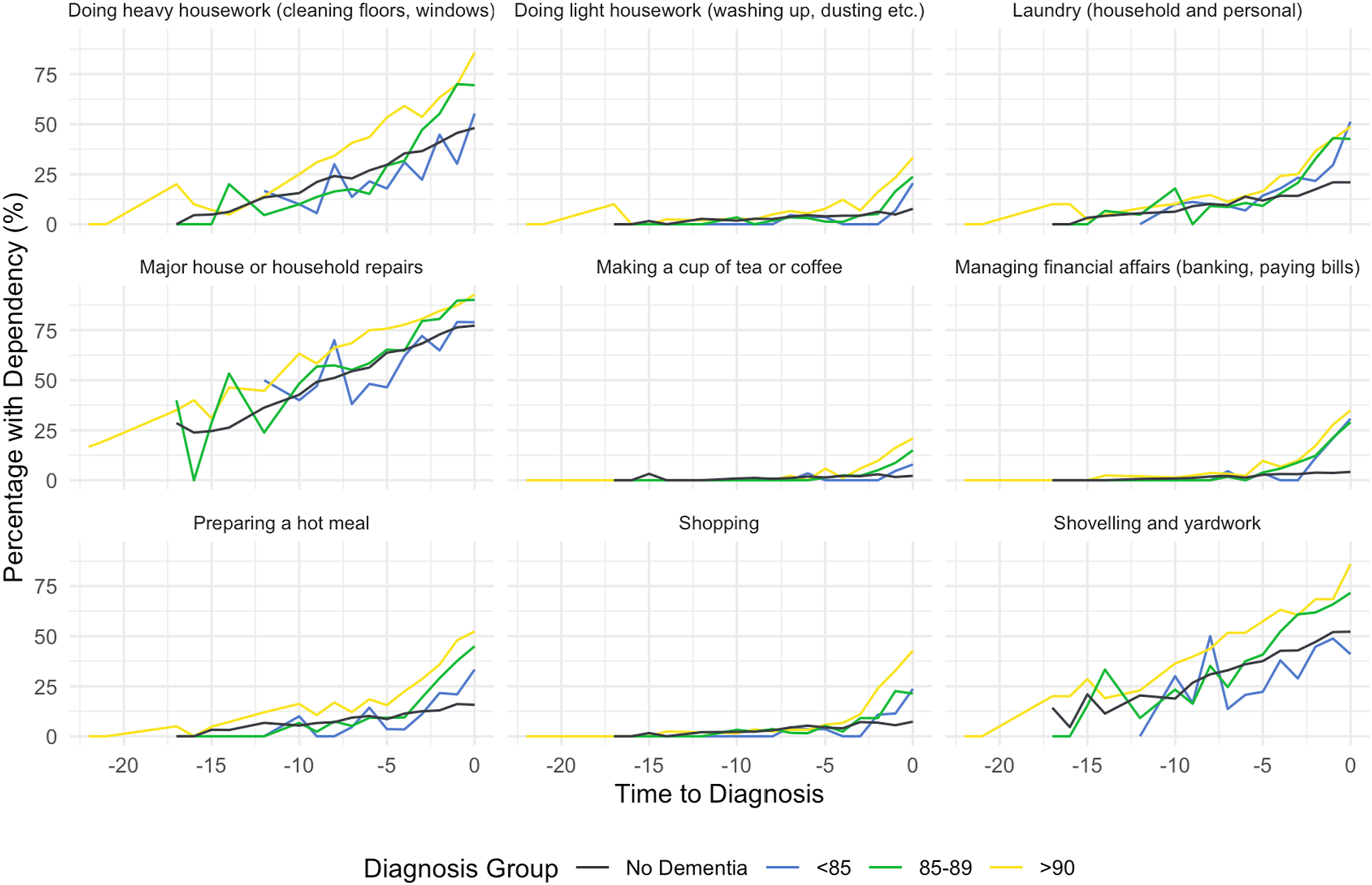
Item Contributions to Overall Functional Decline
To statistically confirm which functional items contributed most to overall decline, we conducted mixed-effects random forest analyses (Supplemental Figures S1 and S2). For BADL, “cutting toenails” and “going up and down stairs” showed the highest variable importance across all age groups, confirming these complex motor tasks as primary drivers of BADL decline. For IADL, “doing heavy housework” consistently ranked as the top contributor across all groups, followed by “shovelling and yardwork” and “shopping.” These findings corroborate the item-level dependency patterns observed in Figures 2 and 3.
Recovery Patterns Vary by Age at Diagnosis
Given the distinct decline patterns by age at diagnosis, we examined whether recovery potential also varied by diagnosis age. Overall, IADL items showed higher recovery rates than BADL items across all groups, and recovery potential increased with older age at diagnosis (Figure 4). Percentage of functional status changes in BADL and IADL items by diagnosis age group compared to no dementia group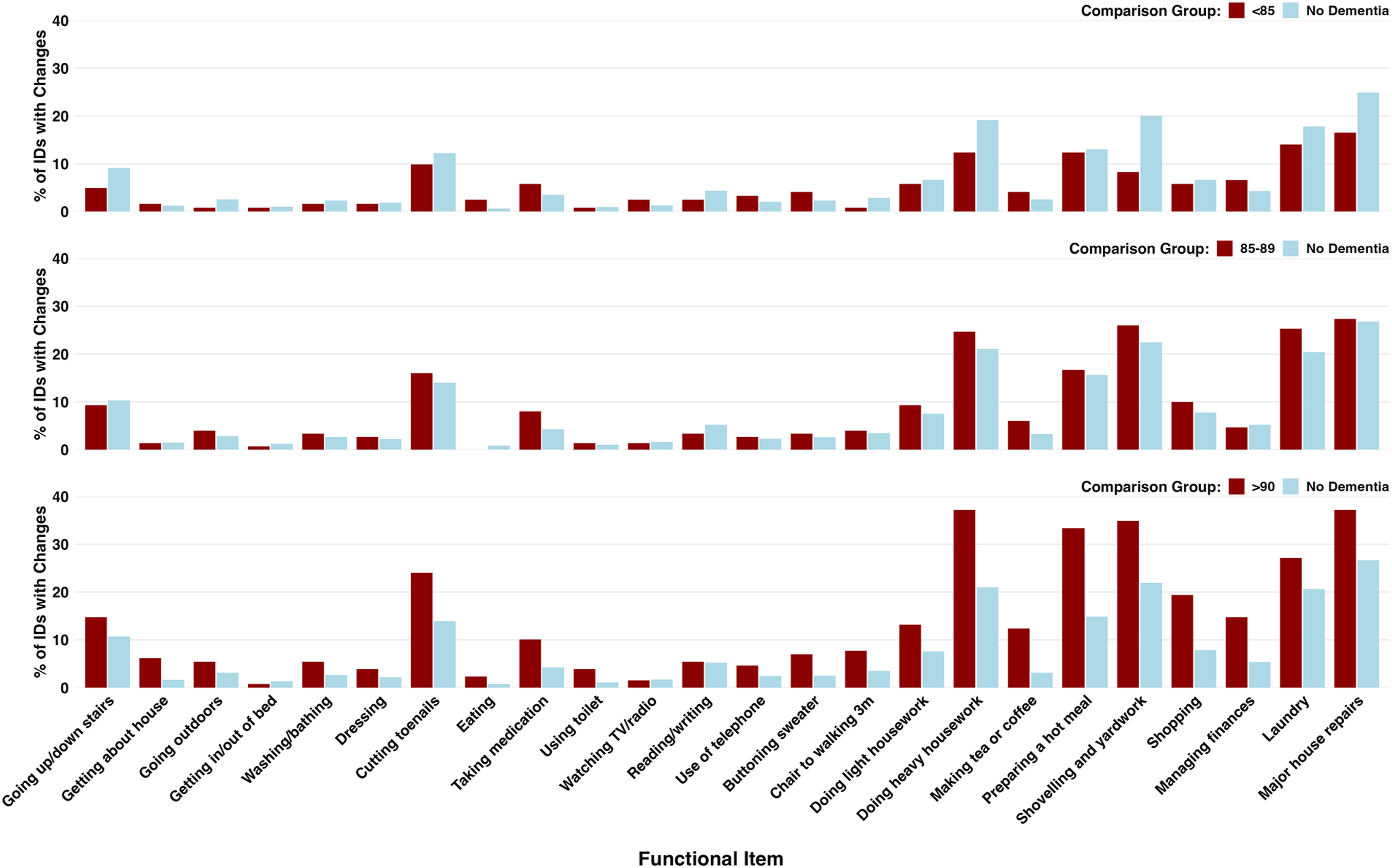
Among those diagnosed before age 85, functional recovery rates were the lowest in both domains. For IADL, the highest recovery rates were observed for “major house or household repairs” (16.5%), “laundry (household and personal)” (14.0%), “preparing a hot meal” (12.4%), and “doing heavy housework” (12.4%), but all were substantially lower than the non-dementia group. For BADL, recovery was limited across most items, with “cutting toenails” showing approximately 10% recovery and most other items below 5%, including core self-care activities like “getting in and out of bed,” “using toilet,” and “getting up out of a chair and walking 3 meters.”
Those diagnosed at 85–89 years showed substantially higher recovery rates than the <85 group. For IADL, several items showed recovery rates comparable to or exceeding non-dementia controls, including “major house or household repairs” (27.3%), “shovelling and yardwork” (26.0%), “laundry” (25.3%), and “doing heavy housework” (24.7%). However, “managing financial affairs” remained low (<5%). BADL recovery remained limited below 5%, except for “cutting toenails” (16.0%) and “going up and down the stairs” (9.3%).
Those diagnosed at age 90 or older showed the highest recovery rates across multiple domains. For IADL, this group showed recovery rates substantially exceeding those of non-dementia controls in several items: “major house or household repairs” (37.2% vs 26.7% in non-dementia), “doing heavy housework” (37.2% vs 21.0%), and “shovelling and yardwork” (34.9% vs 21.9%). For BADL, the ≥90 group showed recovery rates for “cutting toenails” (24.0%), “going up and down stairs” (14.7%), and several other mobility-related items that were comparable to or exceeded those in younger diagnosis groups.
Quality of Life Trajectories
Given the age-specific differences in functional trajectories and recovery potential, we further examined whether objective functional impairment translated to lower subjective quality of life. Physical and mental quality of life showed distinct trajectories (Supplemental Figures S2). PCS declined steadily with age across all groups, from approximately 55 at age 65 to about 35 by age 100. The ≥90 group maintained slightly higher PCS in their early 90s than other dementia groups, though all groups converged to similar levels by age 95. By diagnosis timeline (Figures S2b), all groups showed parallel declines in PCS from approximately 50–55 points at 20 years before diagnosis to about 35–40 points at 10 years after diagnosis.
In contrast, MCS remained remarkably stable and parallel across both age (Figure S2c) and diagnosis timelines (Figure S2d). The <85 group had slightly lower MCS scores than other groups.
Discussion
Age at dementia diagnosis showed differences in functional trajectories in the oldest-old, particularly in the rate, timing, and recovery of functional decline. Individuals diagnosed before age 85 showed a gradual but sustained decline with minimal recovery potential. Those diagnosed at 85–89 years experienced abrupt deterioration after a long period of preserved function, reaching the most severe impairment. Those diagnosed at age 90 or older showed mild age-related decline and greater recovery capacity that can exceed that of non-dementia controls. These findings suggest that functional loss in dementia does not follow a uniform course that age at diagnosis may reflect underlying differences in physiological reserve and compensatory capacity.
Those diagnosed at younger old (<85 years) had gradual functional decline but the lowest recovery rates. This trajectory likely reflects a classic neurodegenerative phenotype of dementia that chronic, progressive neurodegeneration beginning years before diagnosis (Amieva et al., 2008). Prior work suggests that younger-old individuals may carry stronger genetic risk (Farrer et al., 1997), and that higher pathology burden together with lower cognitive reserve could accelerate neurodegeneration and limit functional recovery (Savva et al., 2009; Stern, 2012).
The 85–89 group demonstrated preserved function until age 85, then precipitous deterioration to the highest impairment levels (6–6.5 BADL, 8-9 IADL points). This pattern likely represents a critical threshold where compensatory mechanisms, maintained throughout the 80s through recruitment of additional brain regions (Bishop et al., 2010; Cabeza et al., 2002; Reuter-Lorenz & Cappell, 2008), eventually fail when pathological accumulation exceeds compensatory capacity (Stern, 2002). Their intermediate recovery rates (24–27% for key IADLs) might reflect remaining reserve capacity limited by the severity of acute decompensation.
Individuals diagnosed at age 90 or older showed slower age-related functional decline but higher recovery rates, distinguishing them from those diagnosed at 85–89 years. Although diagnosis-anchored trajectories of the two oldest groups appeared broadly similar in shape, age-based analyses revealed substantially milder decline and greater recovery among those diagnosed at age 90 or older. Individuals who reach very old age without dementia diagnosis until 90+ likely represent a selected population who have escaped major diseases or delayed disease onset and duration to the end of life (Kravitz et al., 2012). Genetic evidence suggests that exceptionally long-lived individuals often carry variants that reduce susceptibility to age-related diseases (Perls et al., 2002), which may support the preservation of neural and physiological reserve into late life. This unique longevity phenotype is also supported by neuropathological evidence. Neuropathological studies show the oldest-old with dementia have fewer neuritic plaques and neurofibrillary tangles than younger individuals with dementia (Savva et al., 2009; Yang et al., 2013). This relatively reduced pathological burden, combined with the inherent robustness of a selected survivor group, could enhance the brain’s capacity for functional plasticity and rebound. The fact that their recovery rates exceed those of cognitively healthy age-matched individuals suggests this enhanced resilience extends beyond cognitive domains alone, implying a greater overall functional reserve capacity.
Task Complexity Determines the Sequence of Functional Loss
Our trajectory analysis supported the established hierarchy of functional decline across all age groups. IADL impairments begin earlier than BADL limitations (Judge et al., 1996; Millán-Calenti et al., 2010; Tsai et al., 2021; Verlinden et al., 2016). Most groups maintained BADL stability until advanced ages, while IADL decline occurred earlier and varied in timing and severity by diagnosis age. These findings support previous research identifying IADL deficits as early cognitive decline indicators (Pérès et al., 2008).
Our item-level analysis further showed a hierarchical decline pattern based on physical demands. The decline sequence identified in our study aligned with Ferrucci et al. (1998) disability domains, which categorized activities by similar underlying functions and execution difficulty: (A) complex manual dexterity and balance (highest difficulty); (B) long-distance mobility and balance; (C) upper limb control and standing balance; and (D) upper limb control in a seated position (lowest difficulty). Among BADL items, “cutting toenails” and “going up/down stairs” were primary contributors of functional decline, consistent with previous research findings (Kingston et al., 2012). “Cutting toenails” requires complex manual dexterity and belongs to Ferrucci’s highest difficulty domain. Fine motor functions (such as precise pinching) and gross motor functions (such as gripping and grip strength) decline with aging and are associated with MCI, and to a greater extent with dementia (Kravitz et al., 2012; You et al., 2024). Stair climbing requires physical balance, and strength, belonging to Ferrucci’s second domain. In the IADL domain, “heavy housework,” and “yard work” showed the most rapid functional decline. Heavy housework and yard work similarly belong to Ferrucci’s highest difficulty domain, requiring dexterity, physical balance and strength (Bendayan et al., 2017), which explains why they represent the earliest functional decline items, along with “cutting toenails.”
Between Decline and Recovery
Functional recovery analysis showed patterns opposite to functional decline. Although functional decline followed the classic pattern where IADL impairments occur before BADL limitations, recovery potential showed a different hierarchy. Previous study reported overall ADLs recovery rates (Chiu et al., 2004), but our item-specific analysis across both BADL and IADL found differences in recovery potential. Complex BADL tasks like “cutting toenails” and “stair climbing” showed higher recovery potential. IADL items similarly demonstrated higher recovery rates. In contrast, core self-care activities like “toileting,” “eating,” and “transferring in and out of bed” consistently showed minimal recovery potential across all age groups.
Katz’s ADL theory (Katz et al., 1963) helps explain these findings: older adults tend to lose the abilities to perform daily tasks in reverse order in which they were acquired in childhood, with basic skills lost last and remaining, more complex tasks retain greater recovery potential when conditions allow. This suggests items causing functional decline are not always irreversible (Chiu et al., 2004), and may be important targets for rehabilitation intervention.
Activities of Daily Living as a Measure of Practical Take-away by Age at Dementia Diagnosis
Across all ages, IADLs decline before BADLs, but the timing and magnitude of change differ by age at diagnosis. For those diagnosed before age 85, the decline tends to be steady with relatively limited recovery opportunities, particularly for complex tasks. Individuals diagnosed at 85–89 years show relative stability prior to diagnosis, followed by more rapid post-diagnosis deterioration; importantly, this group demonstrates meaningful recovery potential in several IADL domains when targeted rehabilitation and supportive home environments are provided. Among the oldest group (≥90 years), post-diagnosis deterioration begins earlier but recovery in IADLs can be pronounced, with higher recovery in several tasks compared with younger groups, though BADL recovery remains limited. Overall, IADL domains—especially those involving complex planning, heavy household work, and mobility-related tasks—offer the greatest opportunities for functional recovery and maintenance of independence with appropriate supports. These patterns support a strategy of early, age-tailored monitoring focused on IADLs and timely rehabilitation to prolong independence across the oldest-old with dementia.
Successful Aging
Quality-of-life trajectories analysis provided additional insights. PCS declined with advancing age, while MCS remained relatively stable across all groups, consistent with previous research (Ding et al., 2021). This pattern indicates that dementia does not necessarily result in a decline in quality-of-life, and subjective well-being may protect against cognitive decline (Willroth et al., 2023). These findings suggest the potential for successful aging despite dementia.
Strengths and Limitations
This study has several strengths. First, the Manitoba Follow-up Study provides 27-year follow-up (1996–2023) with low non-mortality attrition and high response rates (St John et al., 2016). This allows comprehensive functional trajectories mapping throughout the aging process and detailed examination of functional change patterns before and after a dementia diagnosis. Second, we used three age groups (<85, 85–89, ≥90) informed by epidemiologic evidence of dementia incidence doubling approximately every 5.5 years in the oldest-old (Corrada et al., 2010). While finer stratification of the 90+ group would be ideal, the sample size (
There are limitations. First, SAQ-derived functional data were available only for participants who survived to 1996 and had at least two SAQ assessments. Of the 3,983 men at baseline, 1,390 met the inclusion criterion for trajectory analysis. The remaining participants were excluded mainly due to death or insufficient SAQ data. Because death is strongly related to poorer physical, cognitive, and mental health status, missingness is likely not random and may introduces survivor bias. This may lead to underrepresentation of the frailest individuals in estimate trajectory and recovery patterns. Future studies could explicitly model death as a competing risk or apply methods developed for data that are missing not at random (e.g., pattern-mixture or joint models) to better evaluate the potential impact of informative dropout on estimated decline and recovery. Second, the MFUS cohort consists of male Canadian aircrew members, which may limit generalizability to women or to non-military populations. Although the MFUS cohort has many comparable features of aging populations in Canada, such as the functional status (Statistics Canada, 2002), mortality, geographic distribution, and marital status (Statistics Canada, 2012), the extrapolation of our findings to other groups should be done with caution. Third, we used an inclusive recovery definition (any transition from dependency to independence) without testing alternative definitions such as sustained recovery or stratification by dependency severity. Future work with standardized, prospectively collected cognitive and functional measures could explore whether recovery patterns vary by initial impairment level or triggering event type. Finally, dementia status in MFUS was ascertained from clinical diagnoses recorded in medical records rather than standardized research-based cognitive criteria or uniformly abstracted cognitive test results. This may introduce misclassification of dementia status relative to true cognitive impairment. Such misclassification is expected to be non-differential with respect to functional trajectories and recovery, which would bias effect estimates toward the null rather than create spurious patterns. Future work with standardized cognitive testing data could help refine these trajectory estimates. Despite these limitations, our study provides valuable insights into the patterns and likelihood of functional recovery in dementia across the aging spectrum.
Implications for Practice and Policy
Implications for Caregivers
Caregivers play a critical role in recognizing that IADLs often fail before basic activities, with signs that may be subtle. Early coordination with occupational or physical therapy can help identify modifiable risks at home, and attention should be given to practical adaptations that support complex IADLs (e.g., managing household tasks, shopping, transportation) and safety (lighting, stair use, grab bars, clear pathways). Maintaining engagement in meaningful activities and breaking tasks into achievable steps, along with simple, consistent routines and clearly organized environments, can reduce cognitive load and support ongoing function. Promptly seeking assistance when difficulties emerge and coordinating with health-care providers to address evolving needs are essential components of care.
Implications for Health-Care Providers
Health services need to consider the expected functional trajectory and match social and health care services that anticipate this trajectory, rather than relying primarily on reactive responses once functional changes become apparent. While it seems obvious, it is also critical that services match the disability pattern, they often do not (Reckrey et al., 2021). Anticipating the needs based upon expected trajectories may be of use in planning services at both individual and population levels. Health-care teams should integrate routine screening for IADL impairment into dementia care, prioritizing individuals aged 85 and above, using brief, feasible assessment tools. Timely referrals to OT/PT and early home safety assessments can slow or prevent functional decline, while applying a diagnosis–timeline framework helps distinguish normal aging from disease-related trajectories and tailor rehabilitation goals accordingly. Multidisciplinary coordination—including rehabilitation specialists, primary care, social work, and caregiver support—should be organized to address the evolving needs of the oldest-old post-dementia diagnosis.
Cost and Policy Implications
Policies that support early IADL-focused supports and rehabilitation may reduce later, higher-cost BADL dependencies by preserving independence for longer and delaying institutionalization. Investment in community-based OT/PT services, caregiver training, and subsidies for home modifications can enhance functioning and quality of life while potentially lowering overall health-care costs. Integrating dementia care with aging services and ensuring access to home safety assessments and adaptive devices across the oldest-old may improve outcomes without requiring large-scale system overhauls.
Conclusion
We identified age-specific patterns of functional decline and recovery around dementia diagnosis in the oldest-old. Individuals diagnosed before age 85 showed gradual decline with minimal recovery. Those diagnosed at 85–89 years experienced abrupt post-diagnosis deterioration reaching the most severe impairment levels. Those diagnosed at age 90 or older showed mild age-related decline with exceptional recovery capacity exceeding non-dementia controls, showing preserved physiological reserve in this select survivor group. Functional decline followed a universal hierarchy across all age groups. IADL impairments preceded BADL limitations, and complex tasks requiring dexterity and balance declined earliest. However, recovery showed a reverse pattern, with complex tasks like “cutting toenails” and “stair climbing” showing higher recovery potential than core self-care activities. Physical quality of life declined with age while mental well-being remained stable, suggesting potential for successful aging despite dementia.
These findings support age-specific monitoring and rehabilitation strategies. Early IADL decline in complex motor tasks should prompt cognitive assessment. Rehabilitation should target complex tasks with higher recovery potential. Understanding these trajectories can inform service planning and conversations with older adults about expected functional changes.
Supplemental Material
Supplemental Material—Age-Dependent Functional Decline and Recovery Patterns After Dementia Diagnosis in the Oldest-Old
Supplemental Material for Age-Dependent Functional Decline and Recovery Patterns After Dementia Diagnosis in the Oldest-Old by Yan Sin Leung, Depeng Jiang, Philip St John, Robert Tate in Journal of Aging and Health
Footnotes
Ethical Considerations
Health Research Ethics Board (HREB) of the University of Manitoba has approved this study involving human participants (HS2019:286).
Consent to Participate
Signed informed consent was not required from study participants in 1948. However, the Human Research Ethics Board of the University of Manitoba has acknowledged that the continued participation of MFUS members in surveys and medical examinations constitutes consent to the study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. St John and Dr. Tate have received CIHR funding for other analyses and projects. Dr. St John serves as an unpaid Board Member of Age and Opportunity and has received speaking honoraria from McMaster University and the Regional Geriatric Program of Eastern Ontario, as well as consulting fees from the University Health Network in Toronto.
Data Availability Statement
Data are available on reasonable request
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
