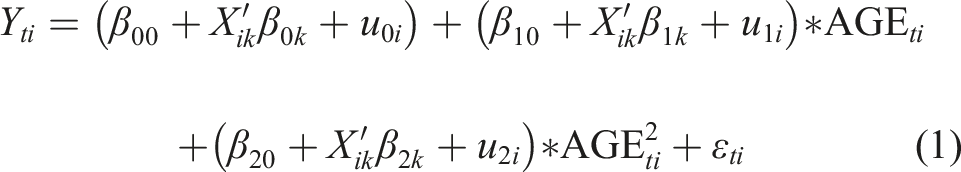Abstract
Keywords
Introduction
Alzheimer’s disease and related dementias (ADRD) are a leading cause of disability and mortality in late life (AA, 2023). The number of Americans affected by ADRD is projected to double to 14 million by 2060 with the greatest burden among older women and racial/ethnic minorities (Nichols et al., 2016). Because extant treatments are limited to symptoms of ADRD after its occurrence, interventions addressing cognitive decline at early preclinical phases are crucial in slowing the progression of decline and delaying dementia onset (Prince et al., 2016). In light of rapid population aging and widening social disparities, assessing life course cognitive trajectories in the overall U.S. population and by population subgroups is essential to national surveillance of ADRD risk and efforts to promote cognitive well-being and delay the disease onset through early intervention.
We identify two major gaps in existing research on cognitive aging. First, characterization of age-related cognitive change over the life span is lacking. Examining the longitudinal nature (timing and course) of cognitive changes preceding late life dementia is a critical first step in understanding the developmental trajectory of cognitive aging and environmental risk factors (Albert et al., 2007; Whalley et al., 2006; Wilson et al., 2002). Previous research shows differences in the shape of trajectories of cognitive change that are predictive of disease risk: not only deficits at baseline, but also a steeper slope of decline in memory and overall cognitive performance at follow-ups increased subsequent risk of dementia (Albert et al., 2007; Walsh et al., 2022). These findings are from older adults and suggest distinctions between normal cognitive aging (stable declines) and pathological cognitive declines (progressive declines) (Alty et al., 2023; Salthouse, 2019) that should be further investigated in longitudinal studies of adults of all ages in that cognitive aging is widely thought to be a lifelong process. The “brain/cognitive reserve hypothesis” proposes that brain functions developed early in life and consolidated in midlife may buffer against later-life neurodegenerative disease and dementia (Gow et al., 2011; Salthouse, 2009; Stern et al., 2023). This hypothesis implies that AD related pathophysiological changes are established long before old age but has rarely been tested empirically. The largest barrier to interrogating the hypothesis is the lack of life course studies of population-based data. Previous studies are limited due to the use of small non-representative samples and cross-sectional or short-term longitudinal analyses within a single life stage (e.g., old age), and confounding of aging and cohort effects (Dodge et al., 2014; Wilson et al., 2002).
Second, social disparities in cognitive aging are not fully documented or understood. Previous research generally finds differences in average cognitive function by gender, race/ethnicity, education, and birth cohort (Diaz et al., 2016; Glymour & Manly, 2008) as well as childhood health and disadvantage (Brown, 2010). However, the influence of each on the trajectory of cognitive change remains unclear. Existing evidence is largely based on older adults and shows mixed results of differential baseline cognitive performance and rates of change with age by population sub-groups (Ardila et al., 2000; Bloomberg et al., 2021; Brown, 2010; Brown & Mutambudzi, 2022; Karlamangla et al., 2009; Levine et al., 2021; Weuve et al., 2018). Discrepancy in previous research may have arisen from differences in sample characteristics, cognitive measures, study designs, and analytic methods, but most importantly, a lack of life course approach. As a result, the link between early-life social disadvantages and later-life cognitive decline has not been consistently established. Key questions remain regarding the underlying life course mechanisms of cognitive decline: when do social disparities in cognitive function first begin to emerge and how long do they last? In sum, a fundamental challenge to bridging the above gaps is that no single, nationally representative study has observed the full life course that begins in the early-life period preceding the occurrence of peak cognitive performance and extends through old age (Whalley et al., 2006).
We address this challenge with an extensive longitudinal life course research design (Figure 1) and a novel application of integrative data analysis to determine for the first time the trajectory of cognitive aging throughout life in Americans aged 12 and older, and main demographic and socioeconomic differentials therein. We integrated four U.S. population-based panel studies of over 50,000 individuals from most 20th century birth cohorts followed for up to 25 years, including the National Longitudinal Study of Adolescent to Adult Health (1994–2018), the National Survey of Midlife Development in the U.S. (2004–2017), Health and Retirement Study (1996–2018), and Americans’ Changing Lives study (1986–2011). Our study aims to (1) describe age trajectories of memory and overall cognitive function from young to old age and (2) examine birth cohort variation, and gender, racial/ethnic, and educational disparities in these trajectories. Interrogating these questions provides new knowledge about natural history of cognitive aging and the earliest life stage of cognitive decline. It also improves an understanding of the secular trends of cognitive aging and lifetime cognitive impacts of key risk factors (e.g., gender roles, structural racism, and educational attainment) in order to develop optimal approaches to minimizing or reducing dementia risk for all Americans. Integrated life course research design for research on cognitive aging: age coverage by study membership. Note. This extensive longitudinal life course design integrates four U.S. population-based cohort studies: national longitudinal study of adolescent to adult health (add health) (waves I–V, 1994–2018), midlife in the US study (MIDUS) (Waves II-III, 2004–2017), health and retirement study (HRS) (1996–2018), and the Americans’ changing lives study (ACL) (waves I–V, 1986–2011) (see details of study samples in Supplemental Tables S2, and S4).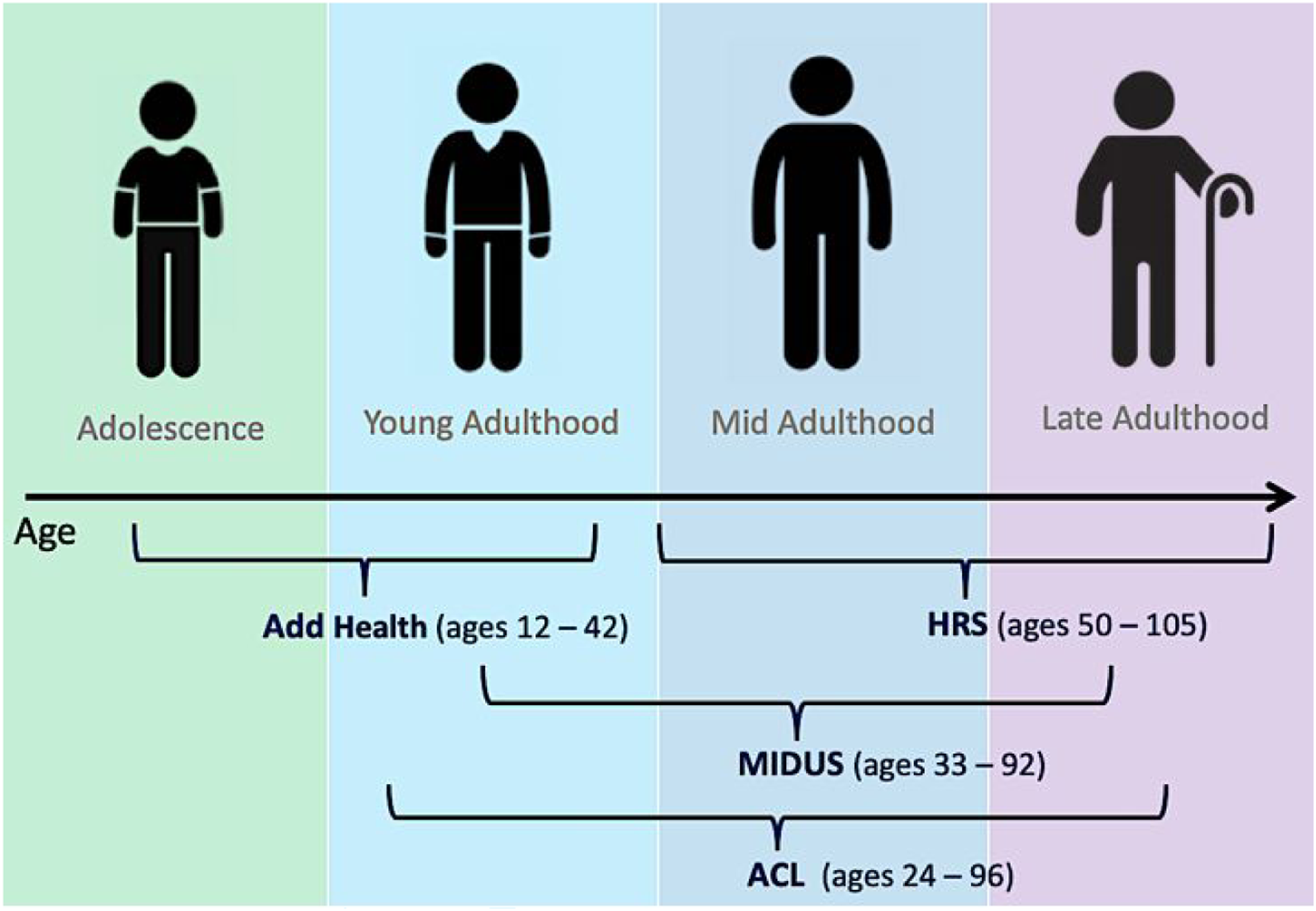
Data and Methods
Data
Data came from four U.S. population-based longitudinal cohort studies that provide prospective cognitive measures and collectively span the adult life course (see Figure 1): The National Longitudinal Study of Adolescent to Adult Health (Add Health) is a nationally representative sample of 20,745 adolescents aged 12–19 in 1994–95 who have been followed into mid-adulthood over five study waves (Harris et al., 2021). Various cognitive measures were collected at Wave I, Wave III in 2001–2002 when respondents were aged 18–26, Wave IV in 2008 at ages 24–32, and Wave V in 2016–2018 at ages 33–42 (Harris & Udry, 2022). National Survey of Midlife Development in the United States (MIDUS) is a national longitudinal study of 7108 English-speaking, non-institutionalized adults aged 25 and older residing in the contiguous U.S. who were first interviewed in 1995–96 (Wave I) and followed in 2004–2006 (Wave II) and in 2013–2017 (Wave III) (Ryff & Lachman, 2017, 2019). Cognitive measures were collected in Waves II and III when respondents were aged 33–92 years. Health and Retirement Study (HRS) is the largest ongoing nationally representative longitudinal survey of U.S. adults aged 50 years and older, conducted bi-annually beginning in 1992 with a sample of 37,000 adults (Sonnega et al., 2014). We used data from the longitudinal RAND files with cognitive measures harmonized across waves from 1996 to 2018 when respondents were age 50–109 years. Americans’ Changing Lives study (ACL) is a nationally representative longitudinal survey that interviewed an initial sample of 3617 individuals aged 24 and older in 1986 (Wave I) and followed them in four waves until 2011 (Wave V) (House, 2018). We used cognitive data available for all waves from participants aged 24–96 years.
Measures
Outcome Variables: Cognitive Function
A variety of tests for cognitive performance were administered in each study to measure both global and domain-specific functions encompassing multiple cognitive domains, including memory, executive function or attention, and orientation. Additional details for all cognitive measures by dataset are found in Supplemental Table S1. We created commensurate variables that are harmonized across studies using previously developed standardization methods for IDA based on the same samples (Yang et al., 2016, 2020). Episodic memory (memory thereafter) was assessed by combining scores from immediate and delayed recall of a word list that were measured in three datasets (Add Health, MIDUS, and HRS). To harmonize these scores using the same scales, we followed previous procedures (Hofer & Piccinin, 2009; Stebbins et al., 2022) and standardized the scores for immediate recall and delayed recall to a mean of zero and standard deviation of one, summed them together, and re-standardized them to derive the final episodic memory score. Overall cognitive function was assessed by the total score of all cognitive tests that include different tests and cognitive domains across studies. In addition to the total score assessing memory available only for adults in Add Health Waves IV and V, we included the Picture Vocabulary Test (PVT) in Add Health Waves I and III in separate analyses to assess cognitive ability (general intelligence and vocabulary) in adolescence and young adulthood. Apart from the Brief Test of Adult Cognition by Telephone (BTACT) in MIDUS which is already standardized (Lachman et al., 2014), we standardized the total cognitive score in each of the other studies (Add Health, HRS, and ACL) to make scores comparable in scale.
Covariates
All measures of covariates are shared across studies and recoded uniformly to be included as controls of baseline characteristics in all analyses. Age was recorded at each wave in single years. We centered age around cohort median and divided it by 10 (see Analytic Methods below and notes of all tables). Birth cohort indicates years of birth that range from 1890–1983. We recoded 10-year cohorts (before 1915 to 1975–1983) into a continuous variable (1–8) and centered it around the 1925–34 cohort. Gender (men or women) and Race/ethnicity (non-Hispanic (NH) White, non-Hispanic (NH) Black, and Hispanic) are based on respondent self-reports. Educational Attainment was measured as the total number of years of schooling completed and categorized into three levels: less than 12 years of school (less than a high school degree), 12–15 years (high school degree or some college) (reference group), and 16 years of school or more (college degree or higher). Immigrant status is a binary indicator of immigrant status was defined based on self-reported nativity: U.S.-born (reference group) and foreign-born. Attrition Status is an indicator of status of lost to follow-up: death or non-response versus remained in the sample (reference group) during the study period. Study membership variable was created to indicate respondent membership in a specific study: Add Health (reference group), MIDUS, and HRS, and to adjust for potential study effects in the pooled analysis (see Analytic Methods).
Study Samples
For each dataset, we included participants with at least one wave of valid measures of cognitive function and complete data on covariates at baseline. We excluded 2616 respondents in Add Health who were older than their grade levels and who dropped out of school due to potential cognitive impairment. We also excluded NH other race participants (ranging 2%–8% across datasets) due to their small sample sizes as well as their large racial/ethnic heterogeneity, which limit statistical power and meaningful results. Less than 1% of respondents with missing data on any covariate were excluded. The sample for the analysis of episodic memory was pooled from three studies and consists of 51,280 participants with 217,139 person-years of observations over a total of 22 years of follow-up across 15 study waves (Table S2). The pooled sample at baseline (Table S3) has a mean age of 53.1 years (standard deviation (SD) = 16.5; range = 24–109) and birth year ranging between 1890–1983 is 56.1% female, 68.2% NH White, 18.9% NH Black, and 12.9% Hispanic. Most respondents reported having the equivalent to a high school degree (55.3%) or higher (25.3%), while 20% did not graduate high school. A small proportion of the sample (9.8%) was foreign-born. The average immediate and delayed recall scores are 6.0 (SD = 1.9) and 4.7 (SD = 2.2), respectively. Among the baseline pooled sample, 41% were lost to follow-up due to non-response (15%) or death (26%), the majority of which were from the HRS. Compared to those who remained in the sample, the lost to follow-up were on average 20 years older, more likely to be NH White and less educated (p <.001).
The samples for the analysis of overall cognitive function include those from all four individual studies. The first three studies are similar in sample size and length of follow-up to those included in the pooled analysis of memory. The ACL further contributed over 3500 participants followed over 25 years across five waves (Table S4). The four study samples (see baseline characteristics in Table S5) include birth cohorts that vary widely in age ranges spanning the entire life course from adolescence to old age (12–109). Each sample is majority NH White, with Add Health being more racially/ethnically diverse than the others and MIDUS being the most homogeneous (88.4% NH White). Add Health and MIDUS also contain the greatest shares of respondents who have attained a high school degree or college education. In contrast, about a quarter of the ACL and HRS samples had less than a high school education. Across study samples with attrition from baselines, the lost to follow-up were more likely to have less education than those who remained in the samples (p <.001).
Analytic Methods
Longitudinal Integrative Data Analysis (IDA)
We used an extensive life-course research design (Figure 1) that integrates four longitudinal samples with an augmented age range from adolescence to old age and also substantial age overlaps between 20 and 30 years across birth cohorts. We modeled age trajectories of cognitive change over the life course with a novel application of existing methods of Integrative Data Analysis (IDA). IDA is a set of statistical tools for analyzing two or more independent samples (Curran & Hussong, 2009; Hussong et al., 2013). In the context of longitudinal analysis, the pooled IDA is based on a combined dataset from separate samples connected by overlapping ages and measures. It augments the accelerated longitudinal cohort design of an individual multi-cohort study using a synthesized cohort sequential design which can allow for inferences across the entire age range of 24 and above from an increased number of cohorts, even though each individual dataset followed participants for a fraction of this time (Bainter & Curran, 2015; Curran & Hussong, 2009) (the term cohort refers to birth cohort here rather than study cohort which refers to membership in a longitudinal study; see more on study membership in Methods). Additionally, a coordinated IDA can be used in which parallel models are fit to each contributing sample separately and results are compared within and across studies in a coordinated fashion (Hofer & Piccinin, 2009; Yang et al., 2016, 2020). Longitudinal IDA offers substantial advantages over conventional studies using single datasets and is well-suited for our investigations (Bainter & Curran, 2015; Curran & Hussong, 2009). First, it enables the test of novel life course hypotheses and can address questions about age dynamics and birth cohort variation over far more extended developmental and historical periods than any single-sample design can allow. Second, it enhances power for statistical analyses due to not only larger combined sample sizes but also greater representation from across-sample heterogeneity. Third, the use of IDA enhances study comparability by directly modeling heterogeneity across studies to strengthen both internal and external validity about resulting population inferences. This creates the opportunity to support data sharing and build a cumulative and replicable science of population health.
Building from earlier work on IDA in life course research (Graham et al., 2017; Yang et al., 2016, 2020, 2021), the current analysis conducted the first longitudinal IDA to examine life course trajectories of cognitive function in the general U.S. adult population. Specifically, we carried out two sets of IDA: (1) pooled IDA of episodic memory from young to late adulthood using the pooled sample from three studies (Add Health, MIDUS, and HRS) that share the same word recall measures; and (2) coordinated IDA of overall cognitive ability from adolescence to old age using varying cognitive measures in all four studies. In combining and comparing independent studies, we drew on the principled and established methodological guidelines for synthesizing large longitudinal data sets (Bainter & Curran, 2015) and adjust for heterogeneity across studies. First, we harmonized variables across studies as described before. Second, the pooled IDA explicitly controls for any between-sample variability by directly modeling study membership as a fixed effect combined with tests of study-by-covariate interactions (Curran & Hussong, 2009; Hussong et al., 2013). Given the range of potential sources of sample heterogeneity (e.g., measurement, sampling mechanisms, and historical period), this option presents the most efficient and effective way to control for any unmeasured sources of study difference. Third, the IDA allows for broader age, period and cohort assessments given the combination of birth cohorts from multiple studies across survey years with increased overlapping of age ranges. We leveraged its strength for age effect inferences by accounting for potential confounding of cohort effects or period effects using established methods for longitudinal cohort analysis (Miyazaki & Raudenbush, 2000; Yang et al., 2021; Yang & Land, 2013).
Linear Mixed-Effect Growth Curve Models
We employed linear mixed-effect growth curve models to conduct longitudinal IDA. These models are well-suited for the present study as they can accommodate time-unbalanced data (i.e., varying number of repeated observations within individual) and permit inclusion of all individuals regardless of the number of waves they contribute, reducing sample selection due to loss to follow-up (Raudenbush & Bryk, 2002). We further followed the parsimonious and effective method to control for attrition type directly in the model to account for potential selection bias due to the possibility that respondents lost had poorer cognitive outcomes than those with complete data (Chen et al., 2010; Yang & Land, 2013). The general model specification is the following:
The models were fit to memory scores for the pooled IDA and separately to overall cognitive scores in each of the four datasets for the coordinated IDA (and separately to PVT data and total score data in Add Health). Given statistically significant gender differences in the majority of analyses, we estimated all models separately for men and women that included key independent variables in a stepwise fashion. We used Bayes Information Criteria (BIC) as the goodness-of-fit statistic to select the best fitting model for each analysis. Our final analyses retained variables with statistically significant coefficients and when their inclusions improved the model fit. Statistical analyses were conducted in Stata 17.0 and SAS 9.4.
Statistical inference of age effects in a single longitudinal study is complicated by two issues which were discussed in previous methodological research (Miyazaki & Raudenbush, 2000; Yang & Land, 2013) and we addressed more effectively in this study using the IDA approach. First, there is potential confounding of age and cohort effects because age and birth cohort variables are highly correlated for non-overlapping segments of the life course. To address this, we centered age around cohort group median to minimize bias arising from systematic variation in age across birth cohorts (Raudenbush & Bryk, 2002; Yang & Land, 2013). Furthermore, the models yield tests of significance of overlapping ages of adjacent cohorts. As waves of data accumulate, the number of overlapping ages across cohort increases, age and cohort become less confounded, and the statistical power increases for the test of cohort differences in age trajectories. Therefore, the IDA further strengthens the reliability and stability of model estimation regarding age and cohort effects due to increased power from the combination of multiple birth cohorts with increased overlapping ages across studies (Bainter & Curran, 2015; Yang et al., 2021). Second, period effects are difficult to estimate separately from age effects in growth curve models since the time metric is set as either age or wave (period), but not both—age and period covary within individuals and cannot be distinguished. To account for this, we followed the methodological guideline to model instead the age-by-cohort interaction effects (β11, β21). Because cohorts vary in age at any historical moment, effects associated with historical time, if any, tend to produce cohort differences in the age-outcome relationship (Chen et al., 2010; Miyazaki & Raudenbush, 2000; Yang, 2007; Yang et al., 2021; Yang & Land, 2013; Yang & Lee, 2009, 2010). Furthermore, the pooled IDA of multiple datasets from different periods allows for the estimation of additional fixed effects of study membership and cohort-by-study interaction to explicitly adjust for historical time (Curran & Hussong, 2009; Yang et al., 2021). Analyses including the study-by-cohort interaction and other study-by-covariate interactions showed no statistical significance for the interaction terms or improved model fit. Thus, we estimated the models without these interaction terms in the final analyses.
Results and Findings
The results showed statistically significant age-related changes in memory and overall cognitive function across the adult life course and within each life stage. We estimated models separately for women and men to investigate social patterning of cognitive trajectories in gender-specific contexts. We found that, for both men and women, the age trajectories of cognitive function varied between groups of individuals defined by year of birth (10-year cohorts), racial/ethnic self-identification, and educational attainment. Below we outline the specific results from the two sets of analyses.
Age Trajectories of Episodic Memory from Young to Late Adulthood
Among participants in the pooled sample, memory performance declined with advancing age, independent of birth cohort effects. The estimated linear rate of decline in standardized memory score was about one SD per decade of life (for women, .95 SD per decade, p < .001; for men, .82 SD per decade, p < .001; Table S6, Model 1). The predicted age trajectories showed a curvilinear life-course pattern of memory performance: peaks between ages 25–35, modest declines beginning between ages 35–40, and accelerated declines after ages 60–65 (Figure 2a). Women performed better on memory tests on average across all ages than men, but experienced faster rates of decline with age (p < .001), leading to a convergence in gender gap after age 75. Predicted age trajectories of episodic memory in adulthood: gender and cohort differences. Note. Predicted age trajectories from linear mixed-effects growth models of standardized memory score in the pooled sample from add health, MIDUS, and HRS (N = 51,280 individuals and 217,139 person-years; Tables S2–S4) adjusting for age, age2, gender, birth cohort, immigration status, attrition status, study membership, and significant interactions (Table S6). (a) By gender. Women displayed a higher average memory test score but steeper slope of decline than men (p < .001, two-tailed test). (b) By birth cohort for women and men (10-year cohorts born before 1905–1914, 1915–1924, 1925–1934, 1935–1944, 1945–1954, 1955–1964, 1965–1974, and 1975–1984). More recent birth cohorts displayed higher average memory scores and slower rates of decline than their predecessors (p < .001, two-tailed test).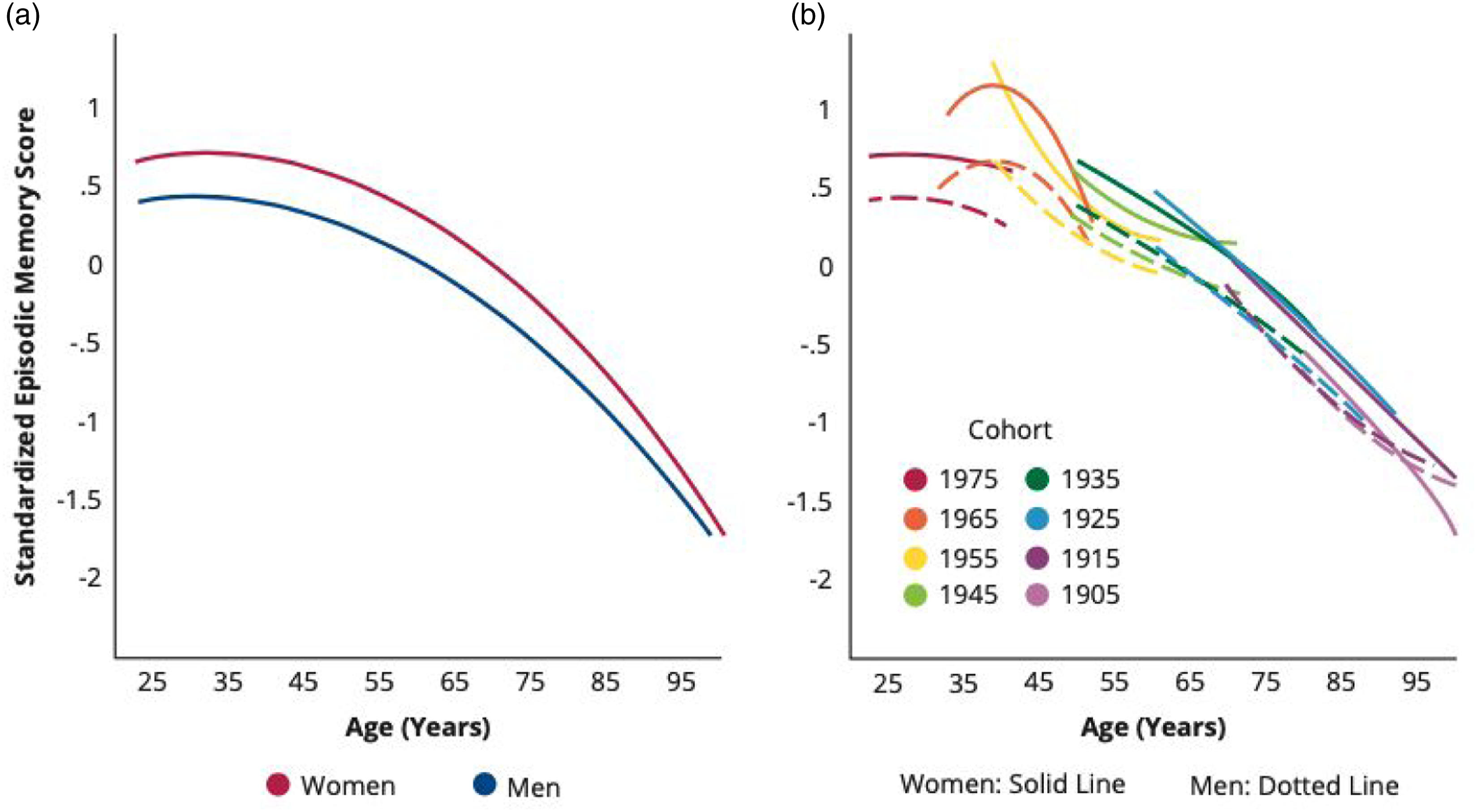
We found significant birth cohort variations in the age trajectories of memory for both women and men. More recently born cohorts tended to perform better on memory tests on average and to experience slower rates of decline than earlier born cohorts. These cohort effects were small in size but statistically significant (p < .001; Table S6, Model 1). The pattern of improving trajectories of memory performance across birth cohorts was largely continuous (Figure 2b), with a slight drop for the most recent cohort (1975–1984) from previous cohorts born in the 1950s and 1960s. The later born cohorts also showed larger gender gaps (increased advantages for women than men) as compared to earlier cohorts (p < .001).
There were pronounced racial/ethnic disparities in trajectories of memory performance for both genders, adjusting for other covariates. NH Black and Hispanic Americans exhibited substantially lower average memory scores across all ages as compared with NH White Americans (for NH Black women, .57 SD lower, p < .001; for Hispanic women, .52 SD lower, p < .001; Table S6, Model 1). The rates of decline in memory trajectories were somewhat slower for these groups compared with NH Whites (p < .05), which diminished but did not eliminate racial/ethnic gaps with age (Figure 3a). NH Black men displayed the lowest memory trajectory of all groups. There were also cohort variations in racial/ethnic disparities in memory trajectories (p < .05). The slower rates of decline for NH Blacks and Hispanics as compared with NH Whites were attenuated in more recent birth cohorts, leading to similar rates of decline across groups and increased disparities in more recent cohorts (p < .001; Figure 3b). Predicted age trajectories of episodic memory in adulthood: racial/ethnic differences. Note. Predicted age trajectories of standardized memory score in the pooled sample from add health, MIDUS, and HRS (Table S6, model 1). (a) By race and ethnicity. Non-hispanic (NH) blacks and hispanics displayed lower memory trajectories than NH white counterparts over the life course for both genders (p < .001, two-tailed test). NH black men had the lowest memory trajectory of all groups. Racial/ethnic gaps decreased but did not diminish in old age. (b) By race/ethnicity and birth cohort. NH black and hispanic deficits in memory trajectories relative to NH whites were more pronounced for more recent cohorts than earlier born cohorts (p < .05, two-tailed test). Only every other 10-year birth cohort is shown for purpose of illustration.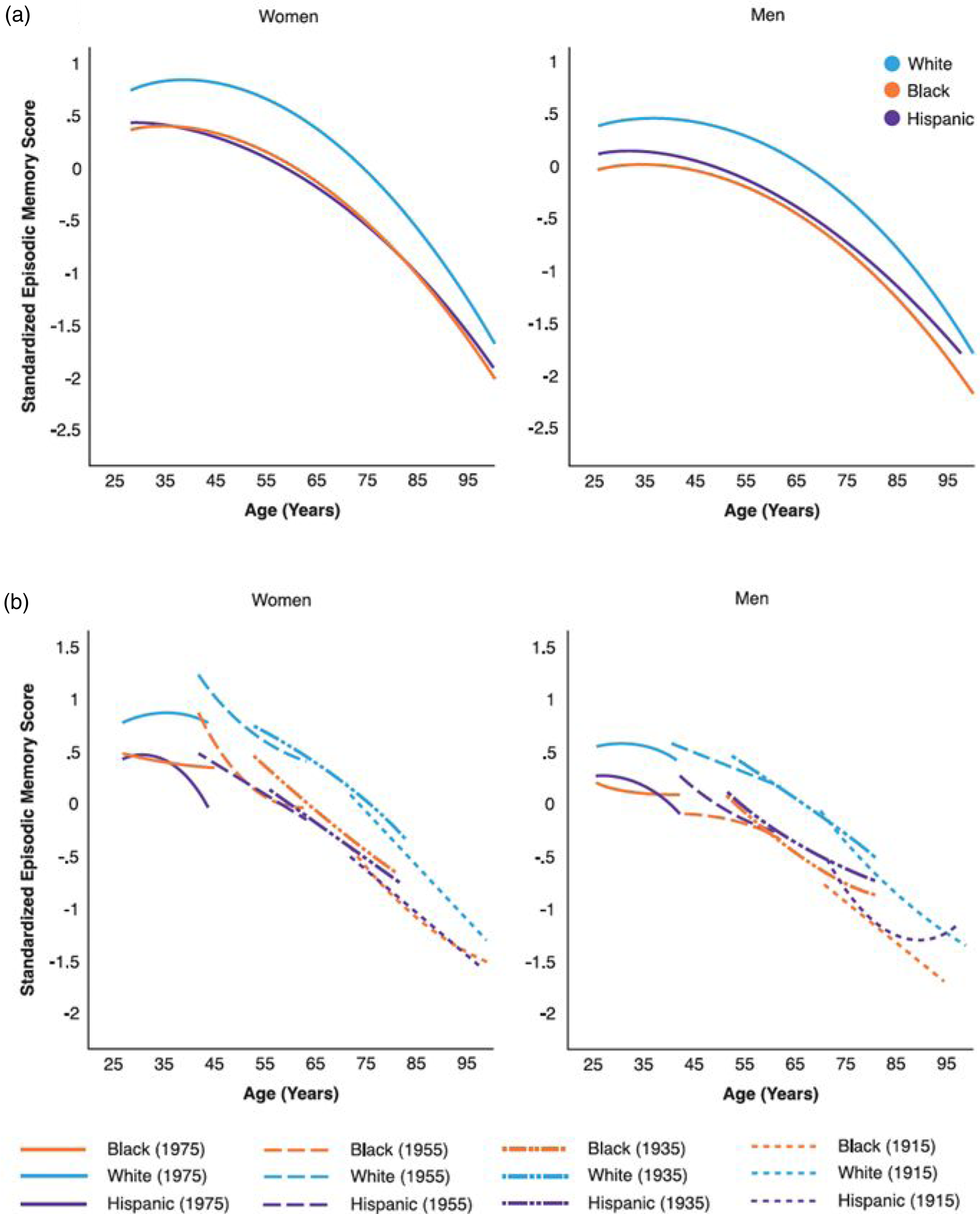
The educational differentials in the memory trajectories were substantial for both genders, controlling for covariates. Compared to those with a high school degree, the mean memory score was substantially lower for individuals with less than 12 years of schooling (for women, .68 SD lower, p < .001; Table S6, Model 2 and higher for those with a college degree or more education (for women, .3 SD higher, p < .001). Among women, those with a college degree or more education showed a slower rate of decline than their less educated peers (p = .026). In men, however, those with no high school degree showed a slower rate of decline than the more educated (p = .008). These differences were small in comparison to educational disparities in average memory performance. As a result, the educational gradient in memory trajectories remained largely constant across life with no apparent convergence: the college educated displayed the highest trajectory, followed by those with a high school degree, and then those without a high school education (Figure 4a). The educational differentials in memory trajectories further varied by birth cohort, with the gaps in increasing in more recent cohorts (p < .001; Figure 4b). Predicted age trajectories of episodic memory in adulthood: educational differences. Note. Predicted age trajectories of standardized memory score in the pooled sample from add health, MIDUS, and HRS (Table S6, model 2). (a) By level of educational attainment. The educational gradient in memory trajectories (p < .001, two-tailed test) remained largely constant across life. (b) Comparison of the highest and lowest education levels by birth cohort. More recent cohorts show larger gaps between education groups than earlier cohorts (p < .001, two-tailed test). Only every other 10-year birth cohort is shown for purpose of illustration.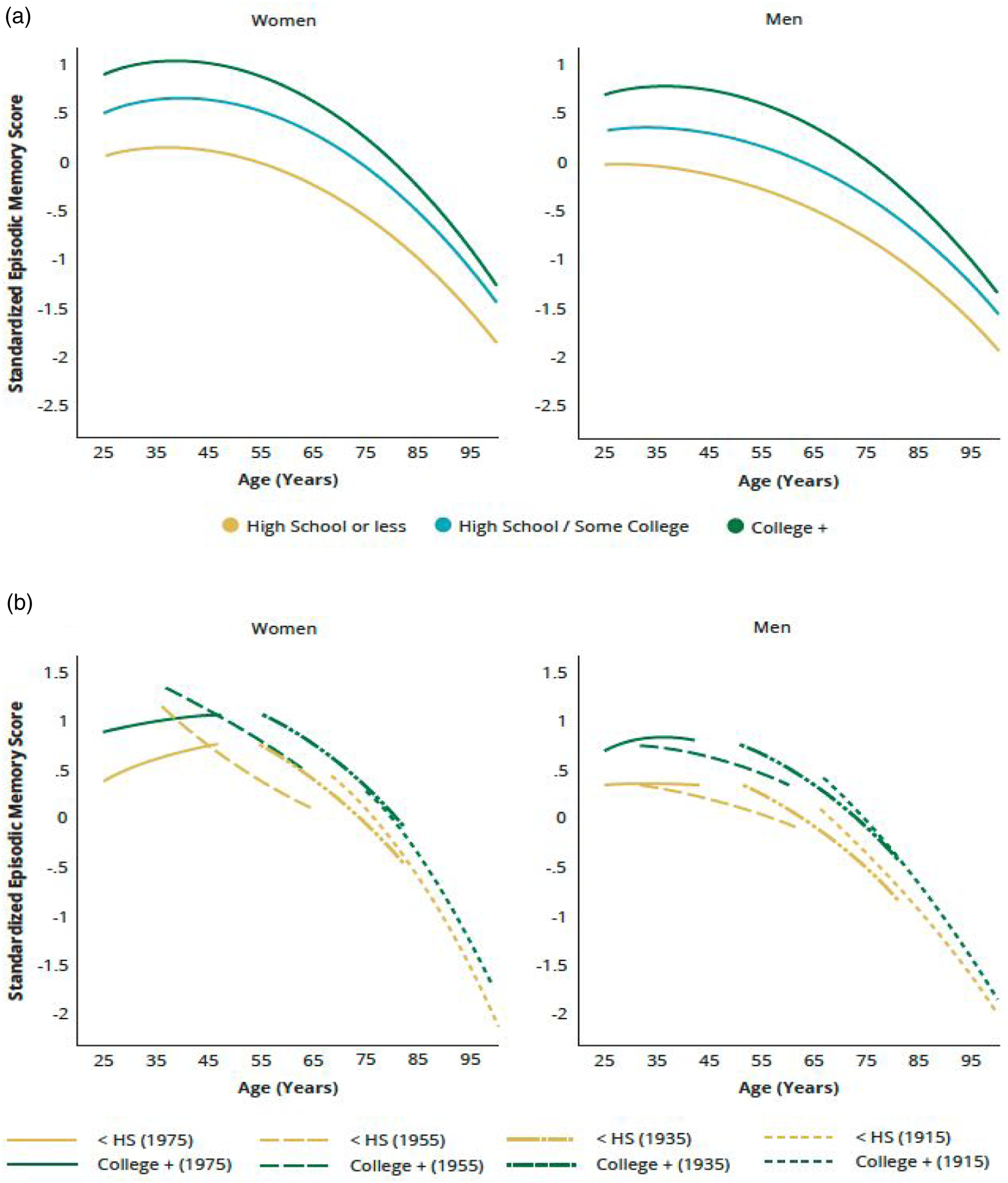
Racial/ethnic differences in average memory score were reduced when adjusting for educational attainment, while both racial/ethnic and educational differentials in cognitive trajectories remained statistically significant in the fully adjusted analysis (p < .05; Table S6, Model 3). The memory trajectories were stratified by both racial/ethnic and education groups (p < .001; Figure 5). In women, NH Whites had the highest memory trajectories regardless of education levels, while NH Blacks with less than a high school education had the lowest memory trajectory. Men showed similar patterns with the exception that the trajectory of the lowest educated NH Whites overlapped with that of the highest educated NH Blacks. For both genders the gap between the lowest educated NH Blacks and the highest educated NH Whites was widest among all subgroups and persisted throughout life with some reduction in old age. These results suggest that educational attainment does not provide an equal benefit for memory function or memory decline across all racial/ethnic groups. Predicted age trajectories of episodic memory in adulthood: racial/ethnic and educational differences. Note. Predicted age trajectories of standardized memory score by race/ethnicity and education levels in the pooled sample from add health, MIDUS, and HRS (Table S6, model 3). For both (a) women and (b) men, the gap between the lowest educated NH blacks and the highest educated NH whites was the widest among all subgroups and persisted throughout life with some reduction in old age. The interaction effects of race/ethnicity and education were significant in both the intercept and linear rate of decline for women and in linear rate of decline for men (p < .05, two-tailed test).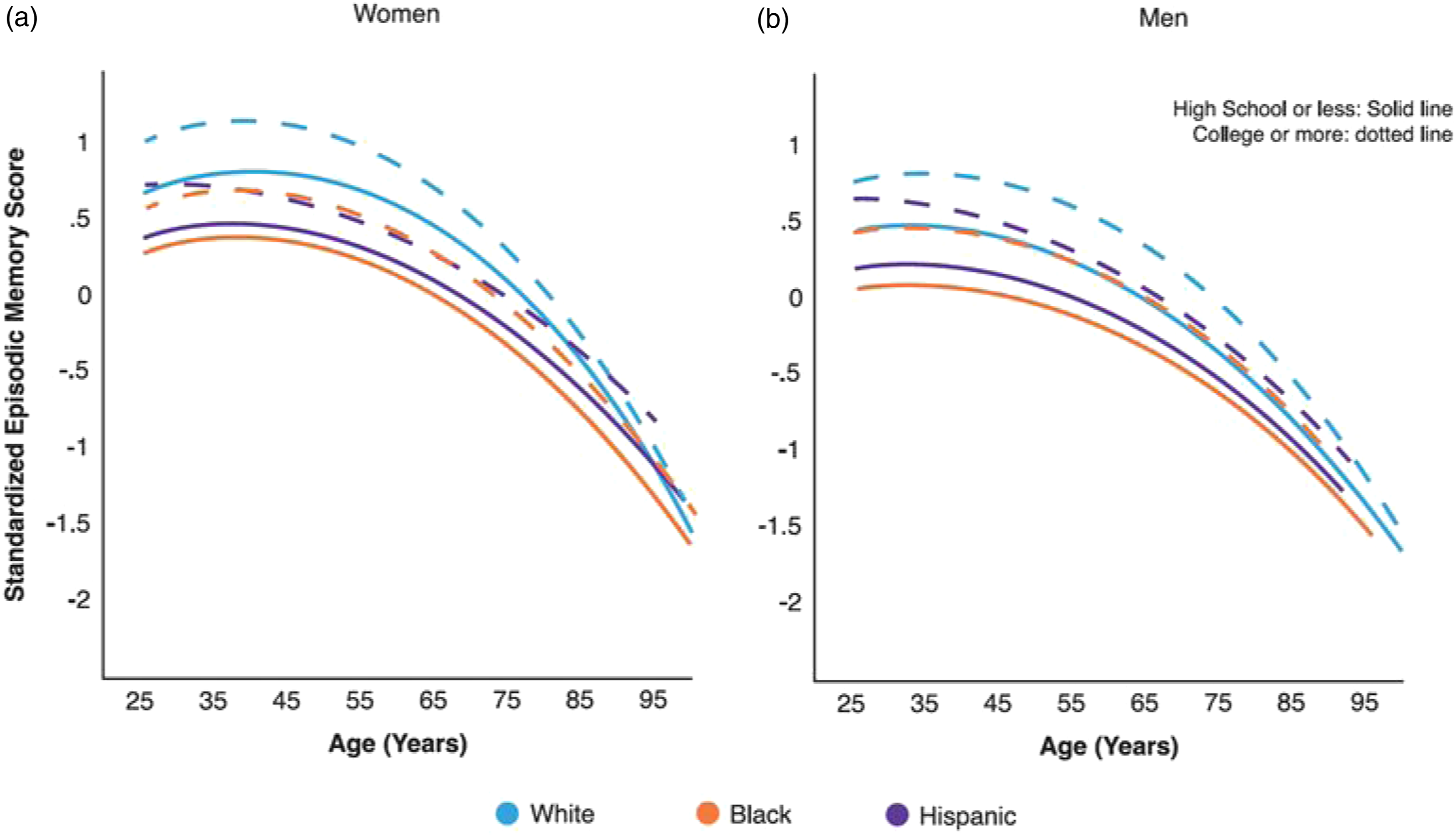
The results for other covariates showed expectedly worse memory trajectories for those lost to follow-up (due to non-response or death) than those with completed follow-up data. Attrition of these participants produced conservative estimates (or underestimates) of cognitive decline. In addition, individuals born outside the U.S. showed poorer average memory performance than U.S. natives. We adjusted for between-sample variability by including study membership combined with tests of study-by-covariate interactions to control for potential sources of study heterogeneity (see Analytic Methods). We found significant differences in only the mean memory scores by study membership and no interactions of study with other variables that were significant or improved model fit, indicating no impact of study-related factors on the results.
Age Trajectories of Overall Cognitive Function from Adolescence to Late Adulthood
The coordinated IDA of overall cognitive ability found consistent results during overlapping segments of the life course across the four studies, which are also highly consistent with findings of episodic memory trajectories. Results that reached statistical significance (p < .05) are summarized below.
Cognitive performance (as measured by standardized PVT in Add Health) increased from adolescence to early young adulthood (ages 12–25) (Add Health Waves I, III; Table S7). Peak performance was then followed by gradual decreases in standardized total cognitive scores from young to late adulthood (ages 25–105), independent of other factors (Add Health Waves IV-V, Table S7; ACL, Table S9; HRS, Table S10). The corresponding predicted age trajectories from these studies collectively depicted a curvilinear pattern of cognitive decline throughout life (Figure 6). Age-related changes were not significant across the two waves of the MIDUS sample (Table S8). Predicted age trajectories of overall cognitive function from adolescence to late life: by study and gender. Note. Predicted age trajectories from linear mixed-effects growth curve models of standardized total cognitive scores in each study sample (Table S7, add health waves I, III, IV-V N = 12,039 individuals and 34,336 person-years; Table S9, ACL waves I–V N = 3532 individuals and 11,623 person-years; Table S10, HRS 1996–2018 N = 33,240 and 131,817 person-years). Results show increases in the picture vocabulary test (PVT) score from adolescence to early young adulthood, followed by gradual quadratic declines in total cognitive scores from young to mid and late adulthood. Gender differences across studies show higher PVT performance for men than women early in life and elevated cognitive trajectories for women relative to men until old age when the gap closed (p < .05, two-tailed test). Results from the national survey of midlife development in the United States (MIDUS) were omitted because of only two waves of data and no significant age change in this study (Table S8, MIDUS waves II-III N = 3820 individuals and 6185 person-years). Abbreviations: add health = the national longitudinal study of adolescent to adult health; ACL = Americans’ changing lives; HRS = health and retirement study.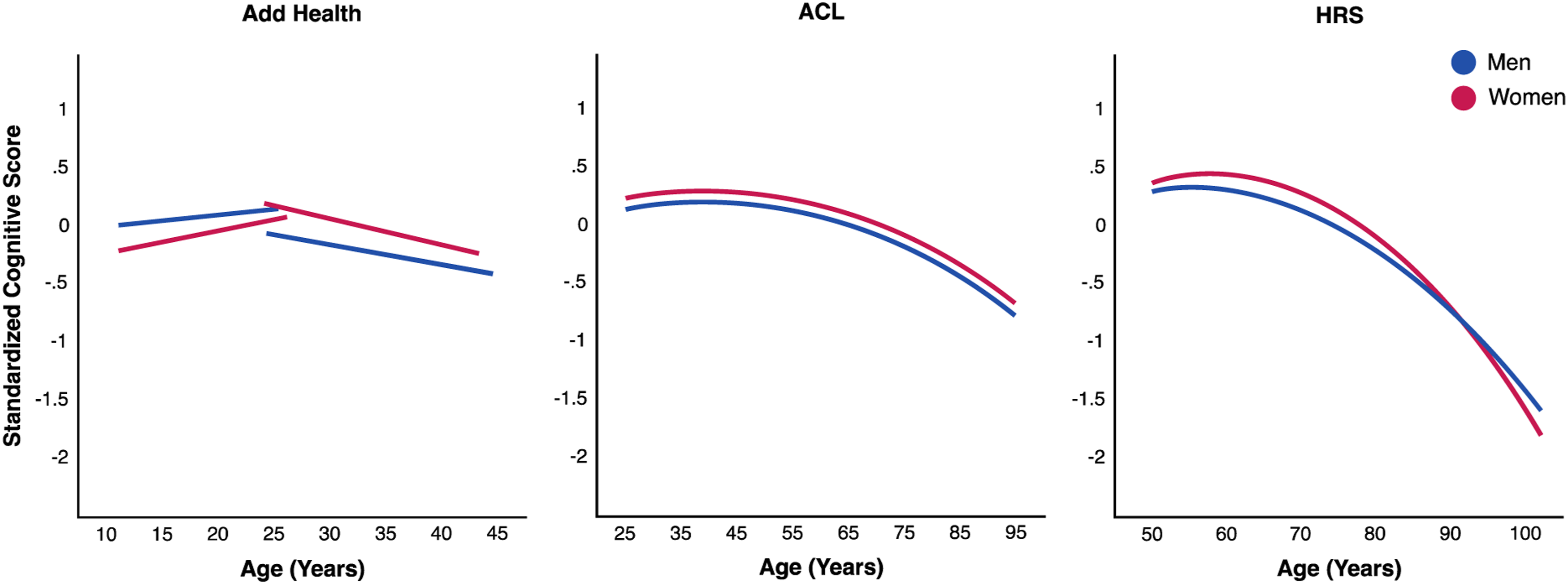
The social patterning of overall cognitive trajectory is similar to that of memory trajectory. There were cohort improvements from mid to late adulthood (Tables S9, and S10). Early in life, men performed better in the PVT than women on average with less increase with age (Table S7). This difference reversed in young and middle adulthood when women displayed better overall cognitive function on average than men (Tables S7, S9, and S10). The steeper cognitive declines for women than men led to a narrowing gender gap and cross-over in old age (Figure 6). The racial/ethnic gaps in the mean cognitive scores were wide (Figure 7), starting early in life (Table S9) and continuing throughout later life (Tables S9, and S10). Racial/ethnic differences in the rates of change with age showed worse profiles for NH Blacks (less increase in PVT, Table S9; faster declines in total score, Tables S9, and S10) and Hispanic women than their NH White counterparts (faster declines in total score, Table S10), but a greater increase in PVT for Hispanic men (Table S7). We further found large educational gradients in the mean cognitive scores but not rates of decline with age (Tables S7–S10), and corresponding persistent educational disparities throughout life (Figure 8). Birth cohort variations in racial/ethnic and educational differentials indicated larger disparities in the mean cognitive scores in more recent cohorts (Tables S9, and S10). Across all studies, race/ethnicity and education were associated with trajectories of overall cognitive function independently and jointly in similar ways as they were with memory trajectories. Taken together, social disparities of cognitive abilities in early life set individuals on cognitive trajectories into adulthood that continued to be highly stratified by social status with no or modest reductions in late life. Predicted age trajectories of overall cognitive function from adolescence to late life: by study and race/ethnicity. Note. Predicted age trajectories of standardized total cognitive scores in each study sample for both women and men (Tables S7, S9, and S10). Results show highly stratified cognitive trajectories by race/ethnicity. The notable gaps between NH blacks and hispanics and NH whites in cognitive abilities (PVT performance) started in adolescence and continued throughout later life. Results from the national survey of midlife development in the United States (MIDUS) were omitted due to a lack of racial/ethnic diversity in this study. Predicted age trajectories of overall cognitive function from adolescence to late life: by study and education (women). Note. Predicted age trajectories of standardized total cognitive scores in each study sample by levels of education for women (results similar for men) (Tables S7–S10). Results show strong educational gradients in cognitive trajectories, with the college educated displaying the highest and those with no high school degree displaying the lowest trajectories. Such differences persisted throughout life with little convergence with age. Abbreviations: add health = the national longitudinal study of adolescent to adult health; MIDUS = national survey of midlife development in the United States; ACL = Americans’ changing lives; HRS = health and retirement study.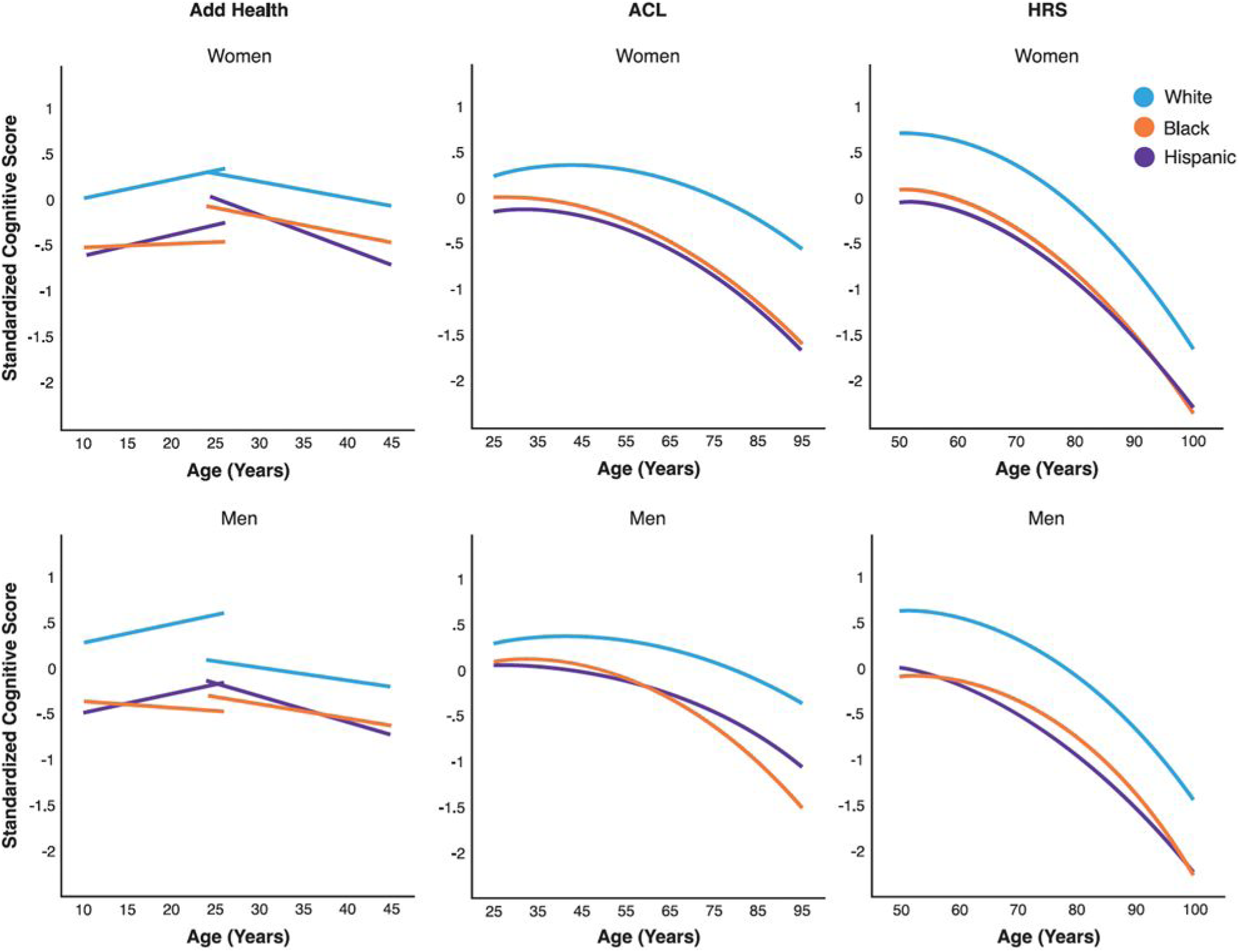
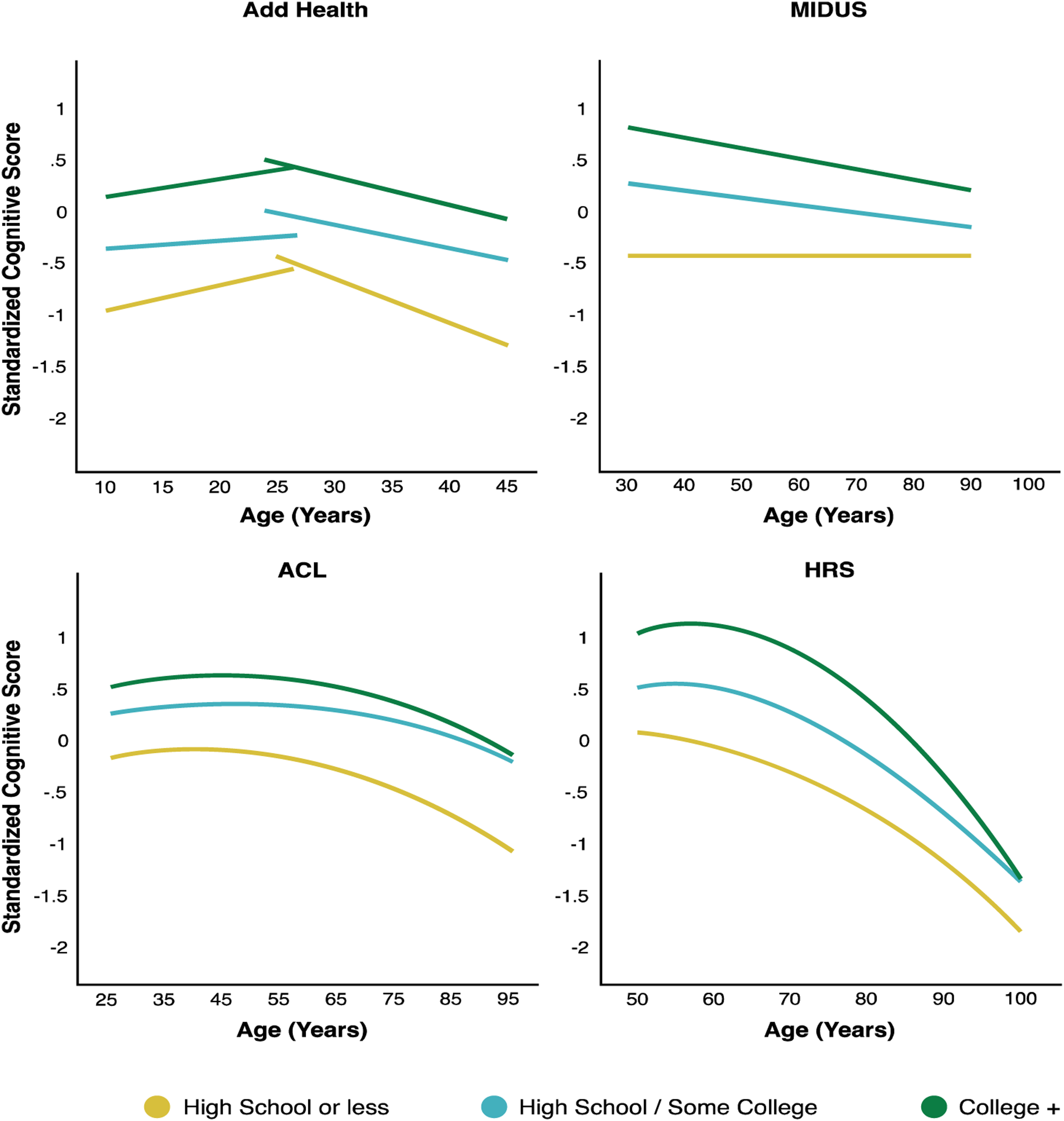
Finally, overall cognitive function varied by attrition status and nativity with some differences across studies. While those born outside the U.S. had lower mean cognitive scores than their native counterparts in most studies, they showed a higher mean score for women in the ACL (Table S9). As the cognitive measures in the ACL do not include tests of memory, this result suggested an advantage in non-memory cognitive function for foreign-born women.
Discussion
Our rapidly aging population faces a world of growing informational complexity where the prospect of healthy life expectancy depends heavily on cognitive functioning. Age is the primary risk factor for cognitive impairment and ADRD (Plassman et al., 2010). Understanding the life course dynamics of aging-related changes in cognitive function is profoundly important to guiding research on the etiology, prevention, and control of cognitive impairment and dementia. The present study contributes to this end by conducting the first and most extensive longitudinal assessment of lifelong changes in cognitive abilities across multiple cognitive domains in the largest and most diverse samples of the U.S. population to date. Through a novel application of the IDA methodology to longitudinal data from four population-based studies, our study yields new findings that did not exist before in studies of single datasets. It also sheds new light on malleability and plasticity of cognitive aging by revealing individual differences by sociodemographic status. There are three primary findings. First, aging-related declines in memory and general cognitive abilities onset in the 4th decade of life and accelerate in the 7th decade. Second, there are marked gender, racial/ethnic, and educational disparities in cognitive aging that originate in early life and vary over the life course. Third, the secular trends show overall improvements across the 20th century birth cohorts on one hand and widening inequalities in more recent cohorts born after the 1950s on the other. We discuss each finding below.
Aging-Related Cognitive Decline
Prior research suggests the potential early onset of aging-related cognitive decline indicated by a slowing in memory and executive function in the 30s (Albert et al., 2007; Salthouse, 2009; Wilson et al., 2002) and mild declines in cognitive performance over time in older adults (Salthouse, 2019; Schaie, 2013; Walsh et al., 2022). The age patterns of performance of cognitive tasks following a “peak-plateau-decline” trajectory are largely implicitly understood (Rosano, 2020) rather than explicitly tested. Our study overcomes previous studies’ limitations to provide direct evidence for the timing and course of aging-related cognitive change in the general population. Our analyses of cognitive change begin as early as possible over life periods as long as possible. The data here initiate in adolescence and young adulthood when cognitive ability is still developing and continue for the remainder of adulthood after cognitive ability matures (Gow et al., 2011). To the extent that early-life influences and environmental exposures affect adulthood cognitive function, and in turn late-life cognitive impairment and ADRD, our identification of the earliest ages of cognitive decline is important in establishing the optimal time window for interventions to minimize or reduce subsequent disease risk.
Furthermore, our precise specification of age-related cognitive change from early to late life provides an essential benchmark to define normal cognitive aging (Salthouse, 2019). Our findings of accelerated cognitive decline with age in presumably healthy community-dwelling adults are of relevance to further research, particularly attempts to understand etiological and developmental processes related to dementia and to distinguish pathological from normal cognitive aging for improved dementia diagnosis.
Social Disparities in Cognitive Aging
Our study reproduced known features of gender, racial, and educational disparities in average cognitive function, and in some cases, rates of cognitive decline with age. We further investigated how these disparities unfold over the full life course and revealed interesting and distinct life course patterns of disparities by gender, race/ethnicity, and educational attainment.
Better memory and overall cognitive performance for women than men on average has been found previously (Levine et al., 2021; Walsh et al., 2022). The direction and size of gender difference, however, vary from early to late life. Consistent with the expectation of “differential preservation” of cognitive reserve (Salthouse, 2006), we found cognitive abilities are better preserved with aging in one gender over the other leading to differential rates of age-related decline by gender. For instance, men lost their initial cognitive advantage during the transition from adolescence to early adulthood, whereas the cognitive advantage for women diminished in late life due to faster declines relative to men. The steeper age-related declines throughout adulthood may explain the elevated dementia risk for older women than men (Alty et al., 2023; Plassman et al., 2010; Walsh et al., 2022). This finding suggests gender differences in sensitive periods of cognitive change and should inform gender-specific prevention and control strategies.
Both racial/ethnic and educational disparities in cognitive function were large and present from early in the life course. Racial/ethnic differences diminished, but did not disappear, with age. Educational differentials largely persisted into old age. These disparities were preserved throughout life because differences in the rates of cognitive declines, if any, were insufficient to offset the substantial disadvantages in average cognitive abilities among NH Black and Hispanic Americans relative to their NH White counterparts and among those with less education relative to the more educated. Previous studies found mixed results regarding racial and/or education differences in the rate of cognitive decline (Karlamangla et al., 2009; Weuve et al., 2018). In addition to limitations in model specifications (e.g., only estimated linear change or including a single attribute of race or education but not their intersection), prior studies focused largely on older samples in which slower cognitive declines among disadvantaged groups can potentially reflect selective survival of healthier individuals. Our study further tested group differences in cognitive trajectories with data from early life during which cognitive change is less likely to be influenced by differential attrition. Our finding lends stronger empirical support to the contention about the specific role of race/ethnicity and education in cognitive reserve: their protective effect against dementia is due to the effect on peak performance rather than on the rate of cognitive decline, resulting in the “preserved differentiation” in cognitive reserve by social status over the life course (Salthouse, 2006).
Our finding of the intersectional influence of race/ethnicity and education suggests fewer cognitive returns from higher education in racial/ethnic minorities. Throughout the life course, NH Whites with a high school degree or less had better or similar cognitive functioning as NH Blacks and Hispanics with a college degree. Our study calls attention to a particularly vulnerable segment of the U.S. population: NH Black men with less than a high school education who received the greatest barriers and fewest resources for improving healthy cognitive function among all subgroups. However, education accounted for some but not all of the racial/ethnic disparities in cognitive function, consistent with previous research showing a partial mediation of education on the total effect of race (Weuve et al., 2018). Together these findings may reflect the strong and persistent influences of a myriad of compounding social structural exposures and cultural bias that inequitably impact people of color in the U.S., including racism, discrimination, neighborhood disadvantage, perceived stress, quality and quantity of education, and access to social and health resources (Glymour & Manly, 2008; Letang et al., 2021). The persistent gaps across racial/ethnic and educational strata in lifetime cognitive trajectories suggest the importance of enhancing cognitive reserves early on for reducing cumulative disadvantages with age and social disparities in late-life cognitive health (Brown, 2010; Brown & Mutambudzi, 2022).
Cohort Improvement and Divergence
The secular trends in cognitive aging showed both signs of improvement and cause for concern. We found evidence for increasingly favorable aging-related cognitive change (higher average cognitive abilities and slower rates of decline) across successive 20th century birth cohorts. The lack of consideration of cohort variations in previous research may lead to biased estimates of age-related change (Dodge et al., 2014). Existing longitudinal studies of cohort effects concentrate on cohort differences in average levels within specific life periods such as in childhood and older adulthood, with varying findings on cohort differences in the rates of change across all ages (Dodge et al., 2014; Finkel et al., 2007; Gerstof et al., 2011; Schaie, 2013). We extended prior studies to include most 20th century cohorts with substantial overlapping age trajectories for more powerful tests of both age and cohort changes. We note that the memory trajectory for the latest cohort born in the 1970s to 80s was estimated to be lower than their predecessors (Figure 2b). This can result from a relatively smaller cohort sample with fewer age overlaps with previous cohorts and incomplete post-secondary education in early young adulthood. The finding needs to be corroborated in future investigations with greater power and a longer observation period beyond young adulthood.
The inter-cohort trend of improvement likely reflects sociocultural influences most notably due to advances in educational attainment during the post WWII period and associated socioeconomic protective factors (Schaie, 2013). The corresponding increased cognitive reserve and resilience to neurodegenerative disease risks bode well for healthy longevity for adults entering old age. Cohort improvement in cognitive function has tremendous societal and healthcare implications. More recent cohorts can be expected to perform at a higher cognitive level and engage in productive employment longer in life. A slower cognitive decline among these cohorts potentially means that they will experience later onset of cognitive impairment and dementia than their predecessors (Dodge et al., 2014). Such improvement will necessitate changes in work and health policies for accommodating the needs of a growing aging population that differs from those born later.
This positive overall trend is accompanied by increased intracohort inequalities across cohorts. That is, gender, racial/ethnic, and educational gaps in cognitive function widened in more recent cohorts. The increased advantages in memory performance in recent cohorts of women are likely attributable to increased access to higher education relative to their male counterparts (Bloomberg et al., 2021) and suggest the importance of a secular increase in attainment of college education for attenuating gender disparities in cognitive function. While increased investments in education for recent cohorts of Americans may enhance cognitive health and reduce the incidence of dementia in the future (Glymour & Manly, 2008), structural racism and associated experiences of discrimination and socioeconomic inequality can lead to persistent cognitive disadvantages and greater dementia risk as more recent cohorts enter old age. Reducing racial/ethnic and socioeconomic disparities may be most effective when focusing intervention efforts at younger ages in post-baby boomer cohorts.
Study Limitations
Despite the strengths of our study, our analyses are highly complex given the large size of all studies as well as study-to-study differences. Several limitations should be noted. First, it was suggested that the standardization method for data harmonization may lead to bias when the underlying assumption that the construct being measured is the same across measures and datasets is not met (Griffith et al., 2013). The individual study samples include cognitive measures that vary in domains and tests (e.g., Add Health PVT is distinct from the other cognitive tests and a proxy to intelligence; ACL does not include memory measures). Our pooled IDA is restricted to the memory measures common to three datasets (i.e., word recalls) and excludes cognitive measures that are not shared across all datasets. The standardization of memory scores of the same measures from the same study population in the U.S. may have largely reduced the potential bias introduced by study differences. Heterogeneity in other cognitive measures across studies, however, presents challenges for data harmonization using simple standardizations in a fully integrated study of all cognitive functions of interest. In this case, we conducted a coordinated IDA within each study and qualitatively compared results across studies. Future research is needed to collect further cognitive data and engage more sophisticated methods, such as latent variable models (e.g., confirmatory factor analysis and item response theory models), to harmonize diverse measures across studies (Griffith et al., 2013; Reason et al., 2020).
Second, our coordinated IDA of individual studies of overall cognitive function are limited in age and birth cohort representations and lengths of follow-up. For instance, only two waves of cognitive data were available for the Add Health and MIDUS analyses, which limited our ability to estimate nonlinear age change and cohort differences in age trajectories in more recent cohorts (Tables S7, and S8). Additional longitudinal follow-ups from young to mid-adulthood, particularly of the Add Health respondents, would provide more cognitive data for a life stage least studied and thus highly valuable for future research of cognitive change.
Third, study variations in sampling designs and compositions restricted direct comparisons across studies in the coordinated IDA. For example, the small numbers of NH Black and Hispanic respondents in the MIDUS and ACL studies limited the power for inference of racial/ethnic heterogeneity in overall cognitive function in the corresponding analyses (Tables S8, and S9). In this case, comparisons of results across studies are not straightforward or complete and call for future research using the pooled IDA that can better address study differences.
Finally, this study is already large in scale and could not include the assessments of structural measures of disparities, potential cultural bias in survey based cognitive measures that may disadvantage minority older adults, or other explanatory factors (biological, behavioral, or psychosocial) underlying individual differences in cognitive decline (Plassman et al., 2010; Stern et al., 2023). These are important topics for future research to examine causal pathways linking social status and cognition.
Nonetheless, this study breaks new ground in understanding the natural history and social disparities of cognitive aging over the full course of human cognitive development. Findings from this study further set the stage for future clinical, demographic, and epidemiological research to lessen disease burden of cognitive dysfunction in the U.S. population.
Supplemental Material
Supplemental Material - An Early and Unequal Decline: Life Course Trajectories of Cognitive Aging in the United States
Supplemental Material for An Early and Unequal Decline: Life Course Trajectories of Cognitive Aging in the Unites States by Yang C. Yang, Christine E. Walsh, Kaitlin Shartle, Rebecca C. Stebbins, Allison E. Aiello, Daniel W. Belsky, Kathleen Mullan Harris, Marianne Chanti-Ketterl, and Brenda L. Plassman in Journal of Aging and Health
Footnotes
Acknowledgments
We thank Dr. Patrick Curran for advice on the statistical methods for longitudinal data analysis. We also thank UNC Creative for producing the high-quality graphics for the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The main funding support for this project comes from National Institute on Aging grant R01AG057800 (PI: Yang). We also thank the support from the University Cancer Research Funds at the Lineberger Cancer Center (PI: Earp), National Institute of Child Health and Human Development grant 2P01HD031921 (PI: Harris).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.