Abstract
Introduction
A growing body of literature supports the significant impact that sensory loss (SL)—hearing, vision, or both—have on health outcomes in older adults. Two-thirds (68%) of adults over age 70 have hearing loss (Goman & Lin, 2016). One in four (24%) adults aged 70–79 and half of adults 80 and older have vision loss (Varma et al., 2016). Dual SL—both hearing and vision loss—impacts 11% of adults 80 and older (Swenor et al., 2013b). These common conditions permeate all aspects of life, resulting in substantial adverse health outcomes. Hearing loss is not only associated with cognitive decline (Lin et al., 2013) but also depression (Lawrence et al., 2020), social isolation (Shukla et al., 2020), and increased mortality (Lin et al., 2019). Similarly, vision loss is associated with cognitive decline (Rogers & Langa, 2010), depression (Kempen et al., 2012), reduced quality of life (Crews et al., 2014), mobility difficulties (Swenor et al., 2013a), and falls and fall-related injuries (Patino et al., 2010). Although the impact of dual SL is an emerging area of investigation, available research supports that dual SL is associated with a greater incidence of depression (Cosh et al., 2018), dependence (Soto-Perez-de-Celis et al., 2018), cognitive loss and dementia (Kuo et al., 2021), and mortality (Gopinath et al., 2013) compared to hearing or vision loss alone. As the population ages, the number of older adults with SL will continue to grow, resulting in a pressing need to understand the complex and interconnected health outcomes and healthcare utilization in those with SL.
Adults age 65 and older are the largest users of healthcare services, representing 36% of inpatient stays (Freeman et al., 2018) and over 85% of home health patients (Alliance for Home Health Quality and Innovation Home Health Chartbook, 2020; Freeman et al., 2018). Similarly, emergency department (ED) visit rates increase with age (Ashman et al., 2020). Over 80% of older adults have at least one chronic illness (Buttorff et al., 2017). Chronic illnesses often require ongoing healthcare interactions and self-care illness management. Yet, hearing and vision loss can make healthcare engagement and communication difficult.
Hearing and vision loss are associated with difficulty managing medications (Crews & Campbell, 2004), low health literacy (Wallace et al., 2022), depression (Cosh et al., 2018; Lawrence et al., 2020; Pardhan et al., 2021), cognitive decline (Lin et al., 2013; Rogers & Langa, 2010), communication challenges with healthcare providers (Agaronnik et al., 2019; Cudmore et al., 2017), not having a usual source of care (Reed et al., 2021), and reduced uptake in preventative screenings (Assi et al., 2020). SL may compromise chronic illness management and engagement with primary or preventative care, leading to a need for acute care services. Current evidence suggests those with SL require more health services, although the results are SL specific. Hearing and vision loss are independently associated with increased hospital readmissions (Chang et al., 2018; Morse et al., 2019), increased risk of hospitalizations (Bal et al., 2017; Reed et al., 2019), and overall greater healthcare costs (Morse et al., 2019; Reed et al., 2019). Hearing loss is associated with increased risk of emergency department (ED) visits (Reed et al., 2019; Wells et al., 2019). However, there is limited research examining vision loss and ED use. For instance, Medicare beneficiaries with glaucoma were not found to have increased odds of ED visits compared to Medicare beneficiaries without glaucoma (Prager et al., 2016). However, Medicare beneficiaries with glaucoma and vision loss have greater ED costs compared to those with glaucoma without vision loss, with costs associated with increasing vision loss severity (Bramley et al., 2008). To date, no studies have examined ED use in older adults with dual SL; however, those with dual SL experienced increased odds of hospitalization and more days of hospitalization compared to those without SL (Deardorff et al., 2019; Huddle et al., 2016).
Vision loss is also associated with increased use of home health services in international studies (Aljied et al., 2019; Hong et al., 2013; Jin et al., 2019); however, in the United States, there is a paucity of research in this area. Only one US study was located reporting Medicare beneficiaries with glaucoma and vision loss experienced 21% increased odds of home health aide visits compared to those without glaucoma (Prager et al., 2016). Limited research exists on healthcare utilization among older adults with SL. Expanding investigation to other health services for all three SL groups—hearing, vision, and dual SL—may add to our understanding of the impact of SL on healthcare use. The purpose of this study is to investigate healthcare utilization for older adults with a diagnosis of hearing, vision, or dual SL compared to those without SL, by examining hospitalizations, ED visits, and receiving home health services among primary care patients within a large academic health system. We hypothesized that older adult primary care patients with a SL diagnosis would have greater odds of having a hospitalization, ED visit, and receiving home health services compared to older adults without a diagnosis of SL.
Methods
Data Source, Setting, and Population
A secondary analysis was conducted using a subset of electronic health record (EHR) data from a parent study examining the mediating effects of medical complexity on the relationship between healthcare resource use and social vulnerability (Hanlon et al., 2021). For this secondary data analysis, we created a cross-sectional dataset by focusing on a subset (N = 45,000) of older adult (age ≥65) primary care patients who made at least one physician or specialist visit, or experienced at least one hospitalization, home healthcare, hospice service, ED, or observation visit in 2017. We used patient characteristic information from 2017. If patient characteristic data from 2017 was not available, we used data from 2015 or 2016, but this accounted for less than 3% of the data. Patients from 27 primary care practices participating within a single academic health system were included in this subset. Resource use variables were generated based on available EHR data from 2017 (see dependent variables below).
Measures
Elixhauser Comorbidity Index—Included Comorbidities.

Independent Variable
Sensory loss status was identified through ICD-10 diagnosis codes. The SL status variable included 4 categories: vision loss only, hearing loss only, vision and hearing loss, and no sensory loss. The following ICD-10 diagnosis codes were used to define vision loss: H25 (age-related cataract); H26 (other cataract); H27 (other disorders of the lens); H36.0 including E08.3, E09.3, E10.3, E11.3, and E13.3 (diabetic retinopathy); H40 (glaucoma); H42 (glaucoma in disease classified elsewhere); H53 (visual disturbances); H54 (blindness and low vision); and H35 (other retinal disorders). The diagnosis codes H90 (conductive and sensorineural hearing loss) and H91 (other hearing loss, including presbycusis, ototoxic hearing loss, sudden idiopathic hearing loss, and other unspecified hearing loss or deafness) were used to define hearing loss. Diagnosis codes were selected to capture common SL conditions experienced by older adults. The SL variable groups were created by dichotomizing (yes/no) if the patient had one of the vision or hearing loss diagnosis codes. Only those diagnosis codes included in the EHR 2017 were captured in the SL variable.
Dependent Variable
Healthcare utilization was measured by three dichotomous variables: hospitalizations, ED visits (without a hospital admission), and home health service use at the patient level during 2017. Hospitalizations were captured as no hospitalizations in 2017 (=0) or one or more hospitalizations occurred during 2017 (=1). Similarly, no ED visits in 2017 were scored as 0, and one or more ED visits that did not result in a hospital admission were scored as 1. For home health service use, if the patient received any home health services (e.g., skilled nursing visits, physical therapy, occupational therapy, and infusion therapy) during 2017, the patient received a score of 1 and a score of 0 if there were no home health services in 2017.
Statistical Analysis
The purpose of this analysis was to examine the relationships between SL and hospitalizations, ED visits, and home health controlling for patient demographics of age, Elixhauser index (Elixhauser et al., 1998), gender, race, and primary care site characteristics.
Descriptive statistics for demographic characteristics and healthcare utilization were generated overall and by the SL group. Demographics and healthcare utilization outcomes were compared by SL using chi-square or Kruskal–Wallis tests. Pairwise comparisons were performed if the overall comparison was significant at p = .05. p-values for pair comparisons were adjusted using the Tukey-Kramer method, and the significance level was set at p < .1.
Multivariable logistic regression models were estimated for binary healthcare utilization outcomes on 4 categories of SL, controlling for Elixhauser index, age, gender, race, and primary care site characteristics. Firth’s regression, as a penalized likelihood estimation method, was applied to account for the rare event nature of healthcare utilization in the population of interest and to produce finite, consistent estimates of regression parameters. Multicollinearity among the independent variables was checked with all variance inflation factors being less than 2. All analyses were conducted using SAS v9.4 (SAS Institute Inc., Cary, NC).
Results
Demographic Characteristics and Descriptive Statistics of Healthcare Use.
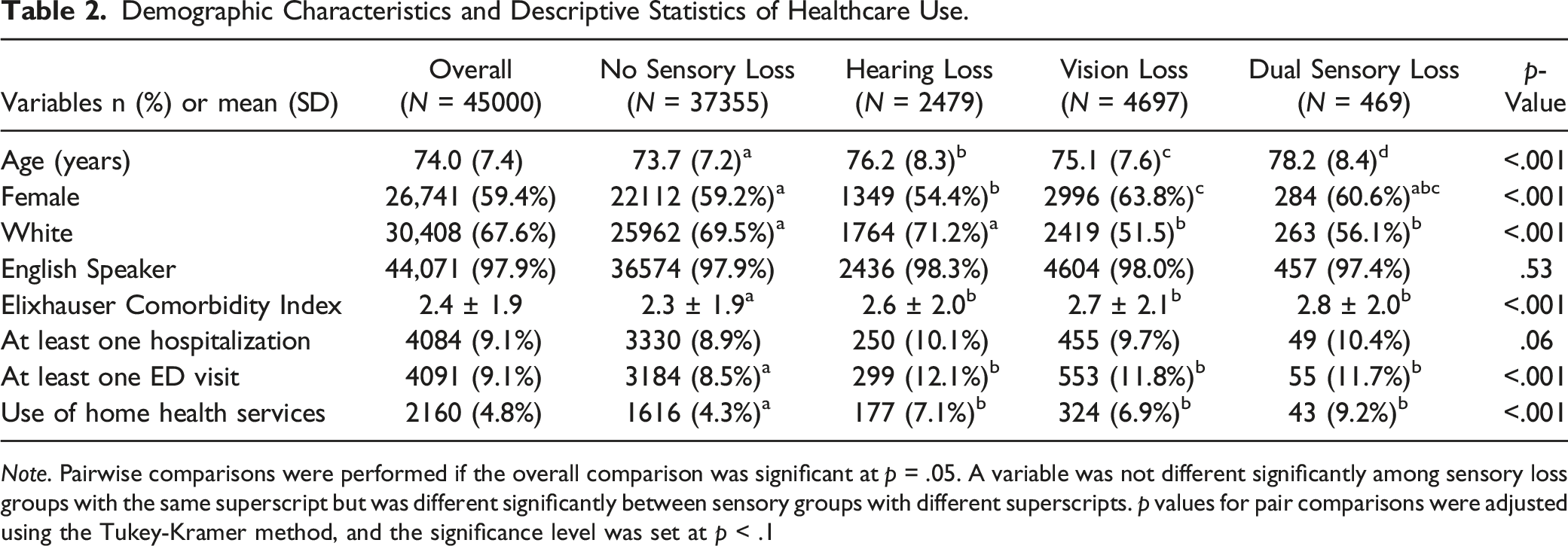
Note. Pairwise comparisons were performed if the overall comparison was significant at p = .05. A variable was not different significantly among sensory loss groups with the same superscript but was different significantly between sensory groups with different superscripts. p values for pair comparisons were adjusted using the Tukey-Kramer method, and the significance level was set at p < .1
Primary Care Site Characteristics in a Large Urban Health System (n = 27).
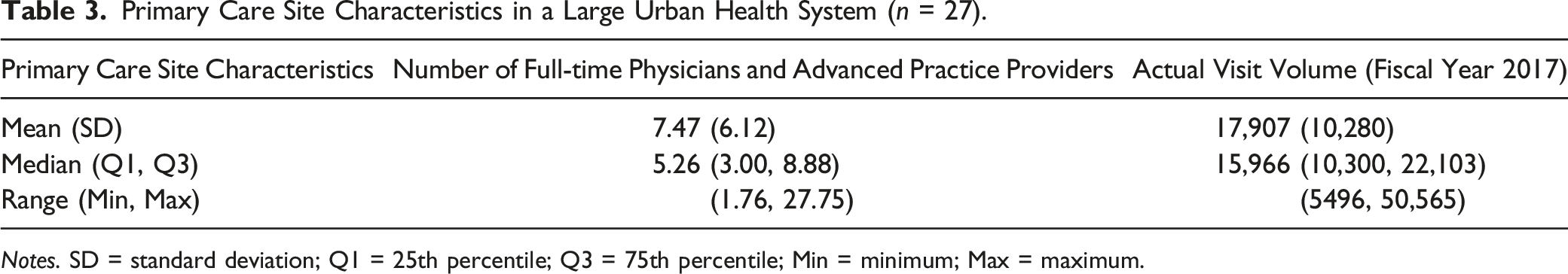
Notes. SD = standard deviation; Q1 = 25th percentile; Q3 = 75th percentile; Min = minimum; Max = maximum.
Healthcare Use Outcomes
Multivariate Binomial Logistic Regression: Hospitalization, ED, and Home Health Utilization for Hearing, Vision, and Dual Sensory Loss, N = 45,000.
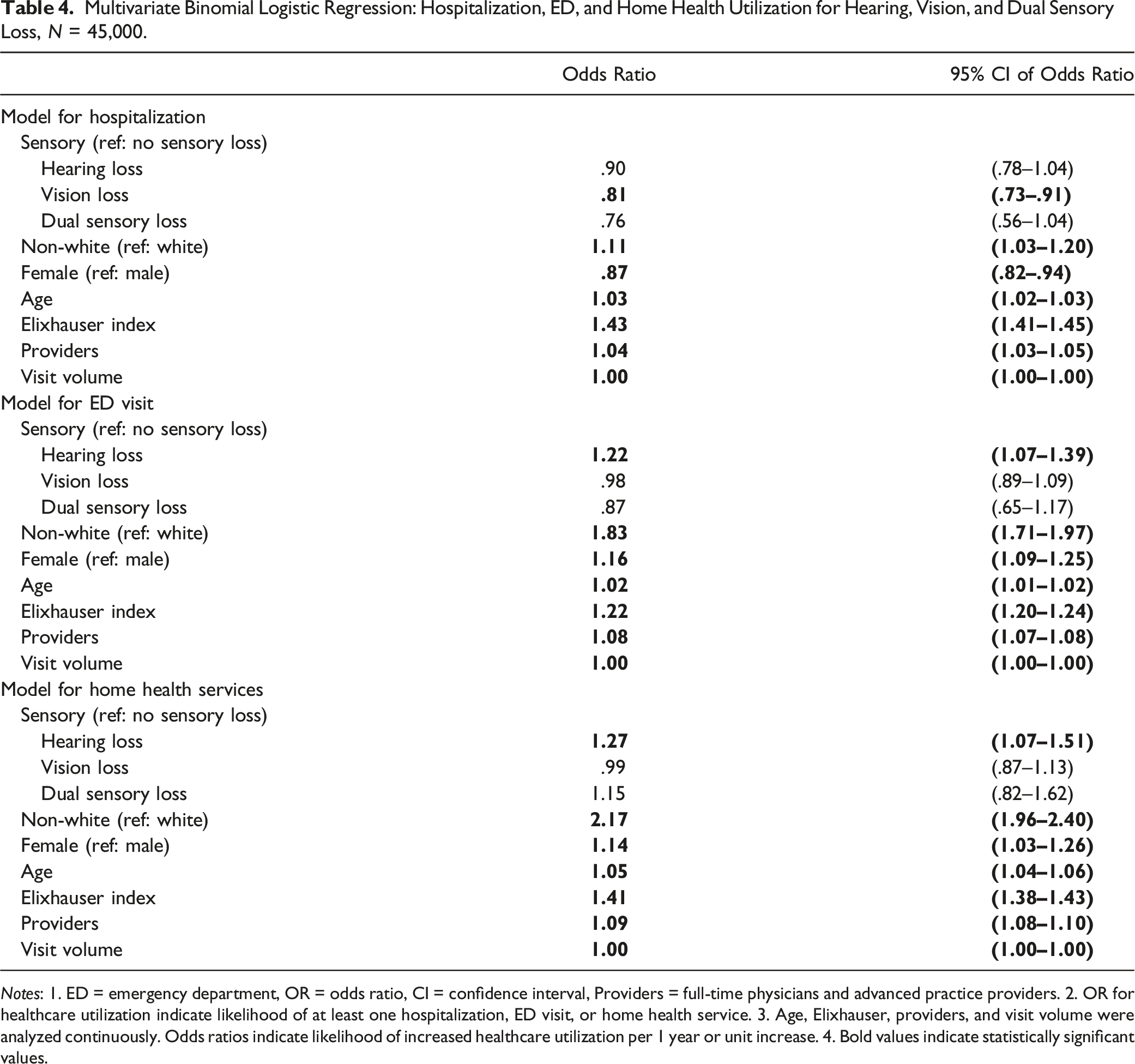
Notes: 1. ED = emergency department, OR = odds ratio, CI = confidence interval, Providers = full-time physicians and advanced practice providers. 2. OR for healthcare utilization indicate likelihood of at least one hospitalization, ED visit, or home health service. 3. Age, Elixhauser, providers, and visit volume were analyzed continuously. Odds ratios indicate likelihood of increased healthcare utilization per 1 year or unit increase. 4. Bold values indicate statistically significant values.
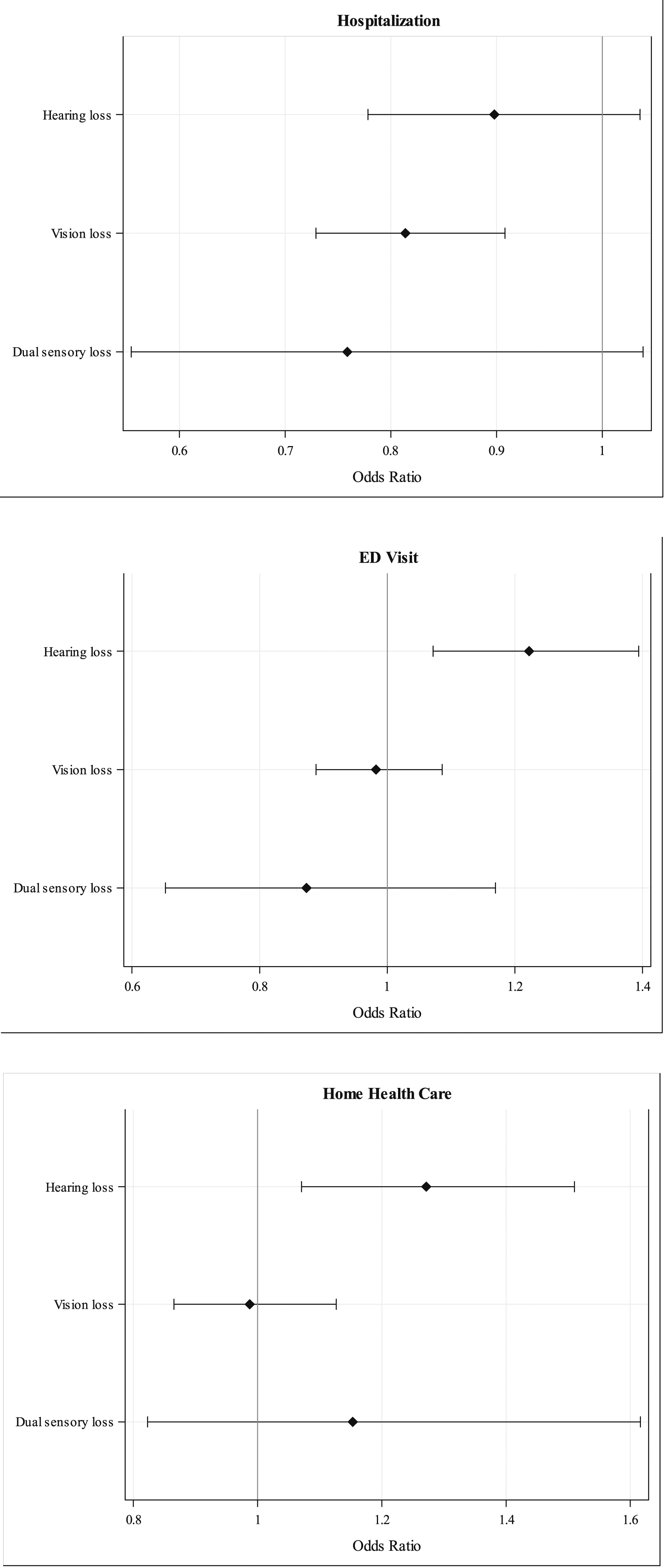
Odds ratio (OR) plots for multivariate binomial logistic regression models: Hospitalization, ED, and home health utilization for hearing, vision, and dual sensory loss.Note. OR for healthcare utilization indicates likelihood of at least one hospitalization, ED visit, or home health service use. Models controlled for race, gender, age, comorbidities, and site characteristics.
Hospitalizations
Hearing and dual SL were not associated with the likelihood of having a hospitalization. Vision loss was associated with 19% reduced likelihood of having a hospitalization compared to those without any SL (OR .81, 95% CI: .73–.91). In the overall sample, non-white race was associated with an 11% increase in likelihood of having a hospitalization (OR 1.11, 95% CI: 1.03–1.20), and older female patients were less likely than males to be hospitalized (OR = .87, 95% CI: .82–.94). Each additional year of age was associated with a 3.0% increase in odds of having a hospitalization (OR = 1.03, 95% CI: 1.02–1.03), and each additional condition on the Elixhauser index was associated with 43% increase in odds of having a hospitalization (OR = 1.43, 95% CI: 1.41–1.45).
Emergency Department
In the model for ED use, those with hearing loss were 22% more likely to have an ED visit than those without any SL (OR = 1.22, 95% CI: 1.07–1.39). Vision loss and dual SL were not associated with likelihood of having an ED visit (OR = .98, 95% CI: .89–1.09 and OR = .87, 95% CI: .65–1.17, respectively). In the overall sample, non-white race was associated with 83% greater likelihood of having an ED visit (OR 1.83, 95% CI: 1.71–1.97), and older female patients were more likely to have an ED visit (OR 1.16, 95% CI: 1.09–1.25). Each additional year of age was associated with a 2% increase in odds of having an ED visit (OR = 1.02, 95% CI: 1.01–1.02), and each additional condition on the Elixhauser index was associated with a 22% increase in odds of having an ED visit (OR = 1.22, 95% CI: 1.20–1.24). Each increase in number of providers at a given practice resulted in an 8% greater likelihood of having an ED visit (OR = 1.08, 95% CI: 1.07–1.08) while the effect of patient visit volume at the primary care practices was negligible in terms of ED use.
Home Health
Hearing loss was associated with a 27% greater likelihood of receiving home health services (OR = 1.27, 95% CI: 1.07–1.51). Vision loss and dual sensory loss were not associated with an increased likelihood of receiving home health services (OR = .99, 95% CI: .87–1.13 and OR = 1.15, 95% CI: .82–1.62, respectively. In the overall sample, the likelihood of receiving home health services for non-whites was 217% times the likelihood for whites (OR = 2.17, 95% CI: 1.96–2.40), and older female patients were 14% more likely to receiving home health services (OR = 1.14, 95% CI: 1.03–1.26). Each additional year of age was associated with a 5% increase in odds of receiving home health services (OR = 1.05, 95% CI: 1.04–1.06), and each additional condition on the Elixhauser index increased the odds of receiving home health services by 41% (OR = 1.41, 95% CI: 1.38–1.43). Each increase in number of providers resulted in an 9% greater likelihood of receiving home health services (OR = 1.09, 95% CI: 1.08–1.10) with the effect of patient visit volume at the primary care practices being negligible in terms of home health services.
Discussion
The purpose of this study was to examine differences in healthcare use among older adults with a diagnosis of hearing, vision, or dual SL seen in primary care of an urban academic health system compared to older adults in the same setting without any diagnosis of SL (hearing or vision). While we hypothesized that primary care older adults with any SL would have greater odds of having a hospitalization, ED visit, or receiving home health services compared to older adults without SL, we uncovered that only hearing loss is associated with increases in the likelihood of having an ED visit or receiving home health services. No relationship was identified between those with dual SL, and vision loss was unexpectedly associated with a reduced likelihood of hospitalization. The findings reported here add to our understanding of the association between resource use and SL among older adults, providing a more comprehensive picture of the needs among this primary care population.
First, this study’s results indicate that, compared to older adults without any SL, hearing loss was the only SL associated with increased odds of having an ED visit among primary care patients. The increased likelihood of having an ED visit for older adults with a diagnosis of hearing loss compared to those without a diagnosis of any SL is consistent with prior literature (Foley et al., 2014; Reed et al., 2019; Wells et al., 2019) despite differences in study populations. Our study included primary care patients, indicating that older adults with hearing loss experience increased odds of having an ED visit despite having an established primary care provider. Further research is needed to determine why older adult primary care patients with hearing loss have greater likelihood of having an ED visit. However, it is possible that the impact of comorbidities and communication barriers may have contributed to the need for ED visits. Hearing loss in older adults is associated with greater morbidity across several health conditions (Deal et al., 2019), including associations with cognitive decline and dementia (Deal et al., 2017, 2019; Lin et al., 2013), depression (Deal et al., 2019; Lawrence et al., 2020), falls (Crews & Campbell, 2004; Deal et al., 2019; Jiam et al., 2016), and mobility deficits (Crews & Campbell, 2004). An increased rate of comorbidities and mobility difficulties may result in a greater risk of acute illness and injury requiring emergency services. ED visits for older adults with hearing loss remained significant in our sample even after controlling for comorbidities. It is possible that not all adverse conditions associated with hearing loss are captured in the Elixhauser comorbidity index, such as falls and mobility deficits (Crews & Campbell, 2004; Elixhauser et al., 1998). Moreover, communication challenges with healthcare providers may increase vulnerability between primary care visits and compromise self-care management. Individuals with hearing loss report communication difficulties with providers during hospitalization and primary care visits (Cudmore et al., 2017; Stevens et al., 2019). Older adults with hearing loss also report greater difficulty with medication management compared to those without sensory loss (Crews & Campbell, 2004) which could lead to increased ED visits, hospitalizations and receiving home health services. Communication difficulties during primary care visits and challenges managing medications may increase the occurrence of acute health issues necessitating emergency services. Future research is needed to elucidate the causes of increased ED use in older adults with hearing loss to support targeted early detection and treatment interventions.
Second, the relationship between vision loss and ED use is not well known. Older adults with vision loss diagnoses in this study were not found to have greater likelihood of ED use, which is consistent with a prior study examining ED use in Medicare beneficiaries with glaucoma (Prager et al., 2016). Conversely, another study showed increased ED costs for Medicare beneficiaries across glaucoma-related vision loss severity categories (Bramley et al., 2008). However, prior studies limited the population to beneficiaries with glaucoma, decreasing generalizability to the broader population of older adults with vision loss diagnoses. Similar to hearing loss, older adults with vision loss have a greater risk of comorbidities (Crews et al., 2017) and adverse health outcomes (Crews et al., 2014; Kempen et al., 2012; Morse et al., 2019; Patino et al., 2010; Rogers & Langa, 2010) yet findings suggest this population does not experience increased need for emergency services, indicating the unique impact of each SL.
Third, only hearing loss was associated with increased odds of receiving home health services. To our knowledge, no studies have examined home health service use in those with hearing or dual SL. However, our study findings on hearing loss and home health service use are consistent when considering the clinical need for home health services. Hearing loss is associated with not just higher rates of common chronic conditions (e.g., heart disease, diabetes, and dementia) (Crews & Campbell, 2004; Deal et al., 2019) but also mobility deficits and falls (Crews & Campbell, 2004; Deal et al., 2019). The need for home health nursing for medication reconciliation, healthcare education, and chronic disease management is essential for those with hearing loss. Additionally, home health physical therapy and occupational therapy may be indicated in this population. We controlled for comorbidities with the Elixhauser index and still identified an increased utilization of home health services independent of their comorbidities in older adults with hearing loss. Despite greater home health use in those with hearing loss, dual SL did not increase likelihood of home health use, possibly due to a lack of statistical power.
Surprisingly, vision loss was not associated with increased likelihood of home health service use, which was unexpected considering the clinical need for home health services and results from the limited number of mostly international studies examining home healthcare service use among individuals with vision loss (Aljied et al., 2019; Hong et al., 2013; Jin et al., 2019; Prager et al., 2016; Wang et al., 1999). An Australian study found individuals with vision loss were three times more likely to use community support services, which included nurse visits (Wang et al., 1999). A Canadian study reported older adults with vision loss were 30% more likely to use home care services than those without vision loss (Jin et al., 2019). One US study found Medicare beneficiaries with glaucoma and vision loss had 21% increased odds of home health aide visits compared to beneficiaries without glaucoma (Prager et al., 2016). Results from our study differ from that of prior studies. One possible explanation of the different findings is variation in the definition and measurement of vision loss across studies.
Also unexpected was that no SL groups were associated with greater likelihood of hospitalization and vision loss was associated with reduced likelihood of hospitalization. Prior evidence suggests that hearing, vision, and dual SL have all been independently associated with greater rates of hospitalization (Bal et al., 2017; Deardorff et al., 2019; Reed et al., 2019). It is possible that by examining these events associated with primary care practices limited our ability to detect a difference. More research is needed to examine rates of hospitalization among those with SL.
Finally, the disparity in healthcare use between whites and non-whites in the overall sample was noteworthy, with non-whites having 83% greater likelihood of having at least one ED visit, and 117% greater likelihood of receiving home health services. However, limited conclusions can be inferred from these numbers as the study was not designed to examine racial disparities. As the study dichotomized race, the precise racial/ethnicity composition of the non-white group is not known. Based on parent study data, the majority of non-white patients were Black. This study also did not include insurance or socioeconomic status as covariates, which would provide greater insight into potential inequities. Further research in this area is warranted including consideration of socioeconomic status, insurance, and equitable access to services.
There are several notable limitations of this study. First, the EHR data were limited to a single large academic health system. Hospitalization, ED, and home health data were available only for those who used the academic health system for such services. While the majority of patients are admitted, go to the ED, and use the primary home health agency affiliated with the academic health system, it is likely that patients had resource use elsewhere. Second, this study only included patients with an ICD-10 diagnosis of hearing or vision loss, which only represented the codes entered for billing if a SL was addressed at a healthcare visit. This does not capture the patients who have SL without an ICD-10 diagnosis documented in their EHR, or patients with a SL ICD-10 diagnosis documented after 2017. As hearing loss prevalence is estimated to be 68% for those over 70 years (Goman & Lin, 2016), vision loss prevalence is estimated to be 24% for those 70–79 years (Varma et al., 2016) and dual sensory loss prevalence is estimated to be 11% of those over 80 years (Swenor et al., 2013b), the 17% of the study sample with any type of SL is a vast underrepresentation within the subpopulation, limiting generalizability. Third, this study only provides information on healthcare utilization; no causal relationships can be inferred. Fourth, despite having a large overall sample, the number of participants with dual SL was small, limiting power to detect differences. Fifth, data on the reasons for service use were not available in the data. Examining reasons for ED visits and home health service use would provide greater understanding into avoidable versus unavoidable service use. Lastly, limitations of the dataset prevented inclusion of important confounding factors, such as smoking and socioeconomic status. The dataset was also limited to 2017, prior to the SARS-COV-2 pandemic, which may not represent current healthcare use patterns.
This study has notable clinical implications. SL care and treatment may address risks associated with hearing and vision loss. Older adults with hearing loss may benefit from interventions that mitigate the risk of ED use, such as patient education, care coordination, or managed care (Morgan et al., 2013). However, such interventions should be implemented in consideration of the needs and accommodations required of the population. Older adults with SL may also benefit from SL-specific interventions, such as visual and aural rehabilitation, and assistive devices such as hearing aids, which support improvements in function and independence. The increased use of home health services may indicate a positive response for those with hearing loss as they have greater needs of assistance with medication management, self-care education, chronic disease management, and physical or occupational therapy. Thus, individuals with SL, notably hearing loss, may benefit from healthcare providers assessing for challenges related to self-care management and mobility prior to an observable need, and outpatient interventions may yield benefit prior to the requirement of home health services.
Additional research exploring healthcare utilization patterns amongst those with hearing, vision and dual sensory loss is needed using comprehensive data to accurately capture the population’s resource use. Future research is recommended to provide insight into the reasons for increased ED and home health service utilization among older adults with hearing loss. Identifying causes of increased healthcare utilization will support development of clinical interventions to mitigate risks and offset utilization costs.
Footnotes
Author Contributions
All authors have reviewed and supported submission of this manuscript. MDN is the principal investigator of original study. KBH and MDN acquired study data. LGW, LH, and PZC contributed to study concept and design. LH conducted statistical analysis and provided interpretation support. LGW, LH, KH, and PZC contributed to analysis and interpretation of data and drafted the manuscript. All authors provided revisions to the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The findings reported in this publication were supported in part by NIH NRSA T32NR009356 and the National Center for Advancing Translational Sciences of the National Institutes of Health under Award Number UL1TR003015.
Disclosures
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Support was also provided by the NewCourtland Center for Transitions and Health at the University of Pennsylvania School of Nursing and the Perelman School of Medicine Center for Therapeutic Effectiveness Research. This study was presented as a poster at the Gerontological Society of America Conference, Nov 10-13, 2021.
Sponsor’s Role
LGW was supported by a grant from the National Institute of Health (NIH) National Institute of Nursing Research (NINR) Institutional National Research Service Award (T32NR009356). NIH NINR had no role in the design, methods, subject recruitment, data collections, analysis, or preparation of manuscript. The parent study was supported by funding from the University of Pennsylvania School of Nursing NewCourtland Center for Transitions and Health, and the Perelman School of Medicine Center for Therapeutic Effectiveness Research.
