Abstract
Spinal cord injury (SCI) represents a major public health issue, as the consequences are often irreversible with no treatment currently available. This results in a growing population living with long-lasting motor, sensory, and/or autonomic impairments directly related to their SCI. Here, we have evaluated the therapeutic potential of a thrombospondin repeats peptide analogue, named NX210, in a mouse hemisection model of SCI. Adult female mice were subjected to a thoracic level 8 dorsal hemisection, and treated with intraperitoneal injections of NX210 starting at 4 h post-injury and then twice a week at 4, 8, or 16 mg/kg. Hind limb motor function was assessed once a week for 10 weeks post-injury using the Basso Mouse Scale (BMS) score and sub-score, the rotarod, and the activity chamber tests. Mice were then sacrificed, and the spinal cords were collected for immunohistochemistry. Interestingly, NX210 improved functional recovery (BMS score and sub-score, latency to fall from the rotarod, spontaneous locomotor activity) with rapid rises in function that were maintained throughout the 10-week study. This was accompanied by a reduction of nociceptive reactivity assessed by the tail flick test. NX210 treatment also increased myelin basic protein and reduced neuron/glial antigen 2 at the injury site 10 weeks post-injury while no significant effects were observed on lesion size, inflammation, and neuron survival. Overall, this study highlights a potential new therapeutic strategy to promote repair and decrease long-lasting functional impairments after SCI.
Introduction
Spinal cord injury (SCI) is a progressive and permanent condition that has a devastating impact on quality of life.1,2 It is the second most common cause of paralysis, following stroke. 3 SCI represents a heavy medical and financial burden to patients, their families, and society, and with an aging SCI population, people are living longer with SCI creating a significant increase in the associated lifetime costs. Currently, there is no cure for SCI, and limited treatment options in the acute phase of the injury to boost neurorepair processes and improve neurological outcomes.4–7 Surgical interventions to stabilize the vertebral column and decompress the spinal cord are employed with some success in acute spinal column injury, 7 but are not as effective if the spinal cord tissue is severely compromised. Additionally, high doses of methylprednisolone administered in the first 8 h post-injury show limited acute and chronic benefits.8,9 There is extensive research focused on reducing the degenerative secondary injury phase, and promoting tissue repair and regeneration; however, no effective treatment targeting the early secondary injury phase is currently available to SCI patients.
SCI consists of two phases that contribute to the observed pathology.4,10,11 The first is the initial mechanical trauma that can result in fractures, breaks, or damage to the vertebral column, as well as extensive damage to the spinal cord itself. This involves disruption of axons, surrounding glial cells, and blood vessels in the cord.11–13 The secondary phase of SCI is a delayed and continuously developing expansion of the injury, which manifests in a broad spectrum of pathologies that exacerbate the injury.4,11,13 A hallmark of the secondary injury phase includes an inflammatory and fibrotic lesion core surrounded by a glial border.14–18 This glial border is composed primarily of astrocytes and, in the acute and sub-acute phases of SCI, functions to segregate the inflammatory lesion core, prevent its detrimental expansion, and reduce tissue loss.15–20 Inflammation is significant in the progression of SCI, 21 however this complex and cascading response has the potential to be both beneficial and detrimental to ongoing outcomes.4,21–23 The inflammatory response to injury in the central nervous system (CNS) includes a central area of inflamed and necrotic tissue at the lesion site surrounded by spreading axonal swelling, Wallerian degeneration, and further inflamed tissue. 23 This involves loss of multiple populations of endogenous nervous cells, including neuronal populations and mature oligodendrocytes. The areas of damage are surrounded by areas of histologically intact tissue and spared axons, which are important for therapeutic developments and potential recovery of function.4,10,22,23 Developing strategies to increase survival of neurons and oligodendrocytes while reducing inflammation and mitigating astrogliosis are of high interest to promote functional recovery afSCI. An acute pharmaceutical approach is one potentially promising approach, especially with the ability for effective systematic delivery.
NX210 is a 12-amino acid residue peptide derived from the most conserved consensus sequence of the thrombospondin type 1 repeats of the subcommissural organ (SCO)-spondin (Axoltis Pharma, France). This glycoprotein strongly contributes to the regulation of cerebrospinal fluid activity, neurogenesis, and axonal guidance, however, only remnants subsist beyond early developmental stages in humans. 24 NX210 robustly increased neuron adhesion and growth of chick cortical and spinal cord neurons in vitro. 25 In animal models, the linear and/or cyclic forms of peptide (NX210 and NX210c, respectively) reduced motor impairments after SCI 26 and cognitive deficits notably in Alzheimer’s disease.27,28 The peptide already reached the clinic, and was shown to be safe and well tolerated in phase 1 single and multiple ascending dose studies after intravenous administrations to healthy volunteers.29,30
In this study, we have examined the effects of repeated intraperitoneal (i.p.) administrations of NX210 peptide in mice 10 weeks after a dorsal hemisection of the spinal cord at the thoracic level. Using a nociceptive stimulation (tail flick) to assess pain, we observed reduced SCI-induced thermal hyperalgesia with 8 and 16 mg/kg doses of NX210. We also found improved functional recovery in mice, using Basso Mouse Scale (BMS), rotarod, and activity chamber tests, most significantly with the 8 mg/kg dose. Finally, we found increases in myelin basic protein (MBP) and reduction in neuron/glial antigen 2 (NG2) at and around the lesion in response to treatment, which may contribute to the therapeutic effect of NX210. The 8 mg/kg dose produced the best recovery and histological outcomes. Overall, this study describes the potential of repeated systemic delivery of NX210 to promote functional recovery, improve some histological outcomes, and alleviate pain hypersensitivity after SCI.
Materials and Methods
Animals
All experimental procedures were conducted in strict adherence to the Institutional Animal Care and Use Committee at Texas A&M University. Texas A&M University is fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care, International (AAALAC). The animal protocol to test the NX210 peptide was approved by Texas A&M - AUP IACUC 2018-0048. Seven to eight-week-old female C57Bl/6 mice were purchased from TACONIC (N = 30). Mice were housed in groups under controlled temperature, humidity, and light conditions (temperature 72°F, relative humidity 52%, 12 h/12 h light/dark cycle) with access to food and water ad libitum.
Dorsal hemisection model of SCI
Mice were subjected to a dorsal hemisection of the spinal cord as described previously. 31 A laminectomy was performed at the thoracic level 8 (T8) to expose the spinal cord. To achieve the dorsal hemisection, the dura was punctured bilaterally with a 30G needle at appropriate locations. The dorsal half of the spinal cord was then cut with a pair of superfine iridectomy scissors at a depth of 0.8 mm. Finally, a micro feather ophthalmic scalpel was used to retrace the lesion to ensure its completeness. The muscles were sutured with 5.0 sutures and the skin was secured with 5.0 sutures and Dermabond. Buprenorphine (0.05 mg/kg) and Penicillin (100,000 units/kg) were injected subcutaneously post-surgery to prevent pain and infection. Beforehand, mice were randomized into four groups: SCI + saline, SCI + NX210 (4 mg/kg), SCI + NX210 (8 mg/kg), and SCI + NX210 (16 mg/kg). Importantly, the surgeon was blinded to the treatment groups.
SCO-spondin-derived NX210 peptide
NX210 (H-WSGWSSCSRSCG-OH) was manufactured by PolyPeptide (Strasbourg, France) and supplied as a good manufacturing practices acetate salt lyophilizate by CARBOGEN AMCIS (Bubendorf, Switzerland) (96% purity). It was reconstituted in its vehicle (water for injection) at 3 mg/mL and aliquoted for storage at −80°C.
Treatment
NX210 and its vehicle were administered i.p. at 4 h post-injury and then twice a week for 10 weeks. Three doses of NX210 were tested: 4, 8, and 16 mg/kg.
Assessment of motor and sensory functions
For all the tests, animals were tested in a random and double-blind manner: all behavioral tests were performed by observers blinded to the treatment groups and quantified by different observers also blinded to the treatment groups.
Basso mouse scale
Mice were placed in an open field and observed for 5 min by two observers. 32 The same observers performed BMS scoring throughout the study, to ensure consistency. Both BMS scores, ranging from 0 (no movement) to 9 (normal locomotion) and sub-scores, ranging from 0 (no movement) to 11 (normal locomotion), were calculated to evaluate limb coordination more precisely.33,34 Mice were tested prior to the injury and then at days 2, 7, 14, 21, 28, 35, 42, 49, and 56 after SCI.
Rotarod
Mice were placed on a rod (Ugo Basile) rotating with increasing speed to 50 rpm over 3 min with constant acceleration. The latency to fall (in seconds), the distance (in meters), and the number of rotations per minute were averaged across three trials per session. 34 Mice were acclimated to the rotarod 1 week before injury for 5 days (2 sessions), with an additional test session 1 day prior to injury (baseline). Mice were then tested on days 2, 8, 15, 22, 29, 36, 43, 50, and 57 post-SCI.
Activity chamber
Locomotor activity is recorded by placing the mice in an open-field arena equipped with light beam arrays in the horizontal X and Y axes. The hardware detects beam paths broken by the animal and determines the location of the mouse within the arena. The total number of movements, distance, and velocity are used as indices of each mouse’s activity, while anxiety-like behavior can be derived from the calculation of the time spent in the center of the chamber. 33 Mice were acclimated to the arena twice for 10 min before testing, and then tested on days 1, 2, 8, 15, 22, 29, 36, 43, 50, and 57 post-SCI. Mouse activity was recorded for 10 min during each test session.
Thermal hyperalgesia
This test provides a measure of spinal reactivity to a nociceptive stimulus that does not rely on communication with the brain. 35 Prior to testing, the intensity of an aversive hot light stimulus (Tail Flick Analgesia Meter, IITC) directed onto the mouse’s tail was set to produce a tail-flick response in naive mice within 2–3 s. Mice were then tested prior to the injury and once weekly post-injury. During testing, if a mouse did not make a tail-flick response within 8 sec, the light stimulus and test session were terminated, to prevent the radiant heat from producing tissue damage.
Immunohistochemistry
Mice were sacrificed on day 73 post-injury by a lethal dose of Fatal plus (pentobarbital), and perfused transcardially with phosphate-buffered saline (PBS)-Heparin (10,000 unit/L, 20 mL, 5 mL/min) followed by a 4% paraformaldehyde fixative solution (30–40 mL/mouse, at 5 mL/min). Following perfusion, spinal cords were post-fixed overnight at 4°C in the same fixative solution. They were then immersed in 30% sucrose for 3 days for cryo-protection. Different segments of the spinal cord (from 4 mm rostral to 4 mm caudal to the injury) were embedded in optimal cutting temperature compound and snap frozen on dry ice. Spinal cords were sectioned with a cryostat (Thermofisher, Microm HM550) at 25-μm thickness (longitudinal and transverse sections), and stored at −20°C in a cryoprotectant solution (30% sucrose and 30% ethylene glycol diluted in PBS) for further processing. After washing with PBS, sections were treated with a blocking buffer containing 5% normal horse serum (Sigma-Aldrich, St Louis, MO, USA) and 0.4% Triton X-100 (Sigma-Aldrich) for 1 h. Incubation with primary antibodies was conducted overnight at room temperature (RT) with dilutions as follows: rabbit anti-glial fibrillary acidic protein (GFAP, 1:500; 130300, ThermoFisher Scientific, Waltham, MA, USA), rabbit anti-neuronal nuclei (NeuN, 1:500; 24307S, Cell Signaling Technology, Danvers, MA, USA), rat anti-cluster of differentiation 68 (CD68, 1:500; MCA1957, Bio-Rad, Hercules, CA, USA), rabbit anti-ionized calcium binding adaptor molecule 1 (Iba1) (1:500; 019-19741, FUJIFILM Wako Chemicals, Richmond, VA, USA), rat anti-MBP (1:500; MAB386, Sigma-Aldrich), rabbit anti-NG2 (1:500; AB5320, Sigma-Aldrich) and rabbit anti-protein kinase C gamma (PKC-γ, 1:500; 59090S, Cell Signaling Technology) overnight at 4°C. After washing with PBS, sections were incubated with goat anti-rat AF 488 (1:500; A11006, ThermoFisher Scientific) and goat anti-rabbit AF 555 (1:500; A32732, ThermoFisher Scientific) secondary antibodies for 1 h at RT, washed again then stained with DAPI (1:5000; D3571, ThermoFisher Scientific) for 10 min, washed, and finally mounted in Fluoromount-G (Southern Biotechnology, 0100-01, Birmingham, AL, USA). Negative controls for nonspecific binding of the secondary antibody were conducted in parallel sections following the same procedure described above, except for the incubation with primary antibodies. Spinal cord sections were imaged in Z-stack at 20×, tiled, and stitched to include 1600 µm rostral and caudal to the injury epicenter on a Zeiss Axio Observer fluorescent microscope. Tissue processing and analyses were performed by observers blinded to the treatment groups. Four animals per group were randomly chosen for immunohistochemistry analyses.
Immunohistochemistry analyses
Images were quantified using ZEN Blue software (version 2.6.76). Eight boxes with a width of 200 μm were drawn both rostral and caudal to the injury epicenter. A contour polygon was made around the spinal cord in each of the 16 boxes. The fluorescent intensity in each segment was measured, and normalized to background intensity, to quantify GFAP, MBP, NG2, and Iba1 expression. Fluorescent intensity ratios were calculated by dividing each individual polygon by the polygon furthest away from the injury both rostral and caudal. The freehand polygon tool was also used to trace and measure lesion size.33,34 CD68 expression was quantified in ImageJ as the percentage of CD68 fluorescence per area (percent positive area) in thresholded black and white images. The number of NeuN-positive cells was manually counted in each individual segment and then divided by the area of each polygon. As there were no viable indicators for which direction was rostral and which was caudal to the injury, and no guarantee that each section was mounted in the same direction, the plus and minus were grouped together for analysis. Lesion depth was analyzed manually for all sections using ImageJ. To do so, a line perpendicular to the length of the cord in the center of the injury was drawn between the outer edge of the cord and the deepest point of the injury in the parenchyma. The length of this line was measured and averaged between sections spanning the mediolateral axis for each animal.
Statistical analyses
Prior to statistical analyses, one mouse from the vehicle-treated SCI group was identified as a significant outlier on the BMS score at 2 days post-injury (using GraphPad QuickCalcs) and was therefore removed from all analyses. Statistical analyses were performed with GraphPad Prism software package 7.0 (GraphPad Software, La Jolla, CA, USA) using one-way and two-way ANOVA repeated measures followed by Ficher’s and Bonferroni’s post hoc multiple comparisons. An alpha level of p < 0.05 was used for the determination of significance in all statistical tests. The data are expressed as mean ± standard error of the mean (SEM).
Results
NX210 promoted functional recovery in a hemisection model of thoracic SCI in mice
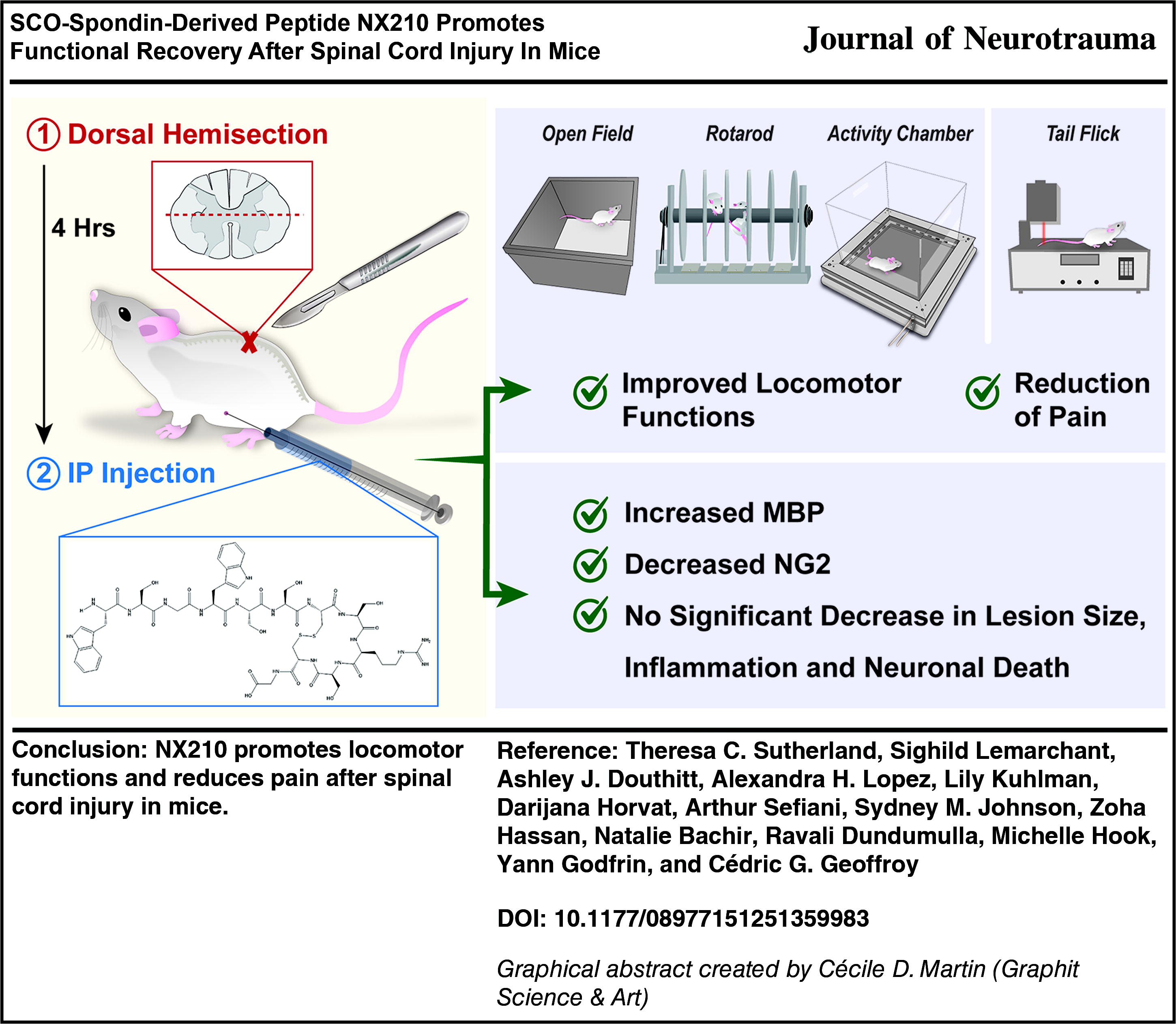
Analysis of locomotor function, using the BMS open-field test, revealed significantly better recovery in NX210-treated mice compared to vehicle controls (Fig. 1A). Mice treated with 4, 8, or 16 mg/kg of NX210, had significantly higher (p < 0.01) BMS scores than vehicle-treated mice, from day 14 post-injury, at the latest, to 56 days post-injury. Treatment with the 8 and 16 mg/kg doses produced even more rapid improvements in recovery (Fig. 1A). Interestingly, 50%, 100% and 75% of mice treated with NX210 at 4, 8, and 16 mg/kg, respectively, had a BMS score superior or equal to 5 at the end of the study (56 days post-injury), a score indicative of plantar stepping and at least occasional limb coordination (Fig. 1B). Eighty-three percent (83%) of NX210-treated mice (8 mg/kg) reached a BMS score ≥5 as early as 14 days post-injury, demonstrating rapid and significant functional recovery. Conversely, only 14.3% of vehicle-treated mice reached a BMS score of 5. The BMS sub-score was also significantly higher in mice treated with 8 and 16 mg/kg of NX210 daily, from 7 days post-injury to the end of the study (p < 0.01 and p < 0.05, respectively) and also nonsignificantly increased in the 4 mg/kg group (Fig. 1C). Mice were weighed weekly as an indicator of general health. The weight of SCI mice treated with NX210 was significantly higher than that of vehicle-treated mice from 13 (8 mg/kg), 20 (4 mg/kg), and 41 (16 mg/kg) days post-injury to the end of the study (Fig. 1D).
NX210 promoted hind limb recovery and general health in a hemisection model of thoracic SCI in mice.
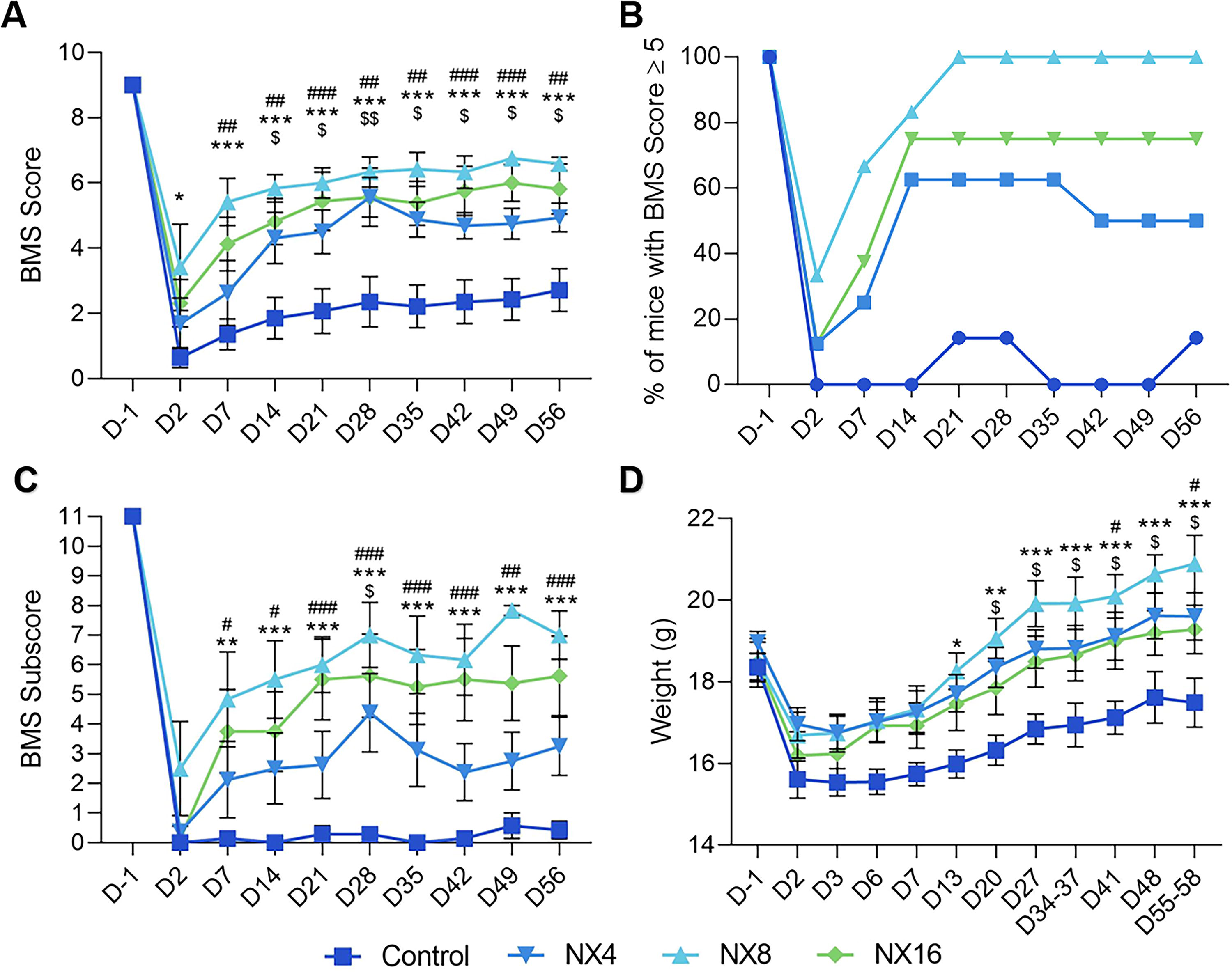
Daily administrations of 8 mg/kg NX210 also increased the latency to fall from the rotarod from 8 days post-injury to the end of the study. Mice treated with 16 mg/kg also stayed on the rotarod longer than vehicle-treated mice (Fig. 2A). Similar results were obtained for the distance (8 mg/kg) and the number of rotations per minute (8 and 16 mg/kg) (data not shown).
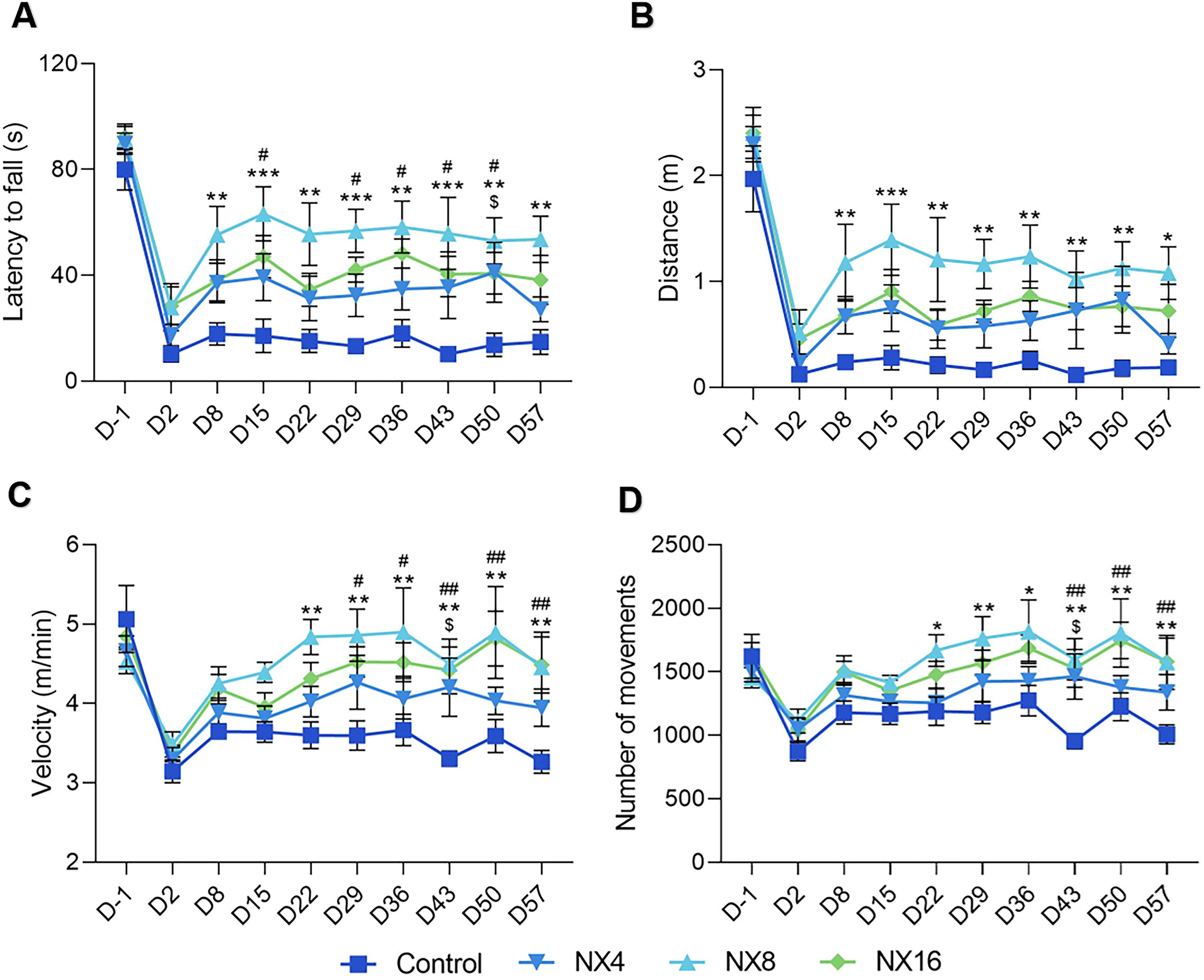
NX210 promoted locomotor recovery in a hemisection model of thoracic SCI in mice.
Analysis of spontaneous locomotor activity using the activity chamber demonstrated that NX210 administrations at 8 and 16 mg/kg increased the distance (Fig. 2B) and the velocity (Fig. 2C) of locomotion from 22 to 29 days post-injury respectively, to the end of the study. Similarly, 8 and 16 mg/kg NX210 treatments increased the total number of movements from 22 to 43 days post-injury respectively (Fig. 2D). NX210 did not influence the time spent in the center of the activity chamber (data not shown).
NX210 prevented SCI-induced thermal hyperalgesia
Pain sensitivity was assessed using the tail flick test (Fig. 3A–B). Vehicle-treated mice and mice treated with 8 mg/kg NX210 displayed significantly reduced latencies, to flick the tail away from the radiant heat stimulus, at 49 days post-injury compared to their baselines, suggesting SCI-induced thermal hyperalgesia (Fig. 3B). However, mice treated with NX210 at 4 and 16 mg/kg did not show any changes in their tail flick latencies (Fig. 3B). This suggests that NX210 prevented SCI-induced thermal hyperalgesia.
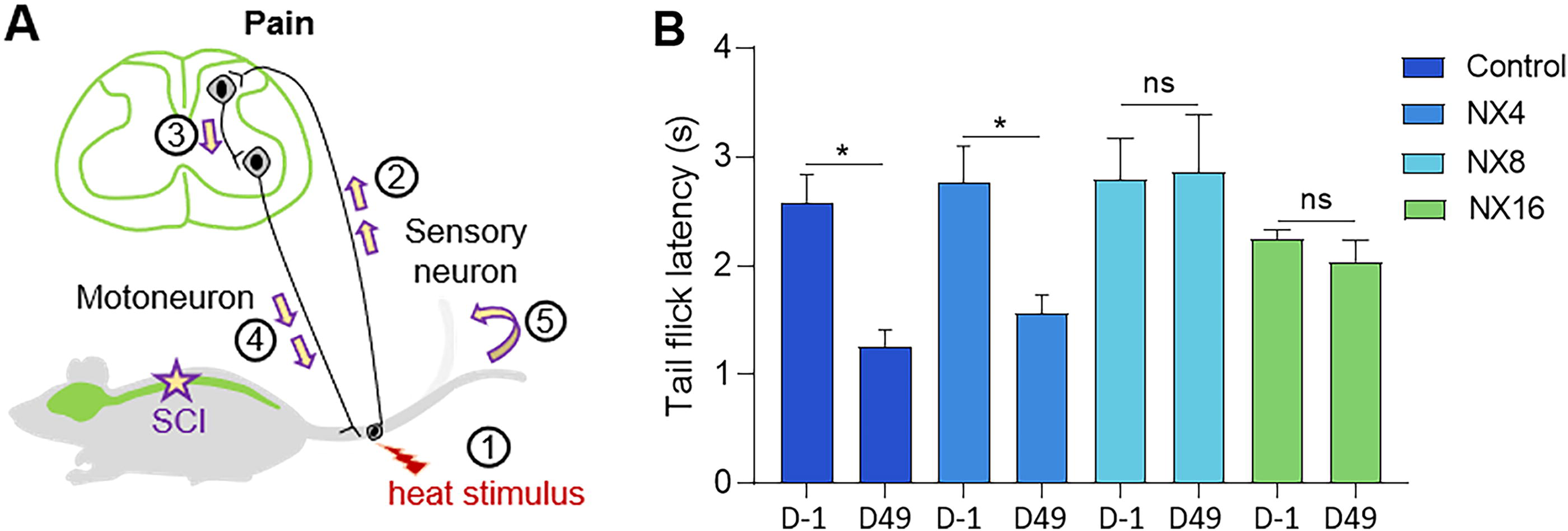
NX210 prevented SCI-induced thermal hyperalgesia.
Consistency of dorsal hemisection lesion
The consistency of the dorsal hemisection lesion was verified by measuring the lesion depth from the dorsal surface, and the efficacy of the lesion was verified using PKC-γ expression in the dorsal corticospinal tract (CST) rostral and caudal to the T8 hemisection. The lesion depth showed no significant difference between groups (Fig. 4A). The PKC-γ staining showed robust expression in the dorsal CST above the hemisection and no expression below (Fig. 4B–Bi). This confirmed effective and similar lesions among groups.
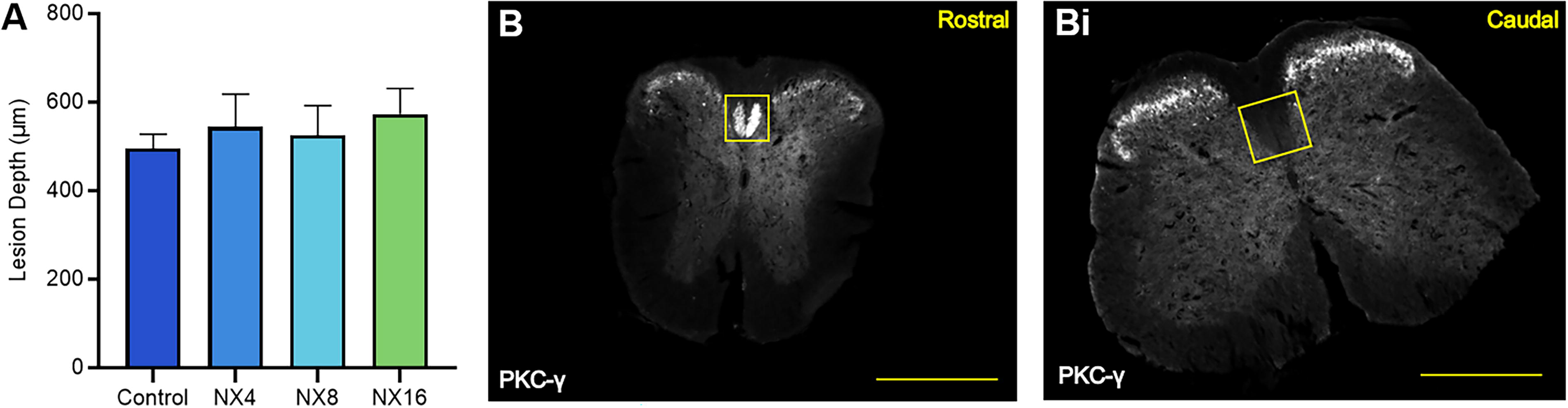
Dorsal Hemisection lesion verification and quality control.
NX210 administration did not significantly reduce the SCI lesion size
The extent of the SCI lesion was assessed by measuring the total area of injured or disrupted tissue, and also the GFAP-negative area within the bounds of the GFAP-positive astroglial border (Fig. 5A–Ai). Due to the inconsistent occurrence of an especially large cavity in one particular animal in the NX210 8 mg/kg treatment group, the total area measure exhibited large deviation within groups. The GFAP-negative area within the injury border, however, proved to be a more reliable measure. This analysis showed a nonsignificant reduction of the injury size (p = 0.259) 73 days post-SCI in mice treated with NX210 (8 mg/kg) (Fig. 5B). There was no effect of 4 and 16 mg/kg NX10 treatment on lesion size.
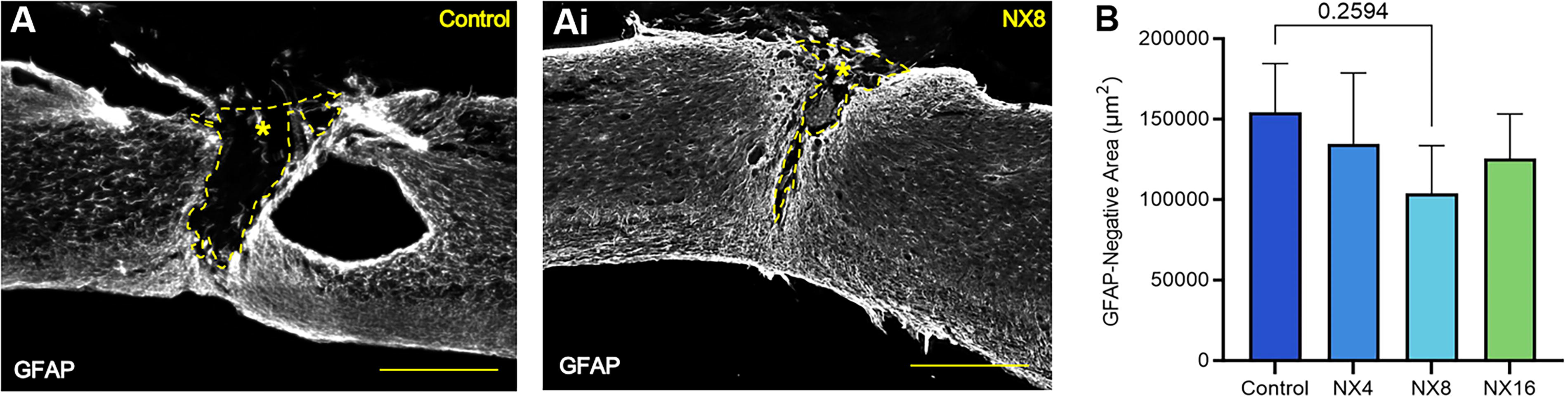
NX210 did not reduce SCI lesion size.
NX210 administration did not significantly reduce inflammatory cell presentation in the spinal cord penumbra after SCI
CD68 is highly expressed by cells in the monocyte lineage. After SCI, this marker is expressed by infiltrating monocytes, blood-borne macrophages, and microglia with phagocytic activity (Fig. 6A–Ai). Treatment of injured mice with NX210 led to a nonsignificant decrease in CD68 expression in the 8 mg/kg group (p = 0.19) but not for the two other treatment groups (Fig. 6B). This suggests NX210 does not significantly reduce the persistent pro-inflammatory response present in the spinal cord at 73 days post-injury.
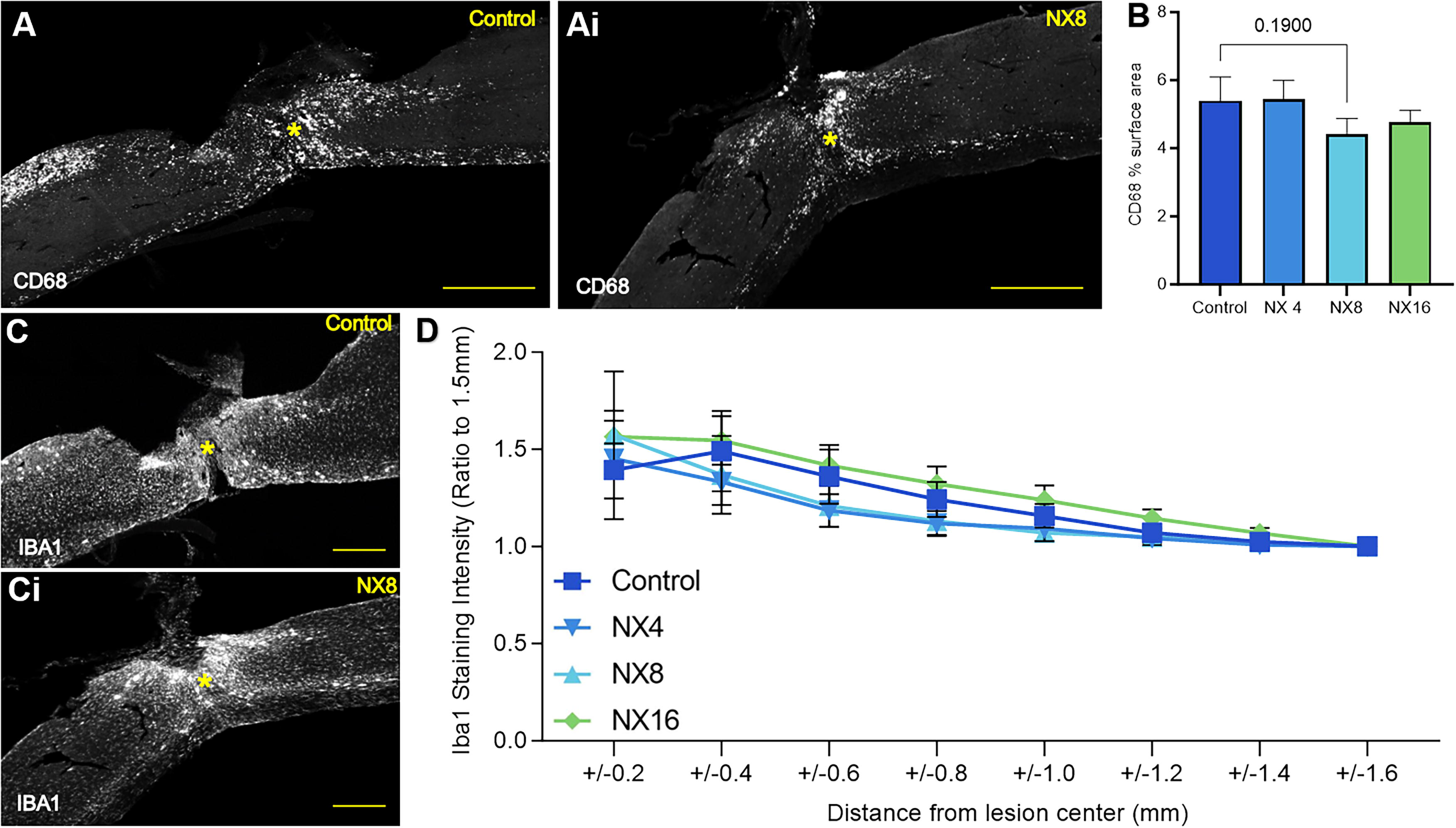
NX210 did not reduce inflammatory cell presentation in the spinal cord penumbra after SCI.
Iba1 is a marker expressed by resident microglia (Fig. 6C, Ci). Iba1 immunohistochemistry showed no difference at the lesion epicenter (±200 µm) for any of the NX210 treated groups compared to the vehicle group (Fig. 6D). No significant differences were observed at ±400–600–800μm in the 4 mg/kg (p = 0.5258, 0.3176 and 0.2969) and 8 mg/kg (p = 0.6831, 0.4135 and 0.3551) groups compared to the vehicle. The 16 mg/kg group followed closely the vehicle control group. This suggests microglial activity was not significantly reduced in the spinal cord at 10 weeks post-SCI for any of the NX210-treated groups.
NX210 administration did not significantly increase neuroprotection
Neuronal nuclear protein (NeuN) is a marker used to identify mature neurons. This marker was used to count the number of mature neurons in each segment moving away from the SCI lesion epicenter to identify levels of neuronal cell loss, and if this is prevented, or rescued, by NX210 administrations (Fig. 7A–Ai). The reduction of the number of NeuN-positive cells at and around the lesion center was not restored with NX210 treatment, regardless of the dose (Fig. 7B). At 600 μm from the lesion center, the 4 mg/kg dosage group showed a nonsignificant increase in NeuN-positive cells (p = 0.1898) compared to the control group while the 8 and 16 mg/kg groups did not show any difference (Fig. 7C). Interestingly this increase in NeuN expressing cells was visible out to 1000 µm both rostral and caudal to the lesion epicenter, though not statistically significant.
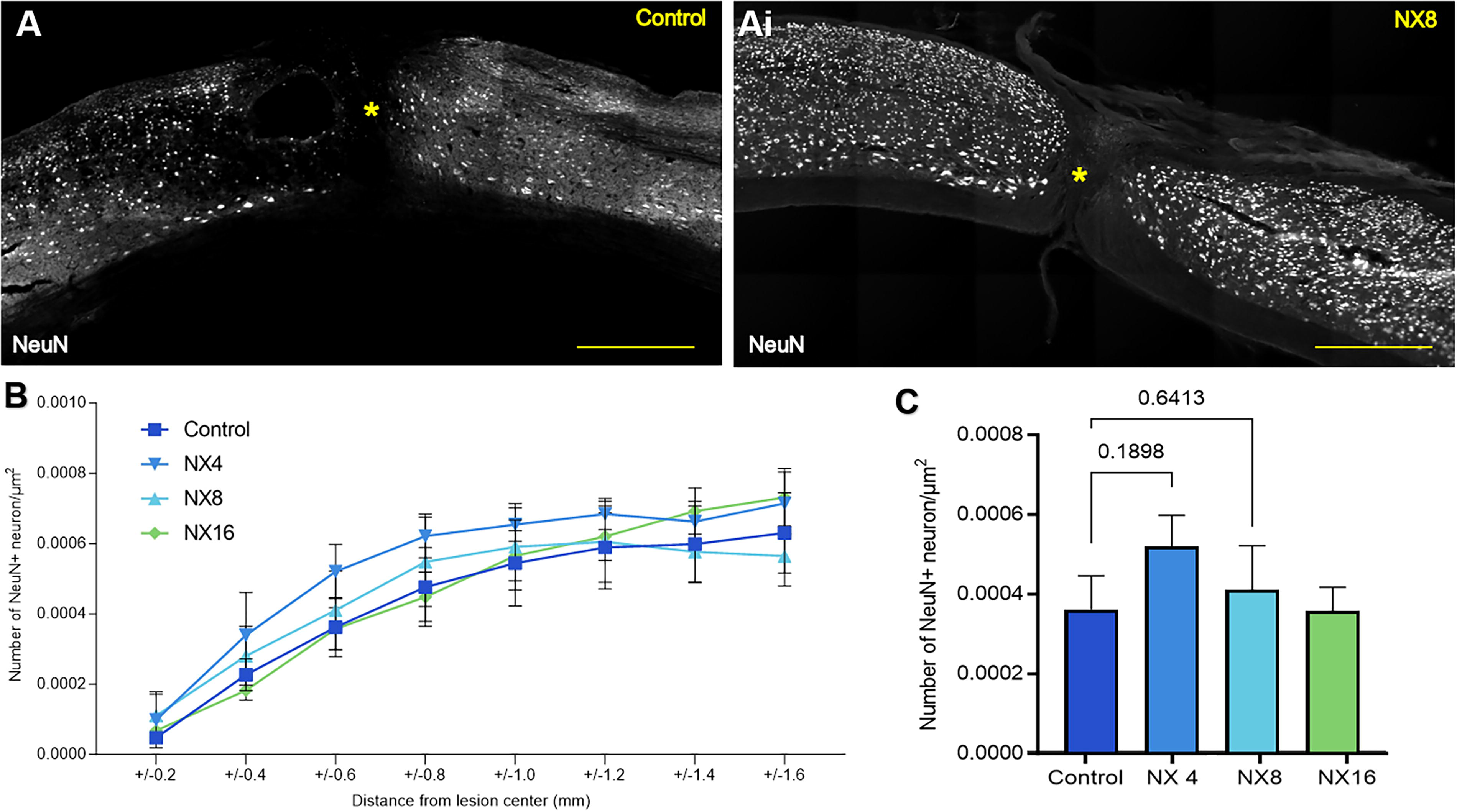
NX210 administration did not show neuroprotective effects at the spinal cord injury lesion.
NX210 administration increased the expression of MBP at the SCI lesion, and reduced NG2-positive cell number and distribution
MBP is a major component of the myelin found in the spinal cord and is associated with both the myelin sheaths around axons and the oligodendrocytes. MBP is lost to the extracellular environment after damage to the spinal white matter, and preservation of MBP levels indicates preservation of myelin integrity and oligodendrocyte numbers (Fig. 8A–B). Immunohistochemistry showed an increase in MBP expression at the lesion center with NX210 administration (8 mg/kg), with a significant increase in MBP expression at 200 and 400 µm (p = 0.024 and p = 0.030, respectively). Nonsignificant increases were observed for the 4 and 16 mg/kg groups at 200 µm (p = 0.122 and 0.106, respectively) and 400 µm (p = 0.193 and 0.109, respectively). MBP expression reached control levels in all the NX210 dosage groups by ±800 μm from the epicenter (Fig. 8C).
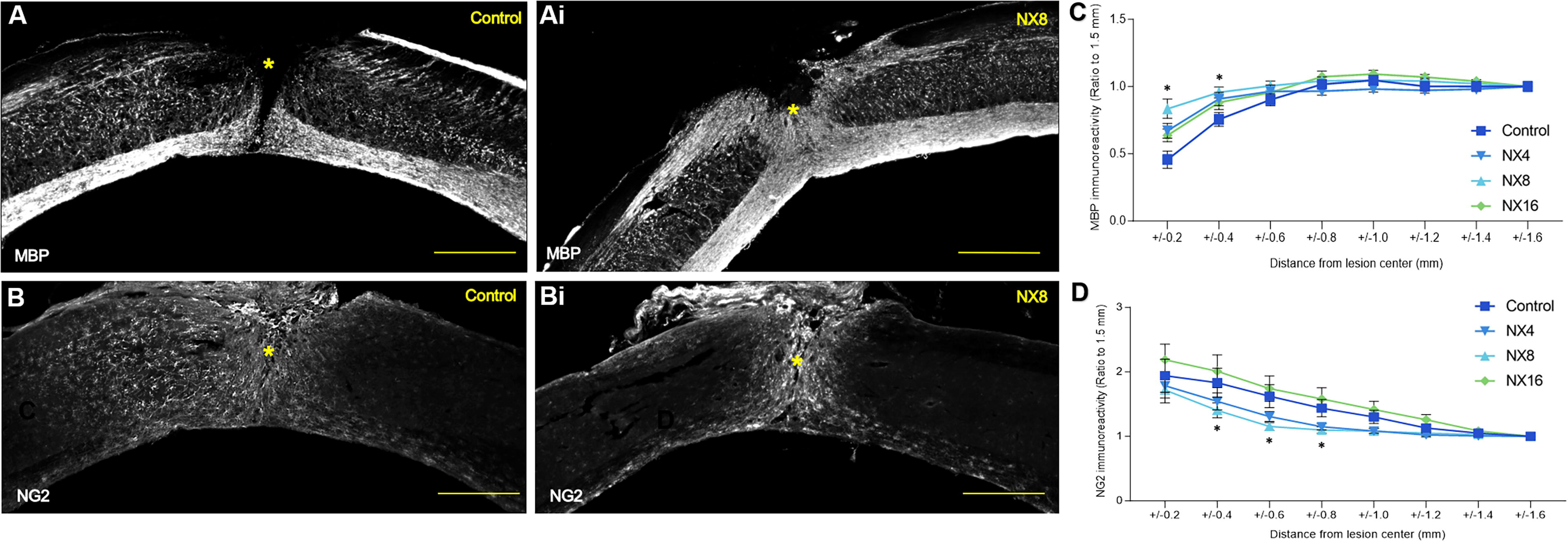
NX210 increased the expression of MBP at the SCI lesion, and altered NG2+ cell number and distribution.
NG2 is a proteoglycan expressed strongly by oligodendrocyte precursor cells (OPCs) and pericytes that contributes to the glial scar and can limit axon growth (Fig. 8A, B). i.p. administrations of NX210 reduced the expression of NG2, and the rostro–caudal spread of NG2-positive cells, in both the 4 and 8 mg/kg treatment groups. This was significant in the 8 mg/kg group at 400, 600, and 800 μm from the lesion center (Fig. 8D). There was a nonsignificant reduction in the 4 mg/kg (p = 0.341, p = 0.2117, and p = 0.1772 at 600, 800, and 1000 µm, respectively). This suggests that NX210 treatment reduces NG2 expression which can have positive effects on locomotor function, either through early OPC differentiation into mature oligodendrocytes or the modulation of the glial scar.
Discussion
Systemic administrations of the NX210 drug candidate peptide significantly increased recovery in the dorsal hemisection SCI mouse model. Improvements were seen in both locomotor recovery and pain sensitivity in SCI mice treated with NX210. These data concur with previous studies showing modest, but significant, effects on locomotor recovery with direct intraspinal injections of NX210 at the injury site, and immediately after a thoracic weight-drop contusion SCI model in rats. 26 The current study extends these findings, using repeated systemic administrations (i.p) of NX210, and demonstrates even more robust recovery with the sustained treatment paradigm. This is important, from a translational perspective, as it demonstrates that this pharmaceutical intervention can be safely and easily administered in the early stages of SCI, to provide long-term neuroprotective effects. As treatment in the current study was delayed until 4 h post-SCI, the data also highlight the potential of this intervention when administered within a clinically feasible treatment window. Due to the mode of administration and the delayed injection paradigms, NX210 could be translated into clinical applications in the future.
The beneficial effects of NX210 were largely seen in the first 2 weeks following injury, with recovery profiles plateauing in all groups, and across all behavioral tests of motor function, after day 21. Recovery was accompanied by an increase in MBP at the site of injury, with NX210 treatment. This increase may be indicative of a reduction in oligodendrocyte death, and a subsequent reduction of demyelination. It is also possible that administration of NX210 peptide could directly stimulate myelination through the activation of the β1-integrin receptor, which is involved in normal myelination in injury-naïve rodents, 36 leading to restoration of white matter tracts. Further work is needed to explore the role of NX210 on oligodendrocyte survival and the increase in myelination.
In contrast to MBP, NG2 was increased at the injury site after NX210 treatment. NG2 is a chondroitin sulfate proteoglycan (CSPG) found in the plasma membrane of diverse cell types in the developing and adult CNS. It is most commonly associated with subpopulations of glia, including OPCs, and pericytes. 37 In injury conditions, NG2 has been seen expressed on OPCs, pericytes, 37 mature oligodendrocytes, 38 astrocytes,39–41 and even macrophages.42,43 Due to the diversity of cells that express NG2, and their varied roles and functions after SCI, the reduction in NG2 immunohistochemistry with NX210 administration may have multiple interpretations. The first is a direct impact on the glial scar and reduced inhibitory signaling. The second is a direct effect on OPCs differentiation. Indeed, NG2-OPCs are a central component of the glial scar, 44 that rapidly respond to SCI with morphological changes and increased NG2 expression. 40 NG2 is known to inhibit axonal regeneration, 45 and is produced by both NG2-OPCs and astrocytes within the scar, 46 and neutralizing NG2 antibodies can improve recovery post-SCI.47,48 However, the role of NG2-OPCs themselves in regeneration is debated, as studies suggest NG2-OPCs may also promote axonal regrowth.38,49,50 Regardless, a reduction of NG2 expression by NX210 could potentially enhance axonal sprouting and plasticity. Furthermore, inhibiting NG2-OPCs results in a less dense, discontinuous glial scar, which, correlates with enhanced axonal regeneration.51,52 Therefore, NX210 may impact the glial scar in ways that beneficially promote locomotor function, which can be paralleled with the nonsignificant changes in lesion size and inflammation we observed. On the other end, NG2-positive glial cells respond to a CNS insult and can proliferate and differentiate into remyelinating oligodendrocytes to restore normal function.38,53,54 Therefore, the lower expression of NG2 observed in the 4 and 8 mg/kg NX210 groups may imply an increase in NG2-positive cells proliferation/differentiation early after SCI into mature oligodendrocytes. This early differentiation could explain the higher MBP expression in these groups at 73 days post-SCI. NX210 may directly promote OPCs differentiation by activating its β1-integrin receptor, a key player in the myelination process.55,56 Therefore, further work is needed to understand the alteration to NG2 expression observed, and how NX210 may promote NG2-positive glial cells proliferation and differentiation.
Along with the results of this study that show a preservation of MBP and reduction of NG2-positive cells, there was no significant increase in the number of neurons, no significant reduction of the lesion size, nor significant decrease in inflammation at the lesion site. However, while not statistically different individually, the decrease of each of these measurements may, combined, contribute to the strong locomotor recovery we observed. Previously, NX210 and NX210c peptides have been shown to reduce excitotoxic neuronal death induced by glutamate in vitro in rat cortical and hippocampal neurons, and human fetal cortical neurons 57 by binding to β1-integrin 58 and Notch receptors, 57 and activating the PI3K/Akt pathway. 57 The PI3K/Akt pathway contributes to neuron survival and axon regeneration after SCI31,59,60 and promoting neuronal survival after SCI is of critical importance to the preservation of neurological function following SCI.61–66 This also suggests that NX210 may enhance axonal growth or plasticity after SCI via the PI3K/Akt/mTOR pathway. In vitro, the effect of NX210 on neurite growth and fasciculation has been confirmed in a variety of neurons25,67–69 including human fetal cortical neurons. 57 In vivo, an increase in axon growth has been demonstrated in an aspiration model of dorsal funiculi in rats. 26 Finally, NX210c enhances synaptic transmission. 28 Altogether, it is therefore conceivable that NX210 and NX210c peptides may have a positive impact on neurons and locomotor recovery. More exploration is necessary to elucidate the mechanism(s) of action of NX210 on neurons after SCI and it remains to be demonstrated that similar mechanisms are involved in adult neurons in vitro and in vivo.
The effects of NX210 on astrocytes appear minimal in the current study, at least in terms of lesion size. In the CNS, astrocytes express β1-integrin70,71 and Notch receptors,72,73 and therefore, may be cellular targets and effectors of NX210 activity. In an in vivo Alzheimer’s model, daily NX210 injections for 11 days reduced astrocytic activation indicated by a decrease in GFAP expression levels.27,42 In addition, it has been shown that ROCK inhibition activates astrocytes and increases deposits of CSPGs. 74 Similarly, activation of the Notch pathway reduces astrocytic response. 72 Therefore, it is still of interest to further characterize how NX210 can potentially modulate reactive astrogliosis after SCI, through activation of Rho/ROCK and Notch, and eventually create a more permissive environment for axon growth and plasticity, and ultimately, contribute to improved functional recovery.
We did not observe a significant reduction in CD68 or Iba1 expression at 10 weeks post-SCI. However, NX210 has been shown to reduce inflammation in vivo in an Alzheimer’s disease model, as assessed by TNF-α pro-inflammatory cytokine levels. 27 In addition, microglia express β1-integrin 75 and Notch receptors, 76 which are involved in microglia migration and activation75,77 and may respond to NX210. Activation of Notch decreases pro-inflammatory cytokine secretion and increases beneficial phagocytic activity in microglia. 77 Increasing phagocytosis and an anti-inflammatory microglial phenotype is important in the acute stages of injury. 78 The phagocytosis activity and the inflammatory cytokine levels were not assessed in this study, and it remains possible that NX210 has an anti-inflammatory effect without changes in CD68 or Iba1 expression. In addition, sustained inflammation is associated with pain in chronic SCI, and inflammation reduction may diminish pain. This study demonstrated a reduction of pain in NX210 treatment groups. This potentially suggests that the reduction of inflammation with NX210 may reduce chronic SCI-associated pain. Alongside the reduction in SCI-induced pain hypersensitivity, general animal health (as indicated by body weight), hind limb locomotor function, and spontaneous locomotor activity were all also improved in animals treated with NX210. Therefore, further studies assessing how NX210 can modulate macrophage proliferation, differentiation, and functional polarization, including their secretion of pro- or anti-inflammatory molecules, as well as the temporal dynamics of these events, are necessary to fully comprehend how NX210 may impact inflammation.
In this study, the significant recovery in the groups treated with NX210 was accompanied by an increase in MBP and a reduction of NG2. However, no significant difference in terms of lesion size, inflammation, or neuronal survival was observed. Histological lesion size has been widely considered as a correlate or reliable predictor of functional outcomes in experimental SCI models, however, in reality, comparably sized lesions can result in very different presentations and magnitude of both initial functional deficits as well as ongoing recovery.79,80 This is due to the complexity of neuroanatomy with factors such as variations in the cells, tracts, or areas directly affected, and those spared, 80 and the level of intrinsic plasticity in the spinal cord, having significant functional implications. The progression of the secondary injury can also have a significant impact, especially the inflammatory response 81 and its impact on neuroprotection or degradation, and axon sparing/growth. 82 In this study, we did not examine the effect of i.p administrations of NX210 on the sparing of axonal tracts, axon sprouting or regeneration, or synaptic plasticity. Any of these factors, or a combination thereof, could contribute to the improved functional outcomes observed without observing a correlating decrease in the histological lesion size and so bear scrutiny.
Overall, whether it is through neuroprotection, improved tissue regeneration and repair, or a combination of both, NX210 is a promising therapeutic intervention in improving outcomes after SCI. Several gaps in knowledge should be addressed in future studies. First, it remains to be determined if NX210 can enhance locomotor functions independently of age and sex, as both factors can impact drug efficacy. Indeed, age and sex can modulate neuron-intrinsic83,84 and extrinsic 14 factors involved in repair and regeneration, even in vitro, 85 which may modulate the efficacy of NX210. Second, further examination of the duration of treatment will determine if a short administration window is enough to increase locomotion. Third, it will be important to assess the effects of an altered treatment schedule in a contusion model, more clinically relevant than the dorsal hemisection used here. Finally, it is important to note that the NX210 peptide takes a cyclic conformation called cyclic NX210 (NX210c), after oxidation and the formation of a disulfide bridge between two cysteines in the peptide sequence. In plasma, only this cyclic form can be measured and may therefore represent the active metabolite of NX210. After initial development of the linear form of NX210, tested in nonclinical settings and a clinical phase 1 healthy volunteer study 29 ) NX210c was also recently tested in phase 1 30 and now a phase 2 clinical trial for amyotrophic lateral sclerosis is ongoing with NX210c (NCT06365216). NX210c has been granted an Orphan Drug Designation in SCI by the European Medicines Agency. It remains to be determined if NX210c can promote functional recovery after SCI to a level similar to NX210.
Altogether, these data demonstrate the therapeutic potential of repeated i.p. administrations of NX210 to protect spinal cord tissue, promote functional recovery, and alleviate pain hypersensitivity after SCI.
Authors’ Contributions
T.C.S.: Investigation, formal analysis, writing—original draft, review and editing, and visualization. S.L.: Writing—review and editing, and visualization. L.K., A.J.D., D.H., A.S., S.M.J., A.H.L., Z.H., N.B., and R.D.: Investigation. M.H.: Investigation, methodology, resources, and writing—review and editing. Y.G.: Conceptualization, resources, writing—review and editing, and funding acquisition. C.G.G.: Conceptualization, investigation, formal analysis, methodology, resources, project administration, supervision, writing—review and editing, visualization, and funding acquisition.
Footnotes
Acknowledgment
The authors would like to thank the Department of Neuroscience and Experimental Therapeutics Behavioral Core for providing support and access to perform BMS, Rotarod, and Activity Chamber procedures.
Author Disclosure Statement
S.L. is employed by Axoltis Pharma. Y.G. is President of Godfrin Life-Sciences, giving advice to Axoltis Pharma, and is also Chief Executive Officer and a shareholder of Axoltis Pharma.
Funding Information
This work was partially funded by the Bryon Riesch Paralysis Foundation (M2002812).
