Abstract
Medication-use evaluations are meant to ensure that medication-use processes are consistent with prevailing standards of care, assure optimal use of therapy, and reduce the risk of medication-related problems. Reversal agents for direct oral anticoagulants are a worthy focus for medication-use evaluations for reasons of efficacy, safety, and cost. A multidisciplinary team of experts developed 2 medication-use evaluation templates illustrating the application of professional society guidelines to the appropriate use of andexanet alfa.
Keywords
Background
One of the US FDA’s primary charges is the process of medication approval wherein a multidisciplinary team of experts weighs evidence of benefit vs risk in the treatment of specific health conditions. There is growing awareness of discordance, however, between outcomes observed in tightly controlled clinical trials vs those from the real-world setting, where health care providers often must contend with patients who often do not conform to strictly defined trial inclusion criteria. Sometimes this gap is addressed through the evaluation of newly published evidence and consensus guidelines developed by a group of experts. Despite these efforts, health care providers sometimes utilize medications for indications or in situations where evidence is limited or the benefit is not clear. While the decision to deviate from the evidence may seem well-reasoned, these variances should be evaluated for efficacy, safety, or cost outcomes relative to prevailing standards of care.
A medication use evaluation (MUE) is a performance improvement tool aimed at improving therapeutic or process outcomes. The objectives of a MUE include: ensuring medication use practice comports with existing standards of care, improving optimal use of therapy, and reducing medication-related problems. 1
Recently updated guidelines for MUE development outline prioritization of medications meriting evaluation and delineation of ways a MUE’s stated objectives may be incorporated into a quality improvement framework. The guidelines recommend using the FOCUS-PDCA model, which involves identifying a process to target and constituting a team of experts qualified to outline what is presently known about the process and causes of variation within it. The model then describes initiating a continuous cycle of designing a change, analyzing the results of enacting it, modifying the process to yield improved results, and then beginning the cycle anew. 1
Reasonable foci for a MUE include medications associated with significant or numerous adverse outcomes or those that incur a significant cost, both of which make antithrombotic reversal agents natural targets. Interest in real-world experience and institutional policies governing the use of antithrombotic agents spans decades2,3 due to the risk of catastrophic outcomes associated with the medication class. 4 Commensurate with changes in prescribing practice, attention has begun to focus on direct oral anticoagulants (DOAC), the factor Xa inhibitors (FXais) in particular. Warfarin use has dropped 39% in US Medicare and Medicaid beneficiaries over recent years, while apixaban and rivaroxaban now account for 46% of oral anticoagulants prescribed. 5 The risk of clinically important bleeding-related outcomes with oral FXais may compare favorably with those of warfarin in non-valvular atrial fibrillation (AF) or venous thromboembolism,6,7 but 2 to 5 DOAC-treated patients with AF per 1000 experience intracranial hemorrhage (ICH) annually, 8 and major gastrointestinal bleeding occurs at an annual rate of 0.76%. 9 Case-fatality rates in DOAC-associated major bleeding are 8% to 15%, 10 and related hospitalization costs run into the tens of thousands of dollars. 11
The literature focusing on the real-world use, outcomes, and costs of DOAC reversal agents is more limited. Andexanet alfa was approved by the FDA in 2018 for reversal of anticoagulation in patients treated with rivaroxaban or apixaban who are experiencing uncontrolled or life-threatening bleeding. Prior to its approval, clinicians managed FXai-associated major bleeding with packed red blood cells, fresh frozen plasma, or prothrombin complex concentrate (PCC),12,13 and they continue to use these products off-label for the same indication today. 14
Two leading quality assurance and patient safety organizations—the Joint Commission and the Institute for Safe Medication Practices—direct health systems to make anticoagulant reversal agents readily available and develop evidence-based protocols governing their use.15,16 Several professional society guidelines and expert panels, such as those from the American College of Cardiology, American Heart Association, and Anticoagulation Forum, favor using a specific reversal agent over a non-specific pro-hemostatic agent for reversal of DOAC-associated major or life-threatening bleeding when available.17–20 However, a recent evaluation of US hospitals providing emergency care shows that fewer than 12% carry andexanet alfa compared to 60% with idarucizumab for reversal of anticoagulation in dabigatran-related bleeding, 21 despite dabigatran’s accounting for a small fraction of oral anticoagulants prescribed in the US. 5 Another survey found 66% percent of hospitals that had not added andexanet alfa to formulary cited cost as a reason. 19
Ongoing questions about the balance of andexanet alfa’s efficacy relative to non-specific pro-hemostatic agents, safety, and cost12,14,19 make andexanet alfa an ideal drug for targeted MUE. To address practical considerations around appropriate use of andexanet alfa, the authors of this paper—a team of pharmacists and physicians who practice in emergency medicine, hematology, and antithrombosis stewardship—formed a multidisciplinary Advisory Panel (AP) that also included a finance representative and represented institutions with and without andexanet alfa on formulary.
As a MUE is an institution-specific performance improvement tool and the AP’s members represented several different health systems across the country, the panel did not conduct a MUE directly. Rather, during 9 meetings over the course of a 4-month period, the committee developed 2 separate MUE templates each addressing a different step of the medication-use process. The AP chose the prescribing step for 1 MUE and dispensing for the other, weighing factors that included the relative priority of health-system needs relating to each step and an interest in illustrating the MUE planning process to the broadest possible audiences: prescribing for clinical staff and dispensing for those involved in the medication distribution process (eg, pharmacy staff). The AP also created a resource guide 22 meant to provide customizable, turnkey templates that may be adapted by health systems locally during MUE implementation.
Medication Use Domains
Approach, Areas of Focus, and Rationale
The first steps in the process are to identify a process to target ( Medication-use evaluation process flowchart: direct oral anticoagulant reversal agentsa.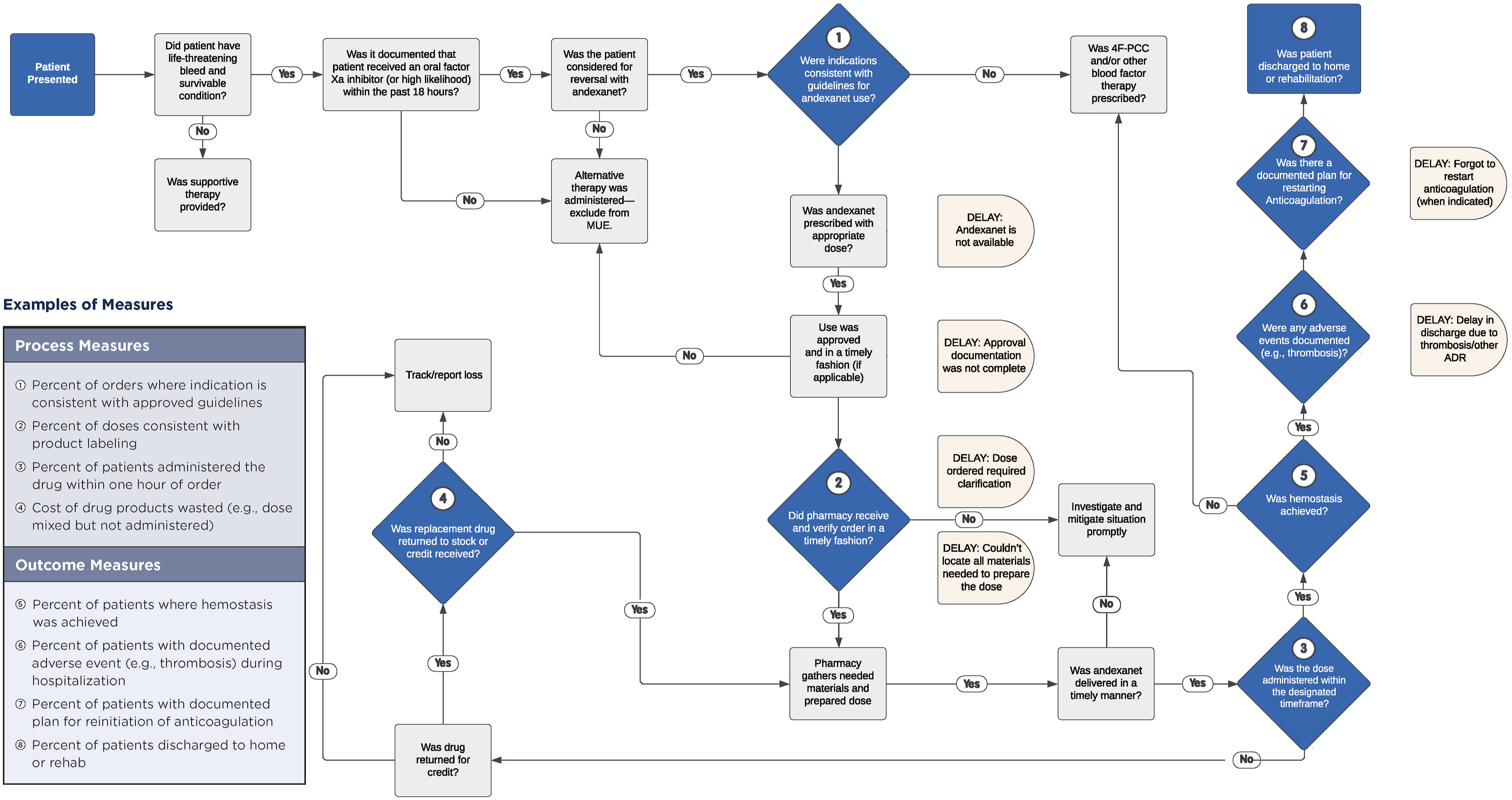
a
bThis is an example set of MUE criteria. Each institution should establish guidelines and MUE criteria appropriate to the setting.
cThe MUE template and guidelines assume that andexanet alfa is available for administration.
dFor assessment of bleeding and definition of life-threatening bleeding, see: Tomaselli GF, Mahaffey KW, Cuker A, et al, 2020 ACC expert consensus decision pathway on management of bleeding in patients on oral anticoagulants: A report of the American College of Cardiology Solution Set Oversight Committee. J Am Coll Cardiol. 2020;76 (5):594-622. doi:10.1016/j.jacc.2020.04.053.
eAssays specific for apixaban and rivaroxaban quantitation are specialized and are not widely available. Assays that are calibrated for each agent may not be available or the results reported in a time period that is useful in treatment decisions. For additional information, see: Levy JH, Ageno W, Chan NC, et al. When and how to use antidotes for the reversal of direct oral anticoagulants: guidance from the SSC of the ISTH. J Thromb Haemost. 2016;14:623-7.
a
bThis is an example set of MUE criteria; each institution should establish guidelines and MUE criteria (eg, parameters for timeliness) appropriate to the setting.
cThe MUE template and guidelines assume that andexanet alfa is available for administration.
Enacting Process Improvement
The latter half of the FOCUS-PDCA model outlines incorporating a MUE’s findings into a cycle of quality improvement to address causes of variation. Questions concerning andexanet alfa use might include: ❖ Has an institution discovered through its MUE process that inappropriate patient candidates were selected for reversal? Potential solutions: ➢ Order sets can be built to include prompts for required criteria (eg, imaging tests) ➢ Expert panels emphasize the importance of clinical decision support tools and standardized electronic order sets embedded with the most up-to-date evidence to identify the best candidates and strategies for anticoagulant reversal18,19 (eg, Glasgow coma scale, ICH score) ❖ Has an institution discovered through its MUE process that incorrect doses of andexanet alfa have been administered or that it has been lost in storage or transit? Potential solutions: ➢ Box kits for preparation by the nurse or pharmacist at the bedside, as has been done with other reversal agents
19
➢ Inventory control measures (eg, RFI technology) or a strong process in place to ensure the medication is returned if not administered.
Considerations and Limitations
A MUE has its share of limitations. It is a tool to identify process variation, design solutions, and measure change. While a well-designed MUE may fulfill the need for real-world data, unlike clinical trials, most MUEs are not designed to establish causality, and, therefore, caution must be used when interpreting and extrapolating the results. For example, the templates developed during this project were not designed to measure factors like the extent to which a reversal agent impacted a patient’s clinical course relative to the patient’s baseline prognostic status, optimal provision of supportive care measures like blood pressure management, and impacts of delays in reinitiating anticoagulation therapy.
Manual chart review should be minimized, as it is time-consuming, resource-intensive, and prone to errors of omission and transcription. MUE guidelines therefore recommend automating the data collection process as much as possible. 1 The AP’s resource guide 22 provides a sample data collection framework.
Finally, those conducting MUEs may encounter several barriers, including a failure to: engage experts in the areas affected; use consistent sources of data; obtain baseline measurements; or follow through with a continuous cycle of change and further analysis that the changes have resulted in an improvement. The MUE guidelines and AP resource guide provide potential solutions to those and other barriers to the effective execution of an MUE. 1
Summary
A multidisciplinary team of experts developed 2 MUE templates illustrating the application of MUE guidelines to the appropriate use of andexanet alfa to serve as a customizable model for health systems interested in evaluating the same question or other medication-use processes locally.
Footnotes
Author’s Note
Jessica Grandoni is also affiliated with the Anticoagulation Management Service, Dana-Farber Cancer Institute, Boston, MA, USA. The First Affiliated Department of Pharmacy, Brigham and Women’s Hospital, Boston, MA, USA.
Acknowledgments
The authors acknowledge the assistance of the ASHP Foundation and of Belinda Lovelace, PharmD, MS, and Terry Dettling, BSN, JD, MPH, MS, both of Alexion, AstraZeneca Rare Disease, at the time of manuscript preparation.
Author Contributions
The following authors reviewed and provided feedback on the manuscript and actively contributed on the advisory panel that developed the medication use evaluations (MUEs) and its implementation guide: Drs. Burnett, Fanikos, Grandoni, Pollack, Rose, Spinler, and Streiff. Dr Fera served as project manager for the advisory panel, prepared the initial drafts of the MUEs and its implementation guide, and incorporated the advisory panel’s feedback into the submitted drafts of each. Ms. Moore actively contributed on the advisory panel in the development of the MUEs and its associated implementation guide. Dr Nussbaum facilitated advisory panel meetings with Dr Fera and reviewed and provided feedback on drafts of the manuscript, MUEs, and implementation guide. Dr Turck prepared the initial draft of the manuscript, incorporated the advisory panel’s feedback into the submitted draft, and reviewed and provided feedback on drafts of the MUEs and implementation guide.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Fanikos reports paid compensation for work on advisory boards for Boehringer Ingelheim International and Vifor, Inc. Dr Fera has received compensation from ASHP for contract work. Drs. Grandoni and Streiff report paid compensation for work on advisory boards for Pfizer Inc. Dr Nussbaum is an employee of the ASHP Foundation. Dr Pollack has received payment for consulting work with AstraZeneca, eXlthera Pharma, PhaseBio Pharma, and Provepharm. All authors report having received honoraria for participating in the meetings whose discussions formed the basis of this manuscript, which was funded by Alexion Pharmaceuticals, Inc. The authors declare no other potential conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The development of this manuscript was funded by Alexion, AstraZeneca Rare Disease.


