Abstract
Introduction
Neuromuscular blocking agents (NMBA) are often administered to patients suffering from a traumatic brain injury (TBI) or spontaneous neurologic injury to facilitate airway management. Unfortunately, the urgent nature of intubation can preclude completion of an accurate neurologic examination prior to administration of the NMBA. The duration of paralysis can impede clinician’s ability to make timely decisions about prognosis and potential management strategies including placement of external ventricular drain (EVD) systems or hematoma evacuation with craniectomy or craniotomy.
Historically, the only reversal agent available for reversal of NMBAs was neostigmine, which comes with several disadvantages including delayed or incomplete reversal, bradycardia, and nausea or vomiting.1,2 Sugammadex was FDA approved in 2015 for the reversal of amino-steroid paralytic agents such as rocuronium and vecuronium. The medication is available in 200 mg ($138.50) and 500 mg ($253.70) vials. 3 Full dose rocuronium can be reversed with sugammadex at a dose of 16 mg/kg if urgent reversal is needed within ∼3 minutes, or a lower dose of 4 mg/kg can be considered if 0 to 2 twitches are present on train-of-four (TOF) monitoring.4-6 Prior to 2018, the institution limited the use of sugammadex to the post-operative setting, failed intubations in the emergency department or intensive care units, or successful intubations of asthmatic patients with poor ventilation. In October 2018, the neurocritical care team at the institution developed a guideline for administering sugammadex to facilitate neurologic examination after a neurologic injury. This case series describes the use of sugammadex for neurologic examinations as a result of a quality improvement analysis after the implementation of a guideline.
Materials and Methods
A list of all adult patients admitted to an intensive care unit or the emergency department at a level 1 trauma center who received sugammadex orders for neurological examination from October 2018 to March 2020 was obtained. Patient records were reviewed for adherence to the guideline and outcomes following the implementation of the guideline. The criteria for use of sugammadex included the following: an uncertain neurologic examination prior to NMBA administration, a computed tomography (CT) head showing an emergent neurological injury, and it was deemed that an accurate post-intubation examination was urgently needed to determine if neurosurgical intervention was indicated. The institutional policy developed to facilitate neurologic examination allowed for alternative dosing depending on TOF assessment and/or time from NMBA administration.4,7-9 If feasible, it was recommended that a TOF be obtained prior to sugammadex administration to guide dosing. de Boer et al found an average time to recovery after a rocuronium dose of 1.2 mg/kg to be 122 minutes. Based on these findings, the guideline suggested the use of sugammadex for patients who received rocuronium within the prior 2 hours. 4 An initial upfront dose of sugammadex without obtaining a TOF was allowed due to the variability in user capability outside the OR and the delay in obtaining equipment. Providers had the option to obtain a TOF if there remained any uncertainty after the initial sugammadex administration. Patient records were accessed to obtain the patient location, weight, mechanism of injury, documentation of NMBA (agent, time administered, and dose), documentation of sugammadex (time administered and dose), any record of TOF documented prior to sugammadex administration, recorded Glasgow Coma Scores (GCS), and confirmation that the indication was for neurologic examination and the outcomes after giving sugammadex. Patients without chart notes connecting sugammadex administration with neurologic status changes and/or clinical decisions were reviewed and ultimately categorized as cases where sugammadex did not clearly contribute to decision making. Descriptive statistics were used to assess outcomes. This research adhered to all ethical guidelines and, as a continuous quality improvement project, was exempt from Institutional Review Board assessment at this institution. Formal consent was not required for this study type.
Results
Characteristics and Outcomes of Patients.
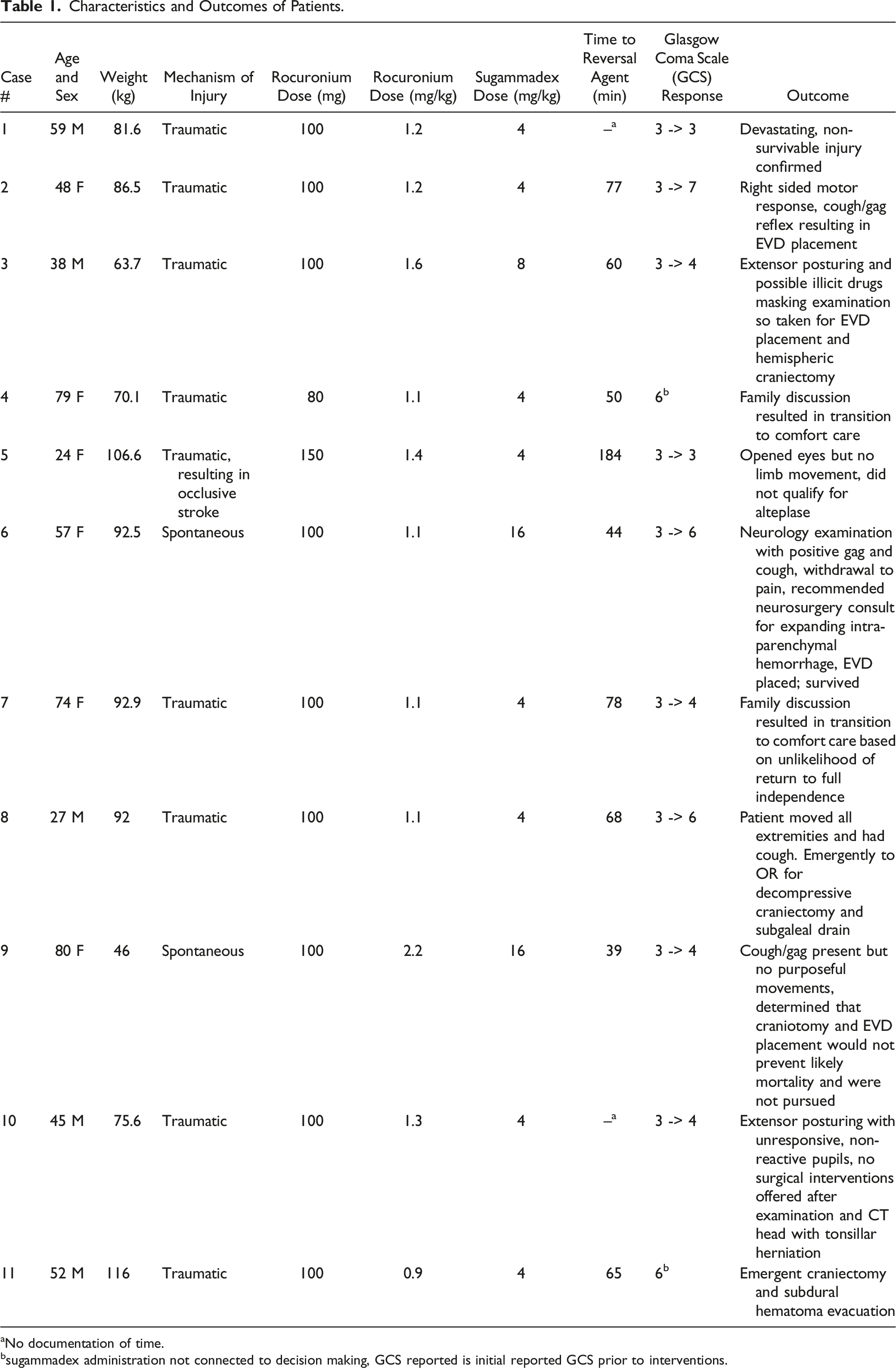
aNo documentation of time.
bsugammadex administration not connected to decision making, GCS reported is initial reported GCS prior to interventions.
Discussion
The institutional policy developed to facilitate neurologic examination allowed for alternative dosing depending on TOF assessment and/or time from NMBA administration.4,7-9 Despite a mean rocuronium dose of 1.3 mg/kg, lower dose sugammadex administration facilitated clinical decision making for most patients. Of those that received a lower dose strategy of 4 mg/kg (72.7% of patients), 5 of the 8 had a documented change in exam. This lower dosing is consistent with a recently published case report on the use of sugammadex to reverse rocuronium 1 hour after administration in a patient with end-stage renal disease and a TOF of 0 out of 4. 10 In addition to facilitating clinical decisions, the lower dosing strategy allowed for cost avoidance when compared to a moderate or high reversal dose strategy (eg, for a 70 kg patient using 200 mg vials, 4 mg/kg = $277, 8 mg/kg = $415.50, 16 mg/kg = $831).
Although rocuronium’s duration of action is expected to be 30-60 minutes, several reports of a longer duration of action have been documented.10-12 The mean time between rocuronium and sugammadex administration was 73.9 ± 41 minutes, suggesting bedside clinicians believed these patients had a longer than expected paralysis. Higher weight-based rocuronium doses may have contributed to a longer duration of paralytic effect. 13 The previously established safety and reliability of sugammadex makes it an ideal agent when clinical assessment may be needed after administration of paralyzing agents. This quality improvement analysis provides information about potential dosing, timing of administration, and effectiveness of the use of sugammadex in the setting of reversing NMBA to facilitate neurologic examination. Although the retrospective design of this quality improvement project limits the ability to assess adverse events as a result of sugammadex administration, none were identified in review of clinical documentation. To the author’s knowledge, this case series and the recent case report by Curley and colleagues 10 are the first to report on the use of sugammadex in this patient population. In other settings, lower doses of sugammadex have been found to be adequate to reverse rocuronium. 14 However, in the operating room, the degree of neuromuscular blockade is measured by post-tetanic count (PTC) and TOF in a controlled setting, at regular intervals, and by personnel trained to perform the assessments. For reversal to facilitate examination, it may not always be practical to perform PTC/TOF monitoring and therefore the dose of NMBA and time from NMBA administration may be useful to determine initial dosing for sugammadex. Due to the time sensitive nature of completing exams in the emergency department, it was common practice to administer sugammadex without TOF. Therefore, it cannot be ruled out that the paralytic effect had subsided. However, clinicians were making decisions at bedside based on high suspicion that the paralytic effect could be contributing. Based on the findings of this case series, further study could explore the use of lower dosing strategies in a prospective manner.
Limitations
Limitations of this report include the small patient population and all patients receiving rocuronium for intubation. The institutional policy suggested TOF monitoring prior to sugammadex to assist with dosing determination, but 81.8% of patients did not have a TOF documented. Therefore, inadequate dosing could have been possible. However, the 2 patients with TOF monitoring had 0 out of 4 twitches and received 4 mg/kg of sugammadex. These 2 patients had a change in neurologic examination after sugammadex administration, which guided clinical decisions suggesting the lower dose strategy was adequate. If there was ongoing concern about an incomplete NMBA reversal, TOF monitoring was available for confirmation after sugammadex administration. No patients received multiple doses of sugammadex. The majority (72.7%) had a change in neurologic examination (change in GCS, return of reflexes) which was the outcome of interest. For the patients who did not have a change in neurologic examination reported (27.3%), 1 had no return of reflexes consistent with the devastating brain injury seen on imaging, 1 had no comment on the examination as the patient was made comfort care irrespective of sugammadex administration, and 1 went straight to the operating room for neurosurgical intervention.
Conclusion
This case series suggests that sugammadex is useful for reversing rocuronium induced paralysis to facilitate completion of a neurologic examination that is needed to determine management strategies for patients who suffered from a neurologic injury.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
