Abstract
Introduction
Dexmedetomidine is a highly selective, centrally acting alpha 2 agonist with anxiolytic, sedative, and analgesic effects. 1 It has emerged as a desirable sedative agent in pediatric intensive care due to its minimal effect on ventilatory drive and reduction in delirium when compared to benzodiazepines.1,2 Due to its sympatholytic effects, common side effects include bradycardia and hypotension. Studies report a variable incidence of bradycardia between 0-42% among patients receiving dexmedetomidine.3,4 There are emerging case reports of more serious cardiovascular events, including sinus arrest and asystole, in the pediatric population.5-11 Most of these case reports have been attributed to high vagal tone, rapid medication infusion, concurrent administration of a negative chronotropic agent, or underlying cardiac conduction dysfunction. All reported events were preceded by severe bradycardia or were associated with a clinical event precipitating a strong vagal response (eg bearing down, stooling, agitation, hypoxia, stimulation of the carina). We present a case of a pediatric patient who developed multiple episodes of sinus arrest associated with a dexmedetomidine infusion without clinical features associated with high vagal tone or intervening bradycardia between events in the pediatric intensive care unit (PICU).
Case Presentation
An 11 month-old previously healthy female presented in acute hypoxemic respiratory failure secondary to Adenovirus bronchiolitis requiring mechanical ventilation. To facilitate sedation, a dexmedetomidine infusion was initiated at .5 mcg/kg/hr and increased to a maximum dose of 1 mcg/kg/hr. During this time, there was a decline in average heart rate from 175 to 135 but was still well within a normal range for age (Figure 1). Timeline reviewing clinical course including medications administered and sinus pause events.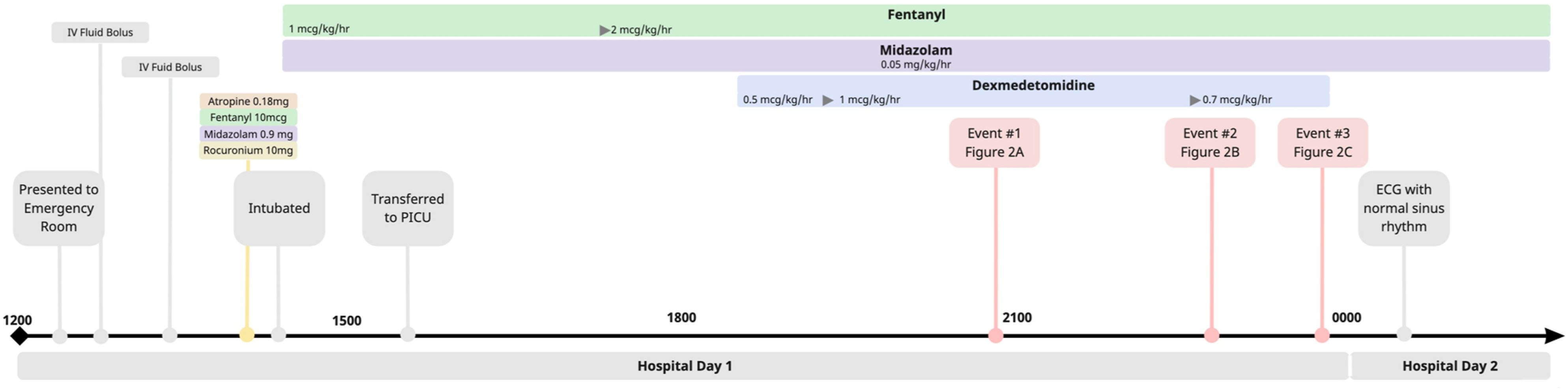
Within 8 hours of initiating therapy, she was noted to have three episodes of sinus arrest. The first episode was 18 seconds with resumption of junctional and then sinus rhythm. The second arrest was 9 seconds with a single junctional escape beat followed by another 13 second pause, with eventual resumption of a regular junctional escape and then normal sinus rhythm. A third episode was 10 seconds in duration. These episodes occurred spontaneously in the setting of an intervening sinus rate >120 bpm throughout (Figure 2). Dexmedetomidine was running at 1 mcg/kg/hr during the first two episodes and .7 mcg/kg/hr during the third. Each episode self-resolved prior to initiation of cardiopulmonary resuscitation. Several hours after the events, the patient required low-dose epinephrine for ∼24 hours for mild hypotension attributed to compensated septic shock; of note, this hypotension was not accompanied by bradycardia or other rhythm disturbance. After discontinuation of dexmedetomidine, there was no reoccurrence of sinus arrest. Displays the ECG and SpO2 tracing during each of the three episodes of sinus arrest (A-C). Note that there is mild bradycardia prior to sinus arrest in the first event, but a normal sinus rate preceding the latter two. The patient did not have any episodes of bradycardia <110 beats per minute in the intervening period between any of these episodes.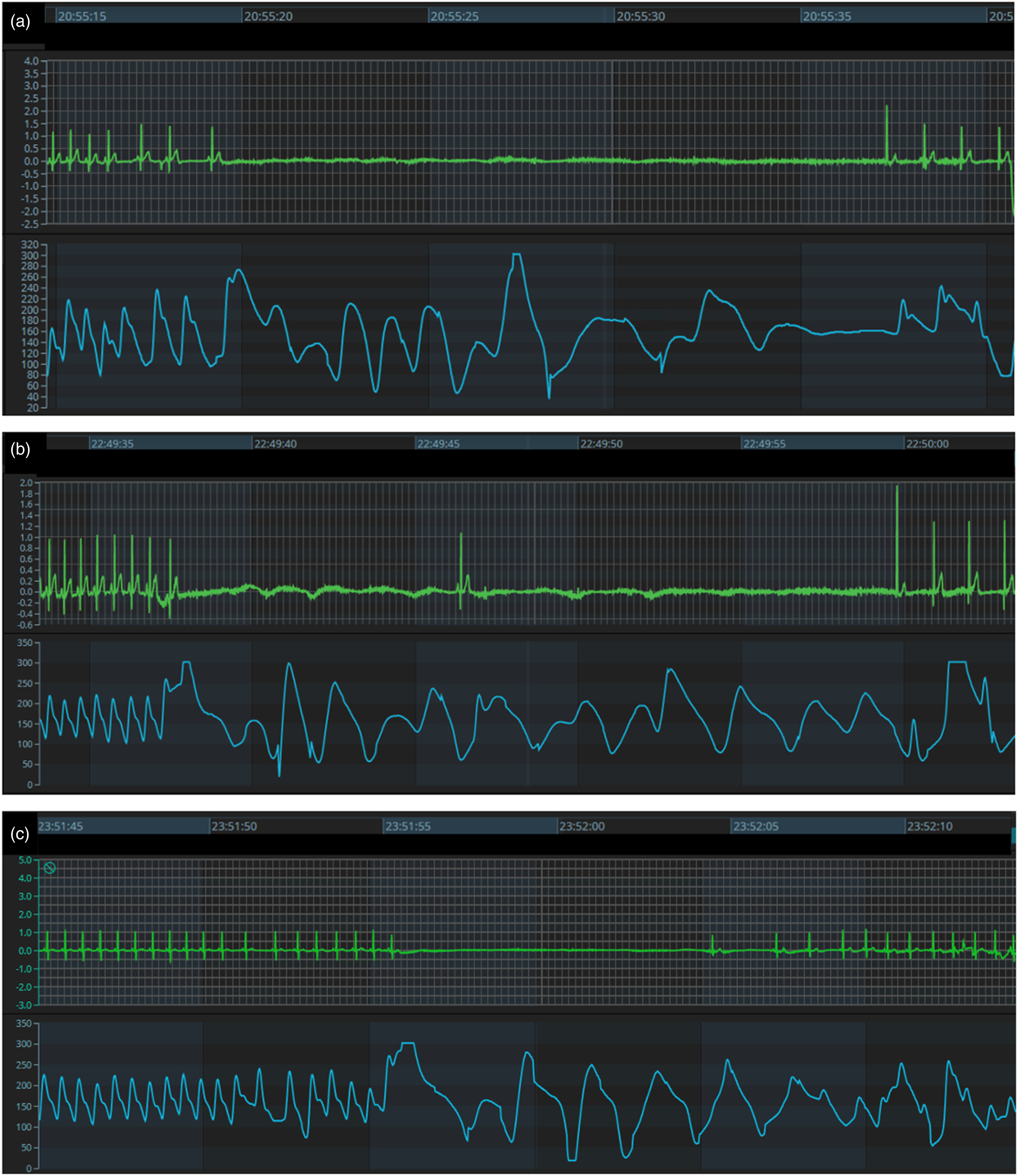
These events occurred with the patient comfortably sedated and without clinical features usually associated with high vagal tone such as increased agitation, ventilator dyssynchrony, hypoxia, coughing, or straining with bowel movement. A chest radiograph was obtained and confirmed placement of endotracheal tube within the upper thoracic trachea, and the patient had not received endotracheal tube suctioning prior to any episode, making carina stimulation an unlikely contributor. Dexmedetomidine was co-administered with fentanyl, midazolam and ceftriaxone, but the patient did not receive corticosteroids or other medications associated with negative chronotropic effects. Laboratory evaluation immediately following the events revealed electrolytes (potassium, magnesium, calcium) within normal limits. An echocardiogram demonstrated normal biventricular systolic function and structural anatomy. She had no other known risk factors for sinus node dysfunction, including congenital heart disease, known cardiac conduction deficits, heterotaxy, family history of cardiac disease, or signs of viral myocarditis.
The remainder of her hospitalization was remarkable for sepsis secondary to
Discussion
We report an infant who experienced three prolonged but self-resolving episodes of sinus arrest while receiving dexmedetomidine for sedation, without recurrence after discontinuation. The case is unique given the duration and characteristics of the episodes. Although the episodes are reminiscent of elevated vagal tone, there was no evidence of sinus bradycardia in between the episodes nor any common clinical precipitants of vagal excess which are commonly reported.
The mechanism by which dexmedetomidine causes sinus and AV node dysfunction remains uncertain. Literature shows dexmedetomidine significantly depresses the sinus and AV node function in pediatric patients as evidenced by an increase in sinus cycle length, sinus node recovery time, PR interval, and Wenckebach cycle length. 12 A recent meta-analysis of side effects in patients receiving dexmedetomidine for ≥24 hours reported a pooled prevalence of bradycardia at 2.6% and hypotension at 6.1%; the authors defined these side effects as hemodynamic changes that required intervention. 4 When bradycardia or hypotension occurs, it is usually within 12 hours of initiation and are reversible with discontinuation of the drug. 4
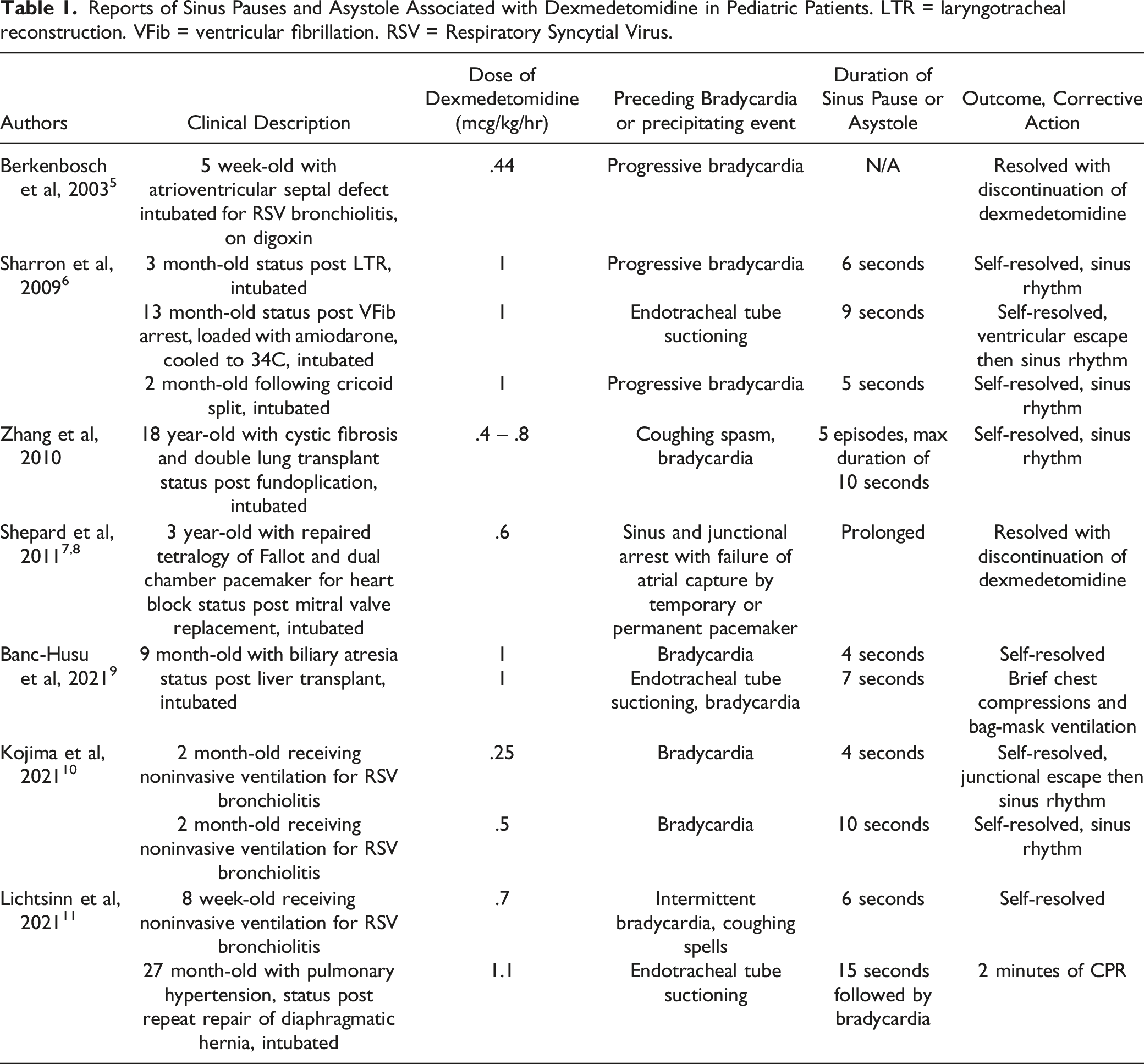
Reports of Sinus Pauses and Asystole Associated with Dexmedetomidine in Pediatric Patients. LTR = laryngotracheal reconstruction. VFib = ventricular fibrillation. RSV = Respiratory Syncytial Virus.
Dexmedetomidine received FDA approval for sedation in 1999, and since that time has become widely used in adult and pediatric critical care. The Clinical Practice Guidelines for the Prevention and Management of Pain, Agitation/Sedation, Delirium, Immobility, and Sleep Disruption in Adult Patients in the ICU suggest using propofol or dexmedetomidine over benzodiazepines for sedation in critically ill, mechanically ventilated adults due to a shortened time to extubation. 13 For mechanically ventilated adults, the guidelines also suggest using dexmedetomidine for delirium where agitation is precluding weaning or extubation. 13 Adult studies frequently note increased incidence of bradycardia and hypotension (and possibility of greater self-extubation). 14 The recent Intensive Care Medicine Rapid Practice Guideline recommends that dexmedetomidine should be used over other sedative agents when desirable effects of reduced delirium and duration of mechanical ventilation are valued over undesirable effects of hypotension and bradycardia. 14
Although only FDA approved in the adult population, dexmedetomidine use has also become more prevalent in the pediatric intensive care unit (PICU). 4 Despite its increased use, there are few prospective and randomized studies analyzing dexmedetomidine use for ≥24 hours in the PICU.4,15,16 One notable recent study was the PROSDEX study, a prospective multicenter study evaluating the efficacy of dexmedetomidine for prolonged sedation (≥24 hours) in 163 critically ill pediatric patients using validated clinical scores to evaluate level of sedation, withdrawal, and delirium. After 24 hours of initiation, dexmedetomidine was associated with improved patient comfort, withdrawal, and delirium scores, and reduced dosages of alternative sedative agents (benzodiazepines, opioids, propofol, and ketamine). 15 Bradycardia was reported in 27% of patients and hypotension in 11%. For hemodynamic adverse effects that required intervention (9% of total patients), a dose reduction was the most common intervention. 15 Banasch et al 16 demonstrated an association between adverse effects with younger age and longer dexmedetomidine duration. A randomized controlled trial is currently ongoing to evaluate the efficacy and safety of dexmedetomidine for prevention of withdrawal syndrome in the pediatric intensive care unit. 17
The 2022 Society of Critical Care Medicine Guidelines on Prevention and Management of Pain, Agitation, Neuromuscular Blockade, and Delirium in Critically Ill Pediatric Patients With Consideration of the ICU Environment Early Mobility (PANDEM) Guidelines now recommend the use of dexmedetomidine as the primary agent for critically ill post-operative cardiac patients with expected early extubation, with a conditional recommendation regarding its use in this setting to prevent tachyarrhythmias. For the general PICU population, the guidelines recommend minimizing benzodiazepine exposure. 18 Although this is not a positive recommendation for dexmedetomidine use, it is commonly favored given the significant drawbacks of alternative agents (propofol, ketamine, barbiturates) has seen success in ICU sedation practices due to its associated reduction in benzodiazepine exposure and reduction in delirium. Despite these advantages, in the pediatric population where FDA approval is lacking, there are considerable hemodynamic risks associated with the use of the medication, with 5-10% of patients experiencing a bradycardia or hypotension that requires intervention. Our case is unique given the duration of events and lack of strong precipitating factors outside of dexmedetomidine use. In addition to previous pediatric case reports in the literature, the authors suggest that dexmedetomidine should be used with careful weight of the associated risks and benefits.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
