Abstract
Introduction
Antineoplastic immunomodulatory and ancillary medications can pose a challenge to patient and hospital worker safety due to the administration schedules, complex steps required for these hazardous sterile compounded medications, and potential cytotoxic exposure. Moreover, errors concerning medications with narrow therapeutic windows, such as chemotherapy, have the potential for serious patient harm and can result in catastrophic reactions or death.1,2 Errors can occur at all stages of the compounding process because entire medical teams are involved with the chemotherapeutic medication process—including physicians, pharmacists, nurses, and pharmacy technicians.3,4 The multicausal nature of errors is large and can include a lack of knowledge, miscommunication, incorrect preparation techniques or other technical errors, and/or urgent requests resulting in rushed preparation. 5 In the last decade, chemotherapeutic and ancillary medication administration has substantially changed, from complete preparation by registered nurses to inclusion of specially trained pharmacy staff and technological innovations, such as remote camera dose verification and barcoding contributing to drug preparation. 6 Despite recent improvements in chemotherapy safety standards, precise compounding and preparation of these medications remain a considerable focus for hospital pharmacies.
Overall, the incidence of chemotherapy medication error rates has a wide range from .004%–41.6%, with the highest for prescription errors (.1%–24.6%), followed by preparation (.4%–.5%), dispensing (.03%), and administration (.02%–.1%). 7 In a 1-year prospective study evaluating chemotherapeutic medication errors in hospitalized patients, pharmaceutical errors occurred in .16% of all anticancer drugs prepared, which was defined as errors in pharmaceutical analysis (pharmacist-generated mistakes or failures to detect prescription errors, data entry in the pharmaceutical software, or preparation, storage, and/or dispensing errors). 3 Notably, the study suggested pharmaceutical errors were underreported due to possible fear of reproach and the time-consuming process for incident reporting. In contrast, in a 2-year prospective study of oncology medication errors in a large community hospital, 38% of errors resulted in incorrect dispensing of correctly ordered medications, with the most frequent medication error reported being late administration due to delayed arrival from the pharmacy; however, when assessing medication errors not due to late dispensing, 25.5% were nurse-dispensing errors vs 8.5% pharmacy-dispensing errors. 8 In a systematic review of compounding errors for all drugs in the US from 1990-2020, the most common errors reported included contamination and concentration errors. 9 Overall, the mean number of patients affected per error was 41 with a mean number of fatalities per error of 3.
The estimated cost of an in-hospital adverse drug event, which is inclusive of medication errors, is estimated to be approximately $4056 per event. 10 Moreover, the projected total cost for US hospitals to treat preventable adverse drug events is $871 million, which does not include the costs of injuries to patients or malpractice costs. 11
One common cause of medication dosing errors is mismanagement of medication overfill volume. Commercially available bags of parenteral solution are labeled with a specific amount of solution; however, the actual volume of solution is larger, as it contains overfill to account for loss depending on storage conditions, specific contents, container permeability, and ratio of fluid volume to the container surface area. 12 Consequently, management of overfill volume is essential for chemotherapeutic medications, since patient-specific dosing and regimen-specific administration sequencing are essential for accurate treatment. Consequently, the Institute for Safe Medication Practices (ISMP) recommends a 10% overfill guideline for the preparation of infusion medications, indicating if the volume of added medication is >10% of the volume listed on the parenteral solution container. If there is overfill, then the volume of the additive plus the potential overfill volume should be removed before combining the additive medication with the bulk solution. Alternatively, upon reconstitution of some medications in lyophilized powder formulations, such as gemcitabine, the vial may actually have less volume than the final amount expected, which may result in a dosing error where a compounder may seek to open additional vials labeled with specific amounts to achieve the desired reconstituted drug volume, but the correct dose is exceeded in the process.
In response to the high medication error rates in US hospitals, Congress passed the Patient Safety and Quality Improvement Act in 2005; this authorized the creation of Patient Safety Organizations (PSOs) and provided unified structure and legal protections for reporting medication errors. 13 PSOs are authorized to conduct safety research to improve patient safety, quality of patient care, and provide education on improved safety practices to use in clinical practice. PSOs focus on data collection and analysis in a standardized manner with common definitions and reporting formats to provide uniformity and confidentiality in reporting patient safety events, thus lending credibility to results obtained.
In this study, the PSO (PSO Advisory, limited liability company) securely acquired data and product visualizations from an intravenous (IV) workflow system and designed a software environment for blinded researchers to evaluate IV chemotherapy preparations. Intravenous workflow systems aim to improve the accuracy of IV-compounded chemotherapeutics and potentially improve safety through reducing medication errors via medication bar code scan confirmation and compounding libraries that ensure the correct medication dose and IV fluids are used. 14 Process errors in IV chemotherapy preparation may not be detected by workflow systems and instead depend on pharmacist assessment using visual identification of the products to determine whether the correct processes have been followed.
Given the unmet need surrounding standardization of delivered chemotherapeutics and improvement for best practices, we evaluated a remote verification process of chemotherapeutic and ancillary medication compounding in a hospital setting.
Methods
Study Design and Objectives
This retrospective, blinded, observational study used the same remote IV workflow verification system to monitor deviations from best practice and/or errors in IV compounding with chemotherapeutic drugs in 2 large hospital pharmacies in the United States over a 90-day period from January–April 2019. Pharmacy technicians were the primary personnel involved in medication preparation at the 2 sites evaluated. Compounding errors were defined using the National Coordinating Council for Medication Error Reporting and Prevention’s definition of a medication error, which describes an error as any preventable event that may cause or result in inappropriate medication use or patient harm while the medication is under the control of a healthcare professional, patient, or consumer. 15
A software user interface was developed to include a picture slide viewer representing images from the remote verification system, IV workflow system outcome codes, and oncology pharmacy services outcome codes. The remote IV workflow system used had specific routes assigned for each medication to guide the compounding process. In particular, safety measures were designed to recognize medication errors, such as drugs scanned with an ordered route that did not correspond to available routes, resulting in an error message display. However, only 1 validation error message is shown at any given scan, which then triggers a stop in the workflow and prohibits dose preparation to continue using the remote verification system.
Primary objectives included identifying undetected errors or deviations from best practice not found by the IV workflow system, assessing time variables—including IV preparation, review and approval times using time stamps, identification and analysis of medication waste, and cost assessments.
Safety Assessments
Assessments using a visual identification system monitor quality control, identify deviations from best practice principles, and demonstrate variations in medication compounding. All images evaluated using the visual identification system were acquired from a combination of 2 separate 90-day data extractions from each of the designated hospital pharmacy facilities. Safety assessments were performed on 5656 images obtained from the remote IV workflow verification system and involved 77 different chemotherapeutic, immunosuppressive, and ancillary medications. All information related to patients and sites analyzed were de-identified to ensure patient confidentiality. Two registered pharmacists blindly reviewed all compounding logs and images of the therapeutic products prepared through the PSO’s remote web-based portal. Typically, 4 images per order were evaluated and included photos of the IV bag and medication vial, reconstitution of the medication with diluent, an image of the syringe showing the volume of the diluent solution, and a photo showing the IV bag with empty syringes after injecting the diluent solution. Deviations from best practice or variations in medication preparation were recorded; however, not all orders analyzed in the study had images to assess.
Assessment of orders based on photo evaluations were categorized as the following: acceptable medication verification, exceeds the ISMP >10% additive volume guideline, inaccurate labeling of medication dose, unable to verify medication, missing vials or syringes, failure to use closed system drug transfer device (CSTD) when appropriate, and inappropriate syringe size to accurately measure medication volume. All orders with images showing correct preparation and final product were classified as “acceptable medication verification.” Orders with inaccurate concentrations, incorrect volumes, and products that were previously compounded and then relabeled were defined as “inaccurate labeling of medication dose,” whereas orders with images of inappropriately sized syringes to exactly measure the stated volume were defined as “inappropriate syringe size.”
Time Flow and Medication Waste Assessments
Mean chemotherapeutic medication compounding time was analyzed for the total compounding time vs number of vials required for preparation per therapeutic product, total compounding time per vial for medications that had similar order frequencies during the study time frame, and total time for STAT orders vs non-STAT orders. Mean compounding time was classified as preparation, verification, sorting, and total time for all steps. Time flow analyses were performed on all orders assessed in this study regardless of the number of vials used. For STAT vs non-STAT orders, mean time for pending orders in the pharmacy was assessed. Additionally, the number of medication orders was analyzed based on time of day between workstations as well as mean time spent on preparation, verification, sorting, or total compounding time.
Analysis of medication waste was evaluated based on the number of vials wasted and the volume of wasted medication, as well as STAT vs non-STAT orders. Vials included in this analysis consisted of both drug vials, as well as admixture vials needed for compounding. Specifically, volume waste for gemcitabine was assessed based on a vial volume size of 200 mg, 1 g, or 2 g.
Time Flow and Waste Analyses
Time flow and waste analyses were performed using Microsoft® Excel® (version 2012) pivot tables to summarize the data. The data was summarized by medication name, number of vials required to compound the final product, and medications ordered with 100-200 instances of ≥1 vial used. Mean time per medication for each preparation, verification, and sorting step and total time to compound the medication were calculated, as well as the mean volume of waste observed per medication and per vial count. This study was not powered to determine statistical significance.
Cost Assessments
Cost analyses were performed using pricing listed for the CSTD provided by a large national purchasing contracting organization. An average fair market value salary of $65 per hour for oncology pharmacists was used to calculate pharmacy costs, with a mean indirect cost of 28% of the salary for benefits and 20% for facility costs. This hourly salary was then converted to $1.60 per minute, including the indirect benefit and facility costs, to analyze the approximate cost per minute to compound a medication. The cost of compounding a dose of gemcitabine per minute was performed using pricing listed for the CSTD consisting of $3.85 for a BD PhaSeal™ Injector (Reference 515003), $1.96 for a BD PhaSeal™ Administration Product (Reference 515306), and $4.09 for 1 BD PhaSeal™ Protector (Reference 515105). For the waste cost analyses for gemcitabine, the concentration of gemcitabine was specified as 38 mg/ml.
Results
Safety Assessments
Among images reviewed, 42.6% demonstrated acceptable verification of the compounded medication. Additional frequently reported results included medications that exceeded the ISMP >10% additive volume guideline (35.9%), had inaccurate labeling of medication dose (28.3%), were unable to be verified (10.4%), had missing vials or syringes (6.9%), did not use a CSTD when appropriate (5.7%), and used an inappropriate syringe size to accurately measure medication volume (4.0%). Notably, many of the images reviewed had ≥1 deviation from best practice detected per dose, which was reflected in the percentages of specific error detection rates.
Images demonstrating an overall medication volume exceeding the >10% ISMP additive rule accounted for a substantial portion of all deviations from best practice observed during this study. Figure 1 shows an example of a photo depicting 1600 mg gemcitabine in a 250-ml bag of sodium chloride .9% solution. However, the total volume noted on the dose label is 292 ml; therefore, the medication volume added accounted for ≥10% of the original 250-ml bag volume, resulting in an imprecise medication concentration. ISMP >10% additive volume guideline deviation from best practice. Example of a deviation from ISMP >10% overfill guideline in a gemcitabine infusion. The dose label for this infusion indicates 1600 mg gemcitabine in a 250-ml bag of sodium chloride .9% solution; however, the total volume is 292 ml, accounting for a volume increase >10% of the original bag volume and resulting in an inaccurate drug concentration. ISMP, Institute for Safe Medication Practices.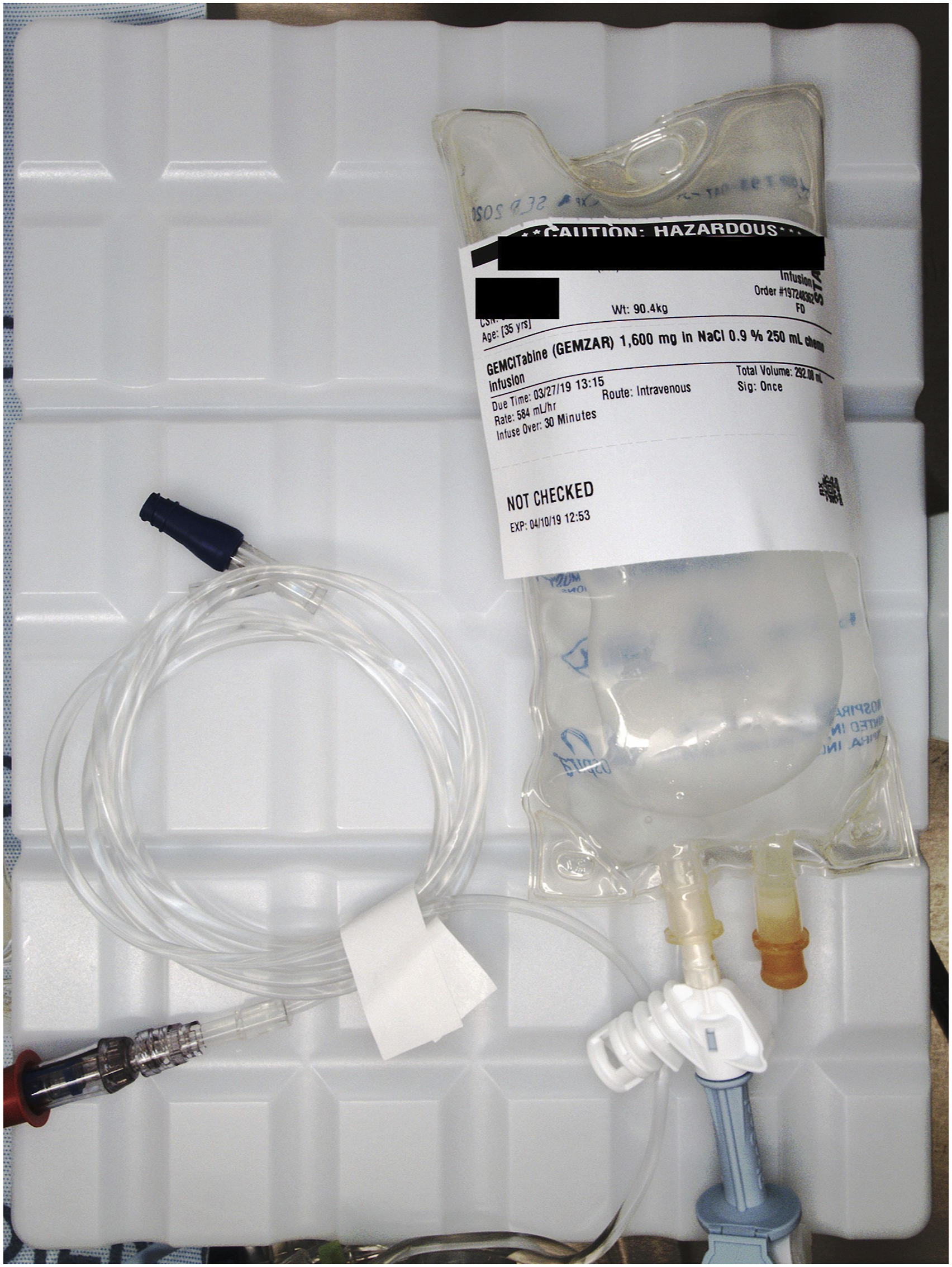
Similarly, several images reviewed showed instances of inappropriate syringe size for the specified dose and volume. For example, in Figure 2, a 3-ml syringe was used for a volume of .76 ml of bortezomib. However, the smallest increment of measurement for 3-ml syringes is .1 ml; therefore, the volume and dose are not able to be accurately verified with this syringe size. Inappropriate use of syringe size deviation from best practice. Image example of inappropriate syringe size use and volume verification. In this photo, a 3-ml syringe was used to measure a volume of .76 ml bortezomib. Yet, the smallest increment of measurement is .1 ml for 3-ml syringes and, consequently, the volume and dose cannot be accurately verified with this syringe size.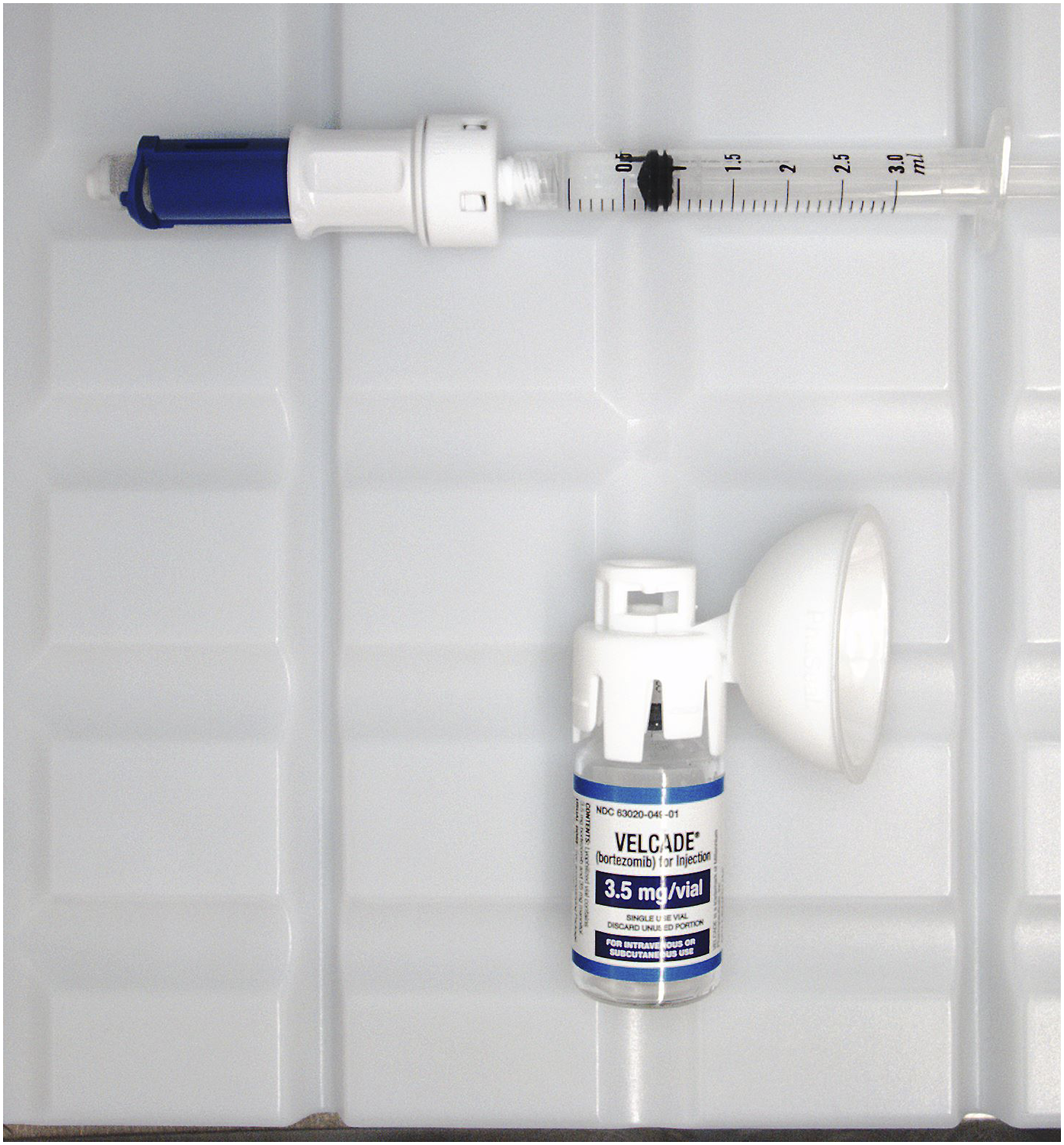
Time Flow and Medication Waste Assessments
The total compounding time for most medications evaluated in this study was between 7.6 and 13.8 minutes. Notably, the greatest time difference observed was between medications using 1 vial vs 2 vials with an increase in time of an additional 4.8 minutes. Analysis of total compounding time per vial for all medications used in this study demonstrated that for medications using >1 vial, each additional vial added ∼50.7 seconds to the total time (Figure 3). For medications using 2-4 vials, there was little difference in total compounding time, but for medications using >5 vials, a total time difference was noted. Overall, iron, methotrexate, and obinutuzumab had the highest total compounding time with an average of >30 minutes (Supplementary Table 1). Total compounding time per medication vial count.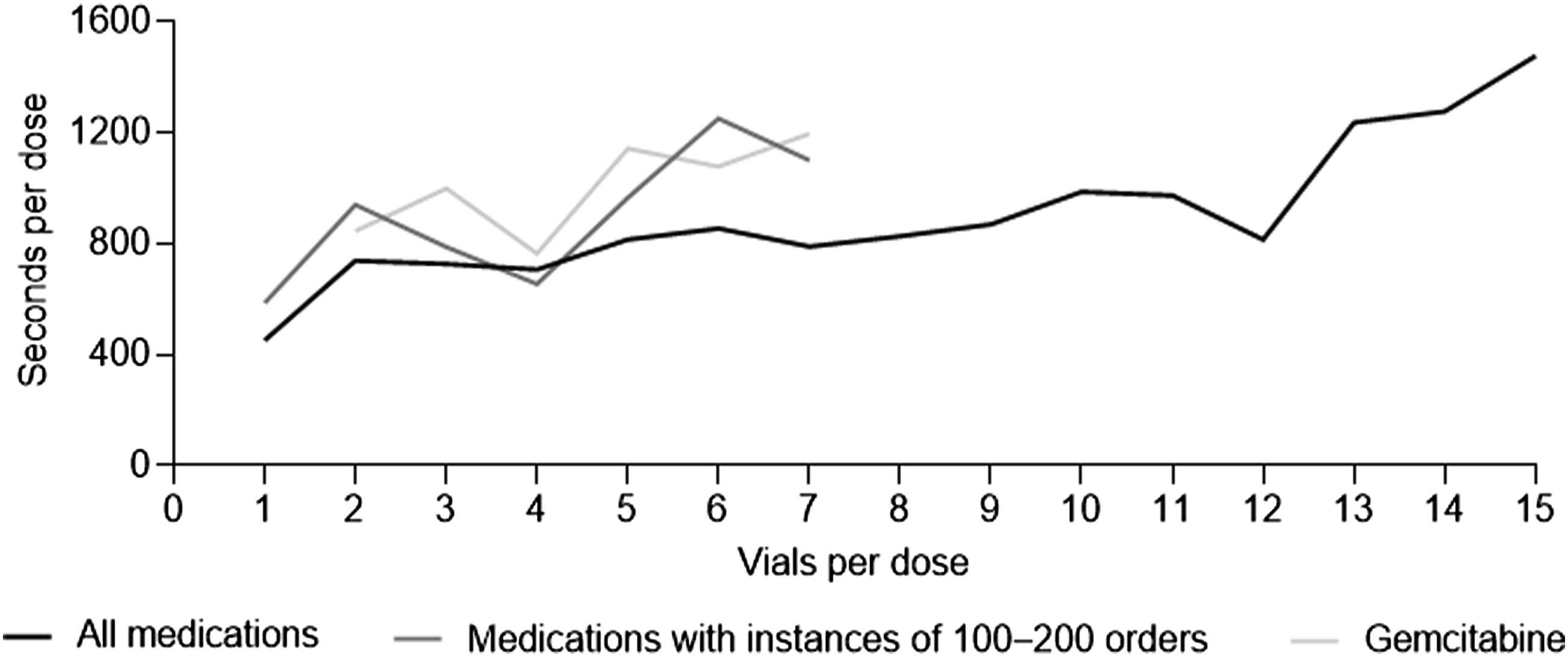
Analysis of medications prepared at a similar frequency during the study time frame showed azacitidine, bevacizumab, dexamethasone, docetaxel, doxorubicin, etoposide, gemcitabine, irinotecan, nivolumab, and oxaliplatin as the highest used medications observed throughout the study with ∼100-200 instances per medication. Notably, a large increase in total time per vial for dose preparation was observed from 1 vial to 2 vials with an average additional time of 5.9 minutes (Figure 3).
When assessing gemcitabine total compounding time per vial, no instances of usage with 1 vial were noted due to the weight distribution of the patient pool. Consequently, time differences for 2-4 vs 5-7 vials were calculated with gemcitabine mixtures using 2-4 vials averaging 14.5 vs 19.0 minutes for doses using 5-7 vials.
In addition, the most likely time for pharmacy orders to be placed was approximately 11
For most medications ordered, the mean number of vials required per dose was 1-3, and the mean number of vials wasted per order ranged from 0-2.16 vials. Most medications had a mean vial waste ranging from 0-.36 vials (Figure 4), and few medications had a mean vial waste count of >1.08 vials. Similarly, most medications averaged <9 ml of waste per dose. Some medications such as atezolizumab and nivolumab demonstrated low overall waste, whereas other medications like bortezomib and dexamethasone showed the highest levels of waste (Supplementary Table 2). Medication waste by mean number of vials wasted per dose. Vials included in this analysis consist of both medication and admixture vials.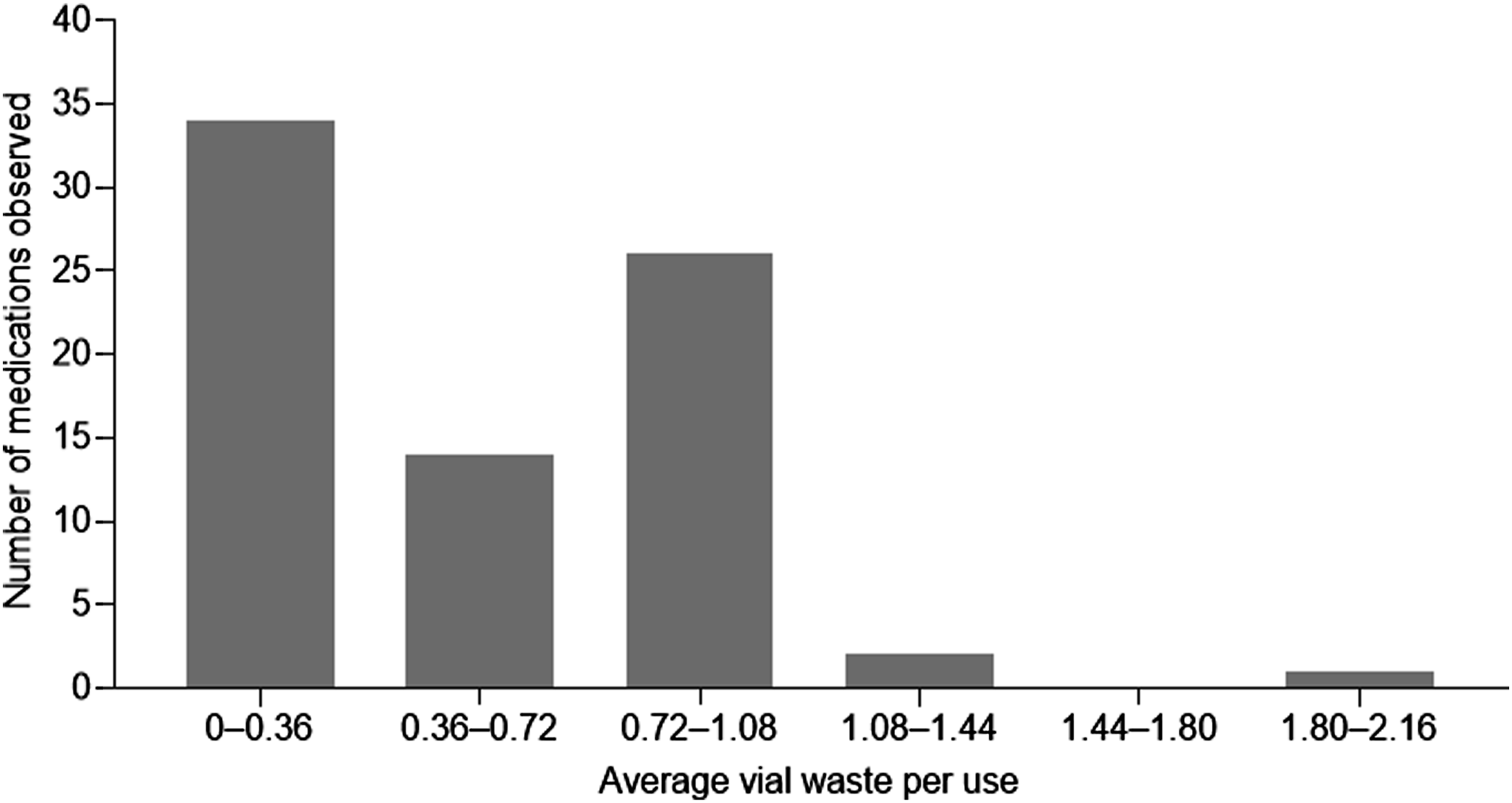
Waste analysis of gemcitabine demonstrated 2-g vials have a larger waste distribution compared with 200-mg vials. For most 200-mg vials of gemcitabine, waste was <5 ml compared with an average of 15-20 ml of waste for most doses using the 2-g vial. Specifically, ∼115 mg of gemcitabine was wasted per patient based on a concentration of 38 mg/ml.
Total mean time per compounding step was similar for STAT orders (9.2 minutes) vs non-STAT orders (12.9 minutes). Notably, STAT orders had a shorter mean pending time than non-STAT orders (1.3 vs 3 hours, respectively). Furthermore, STAT orders wasted more medication than non-STAT orders, with the greatest waste observed from larger volume vials. Most non-STAT orders wasted a mean volume of 3-6 ml vs 9-15 ml for STAT orders.
Cost Assessments
Time and cost assessment of gemcitabine.
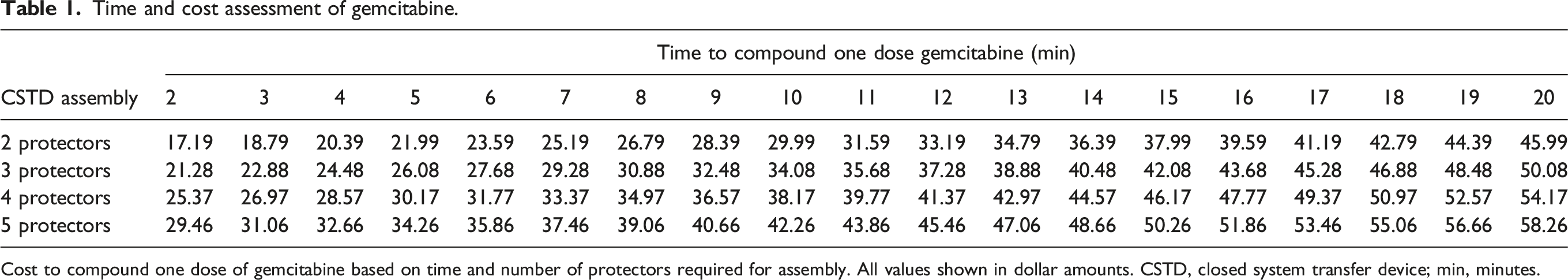
Cost to compound one dose of gemcitabine based on time and number of protectors required for assembly. All values shown in dollar amounts. CSTD, closed system transfer device; min, minutes.
Discussion
Remote verification systems provide important identification of medical errors, resulting in a reduced incidence of medication errors or deviations from best practice reaching patients. Following implementation of a workflow system in a large US cancer center, there was a 74-fold increase in detection of medication errors, as well as a decrease in the average cycle and turnaround time for medication processing and verification. 14 Similarly, in a study assessing chemotherapeutic comparative risk analysis before and after implementation of a remote verification system, the relative risk reduction of compounding error rate was 63% after implementing a workflow system. 16
In this retrospective observational study, use of a remote verification system showed variation between workflow stations and lack of standardization across delivered chemotherapeutics to patients. Image analyses demonstrated that the highest number of orders with a deviation from best practice were those that exceeded the ISMP >10% additive volume guideline. Although the ISMP >10% additive volume guideline is predominantly a recommendation and not part of most hospital pharmacy’s protocols or standard operating procedures, it was considered a deviation from best practice in this study because dose specificity is essential for efficacious chemotherapy treatment. Moreover, healthcare workers may not always be aware of potential overfill in an infusion bag and may stop the infusion prematurely when the indicated volume has been delivered, as programmed in an infusion pump, or alternatively, healthcare workers who recognize overfill may mistake the overfill solution as a pharmacy accommodation for medication lost in tubing. 12 Both scenarios described may result in a potentially considerable loss of the total dose going to the patient—possibly affecting treatment outcome.
Medication waste due to preparation inefficiency can result in substantial economic losses. In a 2-year analysis of chemotherapy waste from a group of oncology service providers in Brazil, a total of 63,824 mg of chemotherapy waste was recorded, with the greatest amounts of waste associated with fluorouracil, gemcitabine, and trastuzumab. 17 This amounted to a significant loss of approximately 602 mg of chemotherapy per patient and 49 mg per order. Similarly, in a study evaluating chemotherapy drug waste at the Oncology Department of the University Hospital of Udine, medication waste initially accounted for 8.3% of the Department annual drug expenditure, corresponding to €179,576, with most of the waste associated with cetuximab, docetaxel, gemcitabine, oxaliplatin, pemetrexed, and trastuzumab. 18 A substantial reduction (45%) in drug waste expenditure was observed after implementing a cost-containment policy that consisted of an expanded selection of multi-dose vials (able to maintain microbiological and chemical stability for at least 24 hours), drug dosages rounded within 5% of calculated dose, choice of most convenient vial size (according to drug unit price and accurate estimate of drug’s daily use), and a per pathology/per drug distribution of chemotherapy sessions protocol over the week. 18 Consequently, strategies aimed at decreasing the amount of chemotherapy waste can be effective and provide meaningful cost savings, as well as improve medication preparation efficiency.
The findings from our study demonstrated variations between specific drugs and their respective average waste—signifying a need for additional assessments on how to minimize waste and economize compounding of medications for use in multiple patients. In particular, several key medications were identified that frequently involved multiple vials per dose—including abatacept, cetuximab, dacarbazine, daratumumab, durvalumab, infliximab, pemetrexed, and trastuzumab—highlighting drugs that may benefit from delivery in different volumes or premixtures. For gemcitabine, smaller volume vials resulted in less medication waste; however, small vial usage was associated with considerably increased total preparation time despite reduced drug waste. Additionally, time taken to prepare medications accounted for the greatest amount of total compounding time, and a considerable difference was observed in total compounding time per vial from 1 vial to 2 vials. Similarly, length of time taken to compound a dose of gemcitabine was associated with greater overall cost per dose. Although cost analyses were only performed for gemcitabine, these findings are likely representative for other chemotherapeutics as well.
The prevalence of STAT orders is an interesting finding because it is rarely necessary for chemotherapy orders to be administered immediately. 19 STAT orders should be reserved for patients needing an immediate medication to address a life-threatening situation.20,21 In contrast, chemotherapeutic and immunosuppressive infusion medications are often prescheduled for administration and are part of a long-term treatment regimen. Moreover, STAT orders for chemotherapeutics may compromise necessary safeguards such as donning personal protective equipment required to handle these drugs, as well as increase the likelihood of compounding mistakes while rushing. We found the amount of time taken to prepare and verify chemotherapy STAT orders was similar to that for non-STAT orders—indicating further lack of benefit for ordering a chemotherapeutic STAT. In general, chemotherapy orders should be scheduled when there is sufficient staffing to guarantee that all safety measures and precautions are taken for compounding, verification, and administration steps to ensure both patient and technician safety. 19
Limitations of this study include missing images of vials or syringes, an inability to verify solutions in some of the images from the remote verification system’s database, orders with incomplete or absent images, and specific pharmacy cost data for the hospital pharmacies studied being unknown and use of estimated costs in this analysis. In addition, percentage agreement or differences in how the 2 blinded pharmacists may have recorded deviations from best practice could be a potential confounder. Another limitation is that 64 of the total 169 gemcitabine orders recorded by the remote verification system had data discrepancies, null values, or standalone bag scans, resulting in extremely low time scores outside of the observed range in this study. These results highlight that remote verification systems are not foolproof and may allow medications to be dispensed despite discrepancies. An additional limitation was that, as an observation study, specific differences and outcomes between workflow stations were not powered to determine statistical significance.
Although the IV workflow system used in this study provided step-by-step instructions for medication compounding, there were large discrepancies in preparation techniques as observed through visual identification assessments. Despite the framework and standardization that remote verification systems aim to provide, variability is inherent in human preparation. In a study evaluating the accuracy of manual chemotherapeutic preparation with controls of either no control, visual double-checking, or gravimetric control, none of the control measures was able to guarantee an accuracy <5% for the final concentrations, and there was large variability between technicians. 22 Similarly, in a study analyzing volumetric dosing accuracies in chemotherapy medications, 71.7% of the preparations were within ±5% of the ordered dose, whereas 87.4% of the preparations were within ±10% of the ordered dose. 23 Although this level of variation in dosing is acceptable for administering chemotherapeutic preparations, the variability between preparations is alarming, given these medications have defined dosing ranges that are essential to achieve desired evidence-based outcomes. 23
The imprecision of manual preparation techniques for chemotherapeutic compounding supports the use of robotics for chemotherapeutic preparation or the use of ready-to-administer medications, which require no further processing. Robotic chemotherapeutic and adjuvant medication compounding is not yet widespread; however, robots may offer safer and more standardized chemotherapy preparations to patients. In a study comparing external contamination for robotic preparation vs human manual compounding of hazardous drugs in a large hospital, robotic systems created considerably safer working environments with less external contamination over manual compounding inside a biologic safety cabinet. 24 Moreover, robotic systems can reduce costs and time required to compound medications, while guaranteeing a high level of concentration conformity. 25 Consequently, benefits of IV robotics vs manual compounding include precision, accuracy, extensive documentation, safety features, and improved sterility.26,27 Ready-to-administer chemotherapy preparations offer similar advantages in that dosing and final infusion product concentrations are standardized across all preparations, improving patient safety. In a pediatric hospital study of high-alert drugs including adrenergic agents and concentrated electrolyte products, implementation of ready-to-administer infusions allowed these medications to be available in an expedited manner, removed the potential for compounding errors, and ensured the medication was prepared in a sterile environment. 28 Approximately 80-200 steps may be involved in the administration of a single dose of medication in a hospital; therefore, ready-to-administer medications eliminate a series of steps where errors often occur. 28 In addition, utilization of a ready-to-administer product may reduce the risk of error, as this would prevent the need to handle and sterilely compound multiple individual vials of a drug for a single preparation. Since the greatest amount of time to compound medications was spent on medication preparation, and medications with longer compounding times tended to have higher associated costs, ready-to-administer drugs may reduce preparation times since they do not require CSTDs and, thereby, also reduce labor and overall costs. Moreover, ready-to-administer chemotherapeutics may provide more accurate dosing of medications and may improve safety and clinical outcomes. Specifically, because of the significant risks to healthcare workers exposed to hazardous drugs, including infertility, miscarriage, skin rashes, birth defects, and cancer, 29 the use of robotic systems and ready-to-administer medications would allow healthcare workers greater opportunity to avoid sterile compounding of cytotoxic medications and, thereby, reduce their risk of contamination and exposure. Moreover, there is the potential for improvement in patient safety with the use of robotic systems and ready-to-administer medications, as both of these medication delivery options provide standardized, accurate dosing as well as reduced external contamination.
Conclusions
The findings from this study highlight the lack of standardization for delivered chemotherapeutics and ancillary medications, the amount of medication waste that occurs during preparation, and the need to establish better quality controls to ensure patient and health care provider safety in the United States. Standardized products, such as ready-to-administer or robotic prepared infusions, may address this unmet need in hospital pharmacies. Further evaluation of ready-to-administer and robotic medication compounding is required to confirm this hypothesis.
Supplemental Material
Supplemental Material - Deviations From Best Practice: Findings From a Certified Patient Safety Organization Remote-Verification Observational Study of Intravenous Compounding of Chemotherapeutic and Ancillary Drugs
Supplemental Material for Deviations From Best Practice: Findings From a Certified Patient Safety Organization Remote-Verification Observational Study of Intravenous Compounding of Chemotherapeutic and Ancillary Drugs by Jeffrey Lombardo, John Coles, Daniel Ryszka, Christine Roussel, and William Smith in Journal of Pharmacy Practice
Footnotes
Acknowledgments
Writing and editorial support for manuscript preparation were provided by Zehra Gundogan, VMD (AlphaBioCom, LLC, King of Prussia, PA), and funded by Sun Pharmaceutical Industries, Inc (Princeton, NJ).
Author’s contributions
All listed authors meet the criteria for authorship set forth by the International Committee for Medical Journal Editors and have significantly contributed to, seen, and approved the final submitted version of this manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JC reports other from Sun Pharmaceutical Industries, Inc., during the conduct of the study. CR serves in a consultancy role for Burns Mechanical; LP3 Network Inc.; Sun Pharmaceutical Industries, Inc.; and University of the Sciences Philadelphia; and has received travel, accommodation, and expense support from Community Oncology Alliance, Evolution Medical Communications LLC, Georgia Society of Clinical Oncology, and Pennsylvania.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Sun Pharmaceutical Industries, Inc.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
