Abstract
Introduction
Mechanisms of clot formation can be disrupted by multiple medical conditions and therapeutic interventions. Cefazolin is a common intravenous first-generation cephalosporin used for a variety of infections. Cephalosporins, including cefazolin, are known to interact with vitamin K antagonist anticoagulants through several proposed mechanisms. 1 Published case reports and related research have also associated cefazolin use with coagulation abnormalities, including hypoprothrombinemia independent of concurrent anticoagulation.2-8 Less is known about the interaction between cefazolin and direct oral anticoagulants (DOACs), including any subsequent coagulation disturbance or bleeding risk. Finally, acute medical comorbidities like coronavirus disease 2019 (COVID-19) and disseminated intravascular coagulopathy (DIC) can independently contribute to hypercoagulability and clotting factor consumption, while a patient’s overall health status or concurrent medications may impact the activity or production of clotting factors.9-19 When these conditions and therapeutic interventions cooccur, the resulting impact on mechanisms of coagulation may be significant.
Here, we report a case of a 74-year-old male patient receiving concurrent apixaban and cefazolin who developed a significant prolongation of prothrombin time/international normalized ratio (PT/INR) in the setting of COVID-19 illness and post-procedural decreased oral intake.
Case Report
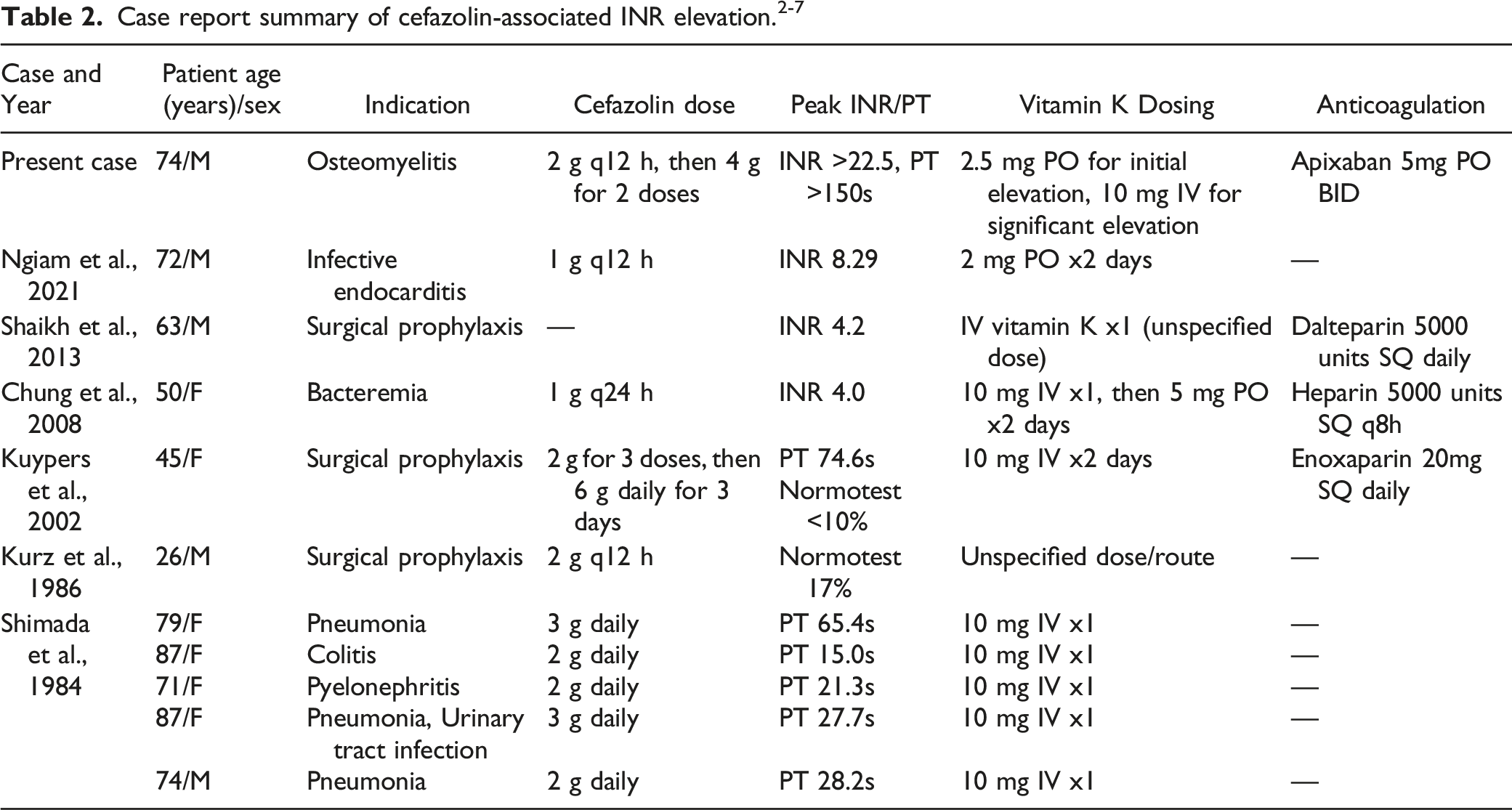
A 74-year-old white male with a past medical history of bullous pemphigoid, heart failure, type 2 diabetes, chronic kidney disease stage 4, atrial fibrillation, and hypertension was admitted for a malodorous right first metatarsal head wound (day 0). Magnetic resonance imaging (MRI) was not suggestive of osteomyelitis and treatment with intravenous (IV) piperacillin/tazobactam plus vancomycin was initiated for a diagnosis of diabetic foot infection. Blood cultures collected prior to antibiotic initiation resulted with methicillin susceptible Staphylococcus aureus (MSSA), therefore, antibiotics were de-escalated to cefazolin 2 grams IV every 12 hours based on renal function and Infectious Diseases (ID) consult service recommendations. On day 5, antibiotics were broadened to IV vancomycin and IV cefepime due to an increased WBC count (19.6 k/µL from 13.9 k/µL on admission) and fever (100.4 °F). On day 8, the patient underwent an angioplasty revascularization procedure. Antibiotics were de-escalated back to cefazolin on day 13 per ID consult service recommendation as patient remained hemodynamically stable and medication penetration to the wound was anticipated to be improved after revascularization. On day 15, the patient underwent right great toe amputation. No cultures were obtained intraoperatively, however, proximal margins of toe resection were noted as concerning for osteomyelitis. ID planned to complete 6 weeks of cefazolin therapy and the patient was scheduled to have a right internal jugular (IJ) tunneled catheter placed for antibiotic self-administration.
On day 17, an INR obtained prior to IJ catheter placement was found to be elevated at 5.1 (with a prior baseline INR of 1.3). In response, the patient was given a single 2.5 mg dose of oral vitamin K. The INR decreased to 2.0 by day 19, after which an IJ tunneled catheter was placed. Upon discharge (day 23), the patient was continued on cefazolin 2 grams IV every 12 hours.
Laboratory test results upon readmission (day 25).
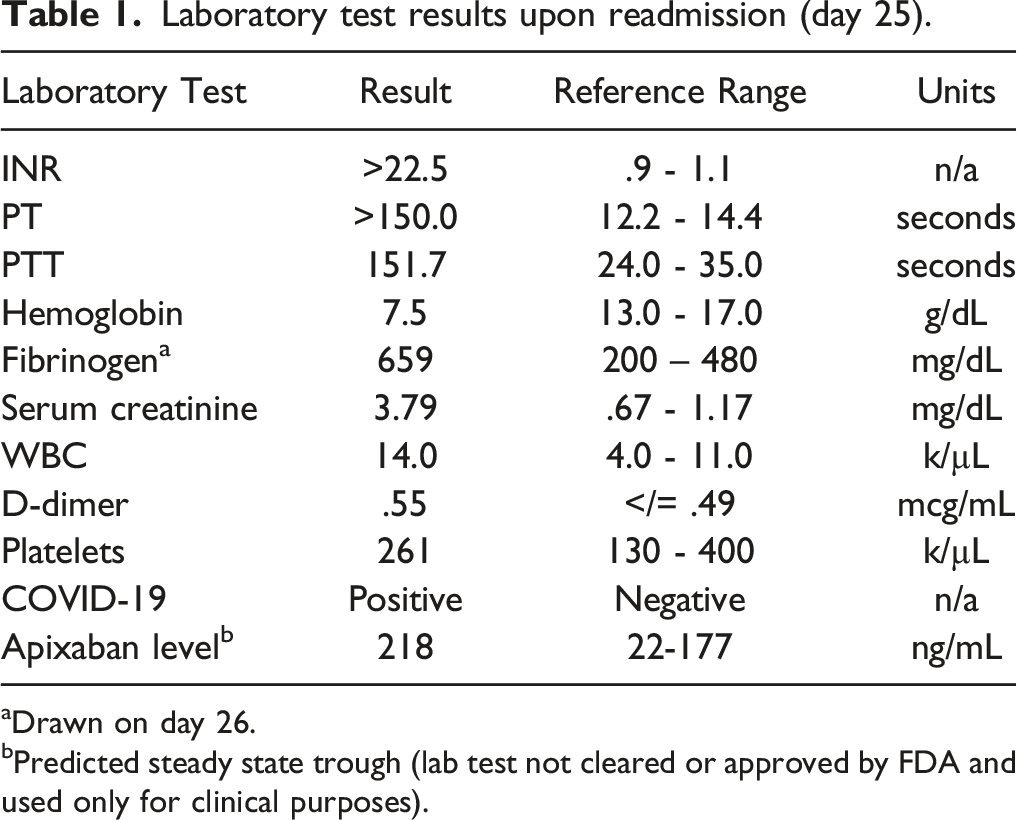
aDrawn on day 26.
bPredicted steady state trough (lab test not cleared or approved by FDA and used only for clinical purposes).
Given available literature, prior INR responsiveness to vitamin K administration, and reported poor nutritional intake, intravenous vitamin K was administered as a one-time dose of 10 mg on day 25. The patient’s outpatient apixaban was held upon admission to decrease risk of further bleeding. An INR ordered for the day following intravenous vitamin K administration returned at 1.6. ID was consulted upon rehospitalization and recommended discontinuing cefazolin and initiating daptomycin while the patient was admitted to the hospital. Daptomycin was subsequently switched to oritavancin to complete the remaining duration of therapy. No further bleeding was noted and apixaban was resumed as outpatient the evening of discharge. An INR drawn one week after intravenous vitamin K administration (day 32) returned at 2.0, demonstrating sustained improvement in coagulopathy. Vitamin K doses and resulting INRs are provided in Figure 1. INR trend throughout cefazolin treatment.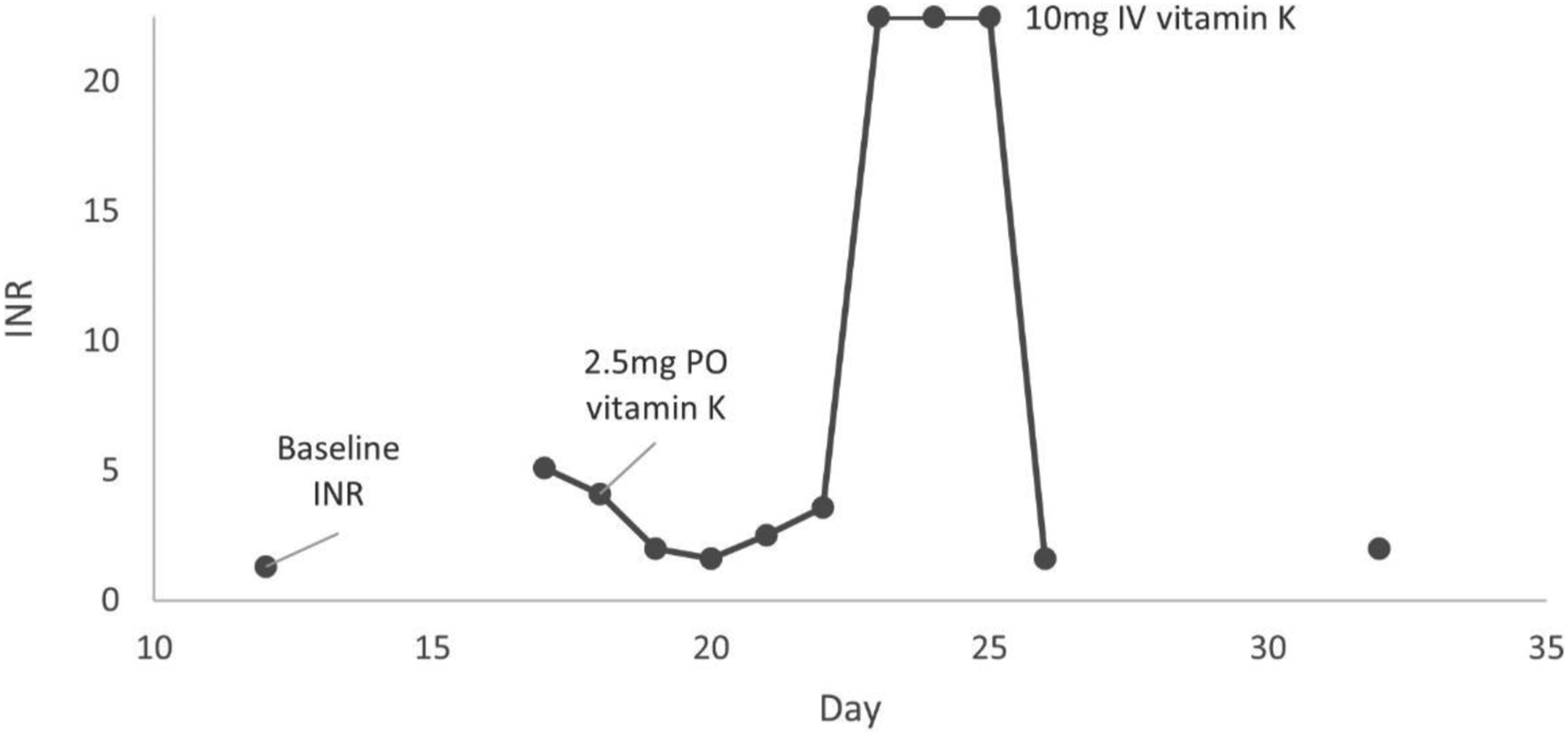
Discussion
Cefazolin and INR
Apixaban and INR
Factor Xa inhibitor DOACs can interfere with standard coagulation assays in a concentration-dependent manner. 9 Due to the mechanism of action, factor Xa inhibitors can prolong PT, INR, and PTT, but observed changes are typically small and variable, especially with apixaban.9,10 A 2017 CHEST systematic review found that PT/INR and PTT were inadequately sensitive to apixaban as results may remain within normal limits even in the setting of significantly elevated apixaban serum concentrations. 10 However, prolongation of PT and/or PTT in a patient with known factor Xa inhibitor exposure should be assumed to be secondary to drug effect until proven otherwise with other laboratory assessments. 9 Because of the variability and lack of sensitivity of PT/INR and PTT to apixaban when administered at approved doses, manufacturer labeling recommends against the routine use of these coagulation labs for clinical monitoring. Apixaban calibrated anti-factor Xa assays provide the best correlation with drug plasma drug levels and can be used as a surrogate marker when drug monitoring is needed (though universally established therapeutic ranges are not available). 10 The current case presented with a PT > 10x ULN, INR > 18x ULN, and PTT > 4x ULN. Apixaban as a sole contributor to these abnormalities is highly unlikely in a patient compliant to the prescribed dose and in the absence of substantial drug-drug interactions. Prior laboratory assessments from past hospitalizations and collection of apixaban steady-state trough support this determination. Other contributing factors were considered as more clinically significant for this case.
COVID-19 and INR
The observed impact of COVID-19 on routine coagulation parameters in published literature is variable. While INR, fibrinogen and D-dimer may be affected, others, like aPTT, often remain normal regardless of illness severity. 11 Of note, D-dimer can also be markedly elevated in a variety of other clinical circumstances. As such, despite the anticipated pro-coagulopathy of COVID-19, the prognostic value of these biomarkers has been explored by and incorporated in therapeutic resources with limited consensus.12,13 Furthermore, differentiation of COVID-19 coagulopathy from other conditions, such as DIC, can be challenging. In principle, early COVID-19 infection is more often characterized by normal platelet values and increased levels of fibrinogen rather than the decreases in both seen with DIC.13-15 However, more severe COVID-19 infections may bear more similarities to DIC with a corresponding worsening of patient outcomes.13-15 Taken altogether, our patient’s laboratory panel and overall mild illness point to contributing factors outside of COVID-19 alone. While the D-dimer is significantly elevated, this lab may be impacted by the patient’s recent operation, current infection, and renal impairment. The high fibrinogen and normal platelet count also do not support DIC as a primary condition. And while alterations in INR and PTT are possible in COVID-19 illness, the substantial elevations seen in this case do not reflect the marginal to absent changes cited in literature as typical of early or mild illness, making COVID-19 an unlikely sole cause of the observed laboratory abnormalities.
Malnutrition and INR
Blood coagulation is controlled by the balance between procoagulant and anticoagulant systems. Several nutrients affect coagulation, including vitamin K which is necessary for the synthesis of most clotting factors with the exception of factors III, IV, and VIII. 16 Vitamin K- dependent clotting factors are divided into anticoagulant (protein C and protein S) and procoagulant factors (II, VII, IX, and X). 17 Vitamin K procoagulant factors undergo vitamin K mediated post-translational modification for conversion into functional proteins. Deficiency of vitamin K from chronic malnourishment can affect all three coagulation pathways, potentially resulting in prolonged PT, INR, and PTT even in the absence of pharmacological anticoagulation. Furthermore, though the bioavailability of apixaban is not principally affected by food, apixaban is approximately 87% bound to plasma proteins, primarily albumin. 18 Albumin concentrations can be affected by various conditions that affect production or clearance of albumin, including heart failure, cirrhosis, nephrotic syndrome, and malnutrition. Chronic nutritional deficiencies affect albumin synthesis through a decreased supply of amino acids and trace minerals. 19 Although hypoalbuminemia caused by chronic malnutrition has not been directly evaluated in patients on DOACs, theoretically, low albumin levels may result in increased drug exposure and in turn affect coagulation assay results. Nutritional deficiencies were not overly apparent in our patient and despite poor oral intake being noted, albumin levels were normal (vitamin K level was not measured). Despite normalization of PT and INR after administration of IV vitamin K, vitamin K deficiency secondary to malnutrition was likely not an independent contributor to this patient’s elevated PT and INR.
Conclusion
Disruptions in mechanisms of coagulation can result from a variety of contributing factors. While literature review can further define possible causes, an individualized evaluation is key to managing co-occurring conditions and therapies. In this instance, early detection of patient reported PICC line bleeding while on cefazolin with assessment of medication adherence and laboratory parameters were essential to care plan development. COVID-19, concurrent apixaban, and malnutrition were all evaluated as potential reasons for observed INR elevations; available literature and patient-specific qualifiers (mild viral illness, prior INR stability on DOAC therapy, and absence of apparent severe nutritional deficiencies) established each as an unlikely primary etiology. As seen in prior case reports and related research, the significant INR prolongation was definitively managed with vitamin K administration and conversion of antimicrobial therapy to an alternative agent, highlighting cefazolin as the most likely contributing factor. For the clinician, it is worth noting that incorporating patient interview questions to detect PICC line bleeding with thoughtful consideration of planned INR monitoring may be important components of extended intravenous cefazolin therapy.
Key Points
• Cefazolin may cause vitamin K deficient coagulopathy resulting in prolonged INR, PT and PTT. • Multiple medical comorbidities can independently contribute to hypercoagulability and clotting factor consumption, making determination of distinct etiology difficult. • Vitamin K may be considered to correct an elevated INR secondary to cefazolin.
Footnotes
Author’s Note
Prepublication review and approval were obtained from the local privacy office confirming appropriate de-identification of patient information using the HIPAA safe harbor method.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.