Abstract
Introduction
Many possible mechanisms have been suggested in the development of ovarian cancer, such as the gonadotropin hypothesis, the androgen/progesterone hypothesis, and the incessant ovulation hypothesis, but the pathophysiology has not been fully explained in the literature to date. 1 Granulosa cell tumor is malignant cancer that develops in the ovarian sex-cord stromal cell. There are two types, adult and juvenile. Call-Exner bodies are mostly detected on pathological examination. Ovarian granulosa cell tumors are developed indolently, and the estrogen effect is an essential factor in its development. Genetic mutations such as FOXL2, PI3K/AKT, GATA4, VEGF, TGF-β, AMH, and SMAD affect the development of adult ovarian granulosa cell tumor (AOGCT) through apoptosis and cell proliferation mechanisms. 2 The FOXL2 genetic mutation is present in 97% of AOGCTs. 3 The FOXL2 gene has an important role in the production of steroid hormones. In the case of mutation in this gene, an increase in steroid production occurs and contributes to tumor formation, although the exact mechanism is not known. 4
The estimated annual incidence of ovarian granulosa cell tumors is 1/100 000, and the rate of locally advanced and metastatic disease in these patients at the time of diagnosis is around 12%. 5 The main therapeutic method is surgery in AOGTCs. Patients with tumor stage IC or above have a significant recurrence risk. Mitotic rate, tumor size, tumor stage, absence of Call-Exner bodies, and cellular atypia were identified as clinicopathological factors affecting prognosis. 6 Also, the presence of residual tumor after surgery has been defined as a very important risk factor for recurrence. 7 Adjuvant chemotherapy should be considered in patients with ovarian granulosa cell tumors if the tumor stage is IC and above or in the presence of risk factors such as tumor rupture, poor differentiation, and the presence of a tumor larger than 10 cm.8,9 It has been shown that chemotherapy, hormonal therapy (HT), and radiotherapy are effective in patients who develop recurrence after surgery and are not suitable for re-surgery. 10
Since AOGCTs are rare, there is no randomized study in metastatic disease. Data on treatment efficacy are generally based on retrospective studies involving a limited number of patients. Although the clinical benefit of chemotherapy in recurrent disease has been demonstrated, retrospective case series have not shown a definitive benefit on overall survival. There are limited data on the efficacy of HTs in the treatment of metastatic AOGCTs. This study, it was aimed to investigate the effectiveness of HTs in patients with metastatic and unresectable AOGCTs.
Methods
Patients and Data Collection
The study was a retrospective cross-sectional study. Ethics committee approval was obtained before the study. The research was carried out in conformity with the Declaration of Helsinki and good clinical practice guidelines. Patients with recurrent or metastatic AOGCTs and who received at least one series of HT between 2014 and 2020 were included in the study. The patients were identified through the hospital information system. Patients who did not have sufficient data for statistical analysis were excluded from the study. The clinical and pathological features of the patients were determined from the hospital patient files. Metastatic disease burden and metastasis distribution characteristics of the patients before HT were recorded. Also, all types and numbers of surgeries undergone by the patients, chemotherapy regimens they received, and HT characteristics were recorded. Staging at the time of diagnosis was done with the eighth Edition of the AJCC-TNM Staging Manual. Inhibin B level was measured using the immunoassay method.
The patients received tamoxifen 20 mg/day or anastrozole 1 mg/day as HT. Also, some patients were given leuprolide acetate 3.75 mg every 28 days as a luteinizing hormone-releasing hormone (LHRH) agonist. Treatment response assessment was done every three months with inhibin B level and imaging. HT responses were defined by the Response Evaluation Criteria in Solid Tumors (RECIST). Treatment-related adverse events were recorded and graded with the Common Terminology Criteria for Adverse Events (CTCAE). Inhibin B level was also measured at each treatment response assessment. The biochemical response was defined as a 30% or greater decrease at inhibin B level, adapted from the RECIST 1.1 criteria. The primary aim of the study was to evaluate the effectiveness of HT. Therefore, progression-free survival (PFS) time was defined as the time from initiation of HT to progression.
Statistical Analysis
SPSS version 25 was used for statistical analysis. Categorical variables were expressed as numbers and percentages. Continuous variables were shown as a minimum and maximum value along with the median value. Survival analyzes were performed using Kaplan Meier analysis, and a survival curve was plotted. Kaplan Meier curve for progression-free survival in the patients.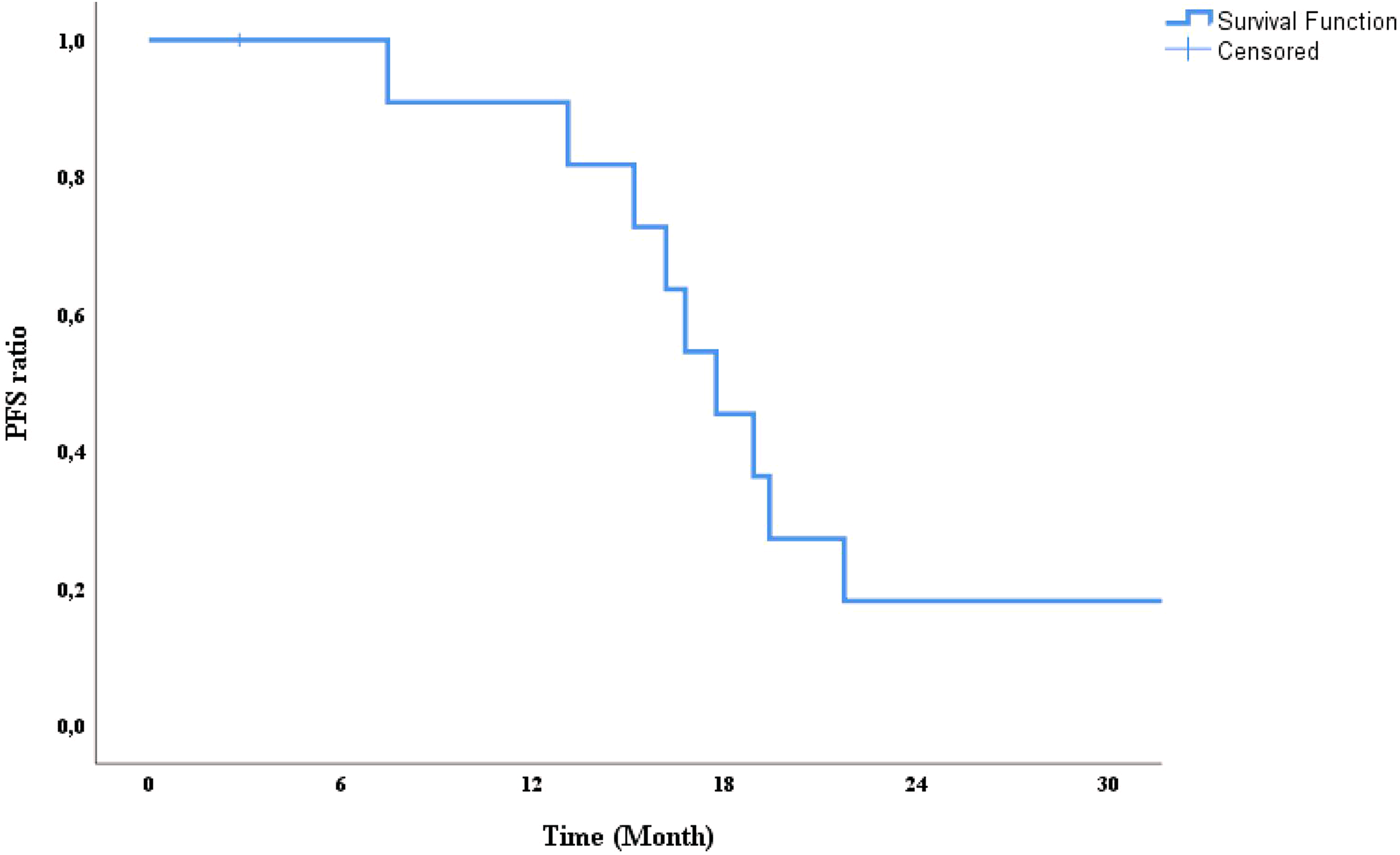
Results
Patient Characteristic and Treatment Approaches
Clinical, Pathological and Treatment Features of the Patients.
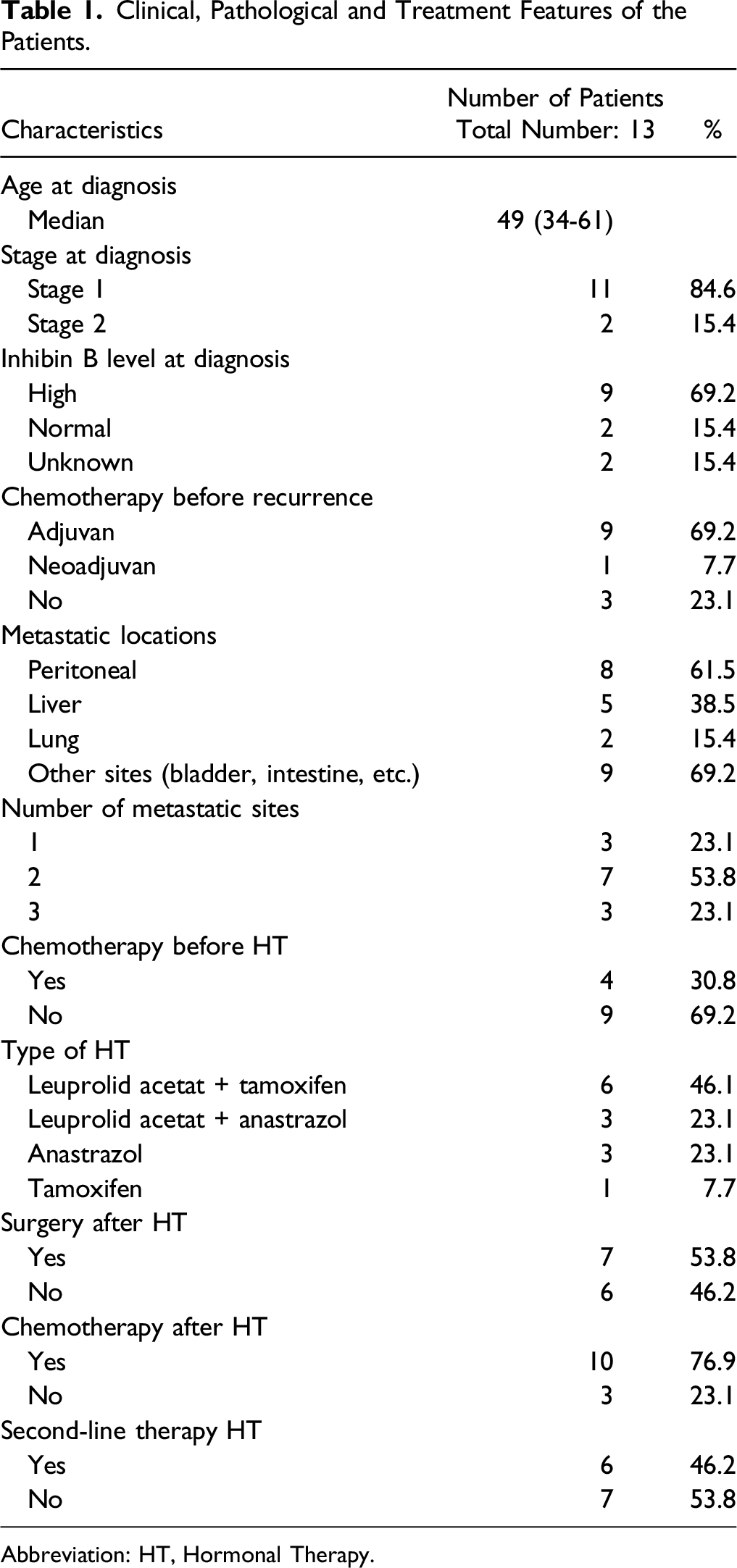
Abbreviation: HT, Hormonal Therapy.
Responses to Hormonal Therapy in the Patients.
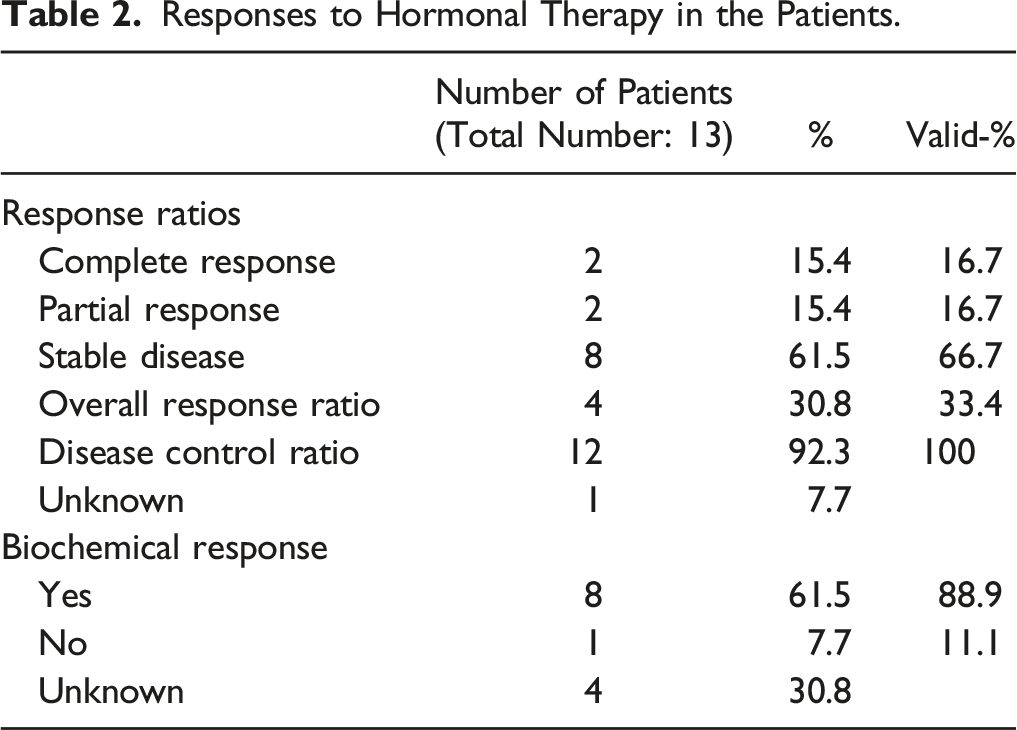
Survival Outcomes and Prognosis
The median time from the first diagnosis to recurrence in the patients in our study was 9 (2-22) years. Seven (53.3%) patients underwent reoperation after progression with HT. Ten (76.9%) patients received different chemotherapy regimens, and six (46.2%) patients received second line HT. The median number of surgeries undergone by the patients during the entire follow-up period was 3 (range, 2-6). Two (15.3%) patients died during the study period. The median follow-up period after recurrence developed was 76 (range, 9-147) months. Median PFS was determined to be 17.7 months (95% CI, 14.7-20.6) (Figure 1).
Discussion
We showed that HT is effective and safe in patients with metastatic or recurrent AOGCTs who have received previous treatment. In a retrospective study involving 35 patients, it was found that the time from diagnosis to the first recurrence ranged from 24 to 141 months. 11 Also, it has been reported in the literature in patients with relapses after 20 years. 12 In our study, we detect late recurrence in some patients similar to the literature. For this reason, long-term follow-up should be performed in patients with AOGCT in terms of recurrence after primary surgery. In the literature, data on the efficacy of HT in patients with AOGCT are primarily based on case reports or pool analyzes of case reports. Tamoxifen, aromatase inhibitors, or GnRH agonists alone or in combination are used as HT in patients with recurrent AOGCT who are not suitable for surgery. There are many case reports showing the efficacy of HT. In a case report, a 65-year-old patient with recurrent AOGCT who progressed after chemotherapy had a partial response to a GnRH agonist, and PFS was provided for more than 8 months. 13 In another case report, a 52-year-old patient who was resistant to chemotherapy had a good clinical and radiological response with the use of anastrozole, and no resistance to treatment developed during the 20-month follow-up period. 14 Similar to the results of our study, 31 patients were evaluated in a systemic review in which different HTs were examined and pool analysis was performed, the overall response rate was detected 71%, and the median PFS value was found 18 months. 15 In a prospective phase 2 study in which 41 patients were evaluated, anastrozole was given to patients with recurrent ovarian cancer, and the clinical benefit rate was found to be 78.9%. Also, the median PFS was determined 8.6 months. 16 In this study, it was emphasized that the results obtained with anastrozole treatment in patients with AOGCT were below the efficacy data reported in the literature. In our study, different HT options were used alone or in combination. There was a good response to different HT options, and no drug-related severe toxicities were detected. In addition, it has been shown in the literature that the use of hormonal drugs from a different group is effective after the developing of resistance to HT in patients AOGCT. In a 46-year-old AOGCT patient who had previously received chemotherapy and progressed under letrozole, disease control was achieved for 20 months with the use of GnRH analog in the second series. 17 In our study, second series of HTs was used in 6 patients, and disease control was achieved in four patients.
Inhibin B level has 98% sensitivity and 88% specificity in detecting recurrence in patients with AOGCT and is an important biomarker for detecting recurrence after primary surgery. 18 In our study, we found a relationship between inhibin B level and treatment response. There are publications in the literature showing a decrease in inhibin B level with response to treatment, similar to our study. In a prospective study that included 123 patients and evaluated the validation of Inhibin B in clinical use, a statistically significant correlation was found between serum Inhibin B level and tumor burden in patients with AOGCT. 19 Also, in a case report, the relationship between the treatments received and inhibin B levels in a 48-year-old heavily pretreated patient with AOGCT was examined; a significant decrease was shown in inhibin B levels in each treatment that reduced the tumor burden. 20
Our study had some limitations. Since the study was of a retrospective nature, the patient group and treatment characteristics were heterogeneous. In addition, due to the fact that it is a rare tumor, the number of patients was low, and the data of some patients were missing.
Conclusions
In this study, we showed that HT is effective and safe in recurrent or metastatic AOGCTs. We found that the inhibin B level was associated with treatment response in these patients. Due to the rarity of AOGCTs, there are limited studies on HT in the literature. In this respect, our study contains important results related to HT efficacy. Our study results need to be supported by multicenter studies with large patient groups. Future prospective studies are needed to understand the recurrence pathophysiology of AOGCTs and to define the factors affecting response in treatment.
Footnotes
Authors’ Note
Presentation at a meeting: This study is presented as a poster at ESMO Congress 2021.
Author Contributions
Idea and design: ID, EA, AA; Data collection: ID, EA, ST, PS, MS; Statistical analysis and writing: ID, EA, AA; Revision of the article for important intellectual content: ST, PS, MS, AA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The local ethics committee approved this study (Number: 267025).
Data Availability
This published paper contains all of the data produced or analyzed during this investigation.
