Abstract
Introduction
Sulfamethoxazole/trimethoprim is commonly used to treat a variety of gram-negative and gram-positive infections. Use of this agent has been associated with several potential adverse reactions, many of which would be considered mild to moderate severity. Agranulocytosis is a rare but serious side effect of sulfamethoxazole/trimethoprim for which there is a paucity of evidence and risk factors are not well described. Agranulocytosis is defined as an absolute neutrophil count (ANC) below 0.5x103 cells/μL and as many as 70-90% of cases are attributable to medications, also called idiosyncratic drug-induced agranulocytosis (IDIA). The frequency of IDIA ranges from 1.6 to 15.4 cases per million population per year with a reported mortality rate of about 5%. 1 While sulfamethoxazole/trimethoprim is known to be associated with IDIA, it has frequently been linked in other cases to complex comorbidities or concomitant medications. Agranulocytosis may also be caused by autoimmune diseases, such as systemic lupus erythematosus, Sjögren’s syndrome, and rheumatoid arthritis, by myelosuppressive antibodies, or T lymphocytes that suppress granulopoiesis. 2 Here we report a case of a healthy individual experiencing likely IDIA related to sulfamethoxazole/trimethoprim.
Case Report
A 53-year-old Caucasian male (177.8 cm; 84.5 kg) with a recent history of cellulitis presented to the emergency department with concerns for weakness, fever, chills, sore throat, and myalgias. His physical exam was unrevealing except for oropharyngeal erythema, subtle swelling and erythema of left lower extremity. He reported no abdominal pain, nausea, vomiting, diarrhea, shortness of breath, cough, weight loss and no recent travel or known exposure to infectious illness. The patient had no other significant past medical history, was not currently taking any medications, and illicit drug screening was not conducted due to the absence of drug abuse history. His only reported medication allergy included naproxen which he noted had caused facial swelling in the past.
The patient was recently diagnosed with cellulitis related to a laceration on his left thigh approximately 19 days prior to this admission. At that time, he was given one dose of intramuscular ceftriaxone and was prescribed a 10-day course of sulfamethoxazole/trimethoprim 800/160 mg by mouth twice daily which he completed 6 days prior to this hospitalization.
Upon admission to the emergency department, laboratory tests revealed a white blood cell (WBC) count of 0.6 × 103 cells/μL (normal range 4.0-11.0 × 103 cells/μL) with 97% lymphocytes and an absolute neutrophil count (ANC) of 0.0 x 103 cells/μL, a platelet count of 386 × 109/L, and a hemoglobin of 12.6 g/dL. The patient was admitted for new-onset severe neutropenic fever and was started on empiric piperacillin/tazobactam and vancomycin.
Further workup was guided by the patient’s initial presentation and included a chest x-ray, blood and urine cultures, and testing for viral pathogens including COVID-19, human immunodeficiency virus (HIV), Epstein-Barr Virus (EBV), cytomegalovirus (CMV), Hepatitis B and C virus. All tests were negative for acute infectious etiology, however the patient did have evidence of past/distant exposure to both EBV and CMV. The patient did not have risk factors for tuberculosis (TB) therefore testing was not done for this pathogen and his infectious symptoms resolved without TB treatment indicating that this was not likely to be the cause of his symptoms. A CT scan was not done since the patient had an absence of signs and symptoms suggestive of intrathoracic or intra-abdominal pathology. An infectious etiology was not identified on this comprehensive workup and the patient was continued on empiric therapy for neutropenic fever for a total of 7 days (piperacillin/tazobactam for 2 days followed by cefepime and vancomycin for 5 days). A bone marrow biopsy was performed on hospital day 5 and showed normocellular marrow with left-shifted granulocytic hyperplasia (consistent with granulocyte maturation arrest) and no morphologic evidence of dysplasia, lymphoma, or high-grade myeloid neoplasm. The pathology report suggested that drug-induced agranulocytosis was within the differential given his history of treatment with sulfamethoxazole/trimethoprim. The patient’s WBC nadir was 0.4 × 103 cells/μL on hospital day 2 with a subsequent rise to 8.9 × 103 cells/μL on hospital day 7. His ANC remained at 0.0 cells/μL from hospital day 1 through 3, followed by an increase to 3.0 × 103 cells/μL on hospital day 7 (Figure 1). His absolute lymphocyte count also gradually increased from 0.6 × 103 cells/μL on the day of admission up to 4.1 × 103 cells/μL on hospital day 7. WBC and ANC Trend through hospital stay.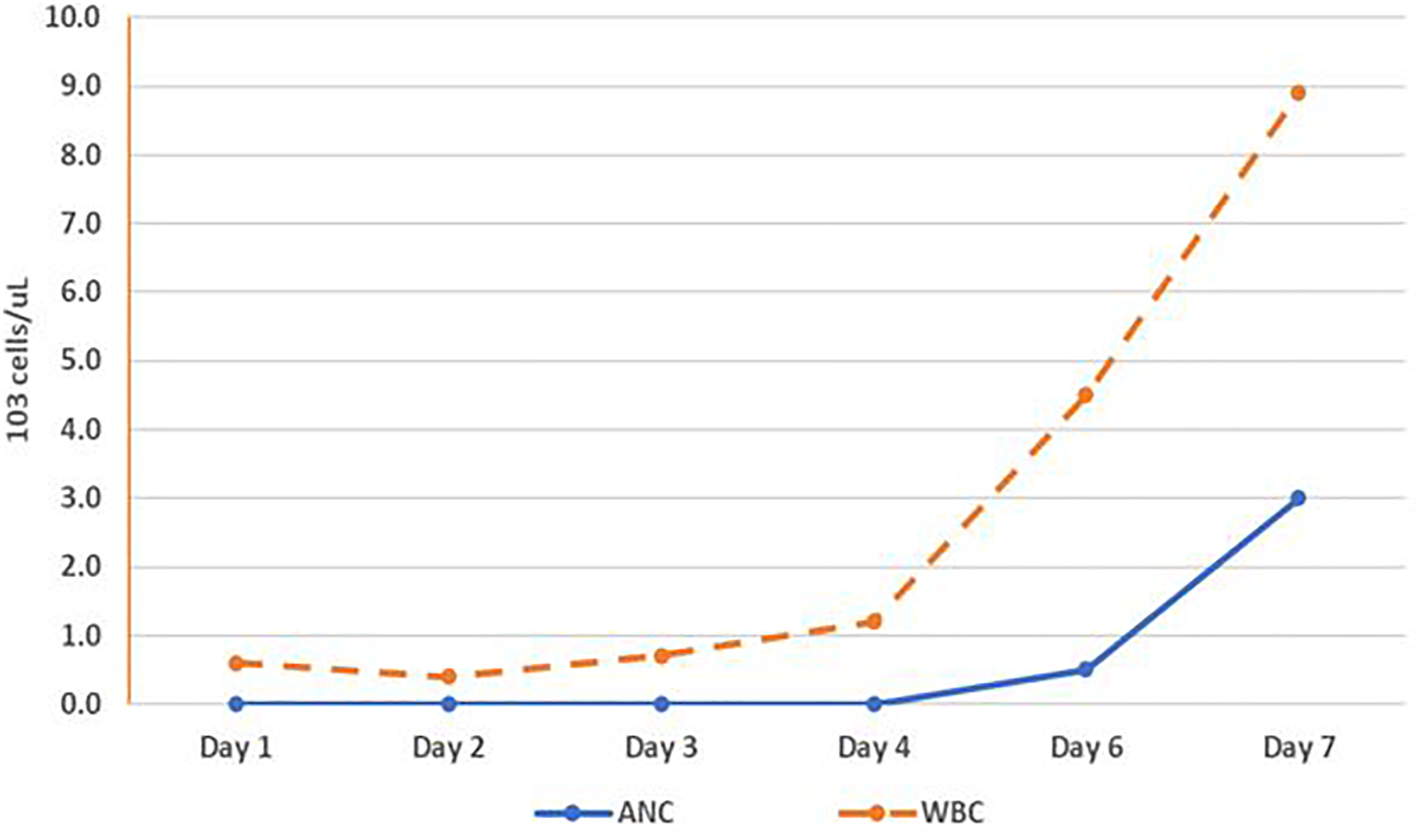
Estimation of Naranjo Adverse Drug Reaction Probability.

Interestingly, the patient reported 2 prior occurrences of sulfamethoxazole/trimethoprim use. Four years prior to this presentation, the patient was given a course of this antibiotic which caused him to “feel unwell” so it was stopped. A year prior to the presentation discussed in this case report, the patient had a similar case of cellulitis from a puncture wound that was first treated with a dose of ceftriaxone and a course of cephalexin. After the cellulitis did not resolve with these antibiotics, he was prescribed a 7-day course of sulfamethoxazole/trimethoprim which resolved his symptoms. His CBC was not obtained after completion of sulfamethoxazole/trimethoprim in either previous course, however the patient reported resolution of both infections with no other noted complications.
Discussion
While hematologic side effects associated with sulfamethoxazole/trimethoprim are rare, this agent is one of the more common offending agents for incident neutropenia. The proposed mechanism for drug induced agranulocytosis is not fully understood, however likely involves an immune-mediated component. This is supported by the fact that in cases of repeat exposure after experiencing IDIA, the onset is often rapid due to the immune system’s memory. 4 Rawson and colleagues 5 conducted a retrospective evaluation of a national health database in Saskatchewan noting an incidence of agranulocytosis of 3.0 per million between 1982 and 1990 and found significant associations between antecedent use of sulfamethoxazole/trimethoprim, beta-lactam antibiotics, erythromycin, propranolol, antithyroid medications, and sulfasalazine. While additional population based and case-control studies have demonstrated an association between sulfamethoxazole/trimethoprim use and agranulocytosis, specific patient level details are not available to determine whether other contributing factors are present.6-8
Published cases of suspected sulfamethoxazole/trimethoprim associated agranulocytosis.
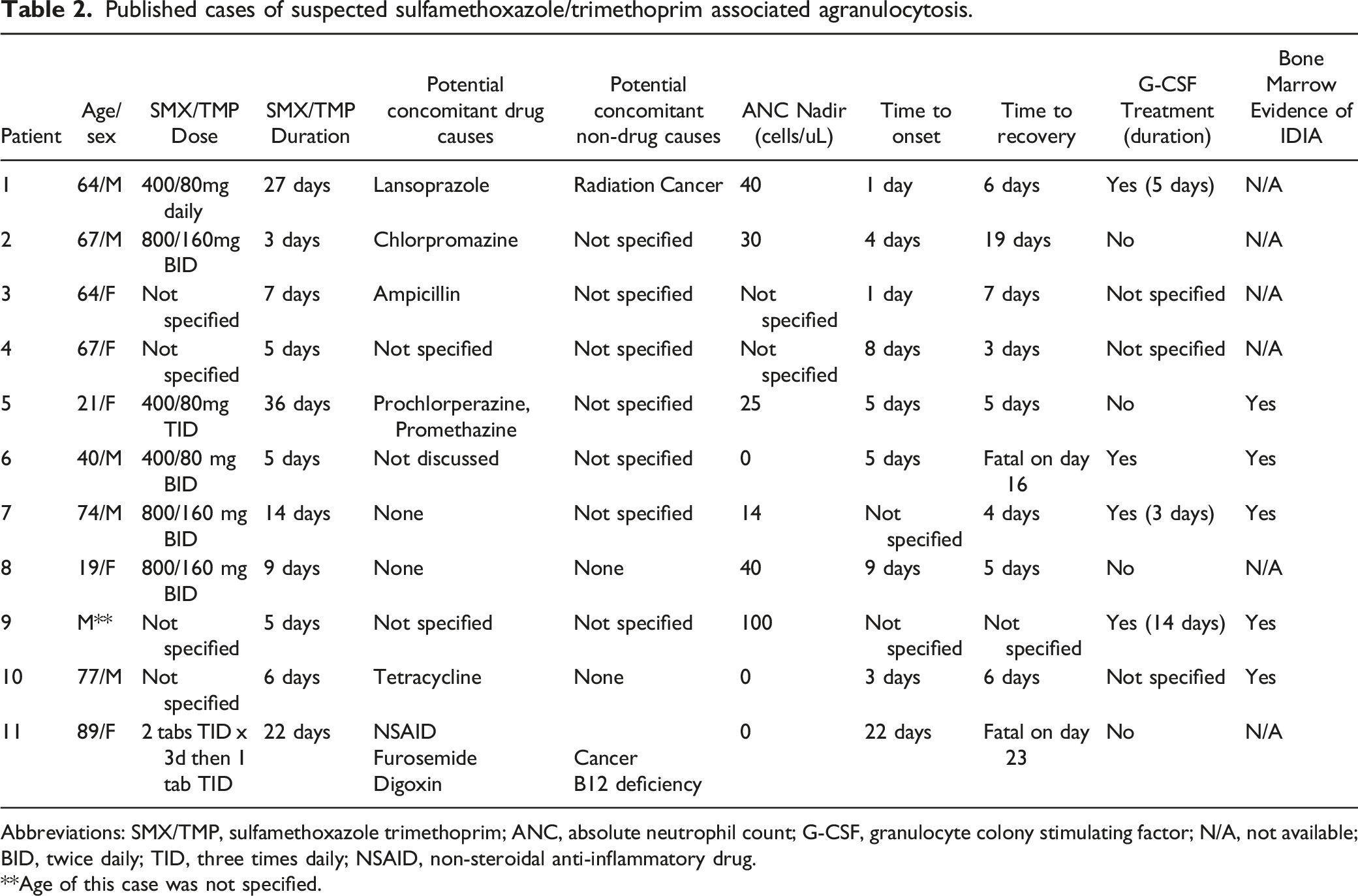
Abbreviations: SMX/TMP, sulfamethoxazole trimethoprim; ANC, absolute neutrophil count; G-CSF, granulocyte colony stimulating factor; N/A, not available; BID, twice daily; TID, three times daily; NSAID, non-steroidal anti-inflammatory drug.
**Age of this case was not specified.
We acknowledge that the patient did receive one dose of ceftriaxone at the start of his sulfamethoxazole/trimethoprim course and this drug has been associated with agranulocytosis. However, since only one dose was given approximately 19 days before presenting with agranulocytosis, it would be highly unlikely that this contributed given the existing literature that ceftriaxone-induced agranulocytosis occurs after prolonged courses with high cumulative doses.19,20 Also, this patient had a bone marrow biopsy and hematologist evaluation which helped to confirm a diagnosis of drug-induced agranulocytosis strengthening this case as a probable representation of sulfamethoxazole/trimethoprim IDIA.
Conclusion
We present a case of agranulocytosis after 10 days of sulfamethoxazole/trimethoprim therapy which resolved after drug discontinuation. After thorough review of published literature, our case shows consistency with available evidence as it relates to time to onset, and adds value for practitioners in that it represents a medically straightforward patient in contrast to many cases where multiple factors may have contributed to agranulocytosis. This case should caution providers to remain aware of the possibility of hematologic toxicity from sulfamethoxazole/trimethoprim, even in otherwise healthy, previously exposed individuals.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
