Abstract
Urinary tract infections are among the most common infections encountered in the clinic and remain a top indication for women to receive antibiotics. Delayed antibiotic prescribing and non-antibiotic symptomatic therapies are treatment paradigms common to other uncomplicated infectious diseases, such as upper respiratory infections. We aimed to review the literature on delayed antibiotics and non-antibiotic treatments as alternatives to immediate antibiotic prescriptions for uncomplicated cystitis. A literature search was performed in PubMed, Google Scholar, and Web of Science to identify relevant clinical trials and reference lists of included articles were examined to find additional studies. All published trials where same day treatment with antibiotics was compared to scenarios where antibiotics were intentionally delayed or withheld or where antibiotics were compared to non-antimicrobial agents or placebo were analyzed. A total of 13 articles were included. Trials were grouped into categories based on their comparator groups: placebo (n = 5), delayed antibiotic therapy (n = 3), and symptomatic treatment (n = 5). Antibiotic delay and ibuprofen, while less effective than antibiotics in early microbiologic and clinical cure, may still be considered plausible alternatives to immediate antibiotic treatment in non-pregnant women with cystitis. Day 7 and later symptomatic resolution ranges from 26-75% with delayed or placebo therapy and 70-83% with nonsteroidal anti-inflammatory drugs. Symptom improvement, however, may be delayed compared to immediate antibiotics. These approaches have shown to decrease antibiotic use in primary care settings with low rates of pyelonephritis. Methodology included in these studies may be considered in stewardship interventions for outpatient clinics.
Keywords
Introduction
Urinary tract infections (UTIs) are among the most common infections that result in antibiotic prescription. More than one third of females experience a UTI at some point in their lives and 74% of women who seek treatment for UTI symptoms will receive an antibiotic prescription. 1 Uncomplicated cystitis is usually diagnosed based on symptoms in the clinic setting, but diagnosis can be aided by urine dipstick test or urinalysis if there is clinical uncertainty or complicating factors.2,3
Antibiotic use for a variety of indications has increased drastically, with a total of 266.1 million prescriptions in the United States (US) in 2014 alone; over 30% of these outpatient prescriptions have been deemed unnecessary. 4 Overuse of antibiotics contributes both to adverse drug events and C. difficile infection as well as antibiotic resistance. In 2016, the Centers for Disease Control and Prevention (CDC) in the US released its Core Elements of Outpatient Antibiotic prescribing to promote antimicrobial stewardship in the clinic setting in order to conserve effectiveness of antimicrobials and prevent antimicrobial associated harms. 5 “Watchful waiting” or delayed therapy is 1 tactic to reduce antibiotic prescribing noted in the CDC recommendations. Delayed prescriptions to be filled at a later date if symptoms worsen or non-antimicrobial symptomatic treatments are tactics that have been used successfully to reduce prescription rates in primary care for acute upper respiratory infections. 6 A Cochrane review reported that there was no difference in clinical outcomes among delayed, immediate, and no prescribed antibiotics in patients with acute bronchitis. 7 This review also revealed that antibiotic exposure decreased significantly in the delayed prescribing groups when compared to immediate antibiotic prescription. When delayed prescription was used as an intervention for acute uncomplicated respiratory infections only 23% of patients used antibiotics compared to 91.1% for immediate antibiotic prescriptions. 8
This same strategy might be considered for uncomplicated UTIs to reduce antibiotic prescribing. Cystitis may not always require rapid treatment as symptoms may resolve spontaneously. 9 While the United Kingdom National Institute for Health and Care Excellence (UK NICE) guidelines incorporate a 48 hour delayed prescribing for treatment of cystitis in non-pregnant women in select situations, this approach is not mentioned in other UTI treatment guidelines. 10 Prior surveys of women with UTIs show that they are willing to delay treatment; 66% of women who have experienced uncomplicated cystitis reported that they would postpone antibiotic use due to the known risks of antibiotic treatment. 11 Furthermore while many health care providers are well educated on first-line antibiotic options, they are often are not aware of non-antibiotic treatment options for UTIs and rarely would delay antibiotic therapy. 12 Conversely, there have been recent reports of increased death related to delay of UTI treatment in UTI in elderly patients.13,14 The increase in mortality rate can be attributed to a significant increase in bloodstream infections which is why early initiation of antibiotics for UTIs in this population is recommended. 14
The purpose of this review is to provide an overview of the literature on non-antimicrobial initial approaches to uncomplicated cystitis in healthy adult females. This review may help healthcare providers provide the best approach to balance treatment success with reduction in antibiotic prescriptions.
Methods
Data Sources
A search was conducted using PubMed, Google Scholar, and Web of Science with no restriction on dates prior to October 2020. The following combinations of keywords were used: urinary tract infection, cystitis, AND delayed prescription, or AND antimicrobial stewardship, or AND placebo. The search was restricted to only articles in the clinical trial category available in English language. Articles found were then analyzed for content and additional articles were found through review of the literature citations.
Study Selection and Data Extraction
Inclusion criteria included clinical trials where same day treatment with antibiotics was compared to scenarios where antibiotics were intentionally delayed or withheld or where antibiotics were compared to non-antimicrobial agents or placebo. Articles not meeting criteria, but pertaining to antimicrobial stewardship or urinary diagnostics were reviewed as background information, but were not included in the review results. See Figure 1. Literature search method.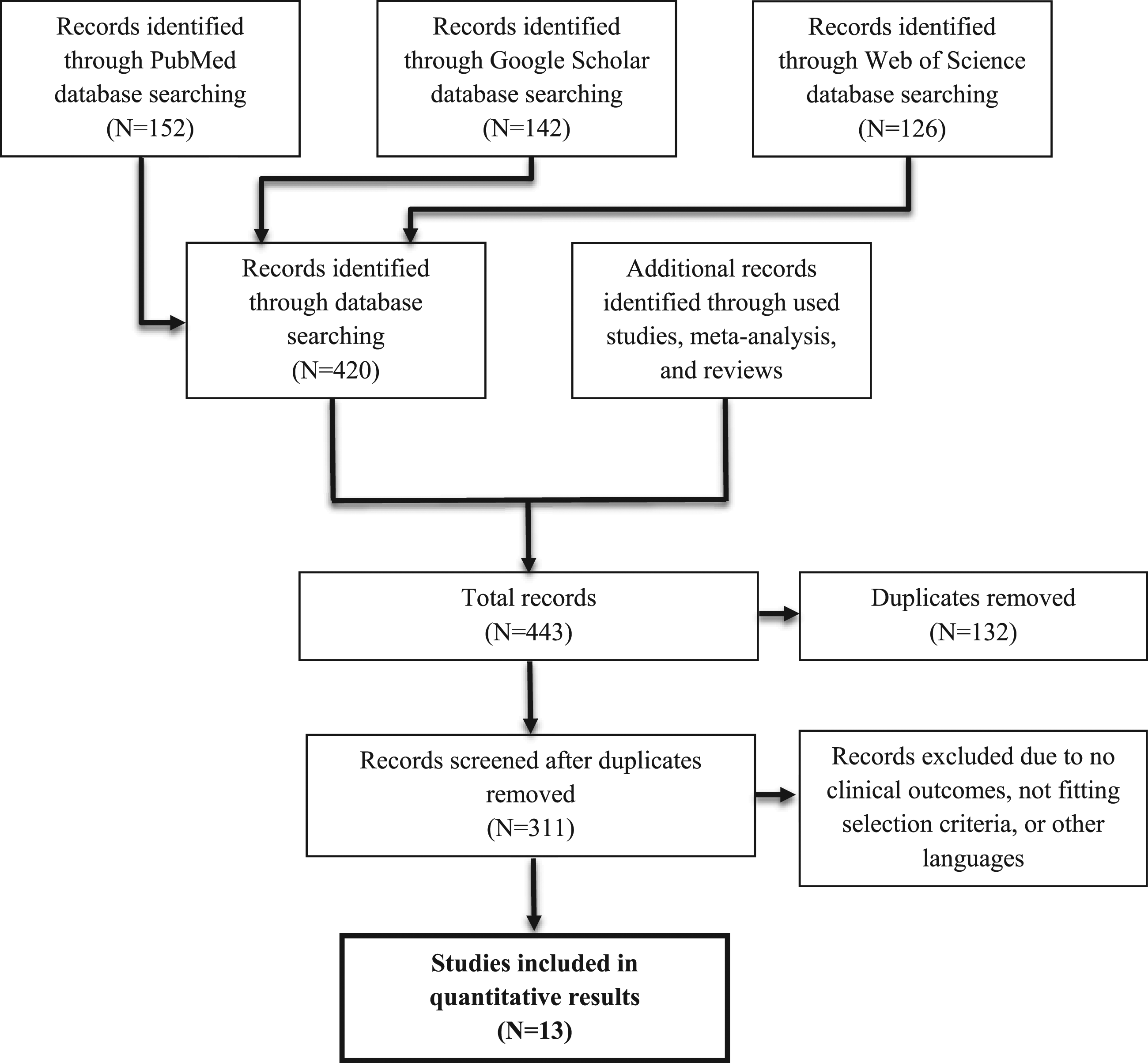
Results
Antibiotics vs Placebo
Results of 5 Trials of Antibiotics vs Placebo.
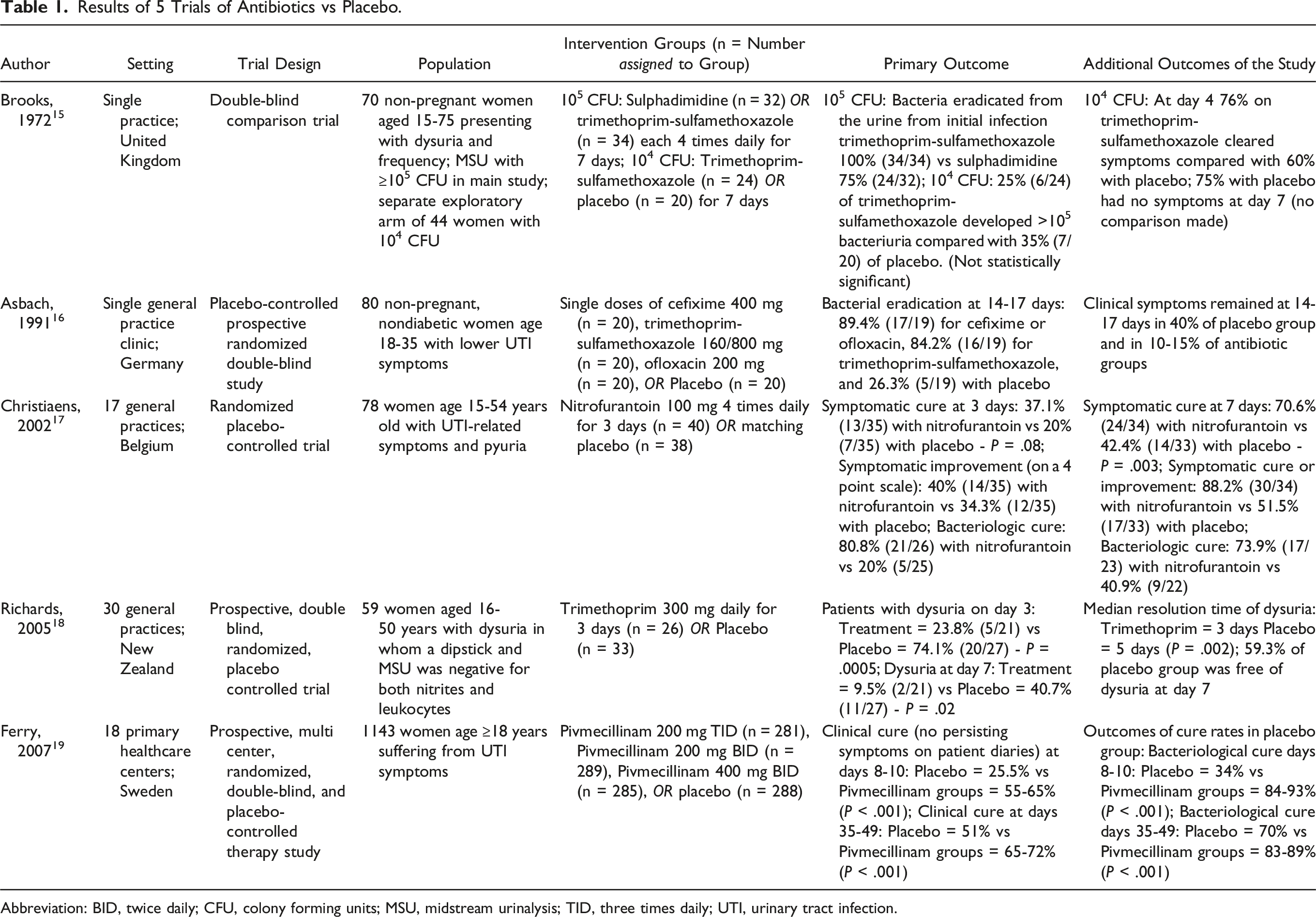
Abbreviation: BID, twice daily; CFU, colony forming units; MSU, midstream urinalysis; TID, three times daily; UTI, urinary tract infection.
Another placebo-controlled trial not included in the meta-analysis addressed treatment in the setting of diagnostic uncertainty. 18 The participants recruited for this study were women with variable UTI symptoms in whom a midstream urine was negative for both nitrites and leukocytes. These women were then randomized to receive trimethoprim for 3 days or a placebo. Urine microscopy and culture was also obtained, but this study focused largely on symptom resolution. Patients completed short written questionnaires at the time of sampling to provide severity of their symptoms. Among women reporting dysuria on randomization, those receiving trimethoprim had quicker resolutions of symptoms than with placebo (3 days vs 5 days, P = .002). At day 7, less patients were still experiencing dysuria in the trimethoprim arm than the placebo arm (10% vs 41%, P = .02). The median time to resolution and proportion of patients experiencing any other symptoms of UTI at day 3 and 7 did not differ. Only 26 participants ultimately had ≥20 leukocytes per milliliter in their urine on microscopy. Thus, many of the patients in this trial did not have a UTI but had a dysuria syndrome responsive to trimethoprim. One additional limitation was use of cranberry juice, urinary alkalinizers, and herbal remedies reported by some patients. Overall, among studies where placebo treatment was used, it is clear that antibiotics shorten duration of symptoms of UTI and significantly improve bacteriologic cure. While estimates vary considerably, placebo treatment still resulted in improvement of dysuria and other symptoms in 25.5 - 59.3% in those who were likely infected (105 colony forming units with symptoms) by day 7.
Immediate Prescription vs Delayed Prescription
Results of 3 Trials of Delayed Antibiotic Prescribing.
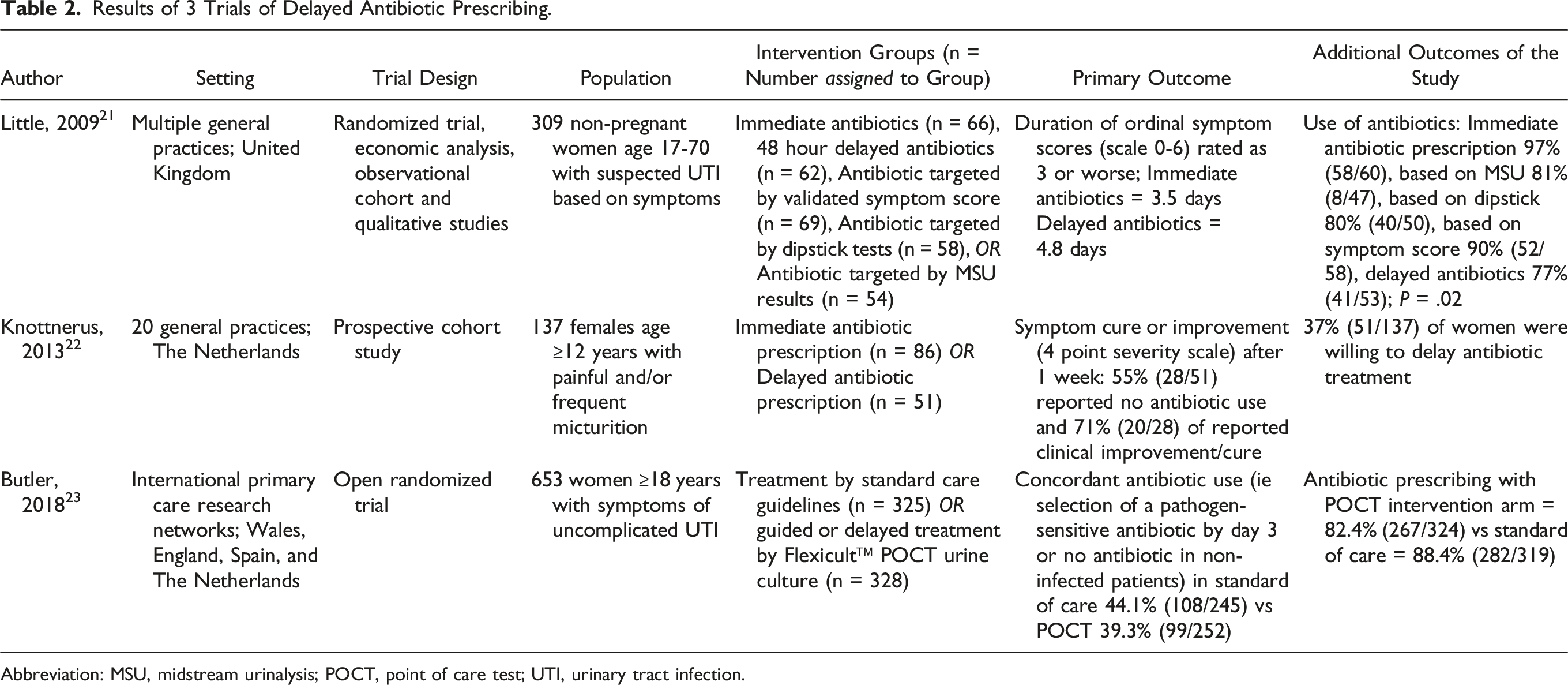
Abbreviation: MSU, midstream urinalysis; POCT, point of care test; UTI, urinary tract infection.
In the first of these, Little et al 21 conducted a series of 6 studies commissioned by the Health Technology Assessment Programme (part of the National Institute for Health Research in the UK); 1 of which was a randomized control trial that included a comparison of delayed and immediate antibiotics. Patients with symptoms of cystitis were randomized into 5 management groups: immediate antibiotics, 48 hour delayed antibiotics, antibiotic targeted by a symptom score, antibiotic targeted by dipstick test score, antibiotic targeted by midstream-collected urinalysis (MSU) results. The authors then measured the antibiotic usage between the patients with the least antibiotics ultimately being used by those who were randomized to 48 hour delayed therapy [immediate antibiotics 97%, MSU 81%, dipstick score 80%, symptom score 90%, and delayed antibiotics 77%; P = .02]. Patients kept a journal to grade the severity of their daily symptoms from 0 (no symptoms) to 6 (as bad as it could be). The average duration of symptoms rated “moderately bad or worse” in the immediate antibiotics group was 3.5 days; which was not significantly different between the antibiotic management strategies. However, patients who delayed antibiotics for 48 hours or longer had a 37% longer duration of symptoms they rated “moderately bad” (RR 1.37, 95% CI 1.11-1.68, P < .001). This study highlights that although duration of symptoms was longer, the use of antibiotics dropped 20%. Limitations to this study include imprecision of symptom scores due to a minority of patients not filling out symptom diaries as well as potential generalizability outside of the national health system of the UK. Furthermore, patients were advised to treat symptoms with over-the-counter remedies, bicarbonate, and cranberry juice. It is not clear how many patients used these options.
Knottnerus et al 22 conducted a cohort trial addressing the willingness of women with symptoms of cystitis to delay treatment as well as outcomes of delayed antibiotic therapy. One hundred thirty-seven women were asked to delay antibiotic treatment as long as they felt they could and 39 women were not asked to delay treatment. Importantly, results of the urinalysis were not available for 1 week. Thirty-seven percent of the women who were asked to delay treatment were willing to do so with 55% of these ultimately not using antibiotics 1 week after diagnosis. Of the women who had not used antibiotics 71% reported clinical improvement or cure of their symptoms. None developed pyelonephritis. More than a third of women with cystitis symptoms in this trial were willing to delay antibiotic treatment when asked by their physician. One major limitation to this study was that the diagnosis was not confirmed in many of the patients. Only 51% of those delaying treatment and 67% of non-delaying patients were found to have a positive culture once the culture became available. This however may demonstrate the utility of delayed therapy in settings of diagnostic uncertainty when point-of-care tests may not be available.
Butler et al 23 published a trial comparing the Flexicult (a point of care test (POCT) that provides urine colony quantification and susceptibility to common antibiotics) to MSU. Prescribers treating patients in the POCT group could prescribe empiric antibiotics and review the POCT results the following day or delay prescribing until the following day when results are available or prescribe a delayed prescription to be taken at a later date based on the results. The primary outcome was to determine antibiotic use concordant to the diagnosis and antimicrobial sensitivity. Antibiotic use concordant with sensitivities and diagnosis at day 3 was similar in the standard and the POCT intervention arm. Interestingly, the POCT group (where delayed treatment was an option) had less prescribing of antibiotics at the initial consultation (82.4% vs 88.4%; OR .56, 95% CI [.35 to .88]). The major limitation for consideration is that the intervention group included both delayed and empiric prescribing approaches. The authors note that while the difference in antibiotic exposure was small, delaying antibiotics at least until POCT results are analyzed might decrease rates of discordant empiric therapy particularly if uropathogen resistance is a local problem or the diagnosis is in question. Taken together, these 3 studies appear to show that delayed therapy for treatment of cystitis appears to be a safe way to decrease outpatient antibiotic use and may be an option particularly if there is a level of diagnostic uncertainty and clinical follow up is possible.
Immediate Antibiotic Prescription vs Symptomatic Therapies
Results of 5 Trials of Symptomatic Therapies vs Antibiotics.
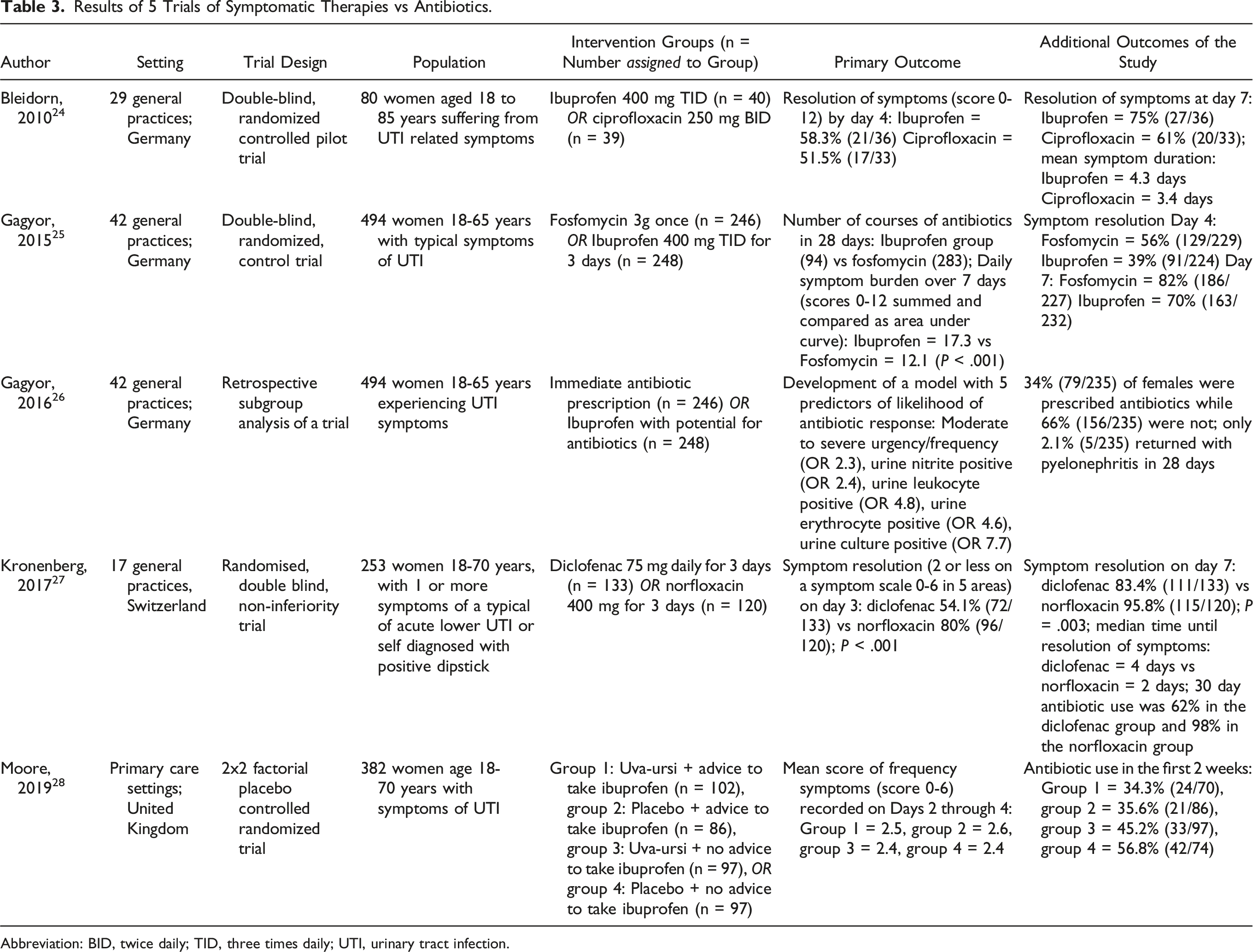
Abbreviation: BID, twice daily; TID, three times daily; UTI, urinary tract infection.
One trial conducted by Bleidorn et al 24 focuses on the difference in symptomatic treatment with ibuprofen and ciprofloxacin. At inclusion, patients completed a subjective symptom score: severity of dysuria, frequency, and low abdominal pain all scored from 0 (not at all) to 4 (very strong) and then summed for a total of 12 points. The patients were randomized into treatment with ciprofloxacin or ibuprofen for 3 days. By day 4, 58.3% of patients in the ibuprofen group and 51.5% in the ciprofloxacin group were free of symptoms, which was not statistically significant. Negative urine cultures occurred more often in the ciprofloxacin group, but this was also not statistically significant. About two-thirds of patients presenting with cystitis symptoms seemed to recover without antibiotics and 1 third of the patients in the ibuprofen group returned for ongoing or recurring symptoms within the first week. The main limitation of this study is it was a pilot study and may not have been sufficiently powered.
Another trial investigated the willingness of women to delay treatment as well as predicting antibiotic use after symptomatic treatment. 25 Gagyor et al developed a prediction model to help determine if which patients suffering from an uncomplicated cystitis could be treated symptomatically; 156 women received ibuprofen and 79 received antibiotics. The final model included 5 predictors for the need of antibiotic treatment: moderate-to-severe urgency or frequency, positive dipstick test results for erythrocytes, leucocytes, and nitrite, and impairment of daily activities. While this study was meant to test the model’s predictive ability, readers can recognize some benefits of a non-antibiotic approach. Only 34% of females in the ibuprofen group were ultimately prescribed antibiotics within 28 days, but 14% who were treated with antibiotics at presentation also required additional antibiotic prescriptions.
A study conducted by Gagyor et al randomized women to fosfomycin once or ibuprofen for 3 days. 26 The trial had 2 major endpoints: the total number of courses of antibiotics on days 0-28 and burden of symptoms on days 0-7. The number of courses of antibiotic treatment within 28 days was significantly lower in the ibuprofen group. The number of all courses in the fosfomycin group was 283 compared with 94 in the ibuprofen group. This corresponds to an incidence rate reduction of 66.5% (P < .001). Women however that were in the ibuprofen group had a mean duration of symptoms 1 day longer than the fosfomycin group. On day 7 only 70% were symptom free compared to 82% fosfomycin being symptom free. There were 5 cases of pyelonephritis among those taking ibuprofen and 1 in the fosfomycin group (P = .12). One patient in the ibuprofen group was admitted to the hospital for gastrointestinal bleeding. Overall, two-thirds of the women who were in the ibuprofen group initially fully recovered with no antibiotic use. This study highlights that in most cases of uncomplicated cystitis if the patient is willing, symptomatic treatment is effective in both resolving symptoms as well as reducing antibiotic use. A major limitation was that inclusion was biased towards patients with less severe symptoms. This could be caused by self-selection, with women with more severe symptoms opting out of the possibility of not receiving antibiotics, which is a clinically-relevant scenario.
A similar trial by Kronenberg et al 27 randomized women with uncomplicated cystitis symptoms to receive diclofenac or norfloxacin for 3 days. Patients were also given a rescue dose of fosfomycin to take at their discretion if their symptoms had not improved after 72 hours. Subjects were asked to rate their severity on a symptom scale in 5 areas on a daily basis. The primary outcome was symptom resolution on day 3 and secondary outcomes included use of antibiotics in 30 days. Norfloxacin symptom resolution was superior in symptom resolution by day 3 (54% vs 80%; P < .001), but at day 7 symptom resolution was as high as 83% in the diclofenac group and was no longer statistically significant at day 10. While the use of antibiotics was lower in the diclofenac group (62% vs 98%, P < .001), many subjects in the diclofenac group took the rescue fosfomycin provided to them by the researchers and did so before 72 hours. Similar to other trials, symptoms responded faster with antibiotics (median 2 days vs 4 days) and mean symptom scores were no longer significant at day 10. Six patients (4.5%) developed pyelonephritis in the diclofenac group compared with none in the norfloxacin group (number needed to harm = 23) and median time to diagnosis in these patients was 5.5 days. A majority of patients ultimately took antibiotics in the first 30 days. A secondary analysis of these patients that took any antibiotic showed greater improvement in symptoms in the diclofenac arm. It is not clear why pyelonephritis was higher in the diclofenac group when this finding is inconsistent with prior ibuprofen trials and even placebo control trials. The authors noted that pretreatment C-reactive protein levels were higher (>10 mg/L) in patients that developed pyelonephritis and high C-reactive protein levels were seen in 50% of those that developed pyelonephritis in the diclofenac group. While this was only an exploratory outcome, it may have also indicated that these patients should be excluded from non-antibiotic treatment as it may have been an early signal of a developing pyelonephritis. It must also be noted that a potential limitation in assessing this trial is early termination of trial recruitment (37% less participants than expected) and acceptance of a lower statistical power may result in a less precise estimate of outcomes.
One final trial examined Arctostaphylos uva-ursi leaf extract, a plant used commonly in western herbal medicine with antimicrobial properties, and ibuprofen as alternative treatments for uncomplicated urinary tract infection in women in the UK. 28 The patients were separated into 4 groups; see Table 3. Each participant was asked to keep a diary recording their symptom severity. The primary endpoint was symptom severity on days 2 through 4. After adjusting for baseline frequency symptom severity, there was no evidence of a difference in symptom severity between any of the factorial groups. Antibiotic use ranged considerably from 34.3% to 56.8% in the 4 groups. Logistic regression showed patients receiving ibuprofen were less likely to need antibiotics (OR .27 [95% CI .10-.72; P = .009]), but the Uva-ursi group was not (OR .59 [95% CI .22-1.58; P = .293]).
Overall, these 5 trials show that NSAIDs may not decrease UTI symptoms as quickly as immediate use of antibiotics and outcomes at day 7 are mixed. While the use of NSAIDs significantly decreases antibiotic use, higher rates of pyelonephritis in patients receiving diclofenac in the Kronenberg et al trial show a need for cautious follow up.
Discussion
While an initial approach with antibiotics appears to improve cystitis symptoms more rapidly and resulting in higher rates of clinical cure in most trials, the response to non-antibiotic approaches is surprisingly high. In the trials that reported day 7 symptom resolution, 26-60% resolution was seen with placebo (excluding “non-infected” cases), 71% with delayed therapy, and 70-83% with NSAIDs. Because of high rates of symptom resolution with non-antibiotic treatments, clinicians should include uncomplicated cystitis among the other conditions where “watch and wait” approaches are used such as bronchitis, sinusitis, and diverticulitis. The combination of the self-limiting nature of many cases of cystitis, the high acceptability of the delayed approach among women, and the low incidence of progression to pyelonephritis seen in these trials, the balance of risks and benefits lends credibility to the initial “watch and wait” approach. 11 There are multiple scenarios in which this approach may provide utility including antimicrobial stewardship initiatives in clinics which aim to reduce antibiotic use and harm, situations of diagnostic uncertainty, or communities with existing high rates of uropathogen resistance result in ineffective empiric therapies.
Study Limitations
While most of the included studies show that delayed therapy and ibuprofen are safe and decrease antibiotic use in uncomplicated UTI, the body of literature is not without limitation and requires consideration when applied to clinical practice. For example, only 8 of the 13 studies reviewed included a formal power analysis or sample size calculation. Some NSAID trials also allowed for rescue antibiotics. Both of these factors could potentially lead to type II errors in comparison to standard antibiotic therapy and a decreased ability to detect small incidences of adverse outcomes. However, when analyzing these studies, generalizability to current practices, issues with diagnostics, and patient demographics must also be considered.
Generalizability
A major limitation of the present literature is generalizability. Every article which met inclusion criteria was conducted in a few European countries or New Zealand leaving practice patterns of many other countries unrepresented. Due to the limited locations of these trials, it is impossible to ignore the impact of extraneous variables such as demographics of the females being treated and the practice patterns in these countries. Additionally, many of the regimens used in these studies are not recommended in the 2010 Infectious Disease Society of America (IDSA)/European Society for Microbiology and Infectious Diseases (ESCMID) guidelines. 29 The antibiotics with the highest level of evidence for uncomplicated cystitis in females according to these guidelines include nitrofurantoin (5 days) and Trimethoprim-sulfamethoxazole (3 days). Other alternatives with lower evidence ranking due to lower efficacy noted in the guidelines include fosfomycin, quinolones, select beta-lactam agents, and pivmecillinam. Pivmecillinam’s availability is limited largely to European countries. Two of the studies focused on the use of ciprofloxacin which has a high risk for collateral damage.24,26 Another utilized trimethoprim without sulfamethoxazole and another used nitrofurantoin for only 3 days; all would be considered suboptimal management contemporarily.17,18 Broad resistance of E. coli in North America to trimethoprim-sulfamethoxazole and quinolone antibiotics may further shift practice patterns away from these agents. Indeed, some of the more recent trials included in this review demonstrated quinolone enterobacterales resistance of >30%. 27
Inconsistent Diagnostics
Among the reviewed studies, urinary symptoms were the main driver for inclusion in the trial. Of the 13 studies reviewed, diagnostics affected the inclusion criteria in 4. Studies either used MSU or urine dipstick (dipstick = 3, MSU = 5, both = 5) for diagnostic confirmation although often the results did not impact the inclusion criteria of participants. Diagnostic thresholds from MSU varied from 102 to 105 colony forming units. One trial even considered 104 colony forming units non-infected regardless of symptomatology. 18 Thus, the reviewed trials likely report a high proportion of patients without UTIs, but with other self-limiting conditions contributing to urinary symptoms. In fact, 1 study found that despite classic urinary symptoms only 32% of the participants enrolled had a confirmed diagnosis. 30
Combining the results both positive leukocyte esterase and nitrites increases dipstick sensitivity (68 to 88%), but has variable effects on specificity depending on the population. 31 Similar studies have assessed the effectiveness of MSU and discovered that in the primary care setting, the sensitivity was found to be 73% (95% CI 66-80) and the specificity 94% (95% CI 88-98). [26] Guidelines differ on when MSU with culture and dipstick tests should be used. The European Association of Urology (EAU) guidelines weakly recommend a urine dipstick test in the setting of uncomplicated cystitis, noting that when patients present with typical symptoms of cystitis there may be little additional diagnostic yield. The role of MSU with culture is limited to symptoms of pyelonephritis, pregnant women, atypical symptoms, or unresolved symptoms. 2 The UK NICE guidelines suggest that MSU or urine dipstick before prescribing antibiotics is considered for children under 16 years of age.
Variability in testing guidelines may lead to unnecessary treatment in symptomatic, uninfected patients, but overreliance on MSU results may also lead to overtreatment in asymptomatic patients. In fact, unnecessary treatment of asymptomatic bacteriuria may be as high as 45%. 32 Urinary diagnostic stewardship measures should be applied in both the inpatient and clinic setting to ensure treatment in only those patients that require it. 30 Delayed treatment policies such as those advised in the UK NICE guideline, also provide time for further investigation in settings of diagnostic uncertainty. 10
Age and Sex Considerations
The mean age of the articles analyzed in the results was 40.4 years of age with the minimum age being 12 years old and the maximum being 85.22,24 All studies excluded males as well. While males are less likely to experience UTIs, they are generally excluded from uncomplicated UTI definitions. Results for these studies therefore can only be applied to women of child-bearing age and middle aged-women and cannot be applied directly to the elderly, children, or males.
There have been a variety of studies that investigate the delay in UTI research in the elderly population. One retrospective population-based cohort study found that outpatients over the age of 65 (mean age = 76.7 years) were significantly more likely to experience a bloodstream infection when antibiotics were delayed or not prescribed at all compared to same day antibiotics. 14 The risk of all-cause mortality was significantly higher with deferred antibiotics and no antibiotics than with immediate antibiotics at any time during the 60 days follow-up (adjusted HR 1.16, 95% CI 1.06 to 1.27 and 2.18, 2.04 to 2.33, respectively). Another similar study found that the risk of bloodstream infections was not increased, but mortality rates increased when treatment was delayed. 13 This is an issue of considerable consequence as urinary diagnostics in older patients can be compromised due to higher rates of bacteriuria and pyuria and because atypical symptoms such as delirium are more common in elderly. Clearly, delirium alone is an inadequate symptom on which to base a diagnosis of UTI. 33 Dasgupta and colleagues 31 in fact found that antibiotic treatment of asymptomatic bacteriuria in older inpatients with delirium was associated with worse outcomes than those that were not treated. In inpatient settings or situations of diagnostic uncertainty delaying treatment in older patients with delirium at least until test results are available to be analyzed is necessary.
There is scant data regarding impact of delayed therapy in children with UTI. 34 A study conducted by Hewitt et al examined 287 children with confirmed pyelonephritis. The children were retrospectively compared as a control group who received treatment initially and a group who received progressive delays in antibiotic treatment of acute pyelonephritis from less than 1 day to 5 days after the onset of fever. The results of this study showed that incidence of renal scarring remained consistent at (30.7 ± 7%). 34 Additional studies are needed to determine the impact of delay of antibiotic therapy in children under 12 years of age.
Application to Patient Care and Future Research
Antibiotic treatment recommendations for uncomplicated cystitis range from 3-7 days. 29 Future treatment guidelines should include delayed therapy with or without ibuprofen, although other NSAIDs cannot be recommended at this time. Such a recommendation for delayed therapy can only be applied to cystitis in the primary care setting in women of childbearing age who are agreeable after proper counseling and can be contacted for follow-up. Consistent with these studies, follow up should occur in 48-72 hours and should include careful screening of symptoms of pyelonephritis as this may present by day 5. For those patients where ibuprofen is initiated, clinicians should recognize that classic symptoms such as fever and flank pain may be diminished. Delayed antibiotic therapy should make use of a post-dated prescription or a call before filling strategy. As seen in 1 study, providing antibiotics at the initial encounter resulted in higher rates of antibiotic exposure than intended. 27 Furthermore, filling antibiotics that are not immediately used may contribute to hoarding and inappropriate use for other infections.
As with other self-limiting infections that may respond without antibiotics, identification of the precise population for the “watch and wait” approach may be needed. Predictive tools for which individuals would benefit from symptomatic treatment as illustrated by Gagyor et al could be useful in the clinic setting.
26
Future research should focus on expanding antimicrobial stewardship initiatives for UTI treatment. Due to the frequency of UTIs in the outpatient setting, the impact of delayed therapy may be an option to decrease antibiotic use and subsequent resistance and C. difficile infections in the community. A potential algorithm for clinicians to consider is in Figure 2. Such a treatment pathway may decrease antibiotic exposure for patients where the infection is uncomplicated or the diagnosis is uncertain. Additionally, if communities experience exceptionally high rates of uropathogen resistance to typical first line antibiotics, culture results prior to delayed antibiotic initiation may become a necessary treatment strategy. Treatment of cystitis may also be amenable to interventions such as shared decision-making tools which may positively impact both patient and prescriber while decreasing antibiotic use.
35
Furthermore, future studies should determine the role of delayed therapies in additional countries where antibiotic overuse might see a greater impact. Antibiotic selection and patient acceptability of delayed antibiotics or non-antibiotic options may differ in other cultures and local health-systems may not always have adequate infrastructure to ensure appropriate patient follow up. Proposed algorithm to reduce antibiotic use in outpatient cystitis.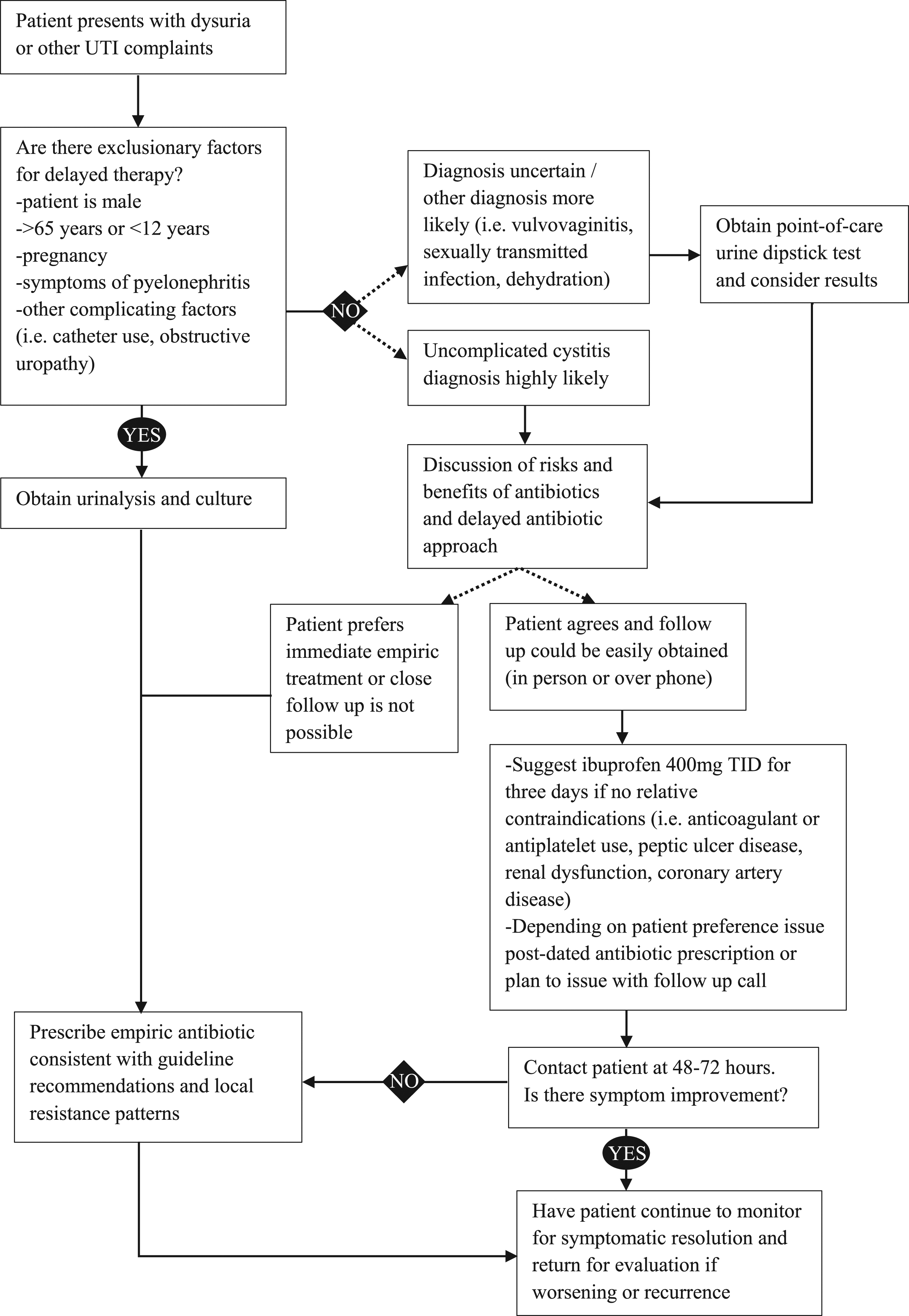
Conclusion
Antibiotic delay and ibuprofen are safe alternatives to immediate treatment with antibiotics in non-pregnant women with cystitis. While symptom improvement may be delayed, infected symptomatic patients who receive placebo will have symptom resolution in as much as 60% of cases by day 7. Those that receive a delayed therapy approach tend to experience a slightly higher burden of early symptoms, antibiotic use is ultimately decreased. Those receiving NSAIDs such as ibuprofen also have slower symptom improvement, but by day 7 will have symptom resolution in 70-83% of cases. These approaches can decrease antibiotic use in the primary care setting with minimal increase in pyelonephritis and may be considered as stewardship interventions for outpatient clinics.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
