Abstract
Introduction
Cardiovascular diseases are the leading cause of death in individuals aged above 40 years of age. 1 Low density lipoprotein cholesterol (LDL-C) has been implicated as a risk factor for cardiovascular events and LDL-C lowering has proven beneficial in prevention and recurrence of cardiovascular events.2,3 HMG-CoA reductase inhibitors or statins have been the mainstay of lipid lowering therapy (LLT) for several decades, although new therapies for LDL-C reduction were approved in the past few years. 4 Results from a nationally representative study evaluating statin use in the year prior to the 2013 American College of Cardiology/American Heart Association (ACC/AHA) guideline changes found that 28% of adults older than 40 years of age were on a statin. 5 The 2013 ACC/AHA guidelines expanded the eligibility of patients recommended to be on a statin by shifting the focus on patient groups that would benefit from statin based on their risk factors rather than LDL-C values. 3
The guideline approved LDL-C lowering non-statin drug classes include cholesterol absorption inhibitors (ezetimibe), bile acid sequestrants, and proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors. 6 The lipid lowering treatment approach has been effective over the past decade with approval of newer treatments and updates in guideline recommendations. 4 PCSK9 inhibitors were approved by the Food and Drug Administration (FDA) in 2016 and at the same time ezetimibe also became available as a generic drug in the US. Inclisiran is a small-interfering RNA treatment approved in the European Union in 2020 for the treatment of hypercholesterolemia or mixed dyslipidemia. The recommended targets for LDL-C have reduced and are based on the completion of large-scale randomized clinical trials that have demonstrated reduction in cardiovascular risk with mean LDL-C achievement as low as 30 mg/dl in the trials.7-9 Results from these trials have been incorporated in the 2018 ACC/AHA guidelines for cholesterol management.10-13
The 2018 ACC/AHA guidelines identify patient management groups for statin therapy which include (a) patients with atherosclerotic cardiovascular disease (ASCVD) (b) patients with severe hypercholesterolemia (LDL-C ≥ 190 mg/dl) (c) patients aged 40-75 with diabetes mellitus and (d) primary prevention adults aged 40 -75 years with an intermediate 10 year ASCVD risk. 6 For patients over 75 years of age, the guidelines recommend continuation of LLT in patients with ASCVD or diabetes who are already on therapy. However, initiation of therapy is recommended only after a discussion of risk and benefits of treatment, and after taking into account patient frailty and preferences. 6
Older adults are especially vulnerable to cardiovascular events and the consequences may be more severe. 14 Two separate meta-analyses of randomized controlled trials (RCTs) found using statins reduced cardiovascular events by 24% to 26% irrespective of age in patients > 75 years.15,16 A real-world study also demonstrated the reduction of all-cause and cardiovascular mortality among older adults using statins for primary prevention. 17 Studies have reported a steady increase in the use of statins among older adults.18,19 In addition, patients above 75 years reported more adverse events to statin than those below 75 years. 20
Existing studies in adults aged 40-65 and older adults >65 years have evaluated the use of specific LLT or focused on specific subgroups such as patients with diabetes, myocardial infarction, or ASCVD.21-23 Due to the potential of bias, most studies have focused on understanding utilization among new users of any LLT.24-28 There is a need to evaluate the use of both statin and non-statin LLT among all older adults, not limited to a certain risk group. More recent data would reflect current clinical practice since the availability of new therapies and publication of their trial results.
Similar to medication use in other chronic diseases, in the real-world setting, LLT may undergo interruptions, switching, dose adjustments, reduction or augmentation of therapy.26,29,30 These modifications correspond to factors such as: side effects or poor tolerance of LLT, need for additional LDL-C lowering, achievement of LDL-C threshold, and formulary changes.31,32 LLT modifications are common and can impact adherence and LDL-C goal achievement. 29 Treatment patterns in older adults are likely to differ from younger adults due to differences in guideline recommendations, increased medication complexity, and altered metabolism in older adults. Thus, in addition to LLT use, assessing treatment patterns in older adults is needed to understand the clinical practice and treatment landscape.
The objective of this study was (a) to describe the utilization of different guideline recommended LLTs and their combinations in older adults and (b) identify the modifications in LLT in older adults (age ≥ 65 years) as well as in the subgroup of patients aged 75 years or older.
Materials and Methods
Study Design and Data Description
A retrospective cohort study was performed utilizing administrative claims data from a regional Medicare Advantage Plan between January 1, 2016 and May 30, 2018. The data included patient health plan enrollment information, patient demographics, medical claims from outpatient visits, and pharmacy claims. The medical files included diagnostic information in the form of ICD-10-CM codes and procedure information as CPT codes. The pharmacy records contained information about drug names, fill dates, dosage information, and days supplied for each prescription. The study was approved by the IRB with a waiver of informed consent.
Patient Selection
This study identified patients with a prescription of any guideline recommended LLT between January 2017 and June 2017. This 5-month duration was the identification period. This period was based on the data availability (January 2016 – May 2018) and the minimum enrollment period required by the inclusion criteria below. The index date was the date of the first prescription for each patient in the identification period. The 1-year period prior to the index date was the pre-index period and the 1-year following index date was the follow-up period. Patients with cirrhosis, rhabdomyolysis, and end stage renal disease (ESRD) were excluded from the study because of additional treatment considerations specific to these conditions. Patients without continuous enrollment in the plan during the pre-index period as well as patients under the age of 65 were excluded from the study. As patients were followed-up for one year after the index date, patients without continuous enrollment in the follow-up period were also excluded from the study. Patients on multiple statins concurrently or three or more LLT at baseline were excluded. The study design is schematically represented in Figure 1. Schematic representation of study design.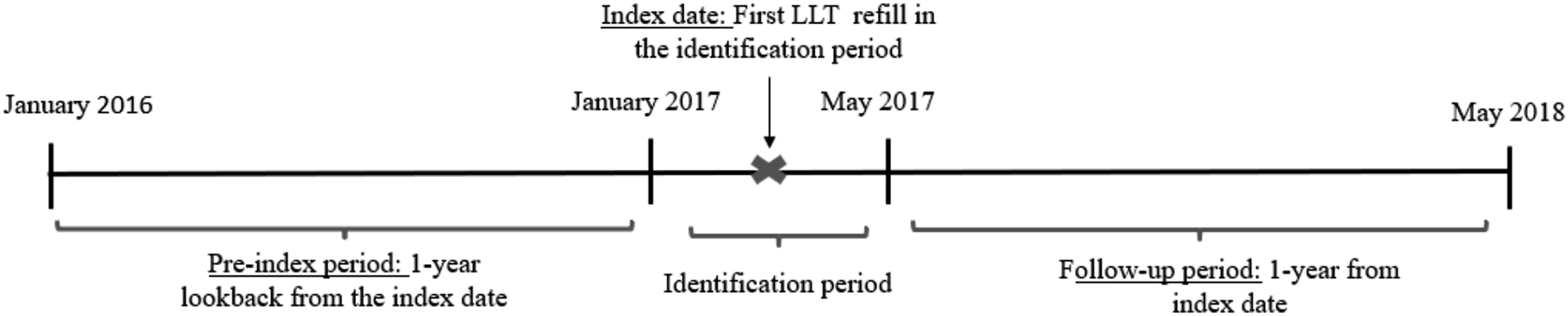
Study Measures
Identification of Baseline Therapy
The type of LLT used by the patients was identified on the index date and was referred to as baseline therapy. The guideline recommended LLT classes evaluated in this study included statins, ezetimibe, bile acid sequestrants, and PCSK9 inhibitors and have been listed in the online supplementary material. Some of these therapies are prescribed in combination, mainly with statins to provide additional LDL-C lowering. Therefore, patients could be on multiple drugs at baseline. The online supplementary material contains details about the algorithm used to identify patients with multiple baseline LLT. Baseline use of triglyceride lowering therapy was also captured.
LLT Use Type
Patients without any LLT prescriptions in the pre-index period were classified as new users of LLT. 26 Patients with prior LLT claims were classified as prevalent or existing users. As new users would not have any pre-index treatment patterns and were expected to be different as compared to patients already on LLT, all treatment modifications were stratified by LLT user type.
Measures of Treatment Modification
The treatment modification in the pre-index and follow-up period closest to the index date was identified.
Up-titration: Measures aimed to increase the potency of the LLT taken by the patient were categorized as up-titration. These include intensification of the statin, increase in the dose, and therapy augmentation as further described.
Dose intensification: This modification could only occur with statin medications based on their classification in the ACC/AHA guidelines. The guidelines identify statins as being low, moderate, and high intensity statins based on the drug and its daily dose. 6 Intensification was the escalation in the intensity (low to moderate, moderate to high, or low to high) of the refilled statin.
Dose increase: Defined as an increase in daily dose of the LLT. If a change for a statin drug was identified as an intensification, then the same change was not considered as a dose increase.
Treatment augmentation (add-on): Use of a new LLT with continued use of the previous LLT. Continuous use was captured as a subsequent fill of the index LLT after starting the new LLT.
Down-titration: Measures aimed to decrease the potency of the LLT taken by the patient were categorized as down-titration. These include statin intensity reduction and dose decrease.
Intensity reduction: Like intensification, this modification could only occur with statin treatment and was defined as a reduction in intensity (moderate to low, high to moderate, or high to low) between consecutive claims for the statin.
Dose decrease: Defined as decrease in the daily dose of the therapy that the patient was using at baseline. A decrease in intensity of statin was not considered as a dose decrease.
Switching: At least one claim for a LLT other than the index drug(s) without continued use of the previous therapy. Switching in the follow-up period was evaluated in further detail as (a) switching from a statin to another statin and (b) switching from a statin therapy to a non-statin therapy or vice versa. Additionally, if a change for a statin drug was identified as an intensification, then the same change was not considered as a switch.
Interruption of therapy: Defined as a gap of ≥ 90 days without any LLT following the end of days of supply from the previous claim. 26
Treatment discontinuation: Defined as the complete discontinuation of therapy in the follow-up period.
In the patterns mentioned above, specifically intensity changes, dose changes, and switching which required discontinuation of previous therapy and initiation of a new therapy, the previous therapy could either be discontinued prior to or within 90 days after initiation of the new therapy. As most LLT are filled as 90-day refills, a 90-day cutoff was used in the treatment pattern definitions.
Statistical Analysis
All baseline demographics and comorbidities were identified for patients in the pre-index period to describe the characteristics of the cohort. Chi-square tests were used to compare the treatment modifications among new and prevalent users. Additionally, treatment modifications were stratified in patients ≥ 75 years of age and compared using chi-square tests to patients 65-74 years of age. The treatment mapping algorithms and descriptive statistics were analyzed using Statistical Analysis Software, version 9.4 (SAS Institute Inc., Cary, NC).
Results
There were 34,506 patients with any LLT use between January 2016 and May 2018 identified in the Medicare Advantage Prescription Drug Plan. Of these, 23,112 patients had a LLT prescription in the identification period, between January and May 2017. Figure 2 represents a flow chart of patient attrition based on the exclusion criteria after which 14,360 patients were included in the final cohort. Flow chart of final cohort derivation.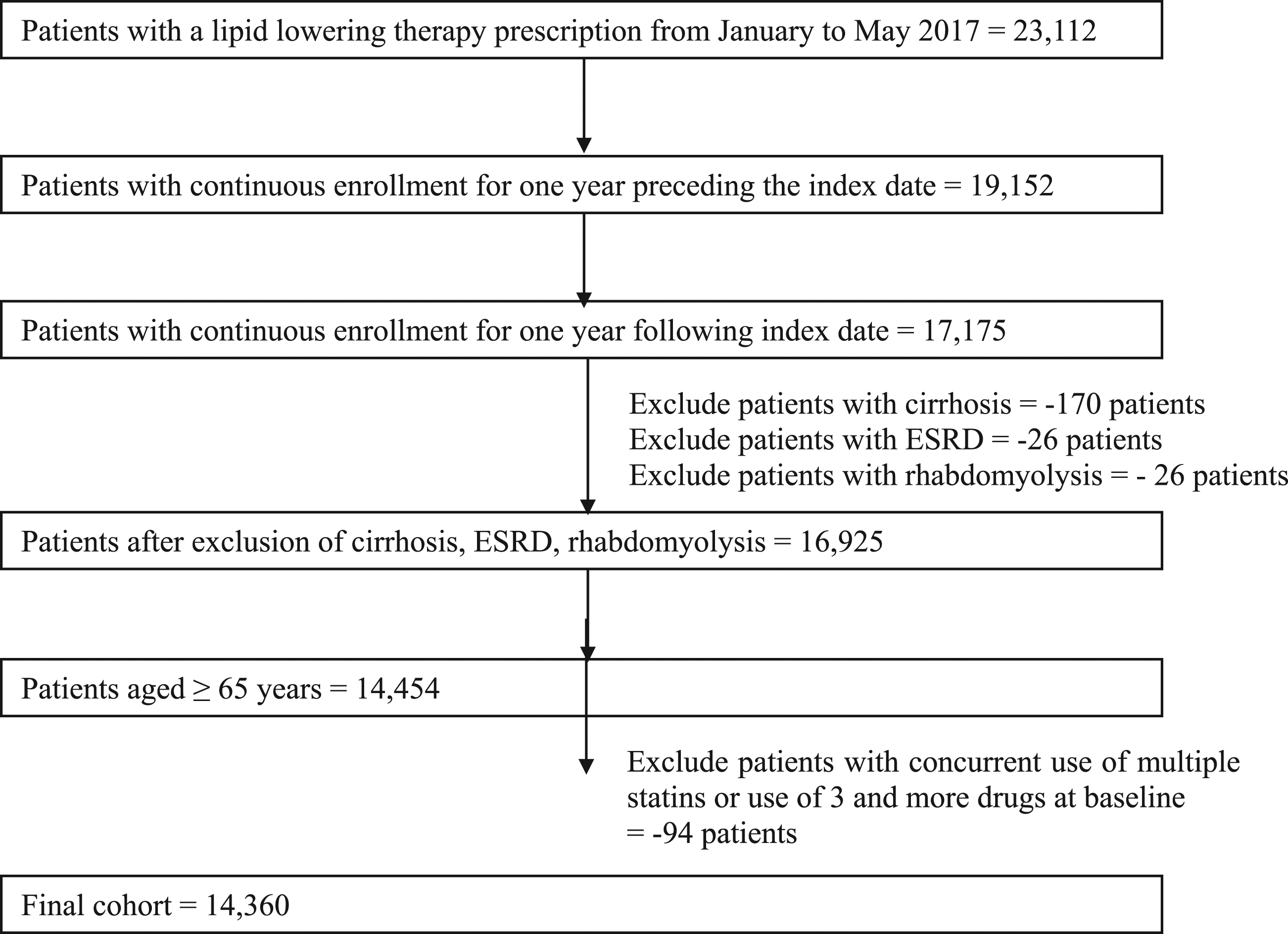
Baseline Demographics and Clinical Characteristics of the Entire Sample of 14,360 Patients.
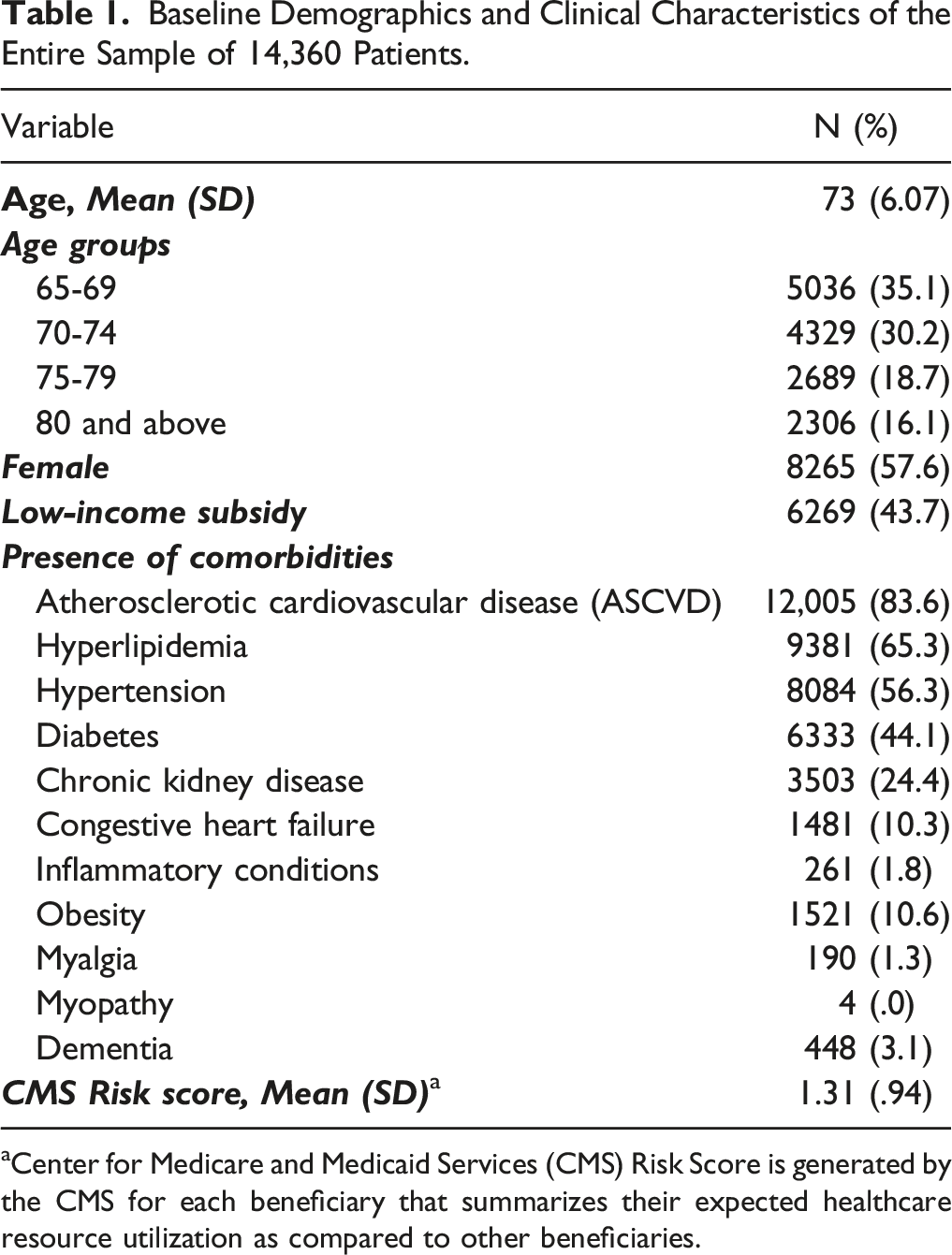
aCenter for Medicare and Medicaid Services (CMS) Risk Score is generated by the CMS for each beneficiary that summarizes their expected healthcare resource utilization as compared to other beneficiaries.
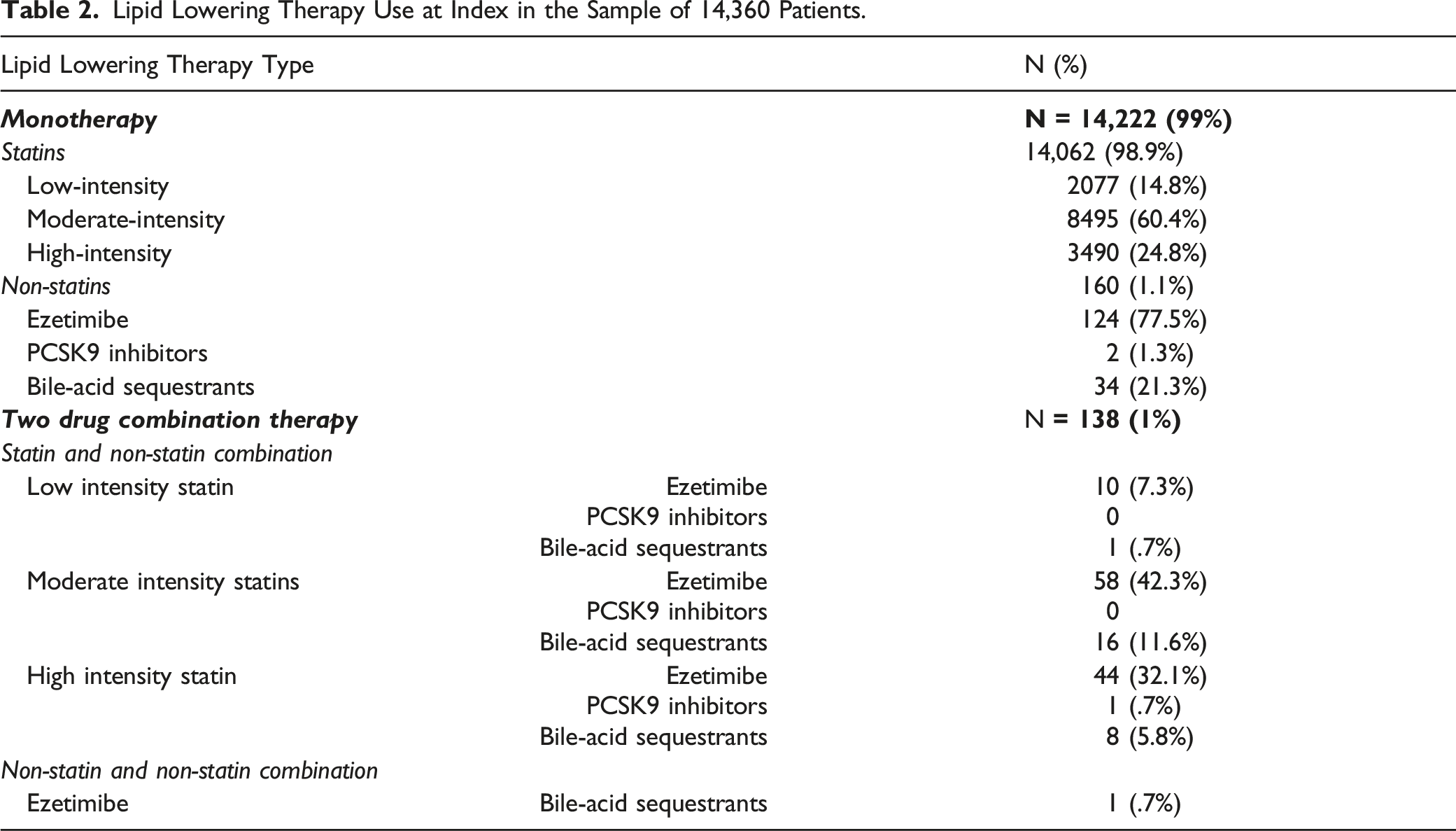
Baseline LLT Therapy Use
Lipid Lowering Therapy Use at Index in the Sample of 14,360 Patients.
Therapy Modifications in the Pre-Index Period
Treatment Modifications in the Pre-Index and Follow-Up Period Among Prevalent (92.6%) and New Users (7.4%) of Lipid Lowering Therapy.
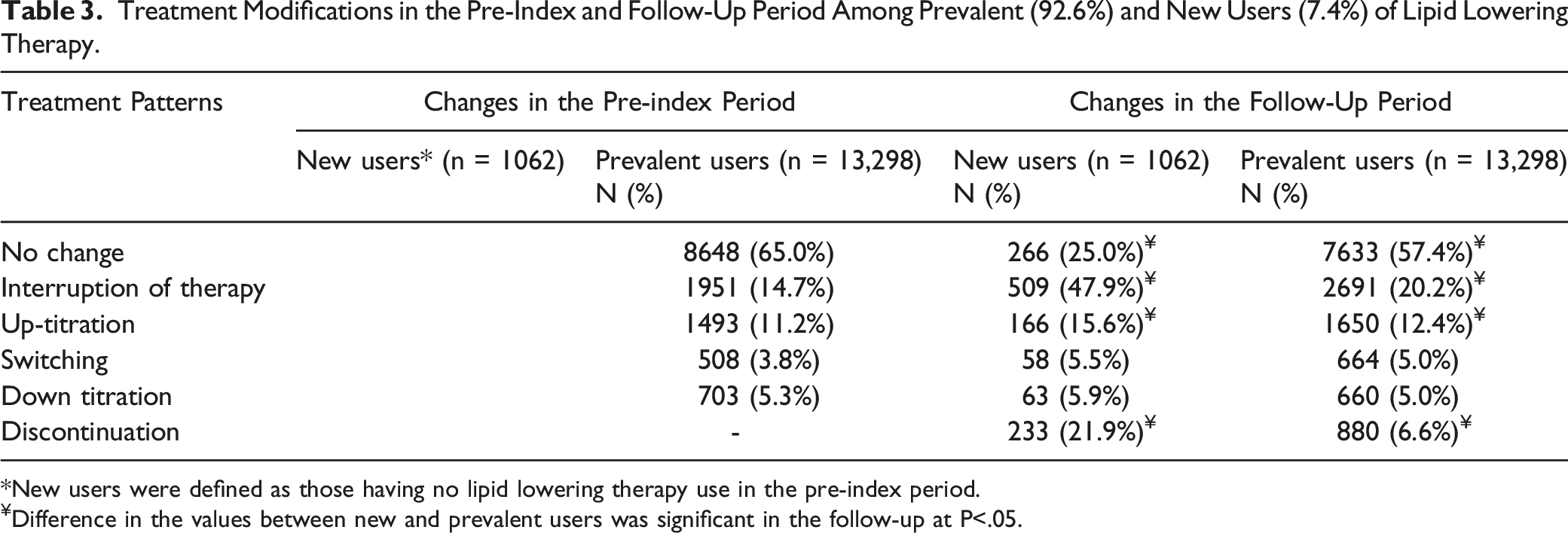
*New users were defined as those having no lipid lowering therapy use in the pre-index period.
¥Difference in the values between new and prevalent users was significant in the follow-up at P<.05.
Therapy Modifications in the Follow-Up Period
Therapy modification in the follow-up period, as presented in Table 3 were stratified for new and prevalent users of LLT. Of the prevalent users, 5881 (44.2%) patients did not have any treatment changes in the entire pre-index and follow-up period. There was a significantly greater number of patients among the new users who had interruption of therapy (47.9% vs 20.2%) as well as up-titrations (15.6% vs 12.4%) when compared to prevalent users of LLT. The discontinuation was significantly higher among new users than prevalent users (21.9% vs 6.6%).
A greater proportion of the prevalent users (57.4% vs 25%) had no change in therapy in the 1-year follow-up. Treatment modifications in the follow-up period were evaluated in greater detail to provide insight into therapy changes and can be found in the online Supplementary Material.
Pre-index and Follow-Up Treatment Patterns in Patients 75 Years of Age and Above as Compared to Patients Aged 65 -74 years.
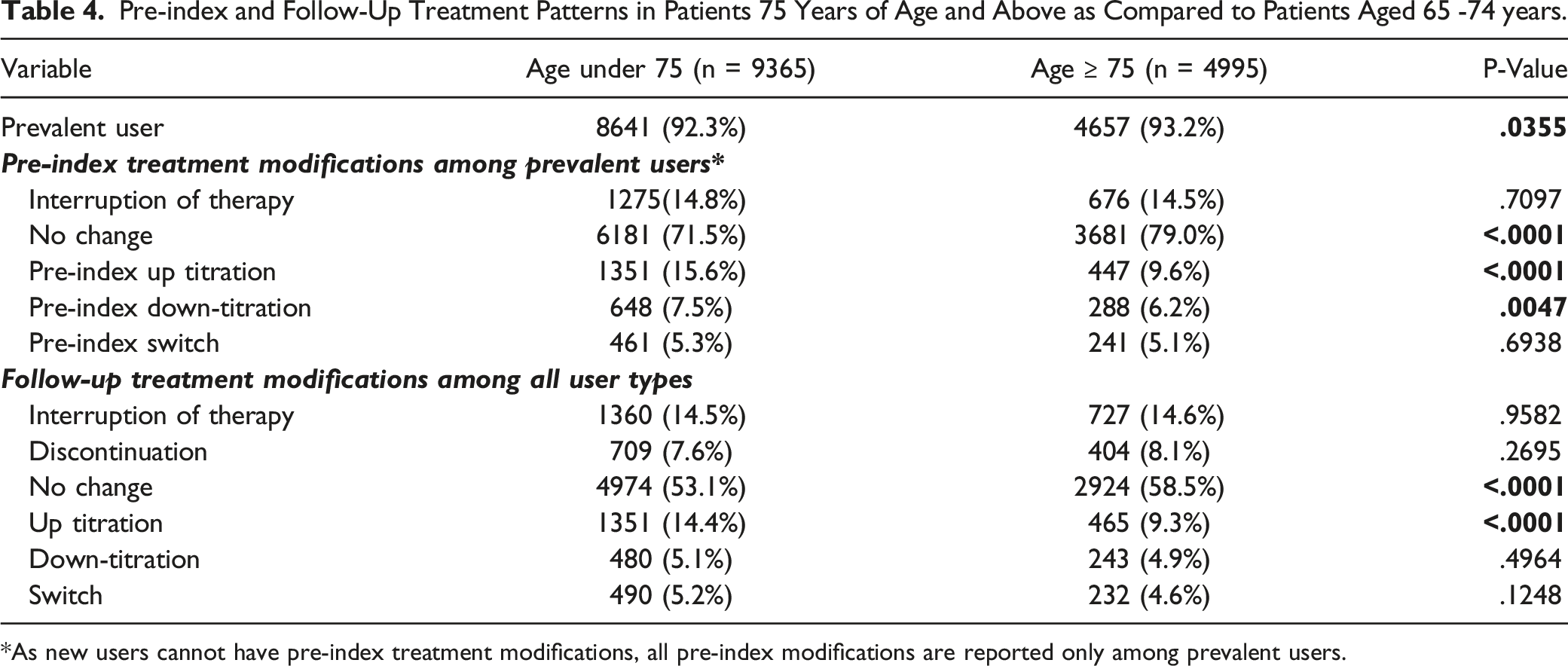
*As new users cannot have pre-index treatment modifications, all pre-index modifications are reported only among prevalent users.
Discussion
This retrospective claims database analysis assessed the real-world use of different LLT and their combinations, and evaluated the LLT treatment patterns in older adults. The study also compared the treatment changes in patients aged 75 and older as compared to older adults aged 65-74 years. Statins were used by 99% of patients either as monotherapy or as a part of a combination. Moderate intensity statins were the most prescribed (60.4%) and their use was more common in the subgroup aged 75 and over. This aligns with the ACC/AHA guidelines for cholesterol management which recommend patients >75 years be prescribed a moderate intensity statin unless they are already taking a high intensity statin without tolerability issues. 6 Despite the availability of newer therapies and the higher risk of statin intolerance in the older adults,33,34 non-statin therapies were used by only 2.1% of patients. Bittner et al. studied the use of ezetimibe in Medicare beneficiaries and found that the utilization of ezetimibe peaked in 2007 and was 4.6% at the end of 2011. 35
There were 143 patients using simvastatin 80 mg daily, the use of which has been limited by the FDA in 2011 due to increased risk of muscle-related adverse events. 36 Therefore, an ad-hoc analysis was conducted to assess if there was any indication of myalgia or myopathy for these patients in the study period, and did not find any such claims suggesting this therapy may be well-tolerated by them. Among patients on combination therapy, patients were pre-dominantly on a combination with a high or moderate intensity statin. This pattern is consistent with the guidelines which suggest up-titrating a patient to a maximally tolerated statin before addition of non-statin therapies. During evaluation of the baseline treatment, 66 patients were found to be using multiple statins concurrently and were excluded. Use of multiple statins simultaneously has not been mentioned in the guidelines, and further research is needed to understand the prevalence of and reasons of multiple statin use.
This study enhances the understanding of lipid management in older adults by describing patterns of LLT modifications over a period of two years in both new and prevalent users and not limiting to a certain risk group. There were 44.2% of prevalent users without any treatment changes, interruption or discontinuation during the entire study suggesting that most of the patients were on stable lipid lowering therapy. Power et al. evaluated treatment patterns among adults with ASCVD newly initiating LLT and found 11.7% of monotherapy initiators up-titration or augmentation during a 1-year follow-up period. 37 In our study 15.4% of new-users and 12.4% of prevalent users had an up-titration of LLT. Despite 92% of prevalent older adults in our study, the comparable rates of up-titration to Power et al indicate active management of LLT in our study population.
Our study evaluated interruption in therapy and discontinuations as separate modifications in therapy. The number of patients who either interrupted or discontinued therapy was 23.9% in the overall cohort. However, the rates of interruption, discontinuation, and up-titration were significantly higher in the new users of statins as compared to prevalent users. The treatment patterns among new users were consistent to a new-user cohort study by Simpson et al which found that interruptions occurred in 46.9% patients with re-initiation in 27.4% of them. Older adults initiating a statin are likely to be on other medications and may be at a higher risk of adverse events. 24 Special attention to new users should be provided to ensure that patients do not have any adverse consequences, understand the importance of the new LLT and take medications as prescribed.
Quek et al performed a retrospective claims-based study of LLT modifications and created an algorithm to group changes into possible statin intolerance or ineffectiveness based on up to two treatment modifications in the follow-up. Dose escalation and add-on were patterns grouped into possible statin ineffectiveness whereas discontinuations, interruption followed by re-initiation, and switching to a non-statin were classified as possible intolerance. 38 Our study mapped treatment modifications in equal detail, among both, new and prevalent users but made no hypothesis about reasons for modifications. Similar to Quek et al. 75% of new users in this study had at least one treatment modification. 38 Understanding reasons for treatment modifications can provide valuable insight into treatment practices and require further research.
This study has several strengths and unique contributions. The study assessed treatment patterns for the years 2016 – 2018 and therefore provides an updated real-world evidence. The study was not limited to new users, a certain type of LLT and a certain risk group; therefore provides a representation of LLT use in the older adult population.
This study has several limitations arising from the use of secondary databases and conducting observational research. The data was obtained from a regional health plan and therefore the generalizability of results may be limited to patients of a similar demographic characteristics. The cohort was drug-based i.e., only information of patients on LLT was available. Therefore, older adults who were eligible for LLT but did not initiate it could not be determined. The comorbidities were identified from one year of administrative claims from outpatient visits only and diagnostic information from hospitalization claims was not available. Use of liver function tests and other information which affects use of statins and other LLT was not available in administrative databases and thus could not be evaluated. Similarly, the reason for the treatment modifications and discontinuation could not be ascertained. This was a descriptive study, and no causal relationships can be established. Lastly, the inclusion requirements of continuous enrollment, use of lipid lowering therapy, and exclusion of certain comorbidities could have resulted in a healthier cohort on stable LLT.
Conclusions
This was a descriptive study aimed at evaluating the landscape of LLT use at a time where the new clinical data as well as updated guidelines had established the importance of statins among all age groups. The study demonstrated that older adults on LLT are more likely to be prevalent users, and new users among older adults undergo greater changes in the therapy. While certain patterns point towards guideline concordant prescribing of LLT there are lingering concerns such as concurrent use of multiple statins and higher discontinuation among new users need further attention. Understanding the treatment landscape is helpful for the providers and health plans to understand the existing gaps and realize the potential of the available LLT. Future research is needed to evaluate the impact of these treatment modifications on medication adherence and improvement in health outcomes.
Supplemental Material
Supplemental material - Patterns of Lipid Lowering Therapy Use Among Older Adults in a Managed Care Advantage Plan in the United States
Supplemental material for Patterns of Lipid Lowering Therapy Use Among Older Adults in a Managed Care Advantage Plan in the United States by Aisha Vadhariya, Manvi Sharma, Susan M. Abughosh, Kim K. Birtcher, Hua Chen, Anjana Mohan, and Michael L. Johnson in Journal of Pharmacy Practice
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
