Abstract
Background
In septic shock, the current Surviving Sepsis Campaign (SSC) guidelines recommend giving fluids and antibiotics, and trending lactate. Guidelines also recommend administering norepinephrine (NE) as the first-line vasopressor to maintain mean arterial pressure (MAP), while delaying vasopressin (AVP) administration unless goal MAP is not achieved on NE monotherapy.1,2 AVP is preferred for supplementary pressor support with NE since it may reduce time to goal MAP, decrease NE dosage requirements and tachy-arrythmias. 3 However, even though AVP is frequently used to help attain MAP, there is conflicting evidence over mortality benefit with AVP, which has resulted in a weak, moderate quality guideline recommendation for its use.1,4 Additionally, the efficacy of AVP also seems to be uncertain in patients with elevated lactate, even though it is presumably less likely to worsen lactate compared to catecholamine vasopressors.5,6
Recently, in 1 study by Sacha et al, 7 these investigators found that high lactate concentration was a negative predictor of hemodynamic response to fixed-dose AVP in patients with septic shock. In a follow up study, they also found that NE-equivalent dose and lactate concentration at AVP initiation were significantly associated with in hospital mortality. 8 However, if AVP initiation occurred at a lower NE-equivalent dose or lactate concentration, there was a reduction in likelihood of in hospital mortality. These studies suggest that AVP may lose efficacy as lactate levels increase, and so other supplementary vasopressors may be needed in addition to AVP and NE. Some limited data is available related to combination supplementary vasopressor use in septic shock suggesting some improvement in outcomes.9,10 But supplementary vasopressor utilization specifically in patients with elevated lactate needs more investigation due to limited research in this area.
The purpose of this study is to describe vasopressor utilization and clinical outcomes in patients with septic shock stratified by lactate level. Understanding vasopressor prescribing practices and patient response to commonly used supplementary vasopressors may help determine the preferred vasoactive agents across a range of elevation in lactate.
Methods
Study Design and Patients
This was a retrospective cohort study of patients 18 years of age or older admitted to an urban academic teaching hospital (Memphis Veterans Affairs Medical Center) from January 2015 through December 2020. Patients were initially identified for inclusion through ICD-9-CM and ICD-10-CM codes for septic shock. Patients were then further screened by chart review in order to include only those patients with sepsis and suspected infection (utilizing systemic inflammatory syndrome criteria for identification of patients with sepsis according to the 2021 SSC guidelines), who were also hypotensive (systolic blood pressure <90 mmHg, or mean arterial pressure <70 mmHg) and requiring NE therapy for hemodynamic support. 1 Patients admitted through the emergency department, or directly to either the medical, cardiac, or surgical intensive care unit were eligible for inclusion into this study. Exclusion criteria included initial vasopressor use other than NE, no available serum lactate level, prior enrollment in the study, and those exclusively with hypovolemic, obstructive or cardiogenic shock. This study’s research methods and procedures were approved by the Memphis Veterans Affairs Institutional Review Board. Informed consent was not obtained or required from the participants since this was a retrospective study.
Patients were separated into 2 groups based on chart review of lactate values. Patients with a baseline lactate level of <4 mmol/L (control group) were compared to patients with baseline lactate level ≥4 mmol/L (lactate group). Baseline lactate level was determined as the highest level before or after (within 12 hours) the start of NE therapy. Patients with a lactate level of ≥4 mmol/L were considered to have severely elevated lactate. The cutoff of 4 mmol/L for severely elevated lactate was chosen since this value has been associated with increased mortality, was a criterion for severe sepsis in the 2012 SSC definition, and Sacha et al found a decreased response to AVP with lactate values of 3.9 mmol/L or greater.8,11,12 We examined the association of lactate values in these 2 categories with supplementary vasopressor administration and patient outcomes. All patients in both groups received intravenous NE for treatment of septic shock, and the only determinant for placement into the study vs control group was baseline lactate level. Vasopressors were initiated based on SSC guidelines and titrated to a goal MAP of 65 mmHg based on clinical judgement of the managing team. Addition or discontinuation of vasopressors was at the discretion of the attending physician, and vasopressor doses were adjusted according to standard published titration references [13,14]. Sepsis bundles were used to treat patients which included guideline based fluid resuscitation, early antibiotic administration, trending of lactate, and vasopressors as necessary. Weaning or down titration of vasopressors was per institutional protocol as described above based on published titration references. The primary outcome was supplementary utilization of AVP, phenylephrine (PE), epinephrine, and dopamine, in addition to background NE therapy between the 2 patient groups. Secondary outcomes included identification of variables associated with multiple vasopressor utilization, as well as duration of vasopressor therapy, maximum dose of vasopressors, time to goal MAP of 65 mmHg (sustained for at least 4 consecutive hours), need for intravenous steroids, need for mechanical ventilation, length of hospital and intensive care unit (ICU) stay, and hospital mortality.
Data Collection
Charts were reviewed for patient inclusion, and data was collected retrospectively and included patient demographics, baseline lactate level, administration of vasopressors, variables necessary for identifying patients with septic shock, information about septic shock treatment and recommended interventions, and source of infection. Other data collected included components of the Charlson Comorbidity Index (CCI) score, SOFA score (data based on worse values within 12 hours of starting NE), vasopressor administration (duration and maximum dose), lactate clearance to less than 2.0 while on vasopressors, baseline arterial pH and while on vasopressors as available. NE equivalent doses were calculated based on an accepted published formula (NE = norepinephrine + epinephrine + phenylephrine/10 + dopamine/100 + metaraminol/8 + vasopressin*2.5 + angiotensin II*10). 15 Baseline hemodynamic variables (worst values within 2 hours of NE initiation) were also collected. MAP was monitored by an arterial line and data was collected every 30 min while on vasopressor support as well. Presumed infection source was determined by physician documentation and adequate empiric antibiotics were determined based on SSC guidelines and the local hospital antibiogram. 1 Patient specific outcomes collected on all included patients were length of stay, need for mechanical ventilation, and mortality. Data was collected until all vasopressor support was discontinued for 24 hours.
Statistical Analysis
Patients were enrolled with a planned equal distribution between each group and screened for inclusion in chronological order based upon admission date. A power analysis was not conducted to determine an adequate sample size since the utilization of supplementary vasopressors across a range of elevation in lactate is not known. Thus, this investigation serves as a hypothesis generating study in this area. The Chi-Square or Fischer’s exact test was used to compare categorical data, whereas parametric continuous data were analyzed using Student’s t-test and non-parametric data with the Mann-Whitney-U Test. Parametric data are presented as mean ± standard deviation, and non-parametric as median (25-75% interquartile range), and nominal data as percentages. Data was determined to be parametric through histogram visualization for normal distribution, and by measurement of skewness and kurtosis. All tests for statistical significance were two-tailed, and significance was established at a P-value of less than .05. All statistical analysis was completed using SPSS version 27 (Chicago, IL: IBM Corp; 2021).
A prespecified multiple logistic regression analysis was performed in order to determine factors predicting the need for multiple vasopressors (such as AVP, PE, epinephrine, and their combinations) in addition to NE. Univariate analysis of patients was performed to identify possible characteristics (lactate group ≥ or < 4 mmol/L, age, CCI, baseline pH, baseline lactate, baseline MAP, maximum NE dose, and SOFA score) for inclusion in the multivariable logistic regression analysis. All variables were entered into a forward multiple logistical regression model and removed stepwise for a value greater than .10. The logistic regression model was calibrated based on a nonsignificant Hosmer–Lemeshow goodness-of-fit P value more than .05, and model discrimination was assessed based upon the area under the receiver operator characteristic curve (AUC ROC). Results were reported as adjusted odds ratio (OR) and corresponding 95% confidence intervals (CI).
Results
Patient Demographics and Baseline Characteristics
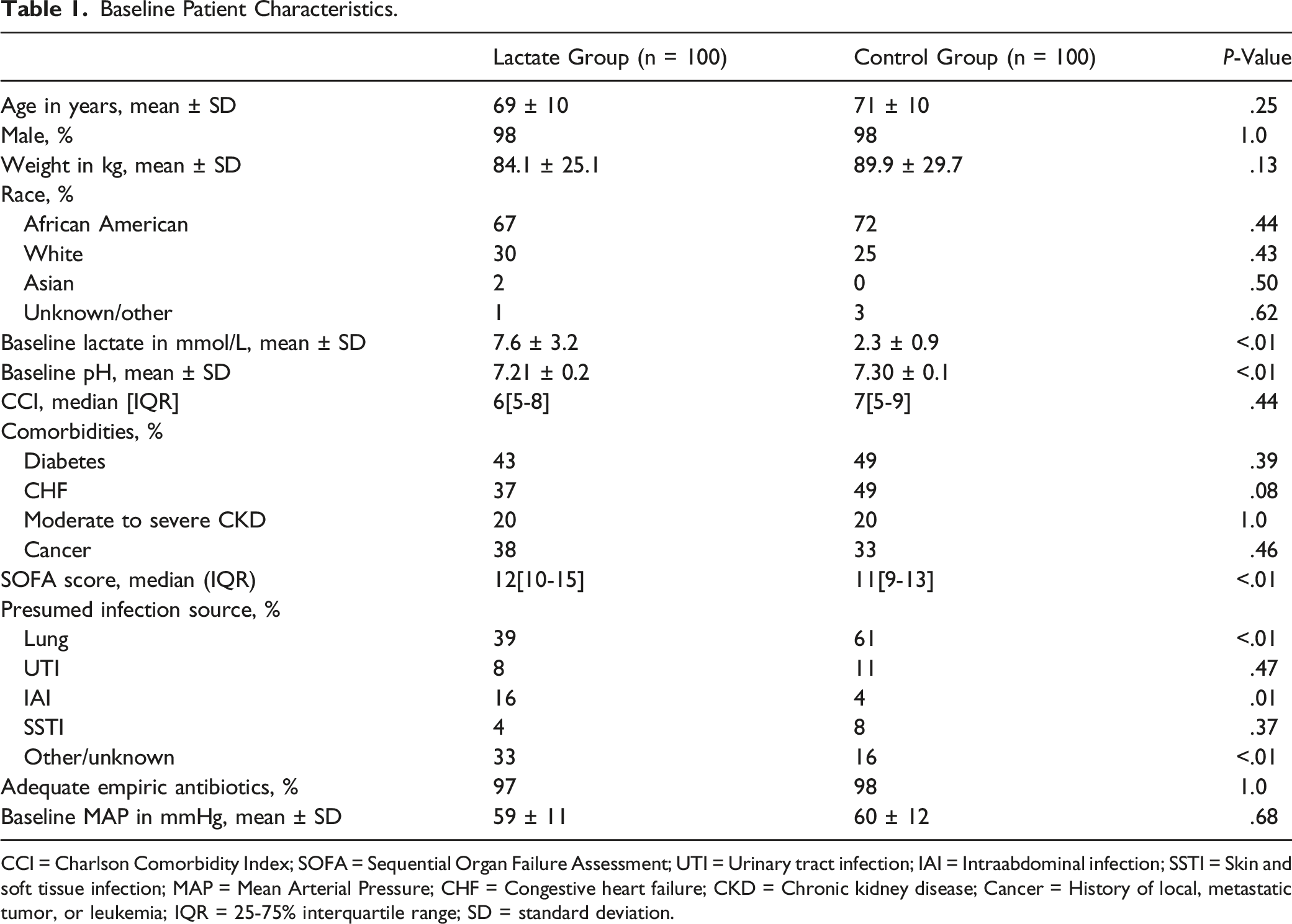
A total of 399 patients were screened for inclusion with 199 patients excluded. The most common reasons for exclusion were use of another initial vasopressor other than NE or no NE administration (Figure 1). One hundred patients were included in the lactate group (baseline lactate of at least 4 mmol/L) and 100 patients were included in the control group (baseline lactate less than 4 mmol/L). Overall, the majority of patients were male (98%) and African American (69%); Table 1. The average baseline lactate in the lactate group was significantly higher compared to the control group (7.6 ± 3.2 vs 2.3 mmol/L ± .9; P < .01), and the lactate group also had a lower mean baseline pH. Baseline MAP (59 ± 11 vs 60 mmHg ±12; P = .68), and adequate empiric antibiotic administration (97% vs 98%; P = 1.0) were similar between groups, while SOFA score (12 vs 11; P = .01) was statistically but not clinically different. Infection sources were similar except that the control group had a higher rate of pulmonary and lower rate of intraabdominal infections. Flow diagram of included and excluded patients; NU, norepinephrine. Baseline Patient Characteristics. CCI = Charlson Comorbidity Index; SOFA = Sequential Organ Failure Assessment; UTI = Urinary tract infection; IAI = Intraabdominal infection; SSTI = Skin and soft tissue infection; MAP = Mean Arterial Pressure; CHF = Congestive heart failure; CKD = Chronic kidney disease; Cancer = History of local, metastatic tumor, or leukemia; IQR = 25-75% interquartile range; SD = standard deviation.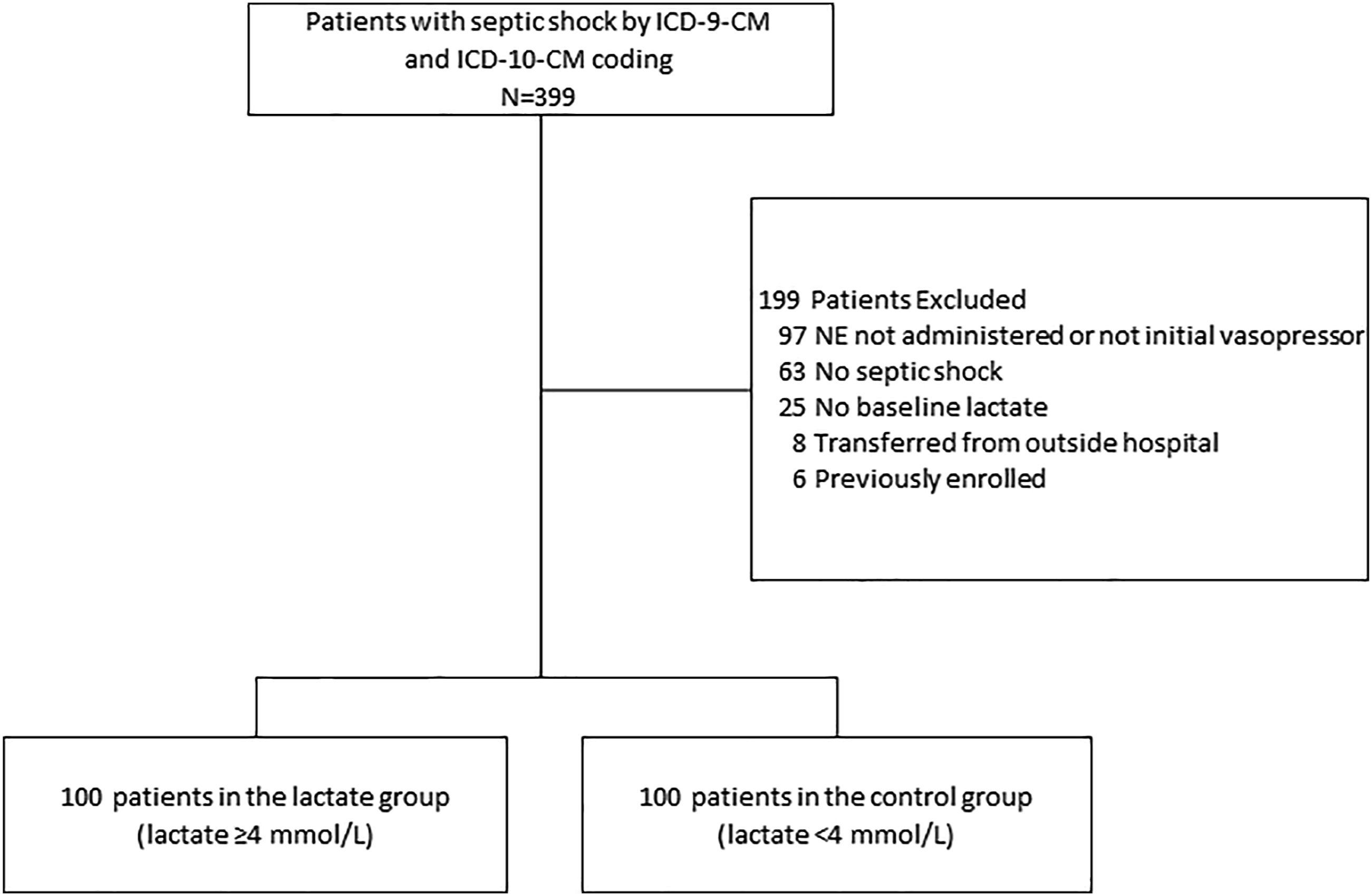
Vasopressor Utilization
Vasopressor Utilization.
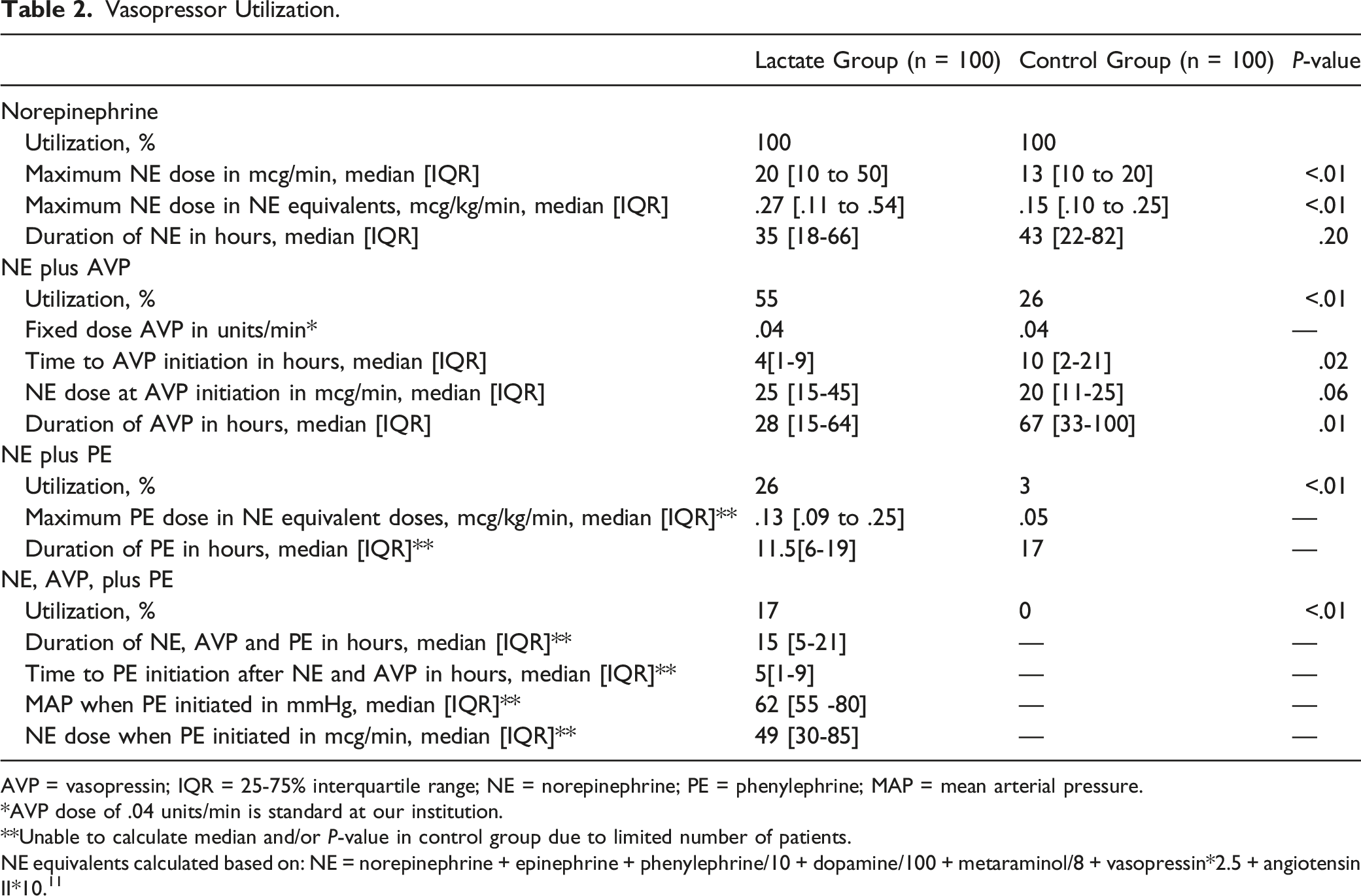
AVP = vasopressin; IQR = 25-75% interquartile range; NE = norepinephrine; PE = phenylephrine; MAP = mean arterial pressure.
*AVP dose of .04 units/min is standard at our institution.
**Unable to calculate median and/or P-value in control group due to limited number of patients.
NE equivalents calculated based on: NE = norepinephrine + epinephrine + phenylephrine/10 + dopamine/100 + metaraminol/8 + vasopressin*2.5 + angiotensin II*10. 11
Secondary Outcomes and Predictors of Vasopressor Utilization
Multivariable Logistic Regression Analysis of Vasopressor Utilization.
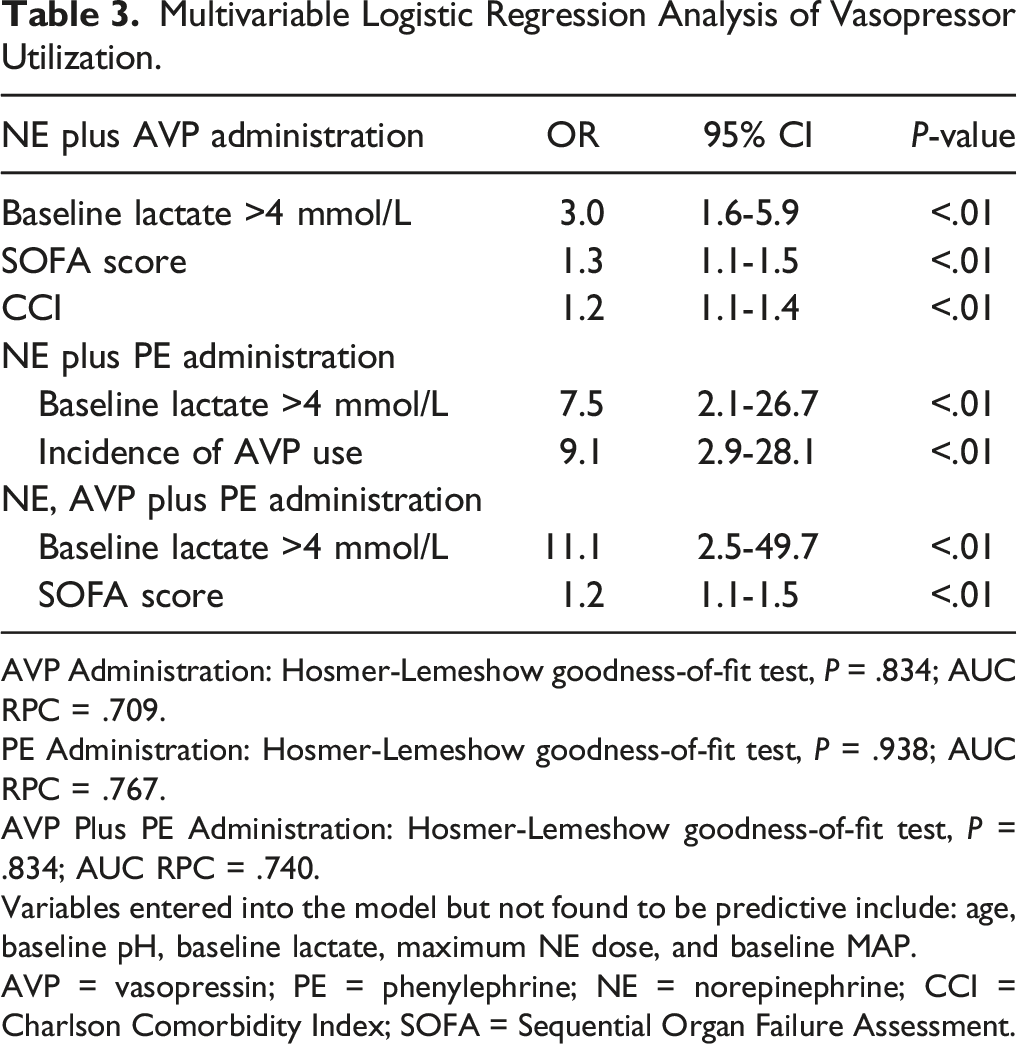
AVP Administration: Hosmer-Lemeshow goodness-of-fit test, P = .834; AUC RPC = .709.
PE Administration: Hosmer-Lemeshow goodness-of-fit test, P = .938; AUC RPC = .767.
AVP Plus PE Administration: Hosmer-Lemeshow goodness-of-fit test, P = .834; AUC RPC = .740.
Variables entered into the model but not found to be predictive include: age, baseline pH, baseline lactate, maximum NE dose, and baseline MAP.
AVP = vasopressin; PE = phenylephrine; NE = norepinephrine; CCI = Charlson Comorbidity Index; SOFA = Sequential Organ Failure Assessment.
Other Secondary and Clinical Outcomes.
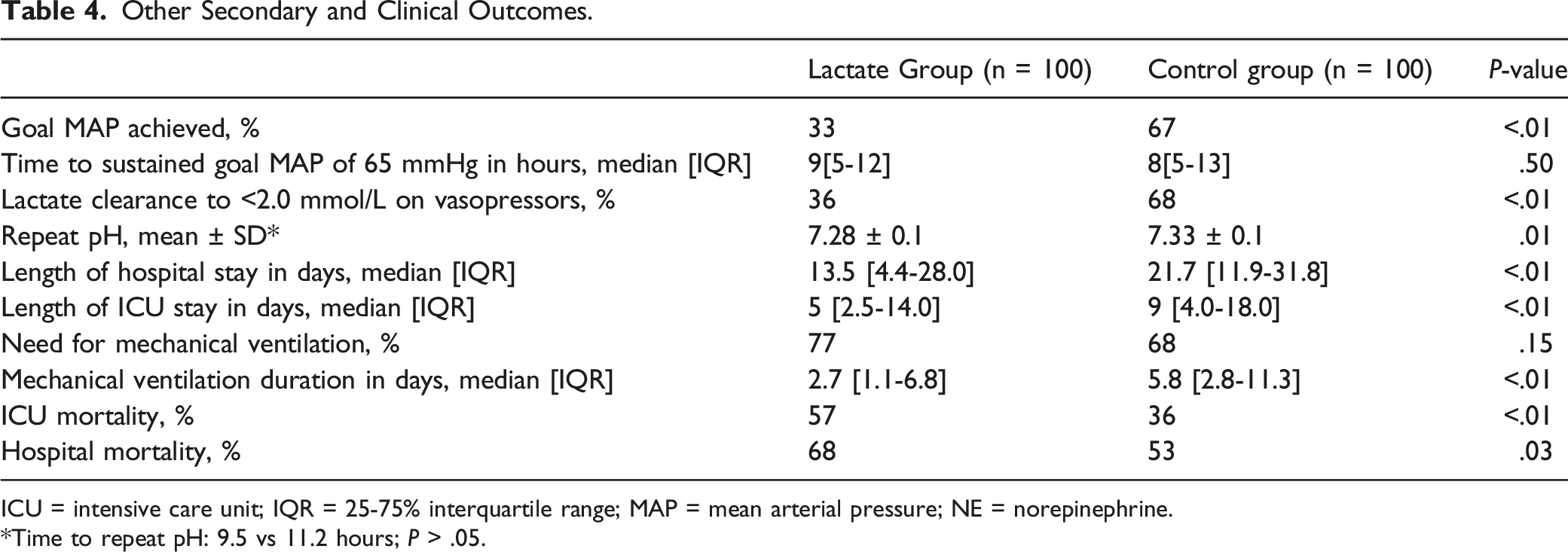
ICU = intensive care unit; IQR = 25-75% interquartile range; MAP = mean arterial pressure; NE = norepinephrine.
*Time to repeat pH: 9.5 vs 11.2 hours; P > .05.
Discussion
To our knowledge, this is the first investigation evaluating supplementary vasopressor requirements beyond NE and AVP in septic patients with severely elevated lactate. In this retrospective study, we observed that patients with septic shock and a baseline lactate of ≥4 mmol/L requiring NE therapy had a significantly higher use of AVP and PE compared to patients with a baseline lactate of <4 mmol/L. We also found severity of illness and baseline lactate of ≥4 mmol/L were independent predictors of requiring multiple vasopressors including concomitant AVP and PE administration. Based on the results of our small retrospective study, patients presenting with septic shock and elevated lactate may be more likely to receive multiple vasoactive agents, but further investigation in larger studies is needed.
The purpose of this study was to describe vasopressor utilization and clinical outcomes in patients with septic shock stratified by lactate level, because it is unknown how the hemodynamic response will be affected by varying degrees of lactate elevation in patients administered different supplementary vasopressors. We found that vasopressors were mostly limited to NE and AVP, which is similar to a previously described study of patients with elevated lactate by Sacha et al. Our study supports previously published data wherein AVP should be initiated in patients with lower lactate, since patients in our study with elevated lactate had higher mortality.7,16 Importantly, our study does provide some insightful information on the additional use of PE in these patients. PE was the most commonly used third-line vasoactive agent added on by clinicians to NE and AVP, which demonstrates preference for AVP as a second line agent in septic shock in accordance with the SSC guidelines. PE use in our patients also seemed to be preferred when MAP was not at goal, despite high NE doses and concomitant AVP therapy.
Advantages of PE include that it has no β-1 adrenergic activity and therefore is less likely to increase lactate further when added on to NE therapy in patients with preserved cardiac output. PE is also an alternative vasopressor used for septic shock for patients who fail to reach goal MAP on pressor support with NE and AVP.6,9 Nevertheless, low quality evidence reveals that it is less frequently utilized because of its uncertain impact on clinical outcomes, and also possible increased mortality compared to NE.5,17 This is the first study to our knowledge evaluating PE administration frequency in this patient population, but this needs further investigation, since our patient population was small. Selection of vasoactive agents for these patients remains challenging since data on outcomes such as goal MAP attainment and mortality are limited, and identifying the most appropriate vasopressor regimen is especially important since elevated lactate is a risk factor by itself for increased mortality.18,19
Future studies should evaluate lactate level at baseline and measure outcomes when multiple supplementary vasopressors are added concurrently. Additionally, upon recognition of elevated lactate in septic shock, consideration should be given for catecholamine sparing strategies and adjunctive therapies such as utilizing steroids, angiotensin II, and midodrine. In regard to other study outcomes, patients with elevated lactate required higher maximum NE doses and more rapid initiation of AVP, even though MAP was similar between groups at baseline. Patients in the lactate group also had a lower baseline and follow up pH, as well as lower rate of lactate clearance, likely due to the severity of lactic acidosis at baseline as well. Finally, we found a higher ICU and hospital mortality rate in the lactate group. This is similar to mortality findings in other studies in patients with severely elevated lactate and may have been driven by the higher rate of intraabdominal infections in the study group.18,19 Likely as a result of a higher mortality rate in the elevated lactate group, we also found a shorter duration of AVP use, mechanical ventilation, ICU and hospital LOS in these patients.
Our retrospective, observational study has limitations, including the small number of patients enrolled and lack of power to determine an adequate sample size, and so we cannot definitively conclude whether there was a difference in multiple vasopressor use. Additionally, patients were included from a single center treating a veteran population, which may limit the external validity of our study and application of our findings to other patient populations. Confounding due to our retrospective assessment of the temporal relationship between lactate and vasopressor use also cannot be excluded. There were uncontrolled therapies inherent to a retrospective study such as administration of vasopressors and antibiotics that could influence outcomes, and also other factors than just lactate that could potentially worsen hypotension and increase the need for multiple vasopressors. Data on fluid resuscitation was also not collected, which is a significant limitation of this study, and so patients in the lactate group may have required more vasoactive agents due to under fluid resuscitation and hypoperfusion. Additionally, the temporal relationship of fluid administration to initiation of vasopressors was also not measured, which may confound our results since delays in vasopressor administration after initiation of fluids may potentially affect time to goal MAP achievement. However, sepsis bundles incorporating fluid resuscitation along with vasopressors are standard at our institution and so it is more likely that adequate fluids actually were administered in combination with vasopressors. APACHE scores were not collected but SOFA scores were calculated for baseline severity of illness comparison between patient groups. Antibiotics were only assessed for appropriate empiric coverage based on the suspected source of infection without final culture data. Data on sedative administration was also not collected which could potentially influence outcomes as well since the majority of patients required mechanical ventilation and many continuous infusion sedatives are associated with hypotension. Finally, data on order of vasopressor discontinuation was not collected, and the AVP rate was fixed at .04 units/min, which may limit the generalizability of our results to other institutions.
This study has many strengths as this is the first study to our knowledge to assess prescribing frequency of multiple vasopressors in patients with severely elevated lactate and septic shock. The study design increases external validity because vasopressors were initiated at the discretion of the primary intensive care unit attending physician similar to a real word setting. AVP is commonly required in treatment of septic shock and our data contributes to the current body of literature on the role of AVP in patients with lactic acidosis. Patients included were a diverse ICU population, including medical, cardiac, and surgical intensive care patients. Sepsis bundles were also utilized to provide consistent care for patients with septic shock. Our study is also unique since we identified patients with elevated lactate at the onset of septic shock in order to measure differences in pressor requirements, providing further support for its initial measurement based on current treatment recommendations. Baseline lactate may therefore be more useful for the intensivist as a predictor of needing multiple vasoactive agents, since it is a readily available test at the onset of septic shock that can potentially direct adjunctive therapies earlier in the patient course.
Conclusions and Relevance
In this small retrospective study, multiple vasopressor use was high in patients with severely elevated lactate. AVP and PE were the most commonly used supplementary vasoactive agents in our study group, and outcomes including goal MAP attainment and mortality were inferior in those patients with lactate of ≥4 mmol/L. Larger studies are needed to confirm our results regarding the effect of elevated lactate on multiple vasopressor requirements, and the optimal vasopressor regimen in this patient population needs further investigation to improve MAP and mortality.
Footnotes
Author’s Note
The results of this study have not been presented previously. A portion of the data from this study has been previously presented in poster form at the American College of Clinical Pharmacy Virtual Poster Symposium, May 25-26, 2021.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
