Abstract
Introduction
Delirium and agitation are common occurrences in patients admitted to the intensive care unit (ICU) with incidences ranging up to 70%.1-3 Delirium has been associated with higher mortality, increased length of stay and long-term cognitive impairment.3,4 Treatment of delirium and agitation is multifactorial and may involve the use of antipsychotic medications when other therapies have been optimized such as pain control, ventilator settings, sleep-wake cycle and other means.
Multiple antipsychotics have been used to attempt to mitigate delirium and/or agitation in the ICU; however, evaluation of these interventions have shown inconsistent results. The largest study to date compared haloperidol and ziprasidone which revealed that neither medication significantly altered the duration of hypoactive delirium. 5 Despite the implications of this trial, antipsychotics are still largely employed in the management of hyperactive delirium as an effort to alleviate the significant morbidity and mortality associated with this condition. Given the paucity of robust data and potential to target multiple pathways implicated in ICU delirium, the Society of Critical Care Medicine’s (SCCM) Pain, Agitation/Sedation, Delirium, Immobility, and Sleep Disruption (PADIS) Guidelines recommend the need for further research on the use of VPA in this setting. 6
Valproic acid (VPA) is a mood stabilizer used in both the acute and chronic treatment of bipolar disorder. The mechanism of action of VPA targets many of the pathways implicated in the pathophysiology of delirium including antagonism of glutamate, potentiation of the effects of gamma-aminobutryic acid (GABA) and it may also decrease dopaminergic activity in the striatum.7,8 Most of the available literature on VPA for treatment of agitation and delirium in the ICU is limited to small retrospective cohort studies, case series, or descriptive analyses.7,9-11 The limited data suggests VPA is associated with a reduction in the incidence of delirium and agitation. However, it remains unknown how VPA compares to other medications used in this setting.
Methods
This was a retrospective cohort study of critically ill patients treated for agitation and delirium conducted at a 525 bed, academic, tertiary care, level 1 trauma center. Electronic medical records were used to identify all patients in the medical ICU (MICU) and surgical/trauma ICU (SICU) from January 1, 2016 to October 1, 2020.
Our standard ICU protocol for pain, agitation, and delirium is in alignment with the recommendations made from the SCCM PADIS Guidelines. 6 This includes a pain control first approach with multimodal pain regimens, use of nonpharmacological measures, promoting the sleep-wake cycle and use of medications if patients experience delirium and/or agitation once other measures are optimized. The nonpharmacological measures include early mobilization with physical therapy, quiet hours that reduce nocturnal noise and light, daytime light promotion, and encouragement of family engagement. For patients not requiring deep sedation, we target a RASS goal of −1 to 0. Spontaneous breathing trials and spontaneous awakening trials are also protocolized and completed every shift with a nurse and respiratory therapist.
Adult patients (age ≥18), admitted to the ICU for ≥48 h were included if they had a Richmond Agitation-Sedation Scale (RASS) score ≥2, were positive on the Confusion Assessment Method for the ICU (CAM-ICU), and received ≥1 of the following medications: haloperidol, quetiapine, olanzapine, propranolol, VPA and derivatives, risperidone, dexmedetomidine, or clonidine during their ICU stay. Patients were excluded if their prior to admission medication list included VPA (or derivatives), had a prior diagnosis of dementia, concomitantly administered a carbapenem, or prescribed VPA for non-study indications. Groups were split into patients who received VPA for treatment of agitation and delirium and a control group who did not receive VPA. Patients in the VPA group who received VPA less than 48 h were excluded to ensure VPA has reached steady state. The index date was the first day the patient was documented as CAM positive and RASS ≥2 in both groups.
Baseline demographics were collected included age, gender, weight, ICU location, new diagnosis of traumatic brain injury (TBI), prior diagnosis of TBI, Sequential Organ Failure Assessment (SOFA) score, Glasgow Coma Score (GCS), prior psychiatric diagnoses, and any substance use disorder. The SOFA score was collected at the index date and all other baseline demographics were collected at ICU admission. Patients were matched 1:1 based on ICU location, age range (18-50; 51-74; ≥75), gender, TBI, and psychiatric history.
The incidence of agitation and delirium was analyzed for 14 days after the index date or until ICU discharge, whichever occurred first. The primary outcome was delirium and coma free days at 14 days or ICU discharge. One delirium and coma free day is defined as documentation of only CAM-ICU negative and RASS > −4 during one calendar day. Secondary outcomes included agitation and coma free days at 14 days or ICU discharge, ICU length of stay, duration of mechanical ventilation, in hospital mortality, and mortality at 28 days. One agitation and coma free day is defined as documentation of only RASS < +2 and RASS > −4 during one calendar day. Descriptive data were also collected in the VPA group after VPA initiation including hepatotoxicity (increase in liver function tests (LFTs) > 3 times the upper limit of normal), thrombocytopenia (>50% drop in platelet count), hyperammonemia (serum ammonia >60 mmol/L), VPA levels >100 mcg/mL, and average VPA dose (mg/day). Additionally, if patients were discharged home with a VPA prescription, the doses at discharge were collected. Our institution does not currently have a VPA dosing, dose titration, and monitoring protocol in place, however, these decisions are made in a multidisciplinary fashion with the clinical ICU pharmacist based on patient-specific factors.
Unpaired t-tests were used for continuous outcomes. Fisher’s exact test was used for categorical outcomes. An alpha error of .05 was considered significant. Descriptive data was reported as percentages and averages.
Results
Baseline Characteristics.
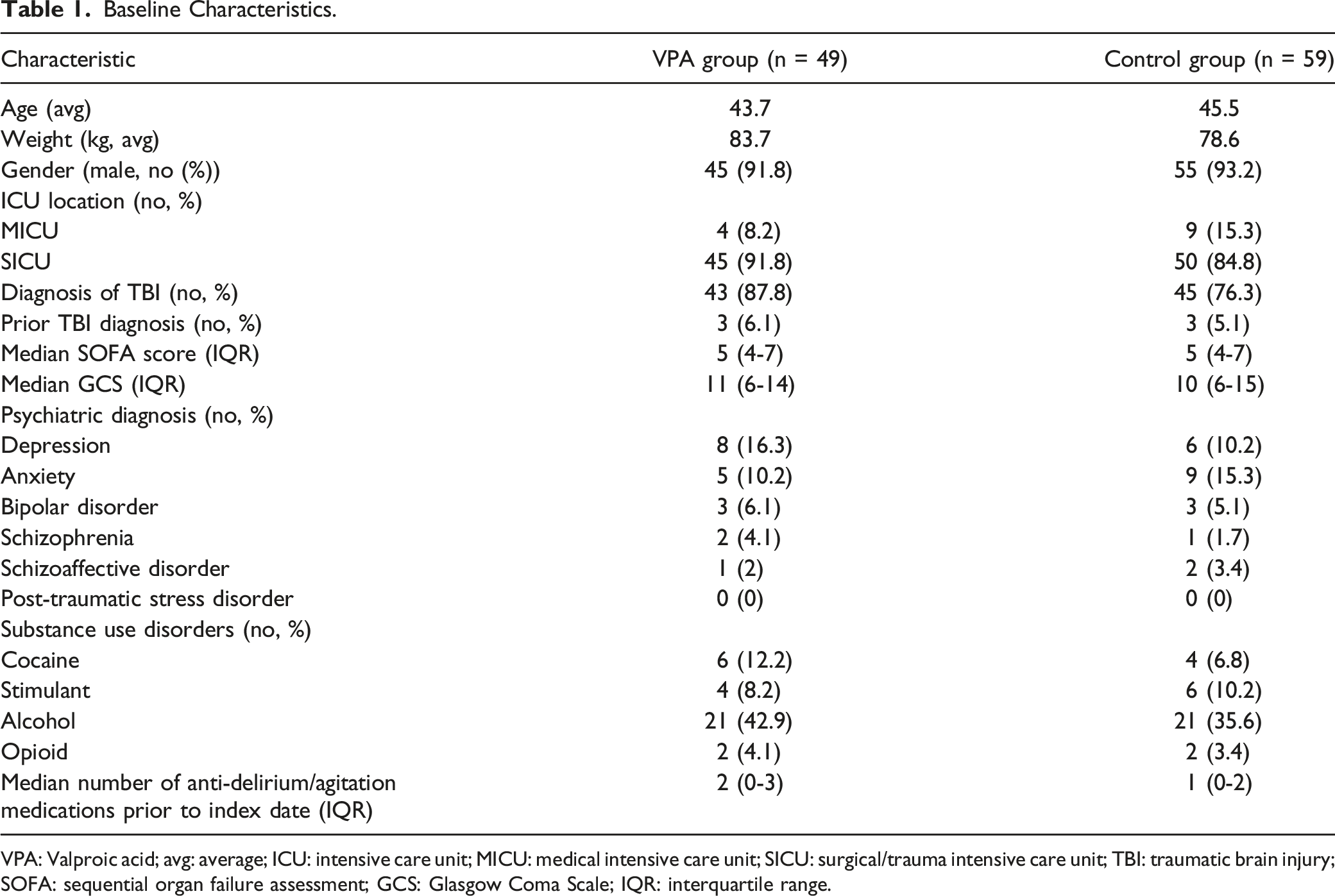
VPA: Valproic acid; avg: average; ICU: intensive care unit; MICU: medical intensive care unit; SICU: surgical/trauma intensive care unit; TBI: traumatic brain injury; SOFA: sequential organ failure assessment; GCS: Glasgow Coma Scale; IQR: interquartile range.
Primary and Secondary Outcomes.
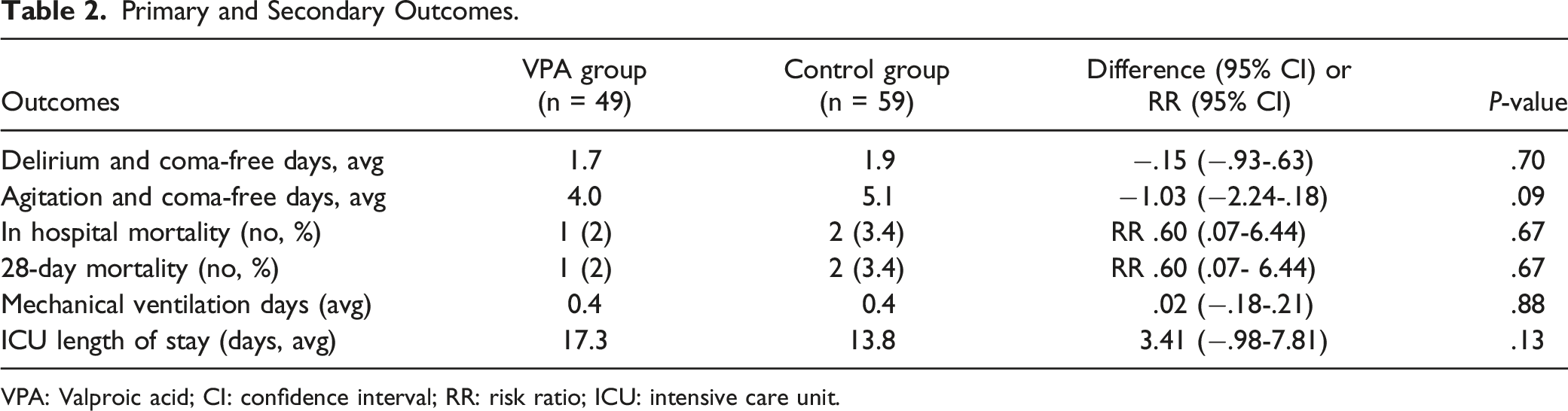
VPA: Valproic acid; CI: confidence interval; RR: risk ratio; ICU: intensive care unit.
There were no significant differences between groups for any of the secondary outcomes (Table 2). In the VPA group, there were 4.0 agitation and coma-free days compared to 5.1 in the control group (difference −1.03, 95% CI: -2.24-.18, P = .09). In hospital mortality in the VPA group was 2.0% and 3.4% in the control group (RR .60, 95% CI: 0.07-6.44, P = .67). Mortality at 28 days in the VPA group was 2.0% and 3.4% in the control group (RR .60, 95% CI: 0.07-6.64, P = .67). Average mechanical ventilation days were .4 in both groups (difference .02, 95% CI: -.18-.21, P = .88). The ICU length of stay was numerically lower in the control group (17.3 vs 13.8 days), but did not reach statistical significance (difference 3.41, 95% CI: -.98-7.81, P = .13).
Safety and Descriptive Outcomes for VPA Group.

VPA: Valproic acid.
Discussion
Our study provides the largest study to date evaluating the use of VPA in addition to non-VPA medications for the treatment of delirium and/or agitation in the ICU. We did not find any significant differences between the VPA and control group with respect to delirium and coma-free days, or agitation and coma-free days. Of note, VPA was always used as an adjunctive medication and often started later in the course of patients’ delirium and/or agitation. Gagnon et al evaluated patients in this setting who received VPA and compared the incidence of agitation and delirium on valproate day 1 and valproate day 3. 9 They found that VPA was associated with a statistically significant reduction in the incidence of delirium (68% vs 49%, P = .012) and agitation (96% vs 61%, P < .0001) from valproate day 1 to day 3. 9 The beneficial effects of VPA seen in the study by Gagnon et al are difficult to extrapolate to our findings due to inherent differences in study design. Our study required patients in the VPA group to be administered VPA for at least 48 h during the intervention period while Gagnon et al conducted their analyses based on 3 days of VPA. Additionally, we included a comparator group, which suggests there is no difference in delirium and agitation when adding VPA compared to non-VPA medication alone strategies.
While there was no statistically significant difference found in the average ICU length of stay, the reduction in 3.4 days in the control group may be clinically relevant when considering the speed of recovery as well as the ICU costs associated with a longer stay. Assessing the severity of delirium and agitation was outside of the scope of this study. However, it may be possible that patients initiated on VPA had more severe delirium and/or agitation leading to longer ICU stays as VPA is often an adjunctive treatment option once other medications have failed at our institution, which may explain why patients were on numerically more anti-delirium and anti-agitation medications at baseline. Although the VPA patients on average required longer ICU stays, it is promising there were no differences with regards to mechanical ventilation days, in-hospital mortality, and 28-day mortality.
There were some adverse effects of VPA noted in our study with hepatotoxicity occurring in 6.1% of patients and thrombocytopenia in 12.2% of patients. Both hepatotoxicity and thrombocytopenia are common in the ICU and given their causes are often multifactorial, it would be difficult to attribute these effects solely to VPA.12,13 However, our findings are similar to what is reported in prior studies evaluating critically ill patients on VPA therapy with hepatotoxicity ranging from 4 to 5% and thrombocytopenia ranging from 9 to 13%.9,11
There were several limitations to this study. The retrospective nature of this study did not allow us to control for non-VPA medication use in either group. This also limited our ability to control for other factors that may cause or exacerbate delirium or agitation such as sub-optimal pain control and administration of benzodiazepines. However, we sought to reduce the influence of potential confounders by matching patients in both groups at baseline. Additionally, we relied on the electronic health record for CAM-ICU and RASS assessments. At our institution, the standard for CAM-ICU and RASS determination is assessed by the bedside nurse and documented at least once per shift (at least twice per day) and as needed with any change in patient agitation or sedation level. However, the assessment documentations were not uniform across patients as some may have had more frequent changes in agitation or sedation levels. As our institution does not have a VPA dosing, dose titration, and monitoring protocol in place, these strategies vary based on the recommendations of the clinical pharmacist and multidisciplinary team. In general, our practice is to initiate VPA at a dose of 250-500 mg every 8 h and is titrated up over the course of several days if needed for ongoing management. The timing of titration depends on patient-specific factors as well as the clinical judgment of the multidisciplinary team. Of note, 88% of our patients were admitted to our SICU and out of the entire study population, 81.5% had a new diagnosis of a TBI upon admission. While the rest of the study population comprised of patients admitted to the MICU, the generalizability of our findings to this population should be validated in future studies.
Conclusion
VPA is an adjunctive treatment option for delirium and agitation in the ICU. Our findings suggest that VPA is associated with similar delirium and agitation-free days compared to other non-VPA anti-delirium and anti-agitation medications, with some adverse effects. Larger, prospective, controlled studies are needed to validate the routine use of VPA for the treatment of delirium and agitation in the ICU.
Footnotes
Acknowledgments
The authors would like to acknowledge Carolyn Valdez, MS biostatistician, for the abstraction and organization of our data set, which greatly improved the accuracy and efficiency of our data collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
