Abstract
Introduction
As we navigated through a second year of the coronavirus disease 2019 (COVID-19) global pandemic, there was a continued abundance of infectious diseases (ID)-related publications. Attempting to identify and analyze important ID/antimicrobial stewardship publications from 2021, in addition to the evolving body of literature related to COVID-19, posed a real challenge for clinicians. In 2021 alone, a PubMed search with the keywords “infectious diseases,” “antimicrobial stewardship”, or “HIV” produced 70,217 results in total. This emphasizes the difficulty of staying up-to-date on new literature, particularly in the midst of an ongoing pandemic.
The Houston Infectious Diseases Network (HIDN) consists of ID clinicians from over 15 different institutions in the Texas Medical Center and the greater Houston area. The network includes ID physicians, pharmacists, microbiologists, and researchers who meet regularly to collaborate on research, education, stewardship, and best practices. HIDN members also participate in the training and mentorship of fellows, residents, and students who are interested in ID. Since 2007, HIDN has compiled an annual review of the top 10 significant publications in ID pharmacotherapy in hopes of providing a useful summary of key ID developments from the previous year.1-14
Article Selection Process
HIDN members were asked to nominate influential ID pharmacotherapy articles published between January 1 and December 31, 2021 (electronic or print publication). Members were asked to nominate articles not of their own authorship, and nominated articles must have been published in peer-reviewed journals. Publications which did not represent original research were excluded from consideration. The list of nominations were compiled and an electronic survey was distributed to the Society of Infectious Diseases Pharmacists (SIDP) members via email. SIDP members were asked to choose 10 articles which contributed most to general ID pharmacotherapy and 1 article that contributed most to HIV/AIDS pharmacotherapy, and members could opt out of voting in either category. Voters were also allowed to nominate additional articles not included in the original survey by using 1 vote per new nomination. The articles were ranked based on the survey results and the top publications were selected.
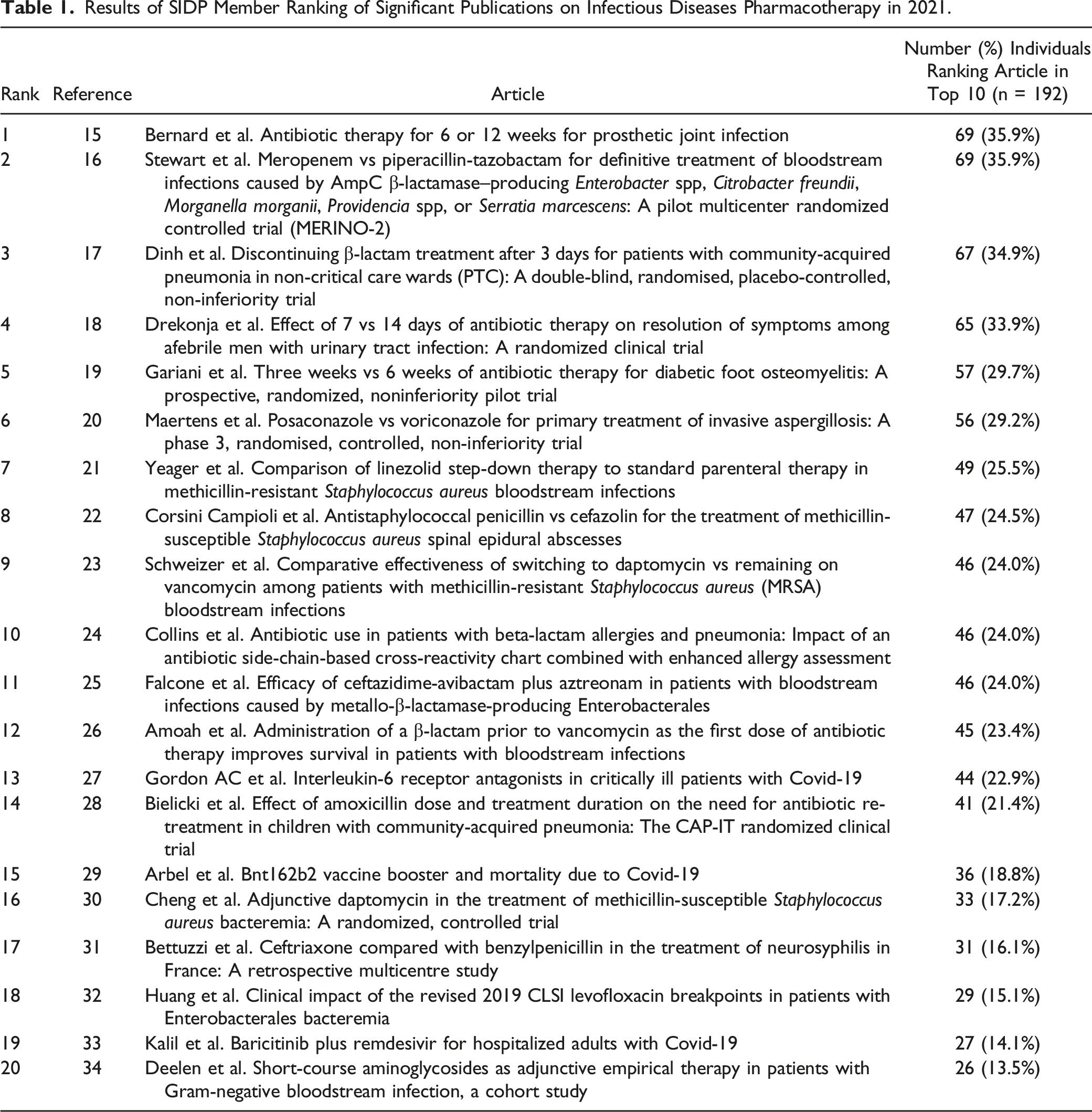
Results of SIDP Member Ranking of Significant Publications on Infectious Diseases Pharmacotherapy in 2021.
Results of SIDP Member Ranking of Significant Publications on HIV/AIDS Pharmacotherapy in 2021.

Primary Takeaways for the Significant Publications on Infectious Diseases Pharmacotherapy in 2021.

Bernard L et al. Antibiotic Therapy for 6 or 12 Weeks for Prosthetic Joint Infection
The management of prosthetic joint infections (PJI) generally includes surgical management in combination with 6 to 12 week courses of antibiotic therapy. However, the duration of antibiotics is largely based on expert opinion. Recent evidence in osteomyelitis demonstrated that shorter durations of treatment were associated with reduced hospital lengths of stay, incidence of adverse events, and emergence of resistance, 40 but evidence supporting shorter durations for PJI is lacking.
Bernard et al. conducted the Duration of Antibiotic Treatment in Prosthetic Joint Infection (DATIPO) trial, an open label, randomized controlled noninferiority trial, in France between November 2011 and January 2015 to compare the efficacy of 6 vs (vs) 12 weeks of antibiotic therapy for PJI. 15 The trial randomized patients ≥18 years of age with a PJI managed by an appropriate surgical procedure to receive either 6 or 12 weeks of effective antibiotic therapy, as determined through susceptibility testing. Randomization was stratified based on surgical procedure (one- or two-stage implant exchange or debridement with implant retention), infected joint (knee or hip), and episode of infection (initial or recurrent). The primary outcome was persistence of infection within two years after completing antibiotic therapy, defined as presence/recurrence of infection by the same causative bacteria as the initial culture, and was assessed by blinded evaluators.
A total of 404 patients were included, of which 203 received 6 weeks and 201 received 12 weeks of antibiotic therapy. The most common surgical management was debridement with implant retention (41.3%) and there were no notable differences in surgical management between groups. Staphylococcus aureus was the most common causative pathogen and caused a higher proportion of infections in those receiving 6 weeks vs 12 weeks of antibiotic therapy (38% vs 30%, respectively). The median duration of intravenous (IV) antibiotic therapy in both arms was 9 days (IQR, 5-15 days) and 51.1% received a combination of rifampin and a quinolone. Based on the primary outcome findings, non-inferiority was not demonstrated with 6 weeks of therapy as compared to 12 weeks of therapy. Persistent infection occurred in 18.1% and 9.4% of patients in the 6- and 12-week groups, respectively (risk difference 8.7%, 95% confidence interval (CI) 1.8-15.6). Sensitivity analyses adjusting for either stratified variables or omitted data from patients who were lost to follow-up also failed to demonstrate non-inferiority of 6 weeks of therapy. Although similar proportions of patients experienced at least 1 serious adverse event (AE) in the 6- and 12-week groups (38.4% vs 35.3%, respectively; P = .52), more patients receiving 12 weeks of therapy experienced nausea and other nonserious AEs compared to those receiving 6 weeks (59.7% vs 47.3%, respectively; P = .01).
Limitations of the trial included the use of a variety of antibiotic regimens and shorter durations of IV therapy than routinely given in the United States. The results of the DATIPO trial were notable for opposing the larger trend in recent stewardship literature supporting shorter courses of therapy and serve to emphasize the importance of selecting appropriate candidates for stewardship interventions.
Collins et al. Antibiotic use in patients with beta-lactam allergies and pneumonia: Impact of an antibiotic side-chain-based cross-reactivity chart combined with enhanced allergy assessment 24
Management of pneumonia may include utilization of β-lactam or non-β-lactam-based regimens. Avoidance of β-lactam antibiotics in patients with reported β-lactam allergies is common despite data showing higher rates of clinical failure and more adverse events with non-β-lactam-based regimens.41-43 Cross-reactivity among β-lactams may be related to R-group side chain homology, and increasing data suggests some β-lactam antibiotics may be safely administered despite a reported allergy to a different β-lactam. An institutional antibiotic side-chain-based cross-reactivity chart was developed at stokes Joseph Mercy Health System and demonstrated improvements in β-lactam prescribing for surgical prophylaxis, 44 but the impact of this tool on the management of other disease states, including pneumonia, is unknown.
Collins et al. conducted a single-center, retrospective cohort study comparing β-lactam utilization in adult inpatients with pneumonia and documented β-lactam allergies before (January 2013 to October 2014) and after (January 2017 to October 2018) implementation of an antibiotic side-chain-based cross-reactivity chart and enhanced allergy assessment in October 2014. The cross-reactivity chart was a color-coded table detailing the likelihood of an allergic reaction based on antibiotic side chains and the patient’s allergy type and severity. Enhanced allergy assessments included pharmacist evaluation of previous β-lactams tolerated, compliance with chart recommendations, and consultation with allergy/immunology for penicillin skin testing, oral challenges, and desensitization if appropriate. The primary outcome was the incidence of β-lactam use per patient encounter between time periods, defined as any receipt of any dose during an encounter. Outcomes were compared with and without propensity-weighted scoring and interrupted time-series analyses.
Overall, 964 patient encounters were included with discharge coding indicating pneumonia and documented β-lactam allergies. Of these, 341 were in the historical cohort and 623 were in the intervention cohort. Several differences were noted between cohorts, including older individuals with higher Charlson Comorbidity Index and internal mortality index scores (derived using laboratory values, past medical history, and admitting diagnoses) in the intervention cohort and higher rates of malignancy in the historical cohort. However, none of these differences were significant following propensity score weighting. β-lactam use per patient encounter increased between the historical and intervention cohorts at 70.4% vs 89.3%, respectively; P < .001, without any change in rates of overall allergic reactions (2.4% historical vs 1.6% intervention; P = .738) or β-lactam-caused reactions (1.3% historical vs .9% intervention; P = .703). There were no differences in the total days of therapy, antibiotic durations, or antibiotic cost per encounter between cohorts. Additionally, although the incidence of hospital-onset Clostridioides difficile infection decreased (1.2% historical vs .2% intervention; P = .032), both inpatient (0% historical vs 6.4% intervention; P < .001) and 30-day (2.4% historical vs 14.3% intervention; P < .001) mortality increased. Neither the change in inpatient nor 30-day mortality appeared related to β-lactam allergies or the study intervention(s).
Limitations of this study include its single-center design and lack of assessment for other variables impacting antibiotic selection, such as culture and susceptibility results. Additionally, β-lactam use was measured throughout the entire encounter and not necessarily for the treatment of pneumonia. As there are multiple high-quality cross-reactivity charts available online and in literature, this publication provides stewardship teams with a model of how to incorporate these charts successfully into practice to improve patient outcomes.
Corsini Campioli et al. Antistaphylococcal Penicillin vs Cefazolin for the Treatment of Methicillin-Susceptible Staphylococcus aureus Spinal Epidural Abscesses
Though uncommon, the incidence of spinal epidural abscesses (SEAs) is rising and remains a significant cause of morbidity and mortality.45-47 Methicillin-susceptible Staphylococcus aureus (MSSA) is the most commonly identified pathogen in SEAs.45,47-49 Treatment includes both surgical and medical interventions, with antistaphylococcal penicillins (ASPs) as first-line therapy and cefazolin as an alternative.49,50 Considerations of cefazolin use include the clinical significance of the cefazolin inoculum effect (CIE), use of clindamycin resistance as a surrogate marker for CIE, and cefazolin’s potentially limited penetration of the central nervous system.51-53 However, recent comparative data show similar efficacy and safety outcomes for cefazolin and ASPs when used for MSSA bacteremia and infective endocarditis.54-56 This study compares the clinical outcomes of cefazolin vs ASPs for the treatment of MSSA SEAs.
Corsini Campioli et al. conducted a single-center, multisite, retrospective cohort study of adult patients (≥18 years of age) from January 2014 to July 2020. 22 Patients with an International Classification of Diseases diagnosis code of SEA were screened and included if all the following criteria were met: SEA findings on imaging via computed tomography (CT) scan or magnetic resonance imaging (MRI), an associated MSSA epidural fluid culture result, and actively receiving treatment with either cefazolin, oxacillin, or nafcillin. Those receiving empiric antibiotics were included if cefazolin or ASPs were administered for >50% of the total duration of therapy. Patients were excluded if additional antibiotic co-administration was >50% of the total duration. Treatment failure was defined as receiving antibiotics longer than 6 weeks, a relapse or recurrence within 3 months following antibiotic discontinuation, and/or SEA-related mortality.
Of the 79 patients included, 45 were treated with cefazolin and 34 were treated with ASPs (29 with nafcillin and 5 with oxacillin). Both groups had similar demographic and baseline characteristics; however, significantly more patients receiving ASPs were admitted to the ICU (29.4% vs 11.1%; P = .048). The rate of S. aureus bacteremia at the time of SEA diagnosis was similar in both groups and all patients received surgical treatment except 1 in the ASP group. While not significantly different, the ASP group had a longer duration of bacteremia (4.5 days vs 3 days; P = .099) and higher rates of infective endocarditis (5.9% vs 0%; P = .18). There was no difference between the ASP and cefazolin groups in the median days of inpatient IV antibiotic treatment (12.5, IQR 5.8-25 vs 9, IQR 5.5-15.5), outpatient IV antibiotic treatment (37, IQR 25-51 vs 38, IQR 30-47), or total days of IV and oral antibiotic treatment (67.5, IQR 45-93.8 vs 55.5, IQR 42.2-96). Extension of antibiotic treatment beyond 6 weeks for treatment failure occurred in 61 patients (77.2%) and was similar in both groups. There were no differences in overall 6-week mortality or SEA-related mortality between groups.
The study’s retrospective nature and small sample size limited its ability to predict treatment failure or discern an optimal treatment duration for those not undergoing surgical treatment. Additionally, the CIE was not measured and could not be investigated as a factor in cefazolin failure. Despite these limitations, this study showed cefazolin may serve as an appropriate alternative to ASPs in the treatment of an MSSA SEA.
Dinh et al. Discontinuing β-Lactam Treatment After 3 Days for Patients with Community-Acquired Pneumonia in Non-Critical Care Wards (PTC): A Double-Blind, Randomised, Placebo-Controlled, Non-Inferiority Trial
Most treatment guidelines for community-acquired pneumonia (CAP) recommend 5-8 days of antibiotics, but the evidence for optimal treatment duration is lacking.57-61 Shorter durations are suggested to limit antibiotic exposure, decrease antibiotic resistance, and reduce adverse events and costs.62-64 This study compares the clinical outcomes of 3 vs 8 days of β-lactam monotherapy in patients admitted for CAP.
Dinh et al. conducted a double-blind, randomized, placebo-controlled, non-inferiority trial with two parallel groups between December 2013 and February 2018, known as the Pneumonia Short Treatment (PTC) trial. 17 Eligible patients recruited from 16 French hospitals were randomized on day 3 of antibiotic therapy to receive an additional 5 days of amoxicillin plus clavulanate (β-lactam group) or placebo. Randomization occurred in a 1:1 fashion after meeting all of the following criteria: ≥18 years of age, admitted for moderately severe CAP, initial receipt of β-lactam monotherapy (IV or oral (PO) amoxicillin plus clavulanate, or parenteral ceftriaxone or cefotaxime), and with clinical response after 72 hours of therapy as evident by select stability criteria. 65 Patients meeting inclusion criteria were discharged on day 3 and received allocated medications. Notable exclusion criteria include severe or complicated community-acquired pneumonia, healthcare-associated infection, suspected aspiration, immunosuppression, and infection due to atypical pathogens. The primary outcome of cure at day 15 was defined as apyrexia, resolution or improvement of signs and symptoms, and no need for additional antibiotics. Secondary outcomes included cure at day 30, all-cause mortality, AEs, pneumonia symptoms and quality of life, and length of hospitalization.
The intention-to-treat (ITT) population included 303 patients (152 in the placebo group vs 151 in the β-lactam group), and the per-protocol (PP) analysis included 291 patients (96%) on day 15. Treatment groups in the ITT population were well matched, with a median age of 73 years (IQR 57-84) and 40.6% female participants. Amoxicillin plus clavulanate was the most common β-lactam given during days 0-3 of hospitalization: 64.5% in placebo vs 64.9% in β-lactam groups. Both ITT and PP analysis indicated non-inferiority of cure at day 15. The ITT analysis showed cure at 15 days in 77.0% of placebo group and 67.5% of β-lactam group patients, with a difference of 9.4% (95% CI, −.4 to 20). The PP analysis showed cure at 15 days in 77.9% of placebo group and 68.5% of β-lactam group patients, with a difference of 9.4% (95% CI, −.2 to 20.3). Analysis of cure at 30 days in both the ITT and PP groups also suggested non-inferiority. In addition, there was no difference in mortality at day 30, proportion experiencing at least 1 AE, or length of hospitalization.
This study has several limitations including the considerable array of exclusion criteria and applicability to patients who have not responded by day 3 of antibiotics. The study also sought to study CAP treatment in a real-world setting and therefore did not attempt to rule out viral etiologies or widely use biomarkers such as procalcitonin or C-reactive protein. Despite these limitations, the authors conclude that discontinuing antibiotics at 3 days of β-lactam therapy was non-inferior to 8 days of treatment in patients admitted for moderately severe CAP if clinical stability was achieved in the initial 3 days of treatment.
Drekonja et al. Effect of 7 vs 14 Days of Antibiotic Therapy on Resolution of Symptoms Among Afebrile Men with Urinary Tract Infection: A Randomized Clinical Trial
Urinary tract infections (UTI) are 1 of the most common infections for which inappropriate antibiotics are often prescribed, especially in the outpatient setting.66,67 Unnecessary and prolonged antibiotic use can lead to a selection for drug-resistant organisms and adverse events. Recently, there has been a growing number of studies shifting the paradigm to use shorter antibiotic courses in infections such as osteomyelitis, pneumonia, and UTI in women.19,68-70 However, the optimal duration of therapy for men with presumed UTI has not been clearly defined.
Drekonja et al. conducted a randomized, double-blind, placebo-controlled, non-inferiority trial at two Veterans Affairs (VA) hospitals from April 2014 to December 2019 which evaluated 7 vs 14 days of antibiotic therapy in afebrile men with symptom(s) suggestive of UTI. 18 Eligible patients included outpatient men aged ≥18 years old with at least 1 UTI symptom (dysuria, frequency, urgency, hematuria, costovertebral angle tenderness, or perineal, flank, or suprapubic pain) who were prescribed 7 or 14 days of either ciprofloxacin or trimethoprim/sulfamethoxazole (TMP-SMZ). Exclusion criteria included UTI treatment in the past 14 days, non-UTI attributable urinary symptoms, fever of ≥38°C, or growth of organisms in urine that were resistant to the study antibiotic (ciprofloxacin or TMP-SMZ). Of note, a urine culture was not required for inclusion. Prior to day 8 of treatment, patients were randomized in a 1:1 ratio to either continue the original antibiotic or receive a placebo for days 8 through 14 of therapy. The primary endpoint was UTI symptom resolution by day 14 after the end of active antibiotic therapy. Secondary outcomes included recurrence of UTI symptoms and adverse events up to day 28 after the end of therapy.
A total of 272 patients were randomized and 254 were included in the primary as-treated analysis set: 131 in the 7-day group and 123 in the 14-day group. There were no statistically significant differences in baseline demographics between 2 treatment duration arms. Of the 239 available urine cultures, 60.7% grew >100 000 colony-forming units/mL of any organism and the most common organism was Escherichia coli (41%). In the as-treated analysis, resolution of UTI symptoms occurred in 93.1% patients in the 7-day arm and 90.2% patients in the 14-day arm, meeting criteria for the noninferiority (difference 2.9%, 97.5% CI -5.2% to ∞). UTI symptom recurrence was observed in 9.9% and 12.9% of patients in the 7- and 14-day treatment arms, respectively (difference −3%, 95% CI -10.8% to 6.2%, P = .7). Similar results were found in the as-randomized analysis. Adverse effects occurred in 20.6% of patients in the 7-day group and 24.3% of patients in the 14-day group. The most common adverse event in both groups was diarrhea (8.8%).
Limitations of this study include the applicability beyond the two study antibiotics, the slightly underpowered sample size, the lack of a urine culture for some participants, and the potential overdiagnosis of UTI which could have affected the statistical differences between two groups. Nevertheless, the study demonstrated that shorter courses of ciprofloxacin or TMP-SMZ may be appropriate for afebrile men with UTI.
Gariani et al. Three Weeks vs 6 Weeks of Antibiotic Therapy for Diabetic Foot Osteomyelitis: A Prospective, Randomized, Noninferiority Pilot Trial
Diabetic foot osteomyelitis (DFO) presents an immense burden for patients due to prolonged hospitalization, high financial costs, and significant morbidity, leading to impaired mobility or amputation.71-74 Although interventions to improve blood circulation and repair foot structure abnormalities may be employed, addressing other underlying factors which contribute to DFO, such as peripheral neuropathy, is not always possible. As a result, many practitioners treat DFO with longer antibiotic courses due to high potential for poor outcomes, including recurrences and complications.75,76 Previous studies, including some highlighted in this review, have shown similar outcomes between shorter and longer duration of antibiotics; however, further adequately-powered trials are warranted to evaluate outcomes of short vs long antibiotic courses for DFO.
Gariani et al. conducted a prospective, randomized, single-center, non-inferiority trial between February 2017 and October 2019 evaluating the outcomes of antibiotic duration for DFO. 19 Adults with a diagnosis of DFO not associated with an implant and with appropriate debridement of all necrotic tissues were included. Partial resection of infected bone was permitted, and subjects were included if residual DFO remained. Patients were assigned in a 1:1 ratio to receive either 3 or 6 weeks of antibiotic therapy for the treatment of DFO. The primary endpoint was clinical remission, defined as absence of clinical and radiological signs of infection, at least two months after the end of treatment. The secondary endpoint was incidence of AE in each study group.
The study included 93 subjects with DFO in the primary ITT analysis, with 44 and 49 subjects randomized to the 3-week and 6-week study arms, respectively. Demographic variables were well balanced between the two groups. The majority of the episodes were osteomyelitis involving the toe (50% in 3-week group vs 63.3% in 6-week group; P=.20), and a similar proportion of patients in each group had partial amputations (36.4% in 3-week group vs 36.7% in 6-week group; P = .97). Although most cases involved polymicrobial infections (51.6%), the most common microbiological etiologies were S. aureus (47.3%) and gram-negative pathogens (30.1%). Among the antibiotic regimens utilized in the study, the most frequent agents used were amoxicillin plus clavulanate (63%), levofloxacin (22%), and clindamycin (17%). Of note, the route of administration was left to the discretion of the treatment provider. Complete remission was achieved in 84.1% of patients in the 3-week arm compared with 73.5% in the 6-week arm (P = .21). Similarly, occurrence of AEs was comparable between the two groups at 38.6% vs 32.7%, respectively; P = .54. With a noninferiority margin of 25%, the short-course group demonstrated noninferiority for both clinical remission (8.5 difference points, 90% CI -24% to 4%) and incidence of AEs (10 difference points, 90% CI -23% to 10%).
Limitations include the small sample size and lack of further stratifications that would identify distinct risk factors impacting treatment outcomes of DFO, such as the presence of limb ischemia. The noninferiority margin of 25% may be too large as opposed to other trials comparing short and long antibiotic regimen durations. Although a larger randomized controlled trial is needed to confirm the results of the study, these findings suggest that 3 weeks of antibiotic therapy, along with surgical debridement, was noninferior to 6 weeks for the treatment of DFO.
Landovitz et al. Cabotegravir for HIV Prevention in Cisgender Men and Transgender Women
Daily oral antiretroviral preexposure prophylaxis (PrEP) with tenofovir disoproxil fumarate (TDF) or tenofovir alafenamide (TAF) with emtricitabine (FTC) has been found to be safe and effective in substantially reducing HIV acquisition.77,78 Medication adherence was found to be a key factor in determining efficacy of oral PrEP agents.79,80 Given its less frequent dosing, the long-acting injectable cabotegravir (CAB-LA) administered intramuscularly into the gluteus muscle every 8 weeks may provide increased adherence and protection as compared to oral PrEP.
Landovitz et al. conducted a phase 2b-3, multicenter, randomized, double-blind, double-dummy, active-controlled trial to evaluate the safety and efficacy of CAB-LA for PrEP between December 2016 and March 2020. 35 Cisgender or transgender women who have sex with men were eligible if they were ≥18 years of age, considered to be at high risk for HIV infection, had a negative HIV serologic test at enrollment and an undetectable HIV RNA viral load ≤14 days before trial entry, had a creatinine clearance of ≥60 mL/min, and were generally in good health. Patients were randomized in a 1:1 ratio to receive either active CAB-LA with placebo TDF/FTC or active TDF/FTC with placebo CAB-LA. The 48-week trial included 3 phases: a 5-week oral-tablet lead-in phase in which 1 active and 1 placebo tablet were given daily to verify safety, an injection phase in which daily oral tablets and a 3-ml intramuscular injection were given at phase start then 4 weeks later and every 8 weeks thereafter, and a pharmacokinetic “tail phase” in which open-label, daily oral TDF-FTC was provided to both groups. The primary efficacy endpoint was incidence of HIV infection at 153-week follow-up. The primary safety endpoint was the occurrence of an AE of grade 2 or higher.
A total of 4570 participants from 43 sites in the United States, Latin America, Asia, and Africa underwent randomization. The ITT population included 2282 in the active CAB-LA group and 2284 in the active TDF/FTC group. Baseline characteristics were similar between the two groups. The trial was stopped early based on efficacy results reviewed in the first preplanned interim end-point analysis at 43 weeks. A total of 52 HIV infections occurred during follow-up, with 12 infections in the CAB-LA arm (incidence rate .4 per 100 person-years) and 39 infections in the TDF/FTC arm (incidence rate 1.2 per 100 person-years). The hazard ratio in the CAB-LA vs TDF/FTC arms was .3 (95% CI .2-.6), which met the statistical criteria for superiority. The direction and overall magnitude of the effect were consistent across pre-specified subgroups and in a per-protocol analysis. Injection-site reactions were reported in 81% of participants in the cabotegravir group and in 31% of participants in the TDF/FTC group, but there were no major safety concerns identified in either group. Although the majority of the participants in the CAB-LA reported injection-site reactions, only 2% chose not to receive further injections as a result.
Limitations include the study’s restriction to transgender women who have sex with men and inadequate TDF-FTC adherence among some participants, which appeared to drive the overall finding of HIV incidence in that group. Despite these limitations, this study shows a long-acting form of PrEP helps to overcome adherence challenges that are common with daily PrEP, and CAB-LA is an effective HIV prevention strategy.
Maertens et al. Posaconazole vs Voriconazole for Primary Treatment of Invasive Aspergillosis: A Phase 3, Randomised, Controlled, Non-Inferiority Trial
Voriconazole remains the standard treatment of invasive aspergillosis, with isavuconazole as an potential alternative due to demonstration of noniferiority to voriconazole in the SECURE trial.81-83 However, voriconazole has notable acute toxicities (eg, visual disturbances, neurotoxicities, QT interval prolongation, and hepatic and psychiatric abnormalities) and long-term toxicities (eg, fluorosis, photosensitivity, and periostitis).84-86 Posaconazole has only been approved for salvage treatment and has not been thoroughly investigated as first-line treatment for invasive aspergillosis. 87 Thus, this study aimed to assess its non-inferiority to voriconazole for primary treatment of invasive aspergillosis.
Maertens et al. performed a risk-stratified, randomized, international, double-blind, double-dummy, controlled trial of posaconazole vs voriconazole for the primary treatment of invasive aspergillosis. 20 A total of 91 study sites in 26 different countries were included from October 2013 to September 2019. Eligible participants were randomized in a 1:1 ratio and stratified based on high risk status (ie allogeneic hematopoietic stem-cell transplantation (HSCT), relapsed leukemia undergoing salvage chemotherapy, or liver transplant) or non-high risk status. Participants received either posaconazole (300 mg IV/PO twice per day on day 1, followed by 300 mg IV/PO once per day) or voriconazole (6 mg/kg IV twice per day on day 1 followed by 4 mg/kg IV once per day, or 300 mg PO twice per day on day 1 followed by 200 mg PO twice per day) for up to 12 weeks. Of note, the voriconazole dose was reduced by 50% in those with hepatic insufficiency (Child-Pugh class A and B). Participants were assessed at weeks 1, 2, 4, 6, and 12 of treatment (including a safety assessment at 30 days after end of treatment), and a survival assessment was conducted 114 days following treatment initiation. The primary efficacy outcome was all-cause mortality until study day 42 in the ITT population with a prespecified noninferiority margin of 10%.
Of the 575 participants included in the study, 288 received posaconazole and 287 received voriconazole. Overall, 62.8% of participants completed the study and 48.9% completed the treatment, with similar proportions in both groups for each instance. The primary efficacy outcome of all-cause mortality occurred in 15.3% of those receiving posaconazole and 20.6% of those receiving voriconazole (treatment difference of 5.3%, 95% CI -11.6 to 1; P < .0001), which met noninferiority. Mortality was notably higher for participants with a median age >57 years, those with extrapulmonary sites of infection, and those with baseline neutropenia. Treatment-related AEs in the ITT participants occurred more often in those receiving voriconazole vs posaconazole (40.1% vs 29.9%, respectively) (difference of −10.2%, 95% CI -17.9 to −2.4). Study drugs were discontinued upon treatment-emergent AEs in 35.5% of those receiving voriconazole and 32.3% of those receiving posaconazole.
Study limitations include a site selection bias for centers able to conduct and transfer diagnostic imaging to study investigators, as well as the lack of voriconazole therapeutic drug monitoring despite it being a standard-of-care practice. Regardless of these limitations, this study supports the use of posaconazole as a first-line treatment for invasive aspergillosis due to its favorable tolerability profile among participants, fewer treatment-related AEs, and similar all-cause mortality rates up to day 42 between groups.
Schweizer et al. Comparative Effectiveness of Switching to Daptomycin vs Remaining on Vancomycin Among Patients with Methicillin-Resistant Staphylococcus aureus (MRSA) Bloodstream Infections
Methicillin-resistant Staphylococcus aureus (MRSA) bacteremia is a serious infection that carries high morbidity and mortality.88,89 Daptomycin is a cyclic lipopeptide antimicrobial that has been approved for MRSA bacteremia and is recommended as a potential first-line treatment option.90,91 In addition, daptomycin use for MRSA bacteremia has become more common, especially in patients with elevated vancomycin minimum inhibitory concentration (MIC) or intolerance to vancomycin (ie, nephrotoxicity).92,93
Schweizer et al. conducted a retrospective, observational cohort study across 124 acute VA hospitals from January 2007 to December 2014 investigating clinical outcomes in patients with MRSA bacteremia who are switched from vancomycin to daptomycin vs those who remain on vancomycin. 23 Patients with blood cultures growing MRSA who were initially treated with vancomycin were included. Patients were categorized in the daptomycin-switch group if they received ≥3 doses for 3-5 consecutive days and were further evaluated for switching early (within 3 days of receiving vancomycin) vs switching at any time during the first hospitalization. Those who received dialysis, passed away within the first 3 days of antibiotic therapy, received another anti-MRSA agent for ≥3 days before starting vancomycin, received vancomycin and daptomycin concurrently for >24 hours, or initially received daptomycin were excluded. The evaluated clinical outcome was 30-day mortality (defined as death within 30 days of vancomycin initiation) in patients who switched to daptomycin compared to those who remained on vancomycin.
A total of 7411 patients with positive MRSA blood cultures and initial vancomycin receipt were included, of which 8.2% switched to daptomycin during their first hospitalization and 1.5% switched to daptomycin within 3 days of vancomycin initiation. The average length of hospital stay was longer for patients who switched to daptomycin compared to those who remained on vancomycin (18 days vs 13 days, respectively; P ≤ .01). Of note, the modified APACHE III score was similar between the daptomycin-switch and non-switch groups. Daptomycin dosing data were available in 589 patients, and the majority (93%) received ≥5 mg/kg dosing. The switch to daptomycin was more likely to occur in younger patients with high vancomycin MICs (≥2 mcg/mL), acute kidney injury, osteomyelitis or endovascular infections, high body mass index, and increased likelihood of receiving another anti-MRSA agent. The multivariate analysis showed that patients who switched to daptomycin early were less likely to die than those who remained on vancomycin (HR .5, 95% CI .3 to .9). However, this association was not observed when comparing those who switched to daptomycin at any time during hospitalization to those who remained on vancomycin (HR .9, 95% CI .7 to 1.1).
Limitations of this study include selection bias and potential confounders due to the nature of retrospective, observational studies. The authors could not assess the adequacy of vancomycin dosing, source control, and/or outpatient antibiotic therapy duration, which could have affected the results. Also, since the study included patients who received vancomycin prior to the start of daptomycin, the results cannot be extrapolated to clinical outcomes in those who are initiated and maintained on daptomycin monotherapy. In conclusion, this study demonstrated that switching to daptomycin within 3 days of starting vancomycin when treating MRSA bacteremia may potentially reduce mortality.
Stewart et al. Meropenem vs Piperacillin-Tazobactam for Definitive Treatment of Bloodstream Infections Caused by AmpC β-Lactamase–Producing Enterobacter spp, Citrobacter freundii, Morganella morganii, Providencia spp, or Serratia marcescens: A Pilot Multicenter Randomized Controlled Trial (MERINO-2)
Expression of AmpC β-lactamase genes can occur in a number of Enterobacterales and non-glucose-fermenting gram-negative organisms by 3 mechanisms: inducible chromosomal resistance, stable chromosomal de-repression, or plasmid-mediated AmpC genes. 94 Inducible chromosomal resistance occurs in the presence of specific antibiotics, particularly third generation cephalosporins, resulting in clinical failure even when the organism appears phenotypically susceptible. Carbapenems and fourth generation cephalosporins demonstrate lower rates of hydrolysis by β-lactamases and are generally considered the β-lactam drugs of choice for treatment of AmpC-producing organisms. 95 Piperacillin and tazobactam are weak inducers of AmpC production while tazobactam demonstrates inhibitory activity against AmpC, making this combination a seemingly viable carbapenem-sparing treatment option for inducible AmpC-producing organisms.96,97
Stewart et al. conducted a pilot international, multicenter, open-label, parallel-group, randomized controlled trial across 7 hospitals in Australia, Singapore, and Turkey from July 2015 to December 2019. 16 Eligible participants were ≥18 years of age and randomized within 72 hours of collection of at least 1 blood culture positive for Enterobacter spp, Klebsiella (formerly Enterobacter) aerogenes, Serratia marcescens, Providencia spp, Morganella morganii, or Citrobacter freundii that demonstrated susceptibility to third generation cephalosporins, piperacillin-tazobactam, and meropenem. Notable exclusion criteria included expected survival of <96 hours and use of concomitant antimicrobials with known gram-negative activity within the first 96 hours. Patients were randomly assigned in a 1:1 ratio (stratified by infecting species and source of infection) to receive meropenem (1 g every 8 hours) or piperacillin-tazobactam (4.5 g every 6 hours). All doses were infused over 30 minutes, and trial drug had to be continued for a minimum of 3 days and a maximum of 14 days after randomization, as determined by the treating clinician. The primary efficacy outcome was a composite of 30-day all-cause mortality, clinical failure (ongoing fever or leukocytosis on post-randomization day 5), microbiological failure (regrowth of index organism between post-randomization days 3-5), and microbiological relapse at 30 days.
Of the 72 patients who were included in the primary analysis population, 38 received piperacillin-tazobactam and 34 received meropenem. Demographic characteristics were similar between groups, except more patients in the piperacillin-tazobactam group underwent surgery in the previous 14 days (14 vs 4) and more patients had liver disease in the meropenem group (4 vs 0). The primary outcome was seen in similar proportions of each group: 28.9% of patients treated with piperacillin-tazobactam as compared to 20.6% of patients treated with meropenem (risk difference 8.3%, 95% CI -12% to 28%). Microbiological failure was more common in patients receiving piperacillin-tazobactam compared to meropenem (13.2% vs 0%; P = .03). This might be due to delayed removal of vascular devices in the piperacillin-tazobactam group (median time to removal 6 vs 4 days). Microbiological relapse rates were not significantly different in those receiving meropenem as compared to piperacillin-tazobactam (8.8% vs 0%, respectively; P = .06).
Limitations to this study include a small sample size, high rate of treatment crossover between the start of empiric treatment and study drug assignment, insufficient definitions and documentation of adequate source control, and lack of β-lactam therapy administration via extended or continuous infusion. In summary, no difference in the primary outcome was seen in those receiving piperacillin-tazobactam or meropenem for the treatment of infections due to AmpC-producing organisms. However, more microbiological failures were seen with piperacillin-tazobactam. A larger clinical trial is required to answer the question of whether piperacillin-tazobactam, when dosed optimally for indication and renal function, is a suitable carbapenem-sparing option for AmpC-producing organisms.
Yeager et al. Comparison of Linezolid Step-Down Therapy to Standard Parenteral Therapy in Methicillin-Resistant Staphylococcus aureus Bloodstream Infections
MRSA-blood stream infections (BSIs) are frequently treated with extended courses of IV antibiotics in an attempt to minimize the risk of relapse and metastatic spread.98-100 These courses, although meant to reduce morbidity and mortality, are also associated with complications (eg catheter-related infections, thrombosis, etc.), decreased convenience, and higher costs compared to oral therapies.98-101 Given these advantages, oral linezolid has recently gained interest as an alternative treatment for MRSA-BSIs. 102 The objective of this study was to assess if linezolid may be a safe and effective oral step-down therapy for the treatment of MRSA-BSIs.
Yeager et al. performed a retrospective cohort study in hospitalized patients with microbiologically confirmed MRSA-BSI that received either linezolid or standard parenteral therapy (SPT) between January 2011 and December 2019. 21 They sought to assess differences in rates of mortality, drug related AEs, and length of stay. The primary outcome was a composite 90-day infection-related re-admission (IRR) due to either a clinical worsening/relapse of any S. aureus infection while receiving therapy or recurrence of any S. aureus infection after therapy completion.
Overall, 215 participants were included with 54 receiving linezolid and 161 receiving SPT (62% vancomycin and 38% daptomycin). Baseline demographics were similar in both cohorts. Participants categorized as 'people who inject drugs’ comprised 35.2% of the linezolid group and 43.5% of the SPT group (P = .284). Acute bacterial skin and skin-structure infection was the most common source of infection (34.4%), followed by bone and joint infections (BJIs) (14.9%), and infective endocarditis (IE) (12.6%). Linezolid was more frequently used in BSI resulting from pneumonia (24.1% vs 7.5% (vancomycin only); P = .001), while SPT was used more in those with IE (15.5% vs 3.7%; P = .023) or BJIs (18% vs 5.6%; P = .026). Severity of illness was similar in both cohorts, except that more participants in the SPT cohort had complicated bacteremia (72% vs 41%; P < .0001) and metastatic foci (45% vs 20%; P = .001). When accounting for this assessment of disease severity, linezolid use was not associated with 90-day IRR (adjusted odds ratio 1, 95% CI .2 to 4.3; P = .986) and there was no difference in 90-day all-cause mortality (3.7% with linezolid vs 6.2% with SPT; P=.487). Additionally, there was no difference overall in drug-related AEs between linezolid vs SPT (16.7% vs 15.5%, respectively; P = .843).
Limitations to this study include its retrospective and observational nature, baseline differences between study groups, potential prescribing bias resulting in more frequent linezolid step-down therapy in lower-risk patients, and initial vancomycin therapy in the majority of patients. Nonetheless, this study contributed to the sparse amount of literature available and found that linezolid step-down therapy achieved similar outcomes to SPT when treating MRSA-BSI. Furthermore, linezolid therapy was associated with no difference in overall AEs and a shorter length of stay.
Conclusion
It is challenging to stay informed on new literature related to ID/antimicrobial stewardship in the calmest of times, much less in the setting of an ongoing pandemic where a rapid influx of additional literature is inevitable. This review aims to aide clinicians by providing summaries of the most impactful ID pharmacotherapy papers of 2021.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
