Abstract
Introduction
Valacyclovir Dosing For Herpes Zoster In Patients With Renal Dysfunction. 3

Case 1
A 66-year old man with ESRD on hemodialysis and a recent diagnosis of herpes zoster infection was admitted to our hospital for new onset lower extremity weakness and gait instability beginning 72 hours prior to arrival. He denied any antecedent trauma, loss of consciousness, fever, chills, shortness of breath, chest pain, numbness, or urinary/fecal incontinence. The patient had been prescribed valacyclovir 1 gram three times daily and hydrocodone/acetaminophen 5 mg/325 mg every 6 hours as needed for herpes zoster, which he had been taking for five days prior. His other medical history included past cerebrovascular accident, type 2 diabetes mellitus, hypertension, peripheral artery disease, and psoriasis. Physical examination was notable for ruptured and crusting vesicles on the right posterior aspect of the back. The patient was alert and oriented but had significant new neurological findings including active tremor, spasticity, slurred speech, loss of coordination, gait abnormalities, dysdiadochokinesia, and abnormal heel-shin and finger-nose tests, indicating cerebellar dysfunction. Laboratory studies were significant for sodium 129 mmol/L, blood urea nitrogen 57 mg/dL, serum creatinine of 9.36 mg/dL and supratherapeutic acyclovir plasma level of 18 mcg/mL (reference range 0.4-2.0 mcg/mL). Computed tomography and magnetic resonance imaging of the brain showed no evidence of acute cerebrovascular accident or other acute pathology. Valacyclovir was held and the patient received emergent hemodialysis on hospital day one. The patient improved dramatically and was able to ambulate by hospital day three. He was subsequently discharged home with outpatient physical and occupational therapy and close nephrology and primary care follow-up.
Case 2
A 72-year-old man with ESRD on hemodialysis and recent diagnosis of herpes zoster infection was admitted to the intensive care unit (ICU) for altered mental status after a fall in his home. The patient denied head trauma but endorsed difficulty with speech the morning prior to the fall. His other medical history included coronary artery disease, heart failure with preserved ejection fraction of 50%, peripheral vascular disease, and obstructive sleep apnea. He had been prescribed valacyclovir 1 gram three times daily for herpes zoster and tramadol 50 mg three times daily as needed for pain one day prior to admission. The patient denied alcohol or recreational drug use, but described dissociative experiences, visual hallucinations, and feeling as if he had ingested alcohol or another substance, all of which began after starting these medications. He denied fever, chills, headache, chest pain, shortness of breath, nausea, vomiting, or diarrhea. In the emergency department, the patient was alert and oriented to person only, pupils were constricted (2 mm) and fixed, speech was slurred, gait was ataxic, and dysdiadochokinesia was noted. Physical exam revealed multiple lesions in various stages of resolution on right side of lower abdomen and back and lateral aspect of right hip.
Laboratory studies were significant for potassium 5.5 mmol/L, phosphate 6.9 mg/dL, calcium 7.7 mg/dL (corrected to 7.9 mg/dL), sodium 134 mmol/L, chloride 93 mmol/L, blood urea nitrogen 55 mg/dL), serum creatinine 9.2 mmol/dL, anion gap 21 mmol/dL, and elevated lactate dehydrogenase of 265 units/L. There was no clinical evidence of infection and computed tomography scan of the brain showed no acute pathology. Rhode Island Poison Control was contacted, and they recommended emergent hemodialysis for suspected valacyclovir toxicity given that the patient was prescribed sixtimes the recommended dose (500 mg daily compared to 1000 mg three times a day). Nephrology was also immediately consulted and agreed with diagnoses of acute valacyclovir toxicity based on symptomatic presentation and valacyclovir dose; subsequently, the patient was transferred to the ICU to receive dialysis.
Valacyclovir and tramadol were held, and the patient received hemodialysis again on hospital days two and three. By hospital day three, the patient’s mental status improved to his baseline level of normal cognition and orientation, and hallucinations resolved.
Discussion
This report describes two cases of VAN. Although the presentation of symptoms differed between the two patients, the onset of neurotoxicity, followed by the return to baseline of clinical and mental status, coincided respectively with the addition and removal of valacyclovir and emergent hemodialysis. Neurotoxicity typically occurs in patients within the first 24 to 72 hours after initiation of therapy, while symptom resolution usually takes two to seven days post-cessation of treatment and hemodialysis. 6 In patients with ESRD, the half-life of valacyclovir can be up to 14 hours, but excretion can be accelerated with hemodialysis. Hemodialysis should be initiated in severe cases of neurotoxicity because acyclovir (the active metabolite) has a low volume of distribution and low protein binding.
Our patients’ clinical presentations were consistent with patterns of valacyclovir-associated neurologic symptom onset and resolution described in prior published case reports. For case two, it is unclear as to why an acyclovir level was not drawn, but it could be postulated the level was not collected due to unanimous diagnosis from Poison Control and nephrology, as well as the critical need for prompt hemodialysis. Medical records stated that the patient presumably had “valacyclovir toxicity exacerbated by tramadol,” which calls to question if tramadol was causative medication. Without an acyclovir level tramadol associated toxicity could not be fully excluded, however tramadol was less likely to be the primary offending agent since it was dosed appropriately. The patient was only prescribed up to 150 mg/day, which is below the manufacturer’s recommended maximum daily dose of 200 mg/day for hemodialysis patients. Additionally, although central nervous system effects such as cognitive dysfunction and lack of concentration are potential significant adverse events of tramadol they are rare and typically dose-related. Most common signs of tramadol toxicity include dizziness, drowsiness, sedated state and headache, none of which were present in this patient. The patient was hypocalcemic at time of presentation, but this calcium level was around the patients baseline of 7.3-8.0 mg/dL secondary to his ESRD. Therefore the dissociative experience, visual hallucinations and ataxic presentation of the patient were attributed to VAN and supported by the inappropriate dose of valacyclovir.
Naranjo Algorithm Scoring of Valacyclovir Potential Role in Patient’s Neurotoxicity 7 .
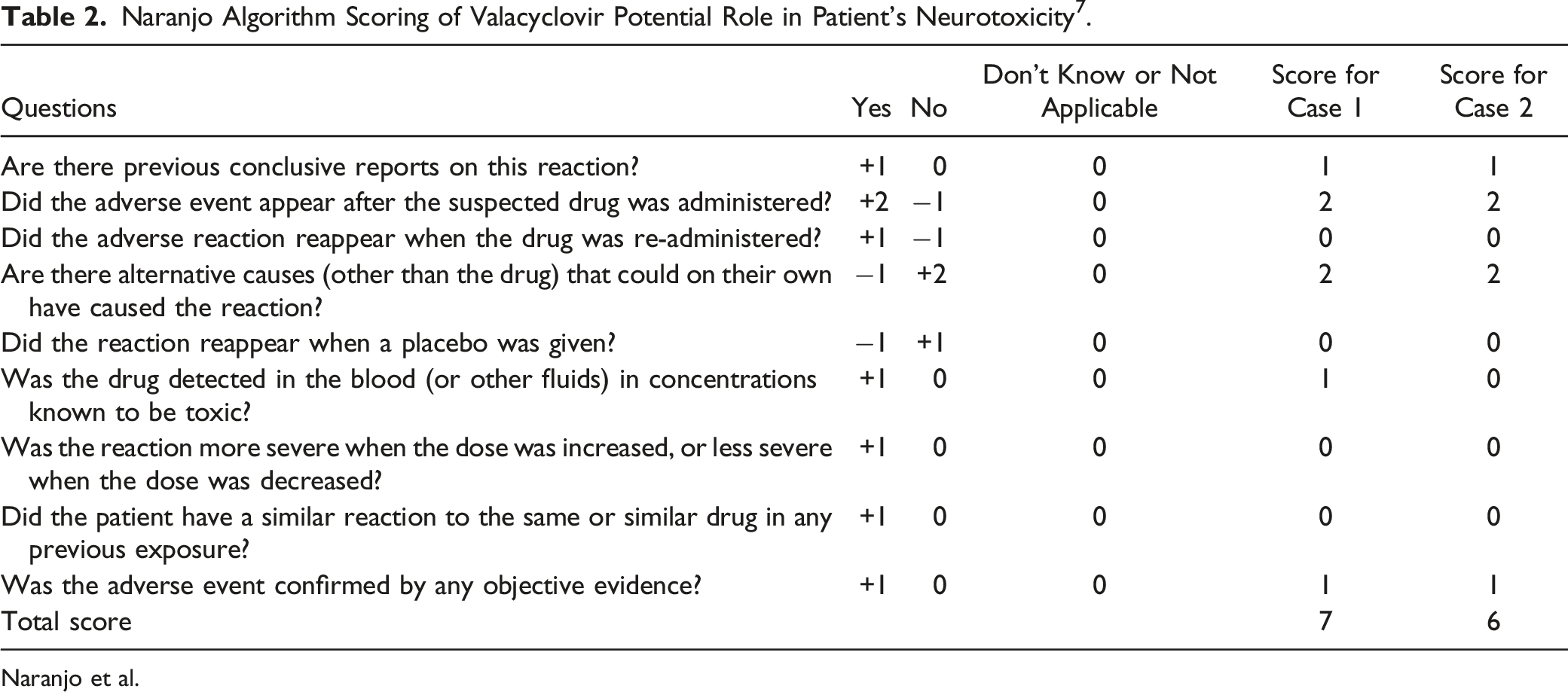
Naranjo et al.
Both of our patients were prescribed a dose of valacyclovir that was six times the recommended dose for their level of renal function (see Table 1 for dosing recommendations). 3 Since valacyclovir relies heavily on the kidneys for excretion, diminished glomerular function can cause a significant increase in the half-life. As a prodrug of acyclovir, valacyclovir results in a 2-fold higher bioavailability which allows for less frequent dosing. In patients with renal dysfunction who cannot clear the drug appropriately, this can lead to an extended half-life resulting in toxic complications. Hemodialysis offers an effective treatment option for patients with ESRD as it can lower readily dialyzable acyclovir plasma concentrations by up to 60% following a 6-hour hemodialysis session. Since there is not a direct relationship between drug levels and symptom resolution, improvement in neurologic symptoms can lag several days after hemodialysis sessions.5,9 Although rare, it is also important to note for patients with preserved renal function who experience VAN treatments can vary and hemodialysis isn’t always the preferred or necessary choice of treatment. In cases of mild to moderate toxicity IV fluid hydration with close monitoring of renal function is an option along with supportive care such as antiemetics for nausea/vomiting. The diagnosis of VAN in the 2 cases detailed above is supported by the inappropriate dosing of valacyclovir and the resolution of symptoms upon removal by hemodialysis and discontinuation of valacyclovir. Additionally, the Naranjo score and previously published literature support this diagnosis. It is prudent to consider herpes zoster encephalitis in the differential diagnosis of patients who present similarly to our patients, especially as the management of this condition warrants continuation, rather than discontinuation, of valacyclovir.
Conclusion
The two cases presented here support previous reports that VAN occurs most frequently in patients with chronic kidney disease or ESRD and can be avoided with dose reductions based on renal function. Patients who develop neurotoxicity as an adverse effect of valacyclovir accumulation should have therapy held or discontinued and hemodialysis initiated emergently. Our case series highlights the absolute importance that patients with renal dysfunction or ESRD receive appropriately dosed valacyclovir in order to prevent the deleterious effects of VAN.
Key Points
(1) Valacyclovir renal dose adjustment is required, especially in ESRD patients, to avoid adverse effects such as neurotoxicity. (2) Common signs of toxicity that may be evident upon patient presentation include disorientation, hallucinations, dysarthria, slurred speech, ataxia, and gait abnormality (3) Hemodialysis can effectively treat excess accumulation of acyclovir and help reverse signs/symptoms of VAN over a period of hours to days.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
