Abstract
Some patients with epilepsy experience seizure clusters, which may be defined as 2 or more seizures occurring within 24 hours. Left untreated, seizure clusters increase the risk for physical injury and may progress to status epilepticus, irreversible neurologic injury, and death. Rescue therapy is based on benzodiazepine treatment. Prompt, appropriate use should be specified in patients’ individualized seizure action plans. Most seizure clusters occur outside the hospital setting. The ideal rescue therapy allows for easy and quick administration by a nonmedical person, which may minimize the need for intervention by emergency medical personnel or transportation to the hospital. In the 2 decades before the approval of 2 intranasal benzodiazepines in 2019 and 2020, rectal diazepam was the only route of administration approved by the US Food and Drug Administration specifically for seizure clusters. Each of the approved intranasal formulations has a unique profile. Both offer a convenient and socially acceptable route of administration. Recognition of seizure clusters and timing of proper administration are key to successful use of rescue therapy. Pharmacists’ counseling plays an important role in reinforcing when and how to appropriately administer rescue therapies and the importance of consistently using rescue treatment when indicated to promote effective management. This review includes resources for pharmacists, patients, and caregivers; reviews currently available treatments; and discusses seizure action plans that support effective treatment of seizure clusters.
Introduction
Patients with uncontrolled seizures are at risk for seizure clusters, a series of seizures that vary from the individual patient’s normal seizure pattern.1,2 Untreated, seizure clusters can increase the risk of physical injury, prolonged seizures, status epilepticus, and death. Seizure clusters also can increase healthcare utilization, emergency-department visits, and hospitalization. 2 Patients with uncontrolled seizures face increased healthcare costs (eg, $23,238/year vs $13,839/year). 3 Additionally, seizure clusters may affect patients’ and caregivers’ quality of life. 4
Pharmacists have an important role in educating patients and caregivers. 5 Patients and caregivers need to understand and recognize seizure clusters as well as understand, communicate, and execute an action plan to deal with these events properly.
The objective of this review is to describe the characteristics of rescue therapies approved for use in the United States for seizure clusters and provide information on effective administration in accordance with an overarching seizure action plan (SAP), or a more focused acute seizure action plan (ASAP). With that goal, literature on seizure clusters, seizure action plans, rescue treatments, and pharmacist counseling was identified and organized to aid pharmacists when they talk with patients and caregivers about appropriate use of seizure cluster rescue medications.
Defining a Seizure Cluster
Roughly half of patients with epilepsy may continue to experience seizures despite administering epilepsy medication,6,7 and patients with frequent seizures are more likely to experience seizure clusters. 8 There is a range of definitions for seizure clusters, 1 including seizure patterns that have been identified across days, weeks, months, and years. 9 One simple definition is the occurrence of ≥2 seizures within 24 hours.1,2,10 Using this definition, 1 analysis found that 58.5% of seizure clusters included a second seizure ≥6 hours after the first, including 21.6% with a seizure ≥12 hours after the first. 10 This highlights the value of 24-hour control. Seizure clusters are recognized as an indication by the US Food and Drug Administration (FDA); however, they are not included in current International League Against Epilepsy classifications or terminology. 11
Commonly Prescribed Rescue Treatments
Benzodiazepine Rescue Therapies by Administration Type.

Abbreviations: AE, adverse event; EMA, European Medicines Agency; FDA, US Food and Drug Administration.
A 2017 study of patients with seizure clusters revealed that only 43.5% had received ≥1 benzodiazepine rescue medication. 25 To address the needs unmet by rectal therapy, intranasal formulations were developed as an alternative. 12 The nasal cavity includes microvilli and an elaborate vascular network, which are ideal for drug absorption. 12 Intranasal delivery avoids first-pass metabolism and can reduce pharmacokinetic variability that occurs with oral or rectal diazepam because of food consumption.26,27 Additionally, the diazepam nasal spray device has been shown to allow for self-administration, 22 which is not feasible with rectal diazepam.
Intranasal midazolam (Nayzilam®, UCB, Inc.) was approved by the FDA in 2019 for patients aged ≥12 years.28,29 If needed, a second dose can be administered after 10 minutes. In a long-term safety study, second doses were used in 38.5% of clusters <6 hours of the first dose. 30 The pH of this formulation developed for intranasal administration is about 5.0 to 9.0, and the drug is packaged as a premeasured dose of 5 mg in a single-use device. 28 To solubilize midazolam, organic solvents including ethanol, PEG-6, methyl ether, polyethylene glycol 400, and propylene glycol are used. 28 Organic solvents may lead to irritation and potentially result in injury to nasal tissue. 31 In the placebo-controlled clinical trial of this formulation, the incidence of nasal discomfort was less than that observed with placebo (5.5% [n = 91] vs 7.7% [n = 2], respectively), with an increase in irritation noted when patients received a second dose (16.3% [n = 43]). 29
Intranasal diazepam nasal spray for patients aged ≥6 years was approved in the United States in 2020 (Valtoco®, Neurelis, Inc.).13,23,32 In a long-term safety study, a second dose was given for 12.6% of clusters within 24 hours, with only 5.8% of patients receiving a second dose in <6 hours. 33 The study permitted enrollment of patients receiving chronic concomitant benzodiazepines (eg, clobazam for Lennox-Gastaut Syndrome), which was also permitted in the rectal gel study but not the midazolam study.19,29,30,33,34 An interim analysis specifically assessed patients who received chronic concomitant benzodiazepines, with 11.1% requiring a second dose of diazepam nasal spray. 34 Of the patients who received concomitant clobazam, only 9.6% of clusters required a second dose. 34 A second dose of diazepam nasal spray can be administered after 4 hours. Diazepam nasal spray is provided in premeasured, single-use sprayers, with dosage based on patient age and weight. 32 The 15- and 20-mg doses require use of 2 sprayers to provide the full dose (ie, 2 sprays of 7.5 or 10 mg). 32 The spray is formulated with the nonionic surfactant n-dodecyl beta-D-maltoside (Intravail® A3) as an excipient for increasing transmission across mucosae, 35 and the formula uses vitamin E to increase the nonaqueous solubility of diazepam. 23
Off-label options (Table 1) have also been used for rescue therapy, 36 particularly when rectal diazepam was the only therapy supported by a clinical trial program. In the 2017 study of patients with seizure clusters, off-label benzodiazepine use was common. 37 Among off-label treatments, oral lorazepam was used most frequently (28.9%), followed by oral diazepam (7.0%), midazolam for injection given intranasally via an atomizer (6.9%), and oral clonazepam (5.4%); in comparison, 7.8% of patients used rectal diazepam. Off-label medications have not undergone FDA review for the indication and can be associated with negative effects. Some off-label oral treatments (eg, orally disintegrating lorazepam or clonazepam) show potentially delayed absorption or bioavailability, the potential for aspiration, and a risk of biting injury.12,38 Midazolam for injection is not formulated in a concentration for delivery to the nasal cavity, which is limited to 100 to 200 μL to avoid leakage and swallowing. Moreover, the pH of the intravenous formulation is low (∼3) and may lead to nasal discomfort. 12
Adverse Events
Long-term safety studies have been completed for the 3 approved formulations. In a study of diazepam rectal gel (n = 149), somnolence was the most common adverse event (AE; 17% of patients, with 9% considered to be treatment-related AEs), but the investigators noted that treatment-related somnolence was difficult to distinguish from post-ictal somnolence. 19 Other treatment-related AEs were mild to moderate. No respiratory compromise or serious AEs were attributed to diazepam rectal gel. Three patients withdrew owing to AEs that were possibly treatment related. 19 The long-term safety evaluation of intranasal midazolam (n = 161) enrolled patients who had completed a prior placebo-controlled trial.29,30 In the safety study, the most common AEs were nasal discomfort (12.4%) and somnolence (9.3%), and there were 2 cases of mild respiratory depression, neither of which led to study discontinuation. 30 AEs related to nasal administration occurred in 17.4% of patients and were not unexpected for this route of administration. Four serious AEs were categorized as possibly treatment related (all “unlikely related”). 30 Of note, before randomization in the parent study, 1.7% of patients in the test-dose phase experienced respiratory depression and were excluded from the comparative phase. 29 Current labeling suggests that intranasal midazolam spray should first be administered under professional healthcare supervision before prescribing it as a treatment for those who are at greater risk of respiratory depression from benzodiazepines. 28 In the long-term safety study of diazepam nasal spray (n = 163), the most common AEs, other than seizure, were nasopharyngitis and upper respiratory tract infection (12.3% each) and pyrexia (10.4%); somnolence occurred in 6.7% of patients, with 1.8% considered treatment related. 33 Only one treatment-related AE occurred in >5% of patients (nasal discomfort in 6.1%), and there were no cases of respiratory depression. No patients withdrew owing to a treatment-related AE, and there were no treatment-related serious AEs. 33 In an interim analysis of concomitant benzodiazepine use, treatment-related treatment-emergent AE rates were similar between patients who did/did not receive clobazam (18.2%, 18.7%, respectively). 34
Benzodiazepines have boxed warnings for concomitant use with opioids owing to increased risk of profound sedation, respiratory depression, coma, and death. Additionally, Medicare requires special medication reviews of individuals who are taking benzodiazepines with opioids or gabapentinoids. If the patient or caregivers are unable to adequately monitor respiratory status with concomitant treatment, the pharmacist should discuss treatment options with the physician. Because benzodiazepines also have an addiction potential, a patient’s risk for addiction and misuse should also be assessed. In addition, if used more frequently than recommended, abrupt discontinuation or rapid dosage reduction may precipitate acute withdrawal reactions, which can be life-threatening.15,28,32,39 Pharmacists should discuss these issues when instructing patients.
Seizure Action Plans
Treatment action plans for chronic conditions can help improve knowledge, confidence, and quality of life. 40 Potential benefits of SAPs, or seizure emergency, response, or management plans, 41 and ASAPs include providing patients and caregivers with an increased sense of control, which may help address stress, helplessness, or fear that may affect their daily activities.4,42 Importantly, SAPs may facilitate transition of patient care from 1 provider to another, including from adolescent to adult care. 43
Pharmacists can help ensure that patients with epilepsy have an SAP and that those with seizure clusters have an ASAP.41,42,44 The SAP is a personalized patient guideline for the prevention and treatment of seizures.44-46 ASAPs specifically focus on prompt treatment during a seizure, from watchful waiting, to appropriate and timely use of rescue therapies, to calling for emergency healthcare services. 44 SAPs and ASAPs should be individualized, considering the patient’s age, abilities, condition, treatment regimen, situation, and support structure. The plans should include seizure presentation and distinguish seizure clusters from the patient’s usual seizure pattern, and they should provide detailed instructions on the steps for administering prescribed rescue medication. 44 They should be concise, with easy-to-follow steps using easily understood terms for caregivers who may be highly stressed. Patients should be encouraged to keep a diary of their seizures and treatment use, and to keep the SAP/ASAP up to date. Plans should be reviewed by the patient and caregiver and their healthcare team yearly or when there is a change in medication. 44
Role of the Pharmacist
Patients’ awareness of the role of the pharmacist outside of distribution of prescriptions may be limited. Surveys indicate that pharmacist follow-up counseling most commonly focuses on questions of frequency of medication use. 47 Pharmacists can support patients and caregivers in achieving and maintaining control of seizures, 48 as they do with asthma and diabetes.47,49,50 Pharmacists have a professional responsibility to educate patients and caregivers on medications and, as essential members of patients’ healthcare teams, can provide ongoing counseling and problem solving.5,37,48-50
In a pediatric setting, pharmacist counseling has demonstrated improved caregiver knowledge, confidence, and perception of epilepsy. 37 Specifically, caregiver scores improved with pharmacist counseling in the following areas: medication administration and AEs, acute management of seizures (including when to seek professional medical attention), and general knowledge of care for a person with epilepsy. Moreover, pharmacist counseling resulted in high levels of caregiver satisfaction with their pharmacist. 37
Somewhat more broadly, patient counseling can increase familiarity with the epilepsy lexicon, including the clinical lexicon developed by The Rescue Therapy Project initiated by the Epilepsy Foundation, 41 which may improve patients’ understanding of their treatment plans.1,41 Patients may refer to seizure clusters by other terms, such as acute repetitive seizures, seizure flurries, serial seizures, or crescendo seizures; others may describe seizure clusters by the way they affect daily life.1,44 Rescue therapies may simply be referred to as “treatment used to stop a seizure or seizure cluster.” 41 SAPs and ASAPs can help ensure consistent use of terminology. 44
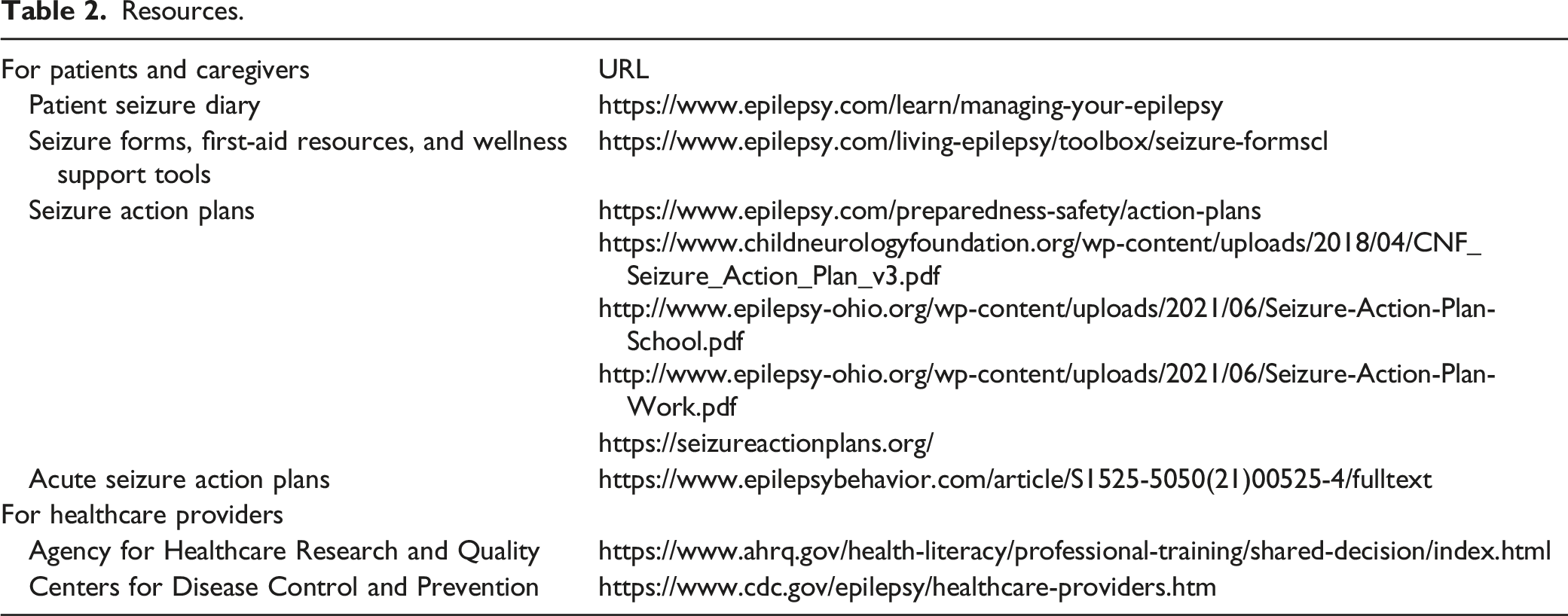
Resources.
Pharmacists can discuss the different treatment options available and clarify why a specific rescue therapy was prescribed. In cases where an off-label drug has been prescribed, pharmacists may counsel patients to talk with their physicians about whether 1 of the newer, approved formulations should be considered. In many states, collaborative practice agreements may allow pharmacists to prescribe and/or renew rescue medications. 51 When a new therapy is prescribed, pharmacists can follow up with patients at intervals to assess side effects and efficacy.
Pharmacists can also communicate the importance of adherence and reinforce how and when to use rescue therapy to help minimize underdosing. Underdosing of benzodiazepines for the treatment of status epilepticus, for example, may occur in the setting of emergency medical services. 52 In a study that examined underdosing of benzodiazepines for treatment of status epilepticus, 21.4% of diazepam, 85.7% of midazolam, and 76.1% of lorazepam doses did not meet dosing guidelines (underdosed), which the authors suggested could be possibly due to caution regarding the risk of cardiorespiratory depression. 52 However, respiratory and circulatory complications are higher in untreated patients. 53
It is important to discuss side effects and safety considerations associated with pharmaceutical treatments, including boxed warnings (risk of concomitant use with opioids, abuse, misuse, addiction, and dependence and withdrawal reactions after used more frequently than recommended) and use in patients with acute narrow-angle glaucoma.15,28,32,54,55 For patients receiving off-label clonazepam, significant liver disease is a contraindication. 55
Pharmacists should ask patients to share their individualized SAPs/ASAPs and ensure that the patient and caregiver understand terms and instructions, know when to administer rescue medication accurately and appropriately, and recognize when emergency medical assistance should be sought. Ideally, pharmacists should evaluate caregiver administration technique as well. 56 Reviewing the SAP/ASAP also provides the pharmacist with an opportunity to facilitate medication and chronic care management, review medication safety, and ensure that the prescription matches the plan, which will help gauge whether the plan is outdated. Pharmacists are in a unique position to monitor increases in rescue therapy usage. Although there may be substantial variability of seizure clusters over time, increased usage may also signal a need to reconsider the patient’s current antiseizure drug regimen, and pharmacists may counsel patients to contact their clinicians to review possible changes in seizure status. Further, pharmacists can contribute to development/revision of SAPs and ASAPs, which may be crucial in transitions of care (eg, from pediatric to adult practice).
Conclusion
Pharmacists participate in the care of patients by educating them about their condition and can help them optimize their treatment. As members of patients’ healthcare teams, pharmacists play an important role in counseling patients to reinforce prompt, appropriate management and use of rescue medication as directed by an SAP or ASAP. Pharmacists can help bridge gaps in communication between patients, caregivers, and other healthcare providers.
Footnotes
Acknowledgments
Editorial support was provided by Laura J. Herold, MA, and Agnes Laskowski, PhD, of The Curry Rockefeller Group, LLC (Tarrytown, NY), and was funded by Neurelis, Inc. (San Diego, CA).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Gidal is a consultant for Aquestive, Eisai Inc., Greenwich, and SK Life Science; is a member of the End Point Review Committee for Sunovion Pharmaceuticals Inc; and has a grant/contract with UCB Inc. Dr. Welty has nothing to disclose. Dr. Cokley is on the speakers bureau for UCB Inc. Dr. Farrell has nothing to disclose. Ms. Shafer has been a consultant for Neurelis, Inc. and for the Epilepsy Foundation. Dr. Rabinowicz is an employee of and has received stock options from Neurelis, Inc. Dr. Carrazana is an employee of and has received stock and stock options from Neurelis, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Neurelis, Inc. (San Diego, CA).
