Abstract
Introduction
Acute kidney injury (AKI) remains a common complication among adult hospitalized patients. Overall, about 20% of patients experience AKI during their hospital stay.1,2 Mortality risk is higher in patients developing AKI compared to those without and incrementally increases with increasing severity. 3 Drug-induced AKI accounts for up to 27% of cases, while contributing to death in 30% of these patients. 4 Clinicians must remain vigilant administering nephrotoxic medications to avoid or mitigate the AKI from transpiring and/or progressing.
The concomitant administration of vancomycin (VAN) and piperacillin/tazobactam (PTZ) has been recently recognized as potentially carrying a higher risk of AKI compared to monotherapy of either agent alone (VAN or PTZ) or combination VAN with other anti-pseudomonal beta-lactams including cefepime (CEF) or meropenem (MER).5-11 However, other studies have not demonstrated an increased risk of AKI with concomitant VAN+PTZ therapy.12-14 Major differences among these studies including AKI definitions and combining critically ill with general ward patients among cohorts as well as including early onset AKI (ie developing ≤48 hours after exposure) may explain these conflicting results.
Most previously published reports comparing the incidence of AKI associated with combination VAN+PTZ and VAN+CEF/MER in the general ward population lacked study design robustness attributed to single-center evaluations with small sample sizes. Unfortunately, none of these studies completely characterized AKI including time to onset, staging of severity including the need for renal replacement therapy (RRT), duration of AKI and/or RRT, and recovery patterns. Therefore, the purpose of this multicenter, propensity score-matched study was to compare the incidence of AKI in the adult general ward population associated with concomitant VAN+PTZ compared to VAN with either CEF or MER as well as characterize AKI development and recovery patterns.
Methods
Patients and Study Design
This was a multicenter, retrospective, propensity-matched, cohort study at a large health-system over a 2-year period (January 1, 2018 to December 31, 2019). Included patients met the following criteria: (1) adults (≥18 years); (2) VAN with anti-pseudomonal β-lactam combination therapy (PTZ, CEF, or MER) started ≤48 hours of each other; (3) combination therapy duration was at least 48 consecutive hours; (4) baseline serum creatinine (SCr) recorded ≤24h after hospital admission; and (5) ≥1 vancomycin level(s) during combination therapy. Exclusion criteria were AKI developing ≤48 hours after initiation of combination therapy; PTZ, CEF, or MER administered in the same hospital admission; baseline end-stage renal disease, or pregnancy.
Vancomycin was managed by clinical pharmacists at the study sites through a standardized protocol. Pharmacists were expected to modify all initial ordered doses to provide 20-30 mg/kg up to a maximum of 2 g as a loading dose, if necessary. Also, pharmacists were able to modify subsequent maintenance doses based on the patient’s body weight and renal function (creatinine clearance) including any renal replacement therapy (eg hemodialysis). The target vancomycin serum concentration was 10-20 mcg/mL; however, a level of 15-20 mcg/mL may be appropriate for certain infections such as meningitis. The vancomycin protocol recommended clinical pharmacists should order the first vancomycin trough level just prior to the third to fifth dose. Pharmacists were permitted to obtain a vancomycin level sooner if the clinical situation warranted it such as significant renal function fluctuations. It was also recommended that pharmacists obtain subsequent vancomycin levels not to exceed 5-7 days since last level.
Broad-spectrum antibiotics including PTZ, CEF, and MER were dosed by pharmacists also using a standardized protocol based on creatinine clearance among all study sites. Furthermore, all hospitals used a standardized approach of dosing extended-infusion PTZ during the study period. The initial PTZ dose was 4.5 g infused over 30 minutes irrespective of renal function. Subsequent PTZ doses consisted of 3.375 g over 4 hours with a frequency of every 8 hours in patients with normal renal function (ie, creatinine clearance ≥20 mL/min) or every 12 hours (ie, hemodialysis or creatinine clearance <20 mL/min). Also, CEF and MER were administered as 30-minute traditional infusions. Although PTZ, CEF, and MER dosing protocols were available to standardized management, pharmacists were allowed discretion to customize dosing based patient specific parameters (infection location, extreme over- or underweight, severity of illness, and/or microbiologic sensitivity information). Lastly, empiric broad-spectrum antibiotics (PTZ, CEF, and MER) including vancomycin was at the discretion of the primary attending physician.
Data Collection
Patients were identified through the electronic health record for potential study inclusion following Institutional Review Board approval. The two study groups consisted of patients administered adjunctive VAN+PTZ or VAN with either CEF or MER. Data collection included patient characteristics (age, sex, weight, comorbidities, etc.) and other clinical covariates such as baseline SCr, antimicrobial duration, nephrotoxic agent(s) and duration, of concomitant, vancomycin characteristics (total daily dose, serum concentrations), Charlson Comorbidity Index, sepsis diagnosis, mortality, and length of stay (LOS). The follow-up period was 7 days total unless antimicrobial therapy was discontinued or the patient died or discharged prior.
Definitions
The same day of hospital admission which both beta-lactam therapy and vancomycin were administered was defined as baseline regardless of whichever of the two was initiated first. The definition and severity staging of AKI was modified from the Kidney Disease Improving Global Outcomes (KDIGO) published guidelines using only the SCr-based criteria since most general ward patient population at the study sites did not have foley catheters routinely placed for urine output collection. 15 Patients identified with a maximum SCr increase of ≥0.3 mg/dL within 48 hours or any SCr during the follow-up period exceeded 1.5 times over baseline were defined as having AKI. The KDIGO staging criteria was modified by removing urine output to classify AKI severity in our study. The following stages were defined as follows: stage 1 AKI was a SCr increase to 1.5-1.9 times greater than baseline or SCr increase ≥0.3 mg/dL; stage II AKI was a SCr increase to 2.0-2.9 times greater than baseline; and stage III AKI was a 3-fold increase in SCr from baseline, SCr ≥4.0 mg/dL, or initiation of renal replacement therapy. Acute kidney injury recovery patterns were classified as fast (<3 days), intermediate (3-7 days), or slow (>7 days) based upon the time-period of SCr returning to either <0.3 mg/dL or <1.5 times from baseline values.16,17 Sepsis was defined using the Sepsis-3 definition in patients with a Sequential Organ Failure Assessment (SOFA) score ≥2 in the setting of known or suspected infection. 18 All patients with a SOFA score ≥2 were considered septic since all included study patients were administered broad-spectrum antimicrobials for suspected infection. The following medications were defined as “nephrotoxic” agents based on previous reports: acyclovir, amphotericin (lipid and deoxycholate formulations), angiotensin inhibitors, carboplatin, cefotaxime, ceftazidime, cefuroxime, cidofovir, cisplatin, colistimethate (systemic), cyclosporine, dapsone, foscarnet, ganciclovir, aminoglycosides (systemic), ifosfamide, non-steroidal anti-inflammatory drugs, lithium, mesalamine, methotrexate, nafcillin, sirolimus, sulfasalazine, tacrolimus, topiramate, valacyclovir, valganciclovir, and zonisamide.16,19
Study Outcomes and Statistical Analysis
The primary outcome was the incidence of AKI associated with concomitant VAN+PTZ compared to VAN+CEF/MER. Secondary endpoints included AKI severity, incidence and duration of RRT, time to AKI onset and recovery. Other secondary aims included clinical outcomes such as in-hospital mortality and hospital LOS. A total sample size of 360 patients equally distributed in each study group (n = 180) was needed to achieve 80% power to detect a 12% difference in AKI rates assuming rates of 27% and 15% for the VAN+PTZ and VAN+CEF/MER groups, respectively. Student’s
Cox competing risk model with cause-specific proportional hazards was used to estimate the AKI risk of VAN+PTZ compared to VAN+CEF/MER. In-hospital mortality was considered a competing risk. The full model included age, gender, baseline creatinine clearance, antimicrobial duration of each specific agent (vancomycin and beta-lactam), vancomycin average weight-based daily dose, ≥3 concomitant nephrotoxic agents, Charlson Comorbidity Index, and sepsis diagnosis. Backward stepwise selection was used to identify significant covariates. A secondary post-hoc Hosmer-Lemeshow test was conducted to assess a goodness of fit test for the robustness of our findings associating VAN+PTZ with AKI risk found in our final prediction model (See Supplemental Table 1). A propensity score balance plot was also constructed to determine equal covariate distribution between both study groups (See Supplemental Figure 1). The propensity score was also included as an additional covariate in the final regression model to address imbalances of baseline characteristics. Statistical analyses were performed using Stata software, version 15.1 (StataCorp. College Station, Texas).
Results
Baseline Characteristics
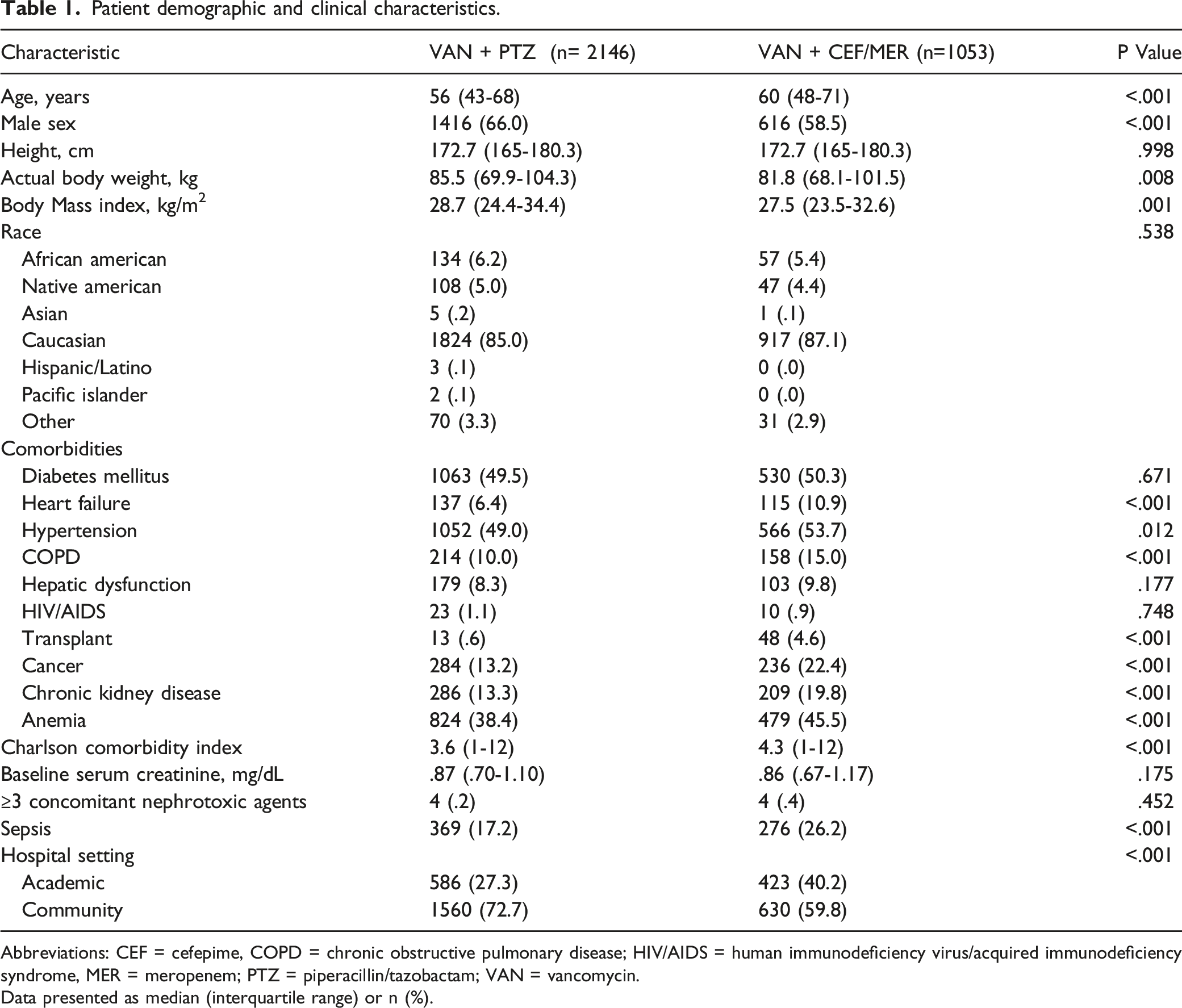
A total of 25 437 patients were identified during the study period with 3199 patients meeting inclusion criteria (Figure 1). A total of 3199 patients were included with 2146 (67.1%) and 1053 (32.9%) patients in the VAN+PTZ and VAN+CEF/MER groups, respectively. Several baseline characteristics were significantly different between study groups (Table 1). The Charlson Comorbidity Index was significantly higher in the VAN+CEF/MER group ( Flow chart of patient screening and selection process. Patient demographic and clinical characteristics. Abbreviations: CEF = cefepime, COPD = chronic obstructive pulmonary disease; HIV/AIDS = human immunodeficiency virus/acquired immunodeficiency syndrome, MER = meropenem; PTZ = piperacillin/tazobactam; VAN = vancomycin. Data presented as median (interquartile range) or n (%).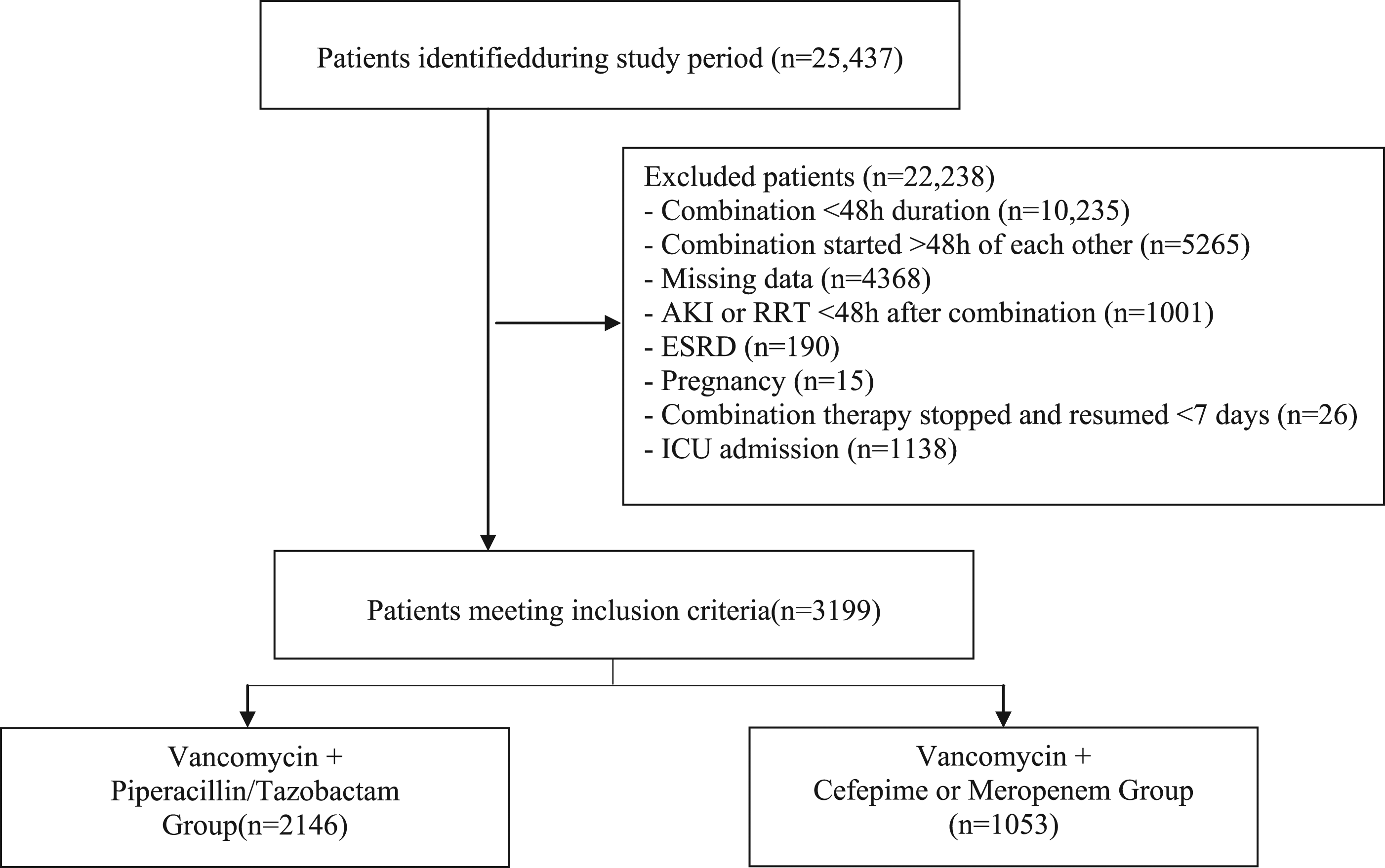
AKI Incidence and Characteristics
Outcomes associated with concomitant vancomycin and anti-pseudomonal beta-lactams.
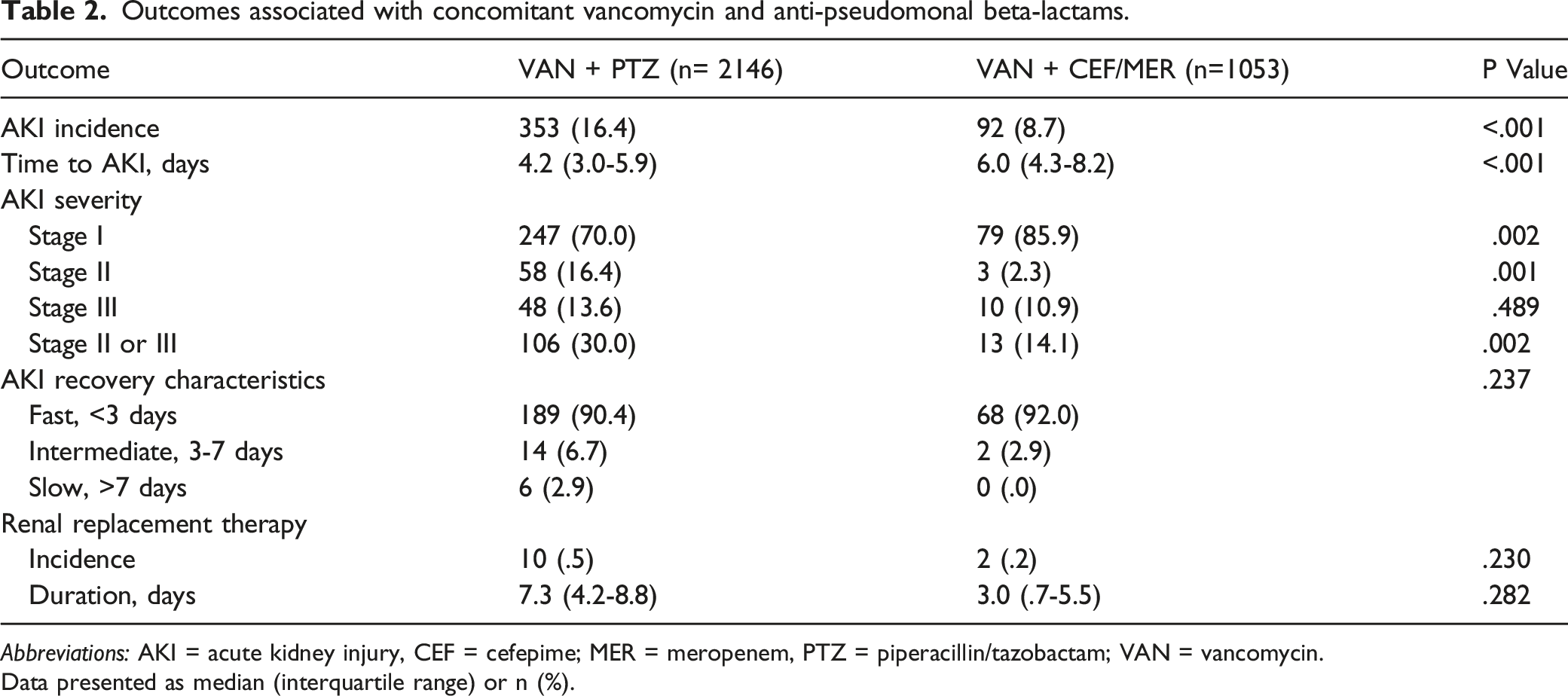
Data presented as median (interquartile range) or n (%).
Stepwise Multivariate Cox Regression Analysis Predicting Acute Kidney Injury.

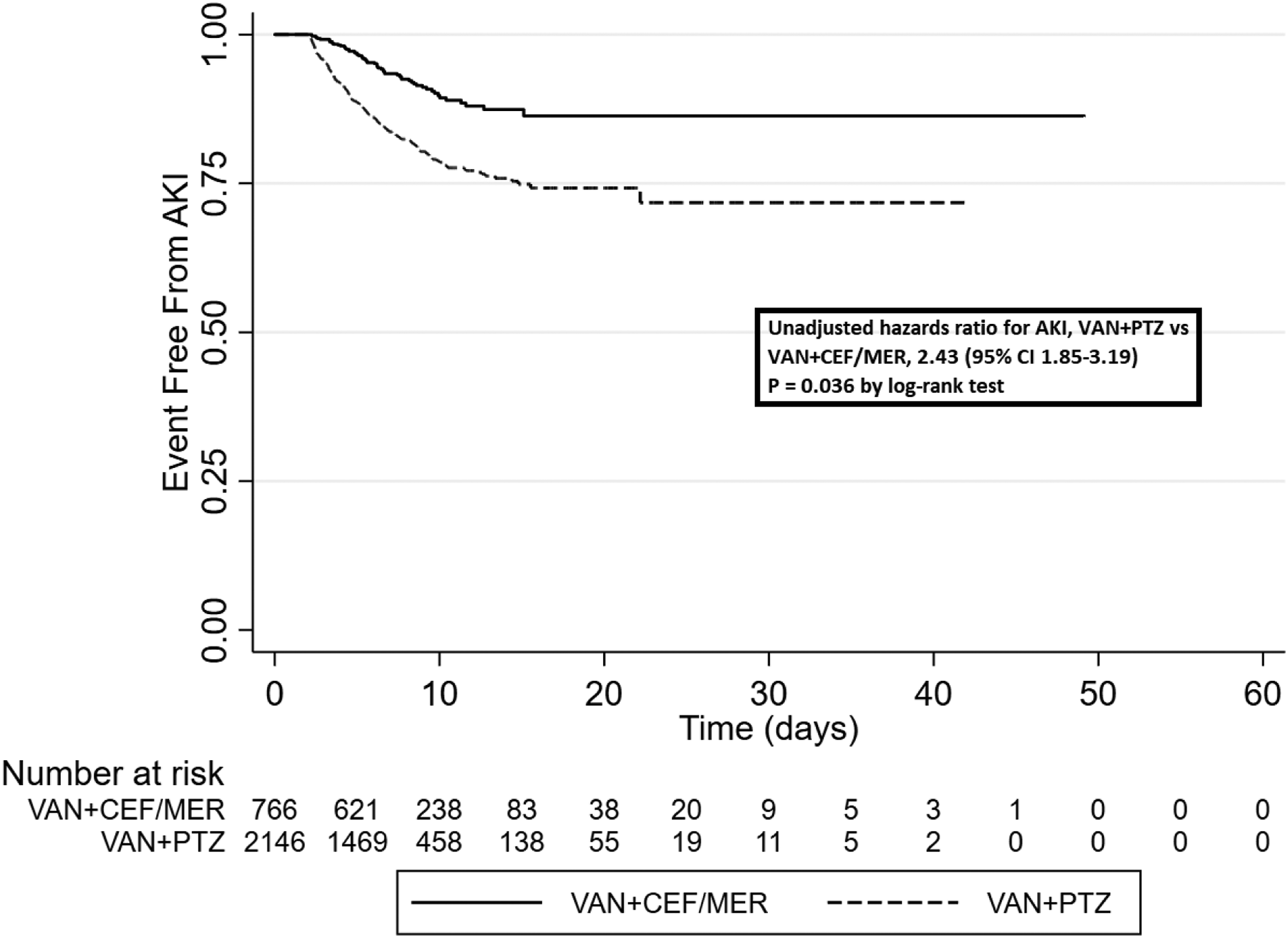
Unadjusted kaplan-meier survival analysis of acute kidney injury.
Mortality and Length of Stay Outcomes
In-hospital mortality was very low although a significant difference was found between VAN+PTZ (0.3%) and VAN+CEF/MER (1.0%) (
Discussion
To our knowledge, this was the first large multicenter, propensity-score matching study conducted exclusively in general ward patients to evaluate AKI incidence, severity, and recovery characteristics receiving VAN+PTZ or VAN+CEF/MER. Concomitant VAN+PTZ was identified as an independent risk factor for AKI in our final model and sensitivity analysis. Although most AKI cases were considered mild (Stage I) for both study groups, higher rates of stage II/III AKI were found with VAN+PTZ over VAN+CEF/MER. In addition, time to AKI was found to have a significantly more rapid onset with VAN+PTZ regimen as compared to VAN+CEF/MER. No differences were noted in the need for RRT nor duration of RRT. Although routine use of extended-infusion PTZ was administered at the study sites, this dosing strategy has not been shown to impact AKI rates compared to traditional 30-minute infusions.13,20 Although clinical outcomes between groups were significantly different favoring VAN+PTZ, caution is warranted in interpreting these results considering the overall mortality rate was low.
Multiple studies have investigated AKI rates associated with broad-spectrum beta-lactam agents, namely CEF and/or MER, against PTZ with concomitant VAN. Although conflicting evidence currently exists associating an increased AKI risk with VAN+PTZ, most published studies have demonstrated a relationship. Moenster et al,
12
first reported the incidence of AKI with VAN+PTZ (29.3%) and VAN+CEF (13.3%) during a retrospective review of osteomyelitis treatment in diabetics, although failing to achieve statistical significance (
Comprehensive characterization of AKI associated with VAN+PTZ is lacking. Our study found the time to AKI was significantly shorter with VAN+PTZ (4.2 vs 6 days), which was a similar to the findings or other previously published reports suggesting onset to AKI may occur 3-5 days after initiation of therapy. 11 Considering the estimated time period for developing VAN+PTZ-induced AKI, patients with early onset AKI <48 hours after initiation were excluded as an attempt to mitigate bias from other causes than the agents being investigated. One other study also excluded patients who developed AKI within 48 hours of hospital admission to potentially reduce AKI resulting from acute illness rather than antimicrobial nephrotoxicity. 10 Our findings further support the importance of de-escalation practices by antimicrobial stewardship programs about 72 hours following concomitant VAN+PTZ therapy as a method to potentially avoid unwarranted nephrotoxic risk. Furthermore, our study found most AKI cases were considered mild (Stage I KDIGO) with more severe (stage II or III) AKI developing with VAN+PTZ. This is consistent to another recent report suggesting a greater risk of more severe AKI (AKIN and RIFLE) with VAN+PTZ was shown irrespective of the severity staging criteria used. 11 The few studies in non-critically ill patients evaluating the need for RRT found no differences between groups, which is also consistent with our findings.11,21,22 Our study found no significant differences between AKI recovery patterns between groups. Also, the majority of AKI was transient with resolution occurring within 3 days. This is in contrast to another study reporting over 60% of patient in both groups did not recover during the evaluation period. 21
This study had several limitations. The retrospective study design may introduce a risk of bias. Another limitation was exclusively relying on SCr to identify AKI. Given our general ward patient population, UOP was not routinely measured and consequently unavailable to compliment AKI diagnosis. Another important limitation of this study was using SCr as the lone biomarker to define AKI. An alternative viewpoint was proposed suggesting SCr elevation may occur with VAN+PTZ in the absence of kidney injury as a result of decreased renal secretion of SCr. 23 This may have impacted our ability to accurately identify patients with AKI rather than VAN+PTZ-induced SCr fluctuations. Furthermore, our study sites only reported vancomycin serum trough concentrations as the primary method of monitoring vancomycin therapy for safety and efficacy. Recently published clinical practice guidelines recommend the area under the curve/minimum inhibitory concentration ratio as preferred pharmacokinetic/pharmacodynamic target for both safety and efficacy. 24 Nonetheless, it should be noted the mean vancomycin serum trough concentration in our study was >15 mcg/mL. This may have biased AKI risk associated with vancomycin over the combination therapy.
Conclusion
Concomitant administration of VAN+PTZ in an adult general ward patient population was identified as an independent risk factor for developing AKI. The incidence of more severe AKI was higher with VAN+PTZ over VAN+CEF/MER. Further research is warranted on AKI risk associated with antimicrobial combination therapy.
Supplemental Material
Supplemental Material - Vancomycin With Concomitant Piperacillin/Tazobactam vs. Cefepime or Meropenem Associated Acute Kidney Injury in General Ward Patients: A Multicenter Propensity Score-Matched Study
Supplemental Material for Vancomycin With Concomitant Piperacillin/Tazobactam vs. Cefepime or Meropenem Associated Acute Kidney Injury in General Ward Patients: A Multicenter Propensity Score-Matched Study by Ivan A. Komerdelj, PharmD, Mitchell S. Buckley, PharmD, FASHP, FCCM, FCCP, BCCCP, Paul A. D’Alessio, PharmD, Delia S. Ziadat, PharmD, BCPS, Emir Kobic, PharmD, BCIDP, Pooja Rangan, MBBS, MPH, Sumit K. Agarwal, MBBS, MBA, Nicole C. Tinta, PharmD, MBA, BCPS, Melanie J. Yerondopoulos, PharmD, BCCCP, and Sandra L. Kane-Gill, PharmD, MSc, FCCM in Journal of Pharmacy Practice.
Footnotes
Author’s Note
This original research was accepted as an abstract for poster presentation at the 2021 American College of Clinical Pharmacy Annual Meeting.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
