Abstract
Background
Over the past decade, the use of direct oral anticoagulants (DOACs) has increased within the United States. 1 As compared to warfarin, growing evidence demonstrates decreased rates of thromboembolism and a similar to decreased bleeding risk with DOAC use.2-5 Prior to the approval of andexanet alfa (AA), the management of major life-threatening bleeding with select DOAC use posed complex challenges involving non-specific reversal strategies. 6 Non-specific agents, such as four-factor prothrombin complex concentrate (4F-PCC), demonstrate variable efficacy in achieving hemostasis in this population.7,8 Approved in 2018 by the Food and Drug Administration (FDA), AA, a recombinant modified human factor Xa (FXa) protein, is the first targeted reversal agent indicated for life-threatening or uncontrolled bleeding in patients treated with apixaban or rivaroxaban. 9 The Andexanet Alfa, a Novel Antidote to the Anticoagulation Effects of Factor Xa Inhibitors (ANNEXA-4) trial was a prospective, multicenter, open-label, single-cohort study, evaluating the effect of AA in 352 patients with acute major bleeding within 18 hours after administration of a FXa inhibitor. 10 While good or excellent hemostatic efficacy at 12 hours was achieved in the majority of patients, an increase in anti-FXa activity four hours after infusion cessation was observed. A relationship between hemostatic efficacy and reduction in anti-factor Xa activity was not demonstrated. Thrombotic complications were observed in 10% of patients within 30 days. Based on the trial results, guidelines suggest the use of AA for apixaban- or rivaroxaban-related life-threatening bleeding. However, factors such as medication cost and safety concerns likely limit widespread institutional adoption.6,11
Due to formulary availability of AA in October 2018, a clinical guideline at our health system was created, incorporating its use in addition to the most frequently used historic reversal agent, 4F-PCC (KCentra®). A multidisciplinary group considered available literature, clinical experience, and operational concerns. While AA is the only agent approved for the treatment of life-threatening bleeding associated with FXa inhibitors, previous clinical experience and literature supports the use of 4F-PCC.7,8 The guideline outlines the appropriate agent with respect to clinical scenario and timing of last FXa inhibitor dose. AA is indicated in intracranial, immediate life- or limb-threatening bleeding (physician discretion), whereas a single dose of 4F-PCC 50 units/kg (U/kg) is recommended for an urgent surgery or procedure. The AA high-dose (800 mg IV bolus, followed by 8 mg/min for up to 120 minutes) or low-dose (400 mg IV bolus, followed by 4 mg/min for up to 120 minutes) regimen is recommended based on the timing of the last dose of apixaban or rivaroxaban. 9 Adjunct blood product use may be prescribed at the discretion of the provider. The purpose of this study was to characterize the use, efficacy, and safety of AA and 4F-PCC for reversal of FXa inhibition.
Methods
This multi-hospital, retrospective, observational cohort study included adult patients aged >18 years administered either AA or 4F-PCC between October 2018 and June 2020 with the indication for reversal of FXa inhibitor-induced coagulopathy. This health-system based study included two hospitals containing level one trauma and comprehensive stroke centers with an urban, university base. Patients administered AA or 4F-PCC for indications other than FXa inhibitor-induced coagulopathy were excluded. Patients were also excluded if pharmacologic reversal was administered but FXa inhibitor-induced coagulopathy was not suspected thereafter. The primary outcome evaluated hemostatic efficacy, assessed by a single reviewer based on the definition from the Scientific Standardization Subcommittee of the International Society of Thrombosis and Haemostasis.(12) Objective criteria, including stabilization of hemoglobin levels at 48 hours after initial treatment; stabilization of hematoma volume or increase by <35% as compared to baseline volume (assessed by computed tomography scan) within 12 hours; absence of further hemostatic or blood product transfusion(s); and absence of neurologic dysfunction or limb loss at discharge were utilized where applicable. Secondary outcomes evaluated adjunct blood product use, incidence of repeat pharmacologic reversal, incidence of thromboembolism, intensive care unit (ICU) and hospital length of stay (LOS) and in-hospital mortality between reversal agents. Protocol driven routine prophylactic screening was not performed during the study period. A drug utilization report was obtained to screen prospective patients. All data points were collected from the electronic medical record, including baseline characteristics, past medical history, indication for anticoagulation reversal, last dose of anticoagulant, imaging, documentation of surgical procedures or interventions, vital signs and laboratory values, and the medication administration record, including receipt of AA and 4F-PCC and timing of anticoagulation re-initiation.
Variables are expressed as values and percentages or median and interquartile range (IQR) where applicable. Categorical variables were compared using a Pearson chi-square test and continuous variables were compared using a Mann-Whitney U test. P values less than .05 were considered statistically significant. All analyses were conducted using Stata (Version 16; StataCorp, College Station, TX). This quality improvement study was considered exempt by the institutional review board.
Results
Baseline Characteristics (N = 85).
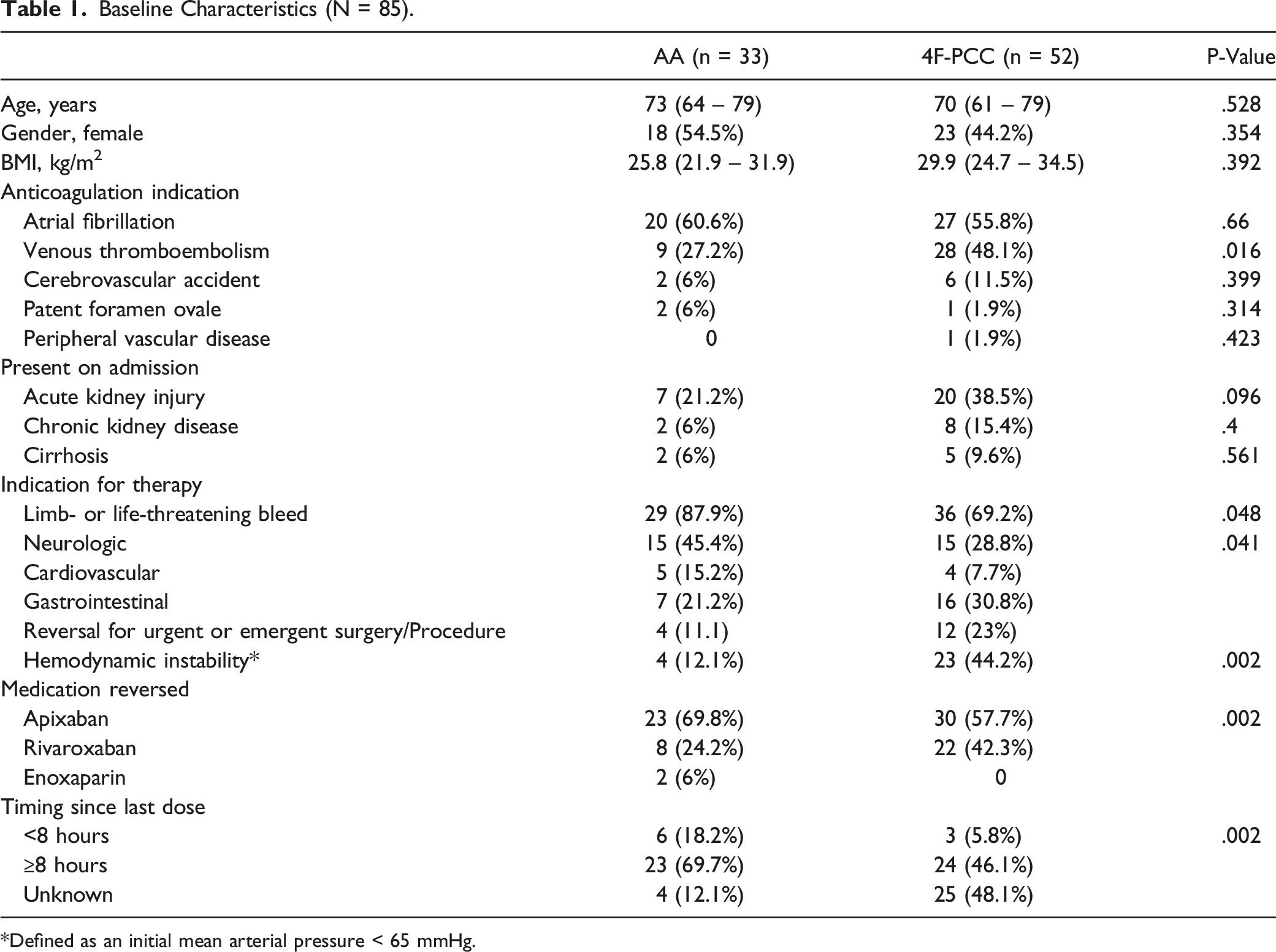
Defined as an initial mean arterial pressure < 65 mmHg.
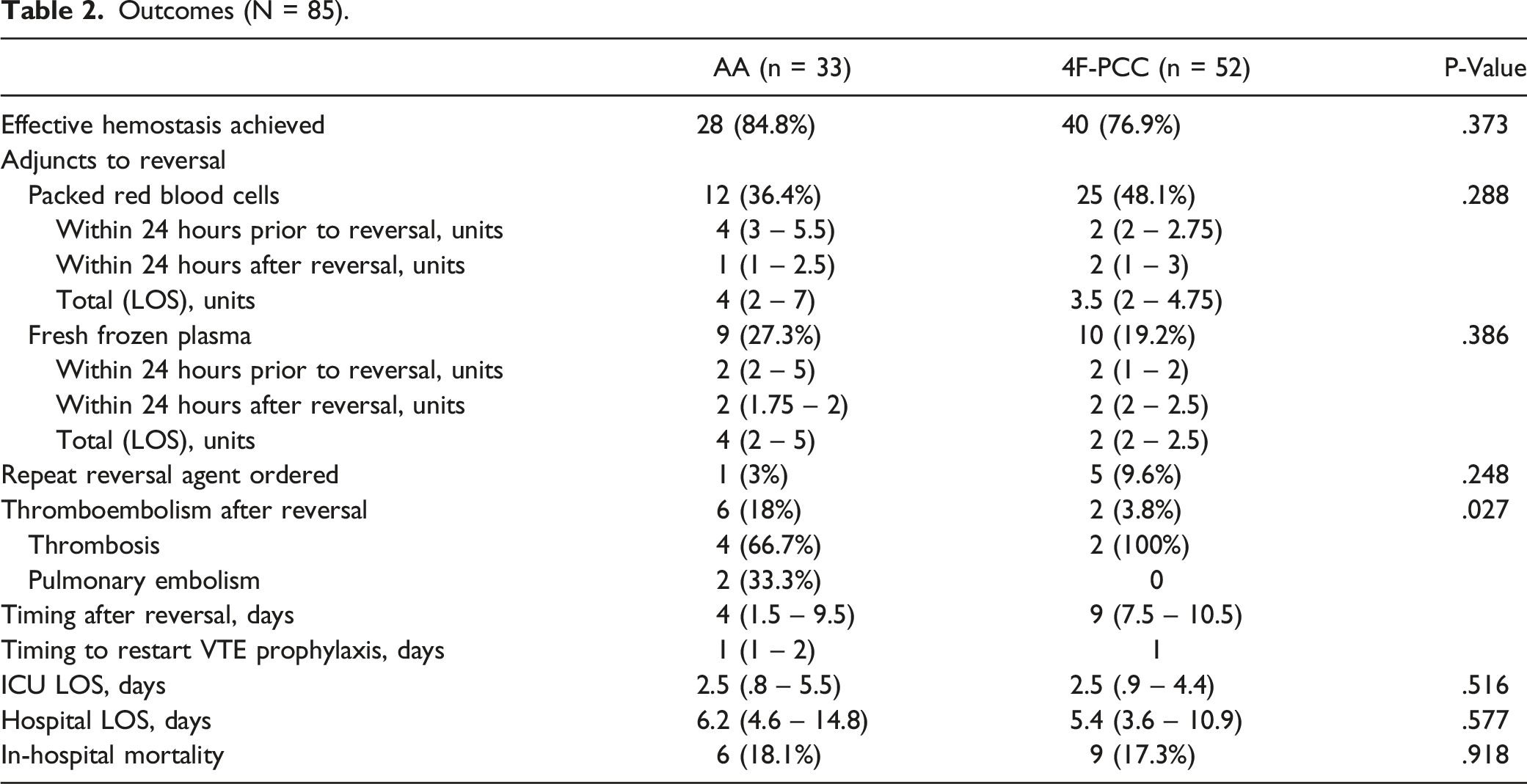
Outcomes (N = 85).
Case Series of Patients Administered a Second Reversal Agent.
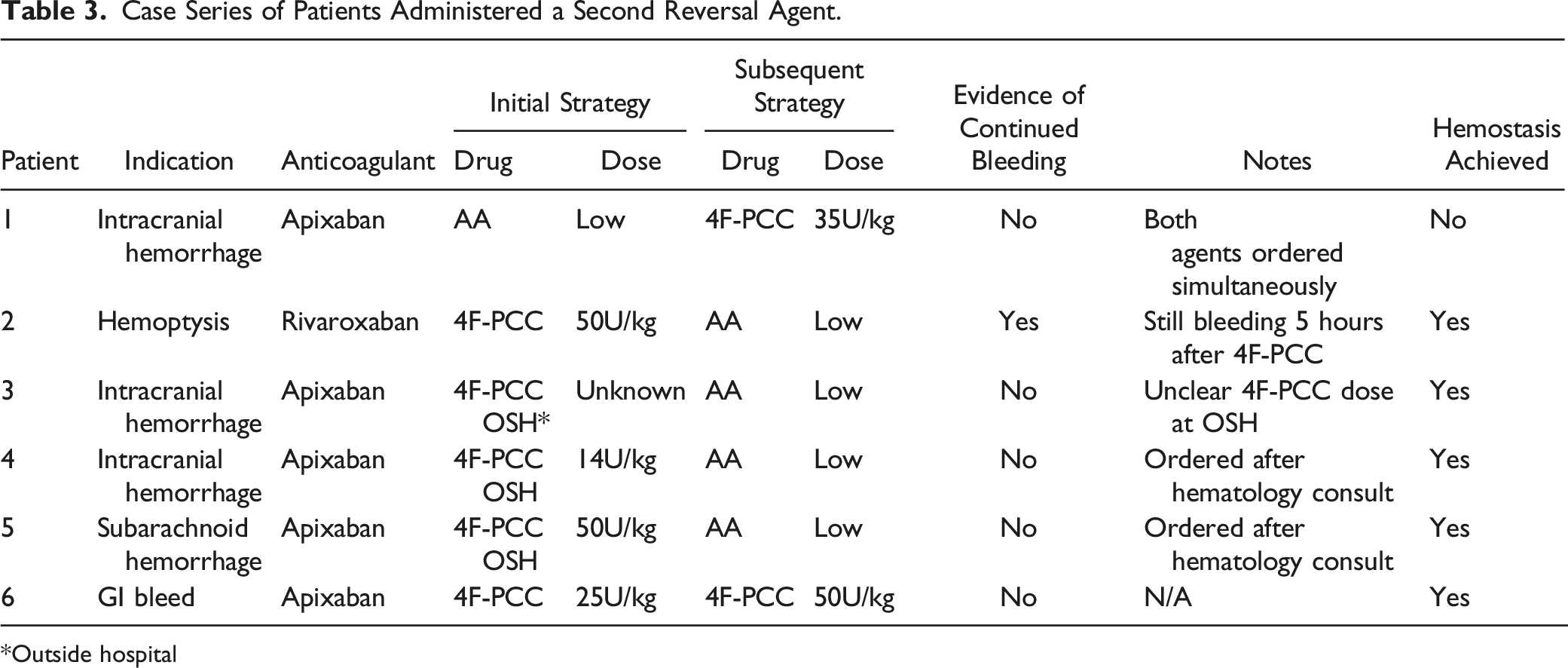
*Outside hospital
Case Series of Thromboembolic Events Occurring After Pharmacologic Reversal a .
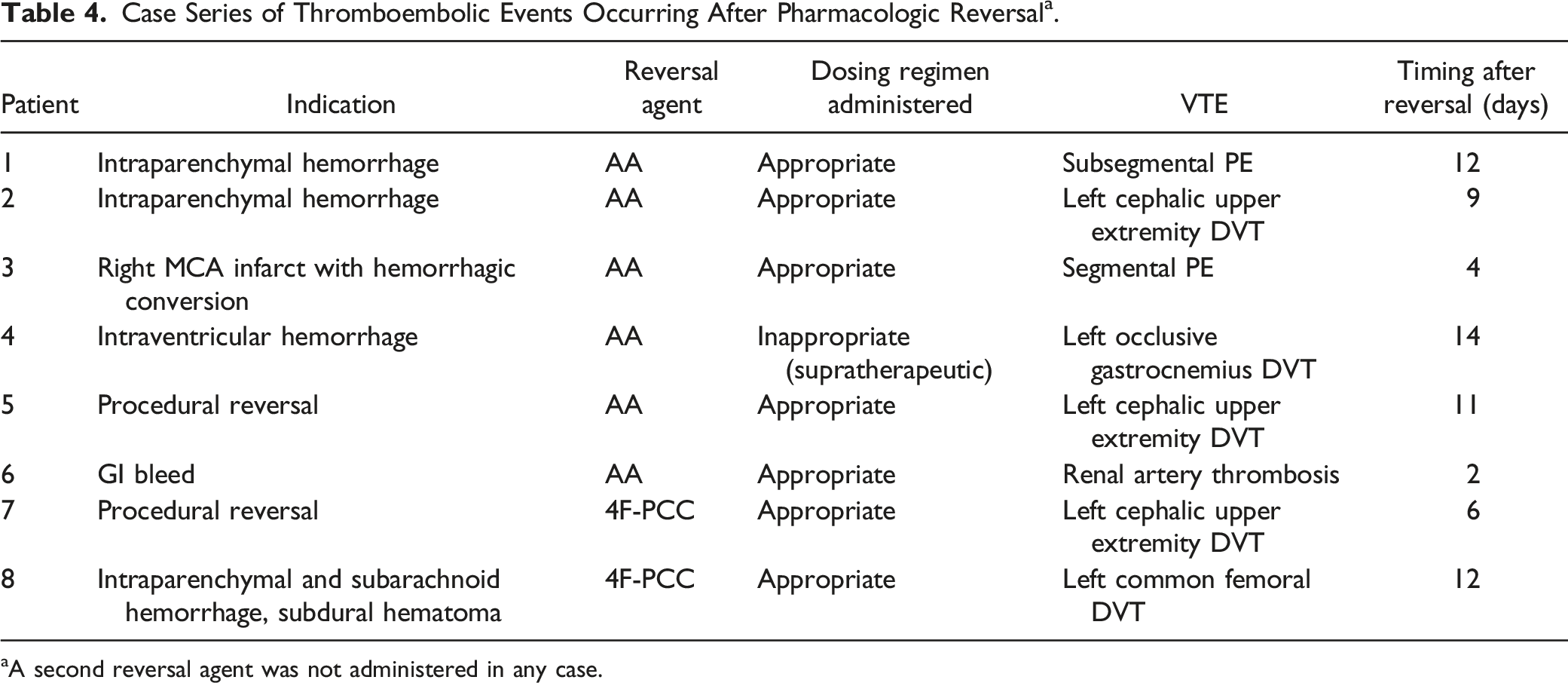
aA second reversal agent was not administered in any case.
Summary of Outcomes with Initial AA and 4F-PCC Administration.

Discussion
This observational study describes the real-world utilization of a therapeutic guideline to aid clinical decision making when emergently reversing FXa induced coagulopathy. While comparisons of reversal strategies have been described in the available literature, many of the published analyses focus on the outcomes for a specific subset of patients.13-15 This analysis uniquely analyzes the outcomes of FXa patients with diverse medical and surgical emergencies.
The combination of the pharmacokinetic and pharmacodynamic properties of each FXa inhibiting anticoagulant, available reversal agents, and patient specific factors (such as underlying indication for the anticoagulation and risk of thrombosis) can significantly complicate decisions regarding anticoagulant reversal. Our analysis demonstrates the effective implementation of a clinical decision-making tool to aid in the management of coagulopathy and bleeding emergencies related to DOAC use. The guideline was implemented in an attempt to mitigate risk of adverse events associated with agents used for reversal, ensure appropriate dosing of reversal agents, and decrease the amount of time spent deliberating optimal reversal strategies. An analysis comparing medication utilization and patient outcomes in the absence of such a tool could prove useful in evaluating the effectiveness of the tool in achieving these goals.
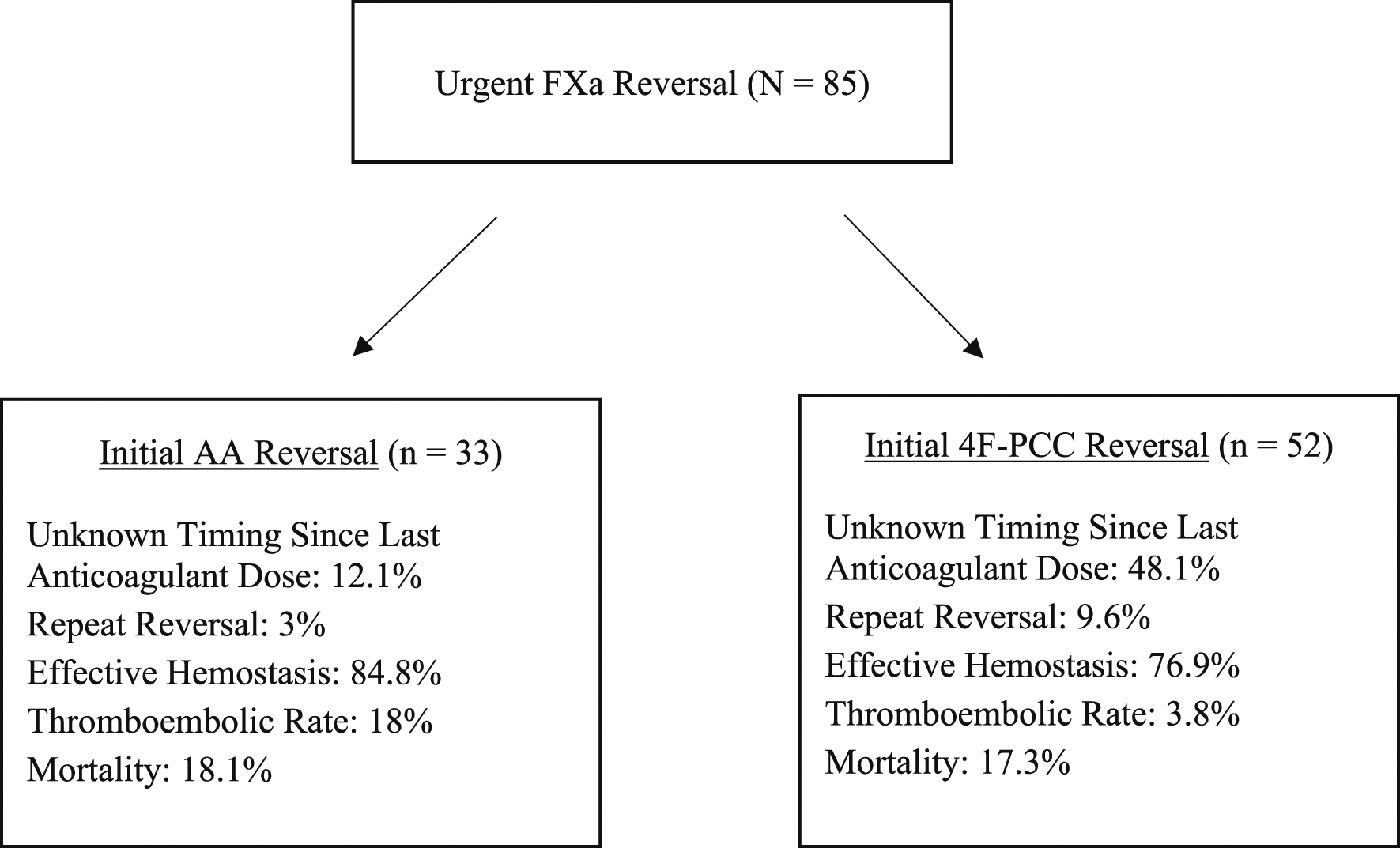
The incidence of patients requiring the administration of repeat reversal agents was low, but more prevalent in the 4F-PCC group (3% vs 9.6%). Interestingly, no VTE events were observed in patients receiving repeat reversal. These scenarios were all unique and reflect the heterogeneity of the patient population being evaluated. However, several instances were the result of a reversal agent being administered at an outside hospital prior to presentation at the included study institutions. Timing between repeat doses varied among these patients; current literature does not address the effect of repeat dosing or the temporal relationship between repeat doses and clinical outcomes. Reviewing outcomes specific to this subset of patients could prove insightful.
A gap in literature exists in analyzing outcomes with respect to the location of bleeding or type of procedure performed. A higher number of patients were observed to have hemodynamic instability at the time of anticoagulation reversal in the 4F-PCC group (12.1% vs 44.2%) despite similar requirements for adjunctive blood products. This could be attributable to recommendations made in the guideline (e.g. preferentially administering 4F-PCC prior to emergent procedure or preference given to andexanet for intracranial hemorrhage). The rates of effective hemostasis achieved in this retrospective analysis correlate with similar analyses that have been previously published.14,16,17 The individual rates of effective hemostasis between groups are difficult to compare as the indications for anticoagulant reversal were different within the cohort. While the number of patients described in this analysis is larger than some published retrospective analyses, our numbers still do not allow for the type of detailed subgroup analysis that would provide clarity on this issue. Further prospective studies in this area, such as analyzing the optimal pharmacologic agent in specific disease states, both medical and surgical, would be helpful in determining the optimal therapy for each indication.
Rates of thrombotic events also correlated to previously published literature evaluating the respective reversal strategies individually.10,16,17 However, it should be noted that the VTE events were not fully adjudicated in terms of ensuring they were not present on admission. Timing of VTE prophylaxis after reversal was similar between the two treatment groups. Each patient with a VTE event after reversal was initiated on prophylactic subcutaneous heparin. All subjects receiving appropriate dosing and the median time to VTE prophylaxis was one day after pharmacologic reversal. It is unknown if an increased VTE risk solely lies with the reversal agent, while modifiable and/or non-modifiable patient-specific characteristics likely represent concomitant factors.
The retrospective nature of our study carries inherent limitations relating to the documentation of information within the patient’s chart. Many ordering providers routinely practice at both study sites, suggesting practice patterns are consistent between institutions. A potential bias may have existed in the selection of pharmacologic anticoagulation and associated cost. Compared to AA, 4F-PCC provides a significant cost advantage. At the time of current writing, the average wholesale price (AWP) of 4F-PCC with dosing based on 100 kg patient (5,000 IU) is $6,582. 18 Conversely, the AWP of the AA low-dose and high-dose regimens is $15,000 and $27,000, respectively. Adjunct blood product use between the two treatment groups may have confounded the association of hemostatic efficacy with specific reversal agents. Further, there are significant differences in patient characteristics between the AA and 4F-PCC groups in terms of bleeding type, which likely interferes with comparison and may explain the lack of differences seen in the outcomes such as LOS and mortality.
Conclusion
This study highlights the real-world challenges associated with the urgent or emergent reversal of FXa inhibiting anticoagulants. The availability of multiple reversal strategies with limited clinical experience or evidence to support one therapy over another makes choosing the optimal agent for each case extremely complex. Similar rates of hemostatic efficacy were observed between the two treatment groups. A greater incidence of thromboembolic events was observed in AA treated patients despite appropriate dosing and timing of VTE prophylaxis after reversal. The data collected herein does not allow for the evaluation of which patients specifically would derive more benefit from one therapy or an alternative. Therefore, it remains critically important to continuously evaluate evidence and real-world experiences to optimize patient outcomes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NDM is a consultant for Alexion Pharmaceuticals. No other conflicts of interest exist.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.