Abstract
Introduction
Crude estimates from 2018 showed that 34.2 million Americans had a diagnosis of either Type 1 or Type 2 diabetes mellitus (DM). 1 This constitutes over 10% of the US population, with approximately 1.5 million new cases diagnosed annually. In 2017, DM was the seventh leading cause of death in the US. The total direct and indirect estimated cost of diagnosed DM in the US was US$327 billion. 1 DM can lead to significant long-term complications including both microvascular and macrovascular complications, as well as substantial healthcare system costs. Estimates from 2013 to 2016 showed that 37% of US adults with DM had chronic kidney disease, and data from 2017 found that 38.6% of all end stage renal disease diagnoses were caused by DM. Further, DM is the leading cause of new cases of blindness, and data from 2018 showed that 11.7% of patients with DM reported vision disability. This data is magnified in the Veterans Affairs (VA) population, where diabetes affects nearly 25% of VA patients. 2
To limit the risk for developing long-term complications of DM and to reduce overall healthcare costs, the American Diabetes Association (ADA) guidelines recommend specific A1c goals based on a patient’s age and overall health status. Most commonly, the ADA recommends targeting an A1c of less than 7% for young, healthy patients and less stringent targets of less than 7.5% or less than 8% in patients who are elderly, who have significant comorbidities, or with hypoglycemic unawareness. 3 Lifestyle modifications are recommended as the first line intervention in patients with pre-diabetes and are recommended in conjunction with medication initiation in patients with diagnosed diabetes to help achieve A1c goals. 3 Crude data from 2013 to 2016 reported that only 24.2% of patients were meeting exercise goals, 77.1% of patients were attempting weight management, and 58.4% were on statin therapy. 1 Overall, there is opportunity to improve DM control and limit risk for developing DM-related complications through goal setting.
Structured goal setting can assist patients with implementing lifestyle changes, which can be related to diet, exercise, glucose monitoring, medication adherence, or other lifestyle goals. Setting goals supports both behavior change and effective diabetes self-care. SMART goals are specific, measurable, achievable, relevant, and time-bound in nature. SMART goals assist patients with setting smaller, more achievable goals in order to work towards achieving a larger, long-term goal. These goals should be individualized based on the patient’s needs and can include both pharmacologic and non-pharmacologic goals. While there is evidence demonstrating successful implementation of SMART goals in both inpatient and outpatient settings, these studies have only looked at the feasibility of goal-setting, but have not reviewed the clinical efficacy and outcomes of these efforts.4,5
Previous studies have established that patients are more successful in achieving lifestyle goals when a provider guides them through the goal-setting process.4-6 However, available literature has not specifically explored the clinical efficacy of SMART goal setting to determine if this process assists patients in achieving health-related goals. Additionally, in the changing clinical climate, where telehealth modalities to deliver patient care are expanding to provide more flexible care, we found that additional studies were needed reviewing specific interventions in this care setting. Our study aimed to contribute to the available body of evidence by not only looking at the successful implementation of SMART goal setting during telehealth visits, but also exploring clinical outcomes as they relate to patients’ diabetes control. The purpose of this study was to assess the clinical impact of SMART goal setting on diabetes management in a pharmacist-led telehealth clinic.
Methods
In June 2020, three clinical pharmacy specialists working at VA Community-Based Outpatient Clinics within a single VA healthcare system began educating patients on the concept of SMART goal setting and assisted patients in goal setting during their diabetes telehealth appointments. These pharmacists worked with patients to identify an area of improvement related to their diabetes care and assisted the patients in setting individualized SMART goals related to adherence, diet, exercise, or self-monitoring blood glucose. Patients who set a SMART goal during these visits were mailed an educational handout explaining more about what a SMART goal is, how it is different from other goals, examples of SMART goals, and outlining the specific SMART goal they chose to work on. At follow-up telehealth appointments, the pharmacists documented each patient’s progress toward achieving their SMART goal, which typically occurred every 4 weeks. Patient progress toward SMART goal achievement was assessed by the pharmacist as no progress, progress, or achieved.
This was an IRB-approved, retrospective, case-control study within a single VA healthcare system. Patients with Type 2 DM managed via telehealth by a Clinical Pharmacy Specialist at a VA Community-Based Outpatient Clinic between June 1 and November 30, 2020 were eligible for inclusion. The electronic medical record was used to identify eligible patients that set a SMART goal for DM management during the study timeframe. These patients were matched to a similar cohort of patients that did not set a SMART goal based on age (+/− 5 years), A1c (+/− .5%), and insulin use (yes/no). The SMART goal group and the control group were managed by separate Clinical Pharmacy Specialists to reduce possible bias, and the same pharmacist conducted each visit with their respective patient. Patients were excluded from the study if they did not have a repeat A1c within the data collection period.
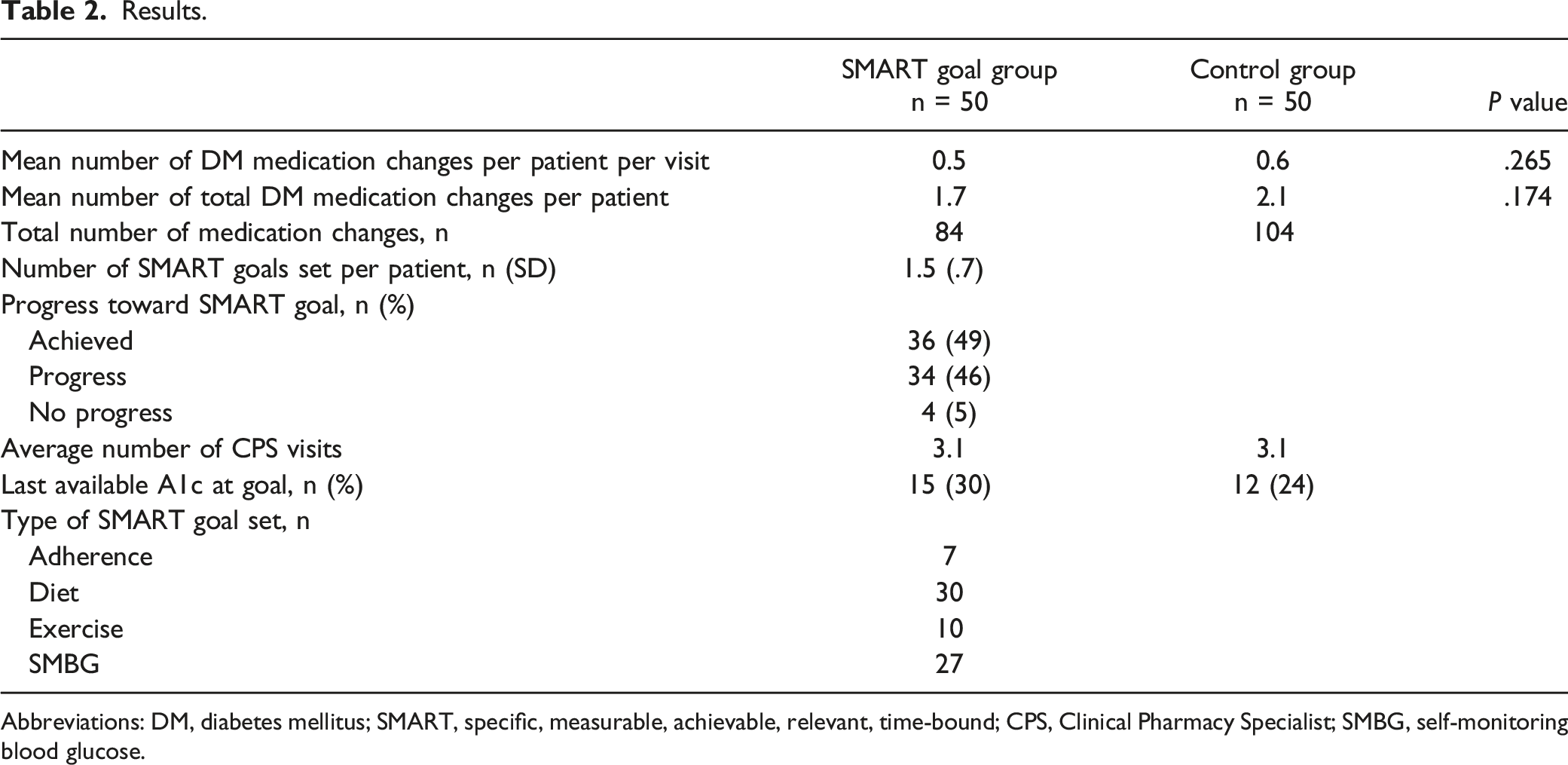
The primary endpoint of this study was the change in A1c from baseline to 3 months in the SMART goal group compared with the control group. Secondary endpoints included change in A1c from baseline to last collected A1c during data collection period, number of medication changes made per patient during each visit, number of medication changes made per patient during the data collection period, number of SMART goals set per patient during the data collection period, and progress toward SMART goal implementation/achievement (ie no progress, progress made, or achieved). A medication change was defined as a dose adjustment, medication initiation, or medication discontinuation.
Descriptive statistics were used to assess the demographics and characterize the primary and secondary endpoints. Primary and select secondary endpoints were analyzed using a Student’s t-test.
Results
Baseline Demographics.

Abbreviations: BMI, body mass index; eGFR, estimated glomerular filtration rate; HFrEF, heart failure with reduced ejection fraction; Alb:SCr, albumin to serum creatinine ratio; CPS, Clinical Pharmacy Specialist; DM, diabetes mellitus; GLP1, glucagon-like peptide-1 receptor.
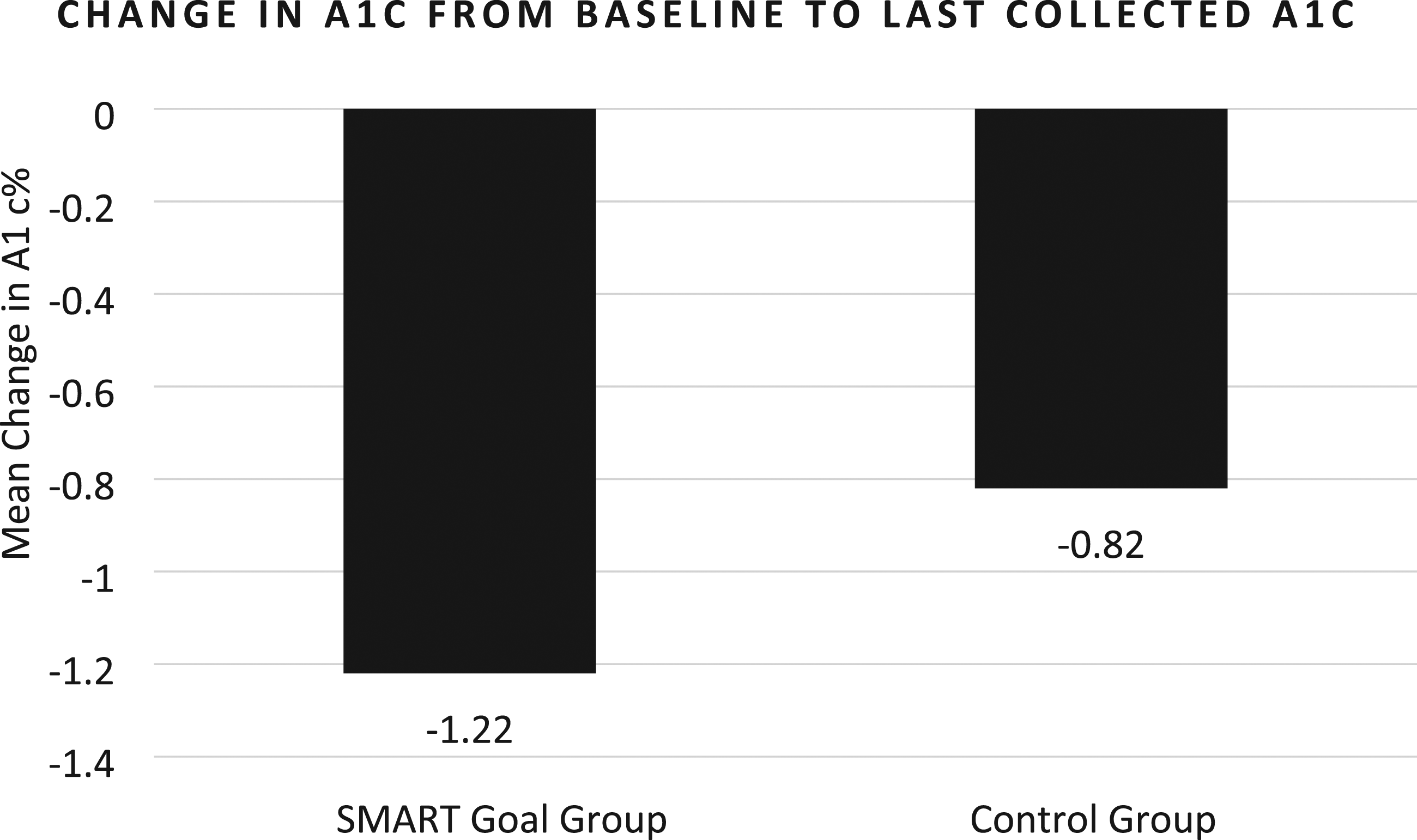
A1c was reduced by 1.2% from baseline to 3 months in the SMART goal group vs .85% in the control group (P = .287, Figure 1). A similar A1c improvement was identified from baseline to last collected A1c during the study period (A1c decreased by 1.22% in the SMART goal group vs .85% in the control group, P = .256, Figure 2). Compared to baseline, 12 additional patients met their A1c goal in the SMART goal group compared to 6 additional patients in the control group (Table 2). Goal A1c was achieved in 30% of patients in the SMART goal group compared with 24% of patients in the control group (Table 2). Overall, the mean number of DM medication changes per patient per visit was similar between the two groups (P = .265; Table 2). Of note, however, the SMART goal group had numerically less DM medication changes per patient over the entire study period compared with the control group (1.7 medication changes vs 2.1 medication changes, P = .174). Patients in the SMART goal group set an average of 1.5 SMART goals throughout the study period. Patients most commonly set goals related to diet and self-monitoring blood glucose. Of all the SMART goals set, 49% were documented as “achieved”, 46% were documented as “progress made,” and 5% were documented as “no progress made.” Primary endpoint: Change in A1c from baseline to 3 months between groups; P = .287. Mean number of months A1c was collected after first clinical pharmacy specialist visit was 3 months in the SMART goal group and 2.6 months in the control group. Secondary endpoint: Change in A1c from baseline to last collected A1c during the data collection period; P = .256. Mean number of months A1c was collected after first clinical pharmacy specialist visit was 3.6 months in the SMART goal group and 3.1 months in the control group. Results. Abbreviations: DM, diabetes mellitus; SMART, specific, measurable, achievable, relevant, time-bound; CPS, Clinical Pharmacy Specialist; SMBG, self-monitoring blood glucose.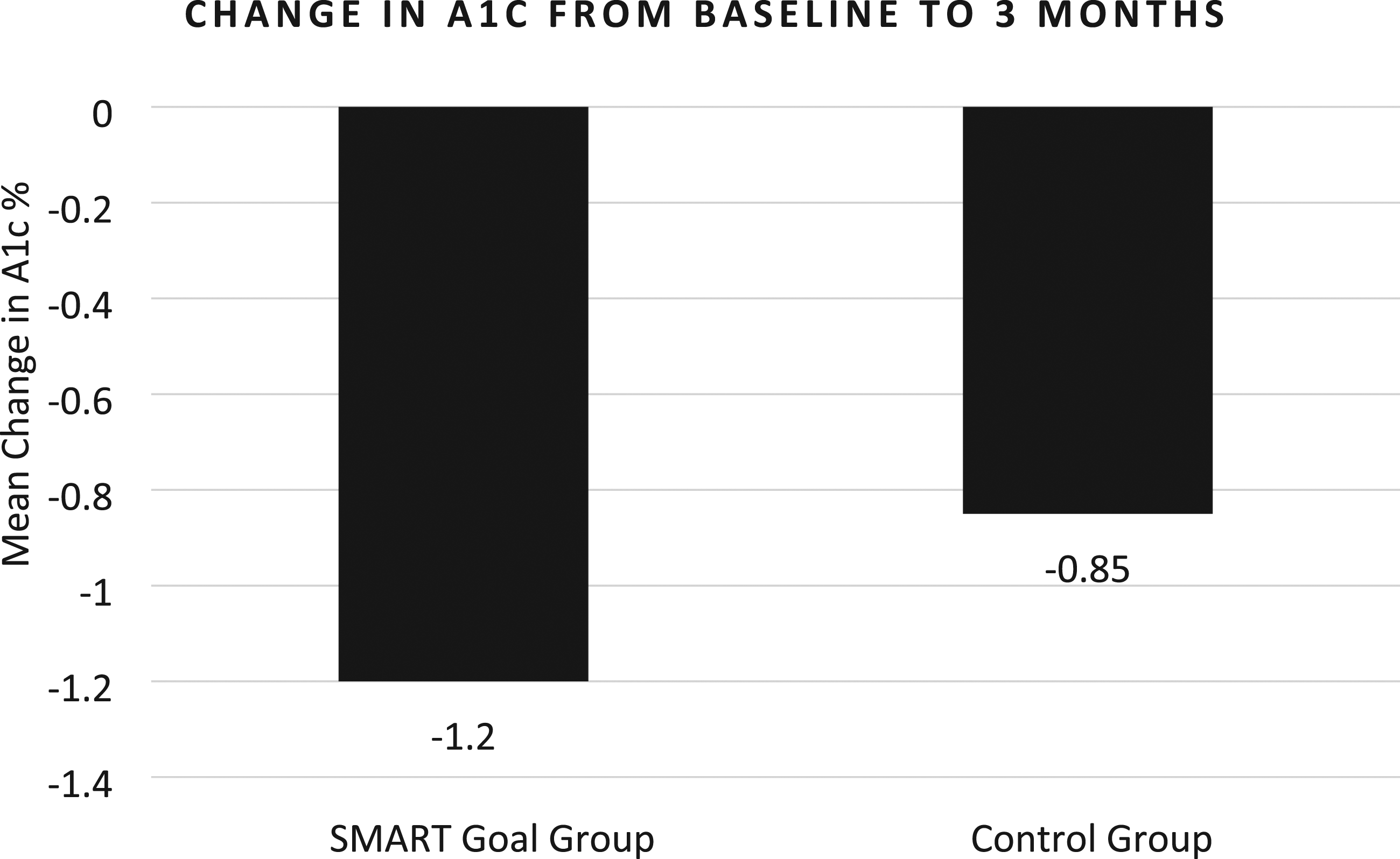
Discussion
We found that SMART goal setting with patients with diabetes during routine telehealth visits with a clinical pharmacist resulted in numerically greater A1c reduction compared with patients who did not set SMART goals, although this result did not reach statistical significance. While this was an exploratory outcome and not a pre-specific endpoint, we also found that more patients achieved their predetermined A1c goal at three months after setting SMART goals than those who did not set SMART goals. Although these results did not reach statistical significance, we believe this study provides important insight into the benefits of SMART goal setting on assisting patients with improving their DM control. While previous studies have predominantly explored the feasibility of successful implementation of goal setting during patient visits, our study fills a current gap in available literature by also assessing the clinical benefit of SMART goal setting by clinical pharmacy specialists using telehealth visits.
In order to better evaluate the implications of our study results, we reviewed previous studies that assessed the effectiveness of telehealth on diabetes self-management and the effectiveness of interventions using a goal-setting approach. A 2013 trial aimed to determine the impact of telehealth programs on self-care behaviors of patients (including activity, health eating, adherence, and self-monitoring blood glucose), which decreased A1c by .16% after 6 months (P = .02). 7 A 2021 meta-analysis evaluating 29 studies looked at the effect of telehealth interventions on A1c levels compared to usual care and found that telehealth interventions reduced A1c by .18% (P = .04). 8 However, a systematic review from 2017 reviewed 7 trials related to effectiveness of telehealth on diabetes self-management and determined that the majority of these studies did not see a statistically significant difference in A1c. 9 Most recently, a 2022 meta-analysis assessing the effectiveness of interventions using a goal-setting approach on A1c found goal setting more effective than usual care at 6 months (mean difference −.32%, 95% CI −.56, −.08) and 12 months (mean difference −.17%, 95% CI −.33, −.01). 10 Based on our A1c results, our study suggests that SMART goal setting via telehealth with a pharmacist may contribute to additional lowering of A1c compared to other types of goal-setting with different professions. Our study provides new information and a foundation that will guide future research in this area with larger populations.
It is noteworthy to point out the overall success that the pharmacists had in helping patients achieve their SMART goals through encouragement and check-ins at each follow-up appointment, with 49% of patients achieving their SMART goal by the end of the study. As a comparison, the Agewell trial (2015) looked at SMART goal setting in the outpatient setting as an intervention to promote healthy aging (N = 75). Visits were conducted by non-pharmacy providers with only 28% of the patients achieving their SMART goals. 4 While the methods and objectives of these two trials differed, the age of our study population was similar (64 years in the current study vs 68 years in the Agewell trial), and it is notable that there may be additional benefit when pharmacists are involved in initiating and following up on SMART goals with patients, compared with other professions.
To ensure the success of SMART goal setting, it is important to anticipate potential barriers to implementation. Certain patients may not be ideal candidates for SMART goal setting, which may impact clinical outcomes. For example, patients with cognitive impairment or patients who are not interested or motivated to make lifestyle changes to improve diabetes control may not fully engage in SMART goal setting. This, in turn, can inhibit meaningful goal setting and implementation. Additionally, we found that patients who are not fully engaged in SMART goal setting due to cognitive or motivational barriers have a higher likelihood of being lost to follow-up, struggling with SMART goal development, and difficulty achieving set goals. Overall, however, we found that SMART goal setting can serve as a catalyst for clinical improvement if implemented in motivated and engaged patients. Additionally, although motivational interviewing is commonly utilized in various clinical settings, providers may need to allocate a larger portion of the initial visit to SMART goal setting to introduce the concept. However, subsequent visits relating to SMART goals should not be expected to add substantial time to the appointment and, in our experience, did not detract from other clinical evaluation or appointment tasks. Despite the required additional counseling, we found that setting SMART goals with agreeable patients during telehealth visits can be an effective way to motivate patients to implement meaningful lifestyle changes to improve glycemic control.
There is a need for larger studies to confirm the clinical impact of SMART goal setting on diabetes management especially within the telehealth setting. The findings from this study suggest that providers should consider incorporating SMART goal setting into clinical practice with agreeable patients who are being followed for diabetes management. Future studies should be aimed at evaluating which patients would be best to target when implementing SMART goals and whether certain types of SMART goals (ie diet focused vs activity) are more effective at improving patients’ A1cs. Additionally, given the growing incorporation of telehealth modalities into clinical practice, additional studies may be needed to explore the most effective methods for implementation and communication of goals and progress with patients.
Although this study showed numerical A1c improvement with SMART goal setting, the relatively small sample size may have impacted its ability to find a statistically significant difference between the study and control groups and to fully evaluate the impact of SMART goal setting on diabetes management. Additionally, this study aimed to implement SMART goal setting during the onset of the COVID-19 pandemic, during a time of significant changes to clinical practice and care delivery models, which may have impacted SMART goal implementation and outcomes. Patients seen by clinical pharmacists during this period were newly transitioning from predominantly face to face visits to entirely telehealth modalities, which may have impacted follow-up and SMART goal implementation. Additionally, due to social distancing requirements, it was notably more difficult to obtain updated labs in a timely fashion, thus impacting the number of patients who met inclusion criteria. Although there was no difference in the number of visits between groups, differing practice styles between clinical pharmacists may have still impacted the results. There may have also been a potential healthy user bias, in which those who set SMART goals may have had greater interest in improving their health. Notably, more patients in the control group had a baseline A1c already at goal, which could have impacted the results. Finally, there is a possibility that clinical pharmacists within the control group may have still implemented SMART goals in some of their patients which may have confounded results.
Conclusion
Overall, patients that set SMART goals had a clinically meaningful reduction in A1c, even though statistical significance was not reached. Setting SMART goals for DM management in agreeable patients may lead to fewer medication changes and improved diabetes control. Larger studies are needed to confirm the clinical impact of SMART goal setting on diabetes management in the telehealth setting.
Footnotes
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Hannah Cook. The first draft of the manuscript was written by Hannah Cook and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
