Abstract
Background
Cefepime is a fourth-generation cephalosporin frequently used in patients with concern for life-threatening or drug-resistant gram-negative infections due to its broad-spectrum antimicrobial activity and generally benign adverse effect profile.1,2 However, beta-lactams, including cephalosporins, are known to cause neurotoxicity, including encephalopathy, myoclonus, and seizures. 3 Moreover, there is increasing literature, mostly among adult patients in the intensive care unit (ICU) setting, to support the existence of cefepime-induced neurotoxicity (CIN). Patients with higher than expected concentrations of cefepime have been reported to experience neurologic events including altered level of consciousness, confusion, encephalopathy, myoclonus, seizures, coma, and death.4-6 The theorized mechanism of neurotoxicity is secondary to antagonism of central nervous system (CNS) γ-aminobutyric acid-A (GABAA) receptors 7 by cefepime after crossing the blood-brain barrier. Cefepime undergoes >85% renal excretion and demonstrates 10% or greater CNS penetration.8,9 It follows that patient-specific risks for CIN identified in systematic reviews have included renal dysfunction, pre-existing brain injury, and elevated plasma cefepime trough concentrations, with 1 study 6 noting universal neurotoxicity with trough concentrations >38.1 mg/L. For context, resistant pathogens such as Pseudomonas frequently have minimum inhibitory concentrations (MICs) as high as 8 mg/L. Since the effectiveness of beta-lactam antibiotics is based on time above MIC and achieving 100% time above MIC is a common pharmacokinetic (PK)/pharmacodynamic (PD) target, 10 a trough of 8 mg/L would have been sufficient in this case. Of note, there is no consensus on the appropriate PK/PD target in critically ill children, but some are in favor of concentrations above 4× MIC for 100% of the dosing interval.11,12 In this latter case, a trough of 32 mg/L would be required, which is close to the neurotoxicity threshold above. There is a relative paucity of pediatric data on CIN, with only 5 cases13-17 reported in the pediatric literature to date, each in patients with renal dysfunction all of whom were 7 years of age or older. Herein we describe a case of CIN developing in a 2-year-old patient with advanced chronic kidney disease (CKD) receiving treatment for S marcescens bacteremia despite pre-emptive dose adjustment for kidney dysfunction.
Case
The patient is a 2-year-old female born prematurely at 28 weeks gestation who had perinatal hypoxic-ischemic injury resulting in encephalopathy and global developmental delay. She also had a history of Stage V CKD with a baseline creatinine of 2.6 mg/dL and an estimated glomerular filtration rate (GFR) of 12 mL/min/1.73 m2 per bedside Schwartz estimation. She had required renal replacement therapy during prior acute illnesses, and thus had a hemodialysis catheter in place, but was not chronically dialysis-dependent. Her home medications prior to admission included calcitriol 1.5 mcg daily, cholecalciferol 400 IU daily, darbepoeitin alfa 12.5 mcg once weekly, diphenhydramine 12.5 mg (approximately 1 mg/kg) nightly, famotidine 2.4 mg daily, ferrous sulfate 70 mg BID, levothyroxine, melatonin 3 mg nightly, sodium chloride 12.5 mEq daily, and sodium phosphate 3 mmol daily.
The patient presented to the emergency department febrile (40.7°C) and tachycardic (250 bpm) though with normal blood pressure for age (91/55 mm Hg), concerning for compensated septic shock. She subsequently experienced her first lifetime focal seizure. Initial laboratory studies revealed creatinine 3.1 mg/dL and albumin 3.3 mg/dL. Seizures were treated with levetiracetam 40 mg/kg loading dose followed by renally-dosed 20 mg/kg/day (10 mg/kg BID) maintenance. She was admitted to the pediatric ICU with septic shock and started on cefepime 50 mg/kg IV q 24 h (recommended dose for a CrCl < 30 mL/min/1.73 m2), 10 with a single dose of vancomycin (20 mg/kg IV) for sepsis with ill appearance in the setting of a central line. On hospital day 2, blood cultures speciated as gram-negative rods, and the patient’s central line was removed for source control. She continued to receive cefepime every 24 h, and no further doses of vancomycin were given.
Admission blood cultures speciated to S marcescens sensitive to cefepime with a minimum inhibitory concentration (MIC) of 0.5 mg/L but resistant to cefazolin and ampicillin suggestive of AmpC production. Cefepime was continued at the same reduced dosing regimen and all subsequent blood cultures had no growth. Despite fluid resuscitation with restoration of hemodynamics and discontinuation of vancomycin, the patient’s serum creatinine worsened, to a peak of 4.2 mg/L on hospital day 3 with a BUN of 86 mg/dL. After initial fluid resuscitation, she was at her neurologic baseline with no further seizures after initial presentation through hospital day 4. On hospital day 5, the patient had a recurrence of focal seizure despite normalization of temperature (maximum 37.5°C that day) which aborted with lorazepam 0.1 mg/kg IV. Electroencephalography (EEG) monitoring was initiated which showed global background slowing without focal epileptiform abnormalities. On hospital day 6, abnormal movements including stretching and tremor were noted without electrographic correlate, so levetiracetam was discontinued. EEG monitoring concluded with normalization in the background rhythm. On hospital days 7-11, the patient experienced agitation, diffuse fine tremor, and behavioral arrest spells lasting seconds to minutes which improved with calming and 2 mcg/kg of enteral clonidine every 8 h as needed.
Due to ongoing neurologic changes as described above, a cefepime trough was obtained 10 min prior to the scheduled dose of cefepime on hospital day 9 that resulted as elevated at 80 mg/L. Thus, clinicians hypothesized that this patient’s neurologic changes were attributable to cefepime-induced neurotoxicity. Serum creatinine on the day the trough was drawn was 2.9 mg/dL, and serum albumin was 2.4 mg/dL. Cefepime was discontinued on hospital day 10 and she was transitioned to ciprofloxacin. By hospital day 12, the patient’s tremor and behavioral arrest spells resolved, >36 h after discontinuation of cefepime. With ongoing hydration, the patient’s serum creatinine gradually improved and was back to baseline by hospital day 14. She completed a total of 14 days of antibiotics with ciprofloxacin and was discharged home with stable BUN/creatinine without the need for dialysis and at her neurologic baseline.
Discussion
Cefepime-induced neurotoxicity (CIN) has been characterized extensively in the adult literature. A recent systematic review of CIN in adults 4 identified renal dysfunction and baseline brain injury, both of which were present in this patient, as independent risk factors for CIN. Five pediatric reports of CIN exist: 2 in patients on chronic hemodialysis,13,16 1 in a patient on chronic peritoneal dialysis, 15 and 2 in patients with acute kidney injury.14,17 Only one of these reports 14 used plasma cefepime concentrations to confirm the clinical diagnosis of CIN, and in this report, the plasma cefepime concentration was 62.6 mg/L 5 days after the last dose of cefepime. Clinical symptoms of CIN in children are ill-defined, and cefepime dosing regimens in children with kidney dysfunction are based on expert opinion with limited pharmacokinetic data to inform dose adjustments. 10
This lack of data implies an unmet need to broaden the use of therapeutic drug monitoring (TDM) to inform the diagnosis of CIN, particularly in medically complex patients such as this one. Indeed, this patient with Stage V CKD and hypoxic-ischemic encephalopathy manifests multiple risk factors for alterations in neurologic status, including infection, metabolic encephalopathy (uremia), drug toxicity including both exposure to cefepime and levetiracetam, the latter of which has been associated with agitation, and an acute worsening of her underlying CNS disorder. However, the temporal association of this patient’s symptoms of agitation, tremor, and decreased alertness with cefepime exposure, including symptom onset after initiation of cefepime and improvement after cessation (see Figure 1), along with an elevated cefepime trough, supports the argument that this patient experienced CIN. In addition, since cefepime is approximately 20% protein bound, hypoalbuminemia can contribute to increased free cefepime concentrations and thus to increased cefepime influx into the CNS
4
; the patient’s albumin was 2.4 mg/dL at the time the trough concentration was obtained. Clinical timeline of events. Green stars denote seizure activity. Blue triangles denote episodes of tremors, agitation, and behavioral arrest spells.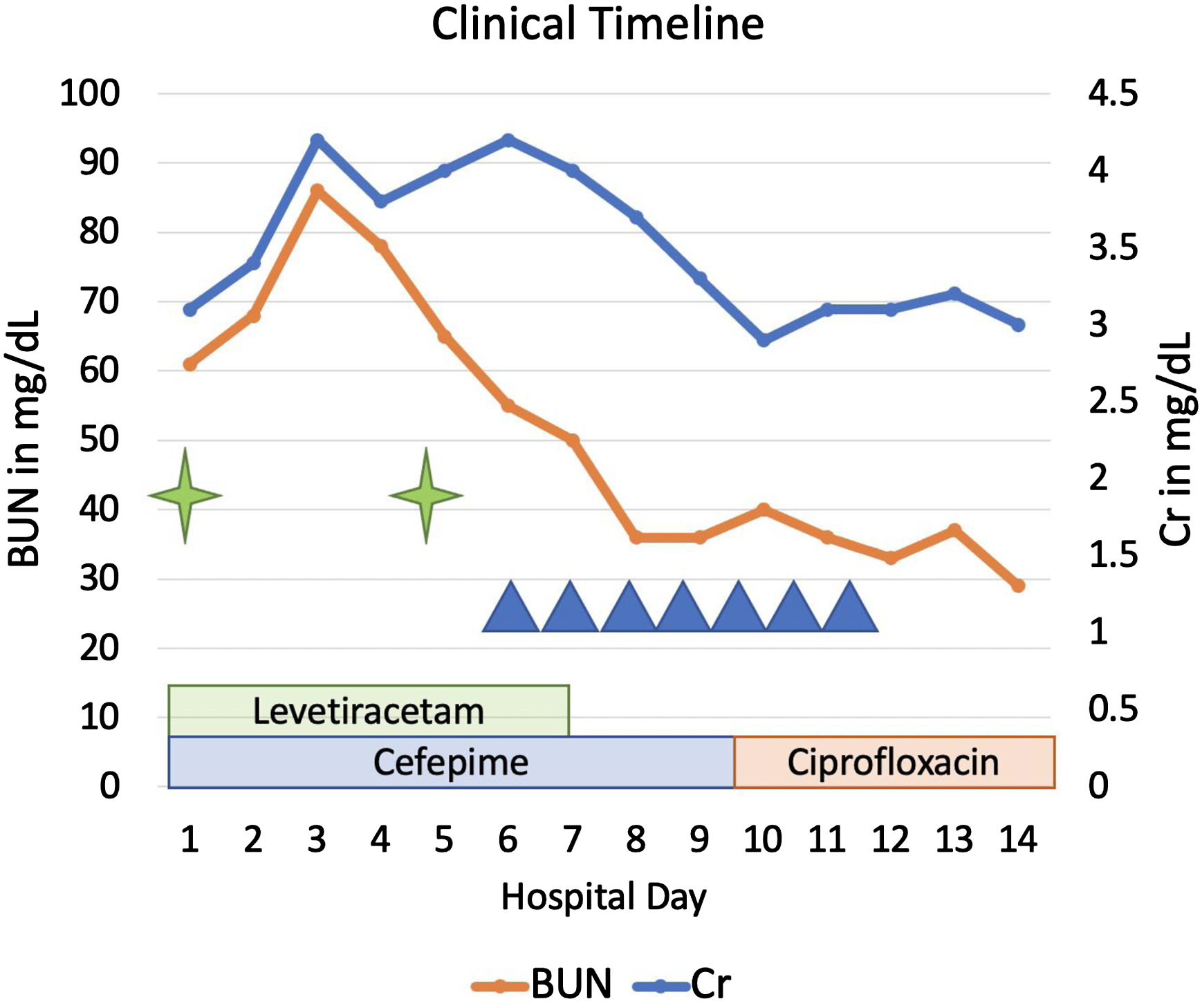
Moreover, prior reports of cefepime-induced neurotoxicity have shown onset of symptoms at a median of 4 (IQR 2-6) days of therapy. The delayed emergence of tremor, agitation, and acute encephalopathy during admission and following an interval of stabilization raises concern for adverse effects from therapeutic interventions. Using the Naranjo Scale for Adverse Drug Reaction Probability, 18 we calculate this patient’s score as a 4: 1 point for previous conclusive reports of this reaction, 2 points for the adverse event (second seizure after being at her neurologic baseline for 4 days and episodes of tremors, agitation, and behavior arrest spells) occurring after the suspected drug was administered, 1 point for improvement in condition after drug discontinuation, −1 point for potential alternative causes of the condition, as described above, and 1 point for the drug being detected in blood in concentrations known to be toxic. A score of 4 is within the range defined as “possibly” due to the drug in question, but the marked elevation of cefepime trough at 80 mg/L with subsequent improvement in neurologic symptoms following discontinuation is strongly suspicious for cefepime as a contributor to her symptoms. Her trough concentration is much higher than 36 mg/L, which was recently reported 5 to provide excellent sensitivity and specificity for CIN in adults, at 95% and 91%, respectively, with an odds ratio of 1.06 times increased odds of CIN for every 1 mg/L increase in cefepime trough concentration. A separate study 6 of adults saw no neurotoxicity with trough concentrations <7.7 mg/L and universal neurotoxicity with trough concentrations >38.1 mg/L. Indeed, CIN is a common conclusion in adult patients with undifferentiated acute encephalopathy, though it is less well-characterized in children. Therefore while all of the above studies were performed in adults and thus of limited generalizability to this patient, there is unfortunately a paucity of data on pediatric CIN,13-17 with even less data about plasma cefepime concentrations in these patients. Future prospective studies to describe the cefepime exposure-neurotoxicity relationship in children are warranted.
This case thus illustrates the potential role of TDM in cefepime in pediatrics. While it is possible to have made a presumptive diagnosis of cefepime-induced neurotoxicity in the absence of an elevated cefepime trough, confidence in this diagnosis was higher given the benefit of having an existing validated assay for cefepime concentration measurements at this institution. While routine TDM for short courses of cefepime may not be indicated, it may prove beneficial for patients such as this 1 who are at high risk for toxicity due to renal dysfunction or baseline CNS disease. In particular, even the most conservative estimates for effective anti-microbial activity in beta lactams suggest targeting concentrations to be above 4× MIC for the entire dosing interval, 19 which in this patient would be troughs above 2 mg/L (for a Serratia MIC of .5 mg/L). It is thus conceivable that early TDM, along with model-informed precision dosing, could have avoided cefepime toxicity while allowing for adequate antimicrobial activity by aiming for troughs close to 2 mg/L. Unfortunately, at this time, to our knowledge, only a small percentage of hospitals in the United States measure cefepime concentrations for research purposes and even fewer can provide TDM results clinically for decision making. Indeed, a recent worldwide survey 20 of the use of TDM in ICU populations found that only 1% of respondents used TDM for piperacillin/tazobactam and 2% used it for meropenem, with no mention of cefepime. Therefore, until low-cost assays for cefepime concentrations are broadly available, clinicians must make the diagnosis of cefepime-induced neurotoxicity on clinical grounds alone, which may lead to misdiagnosis with inaccurate attribution of neurologic changes to cefepime neurotoxicity. More research is therefore needed to help establish cefepime trough concentrations that reliably correlate with neurotoxicity in pediatric patients to justify broader uptake and use of cefepime TDM.
Cefepime-induced neurotoxicity should be included in the differential diagnosis of altered mental status, disorientation, seizures, or tremor, in pediatric patients receiving cefepime, particularly those with renal dysfunction or baseline CNS abnormalities. There is a need for further research to identify plasma concentrations associated with pediatric CIN. Additionally, therapeutic drug monitoring may help inform appropriate antimicrobial dosing and avoidance of toxicity.
Footnotes
Author Contributions
H.R.H. conceptualized the case, drafted the initial manuscript, and reviewed and revised the manuscript.
K.P., S.T.G., D.L. J.M.T., and S.B. conceptualized the case and reviewed and critically revised the manuscript.
All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Consent to Publish
The patient’s parent provided written informed consent for submission of this case report to the journal.
