Abstract
Eravacycline is approved by the U.S. Food and Drug Administration (FDA) for the treatment of complicated intra-abdominal infections. It is a novel, fully synthetic fluorocycline antibiotic belonging to the tetracycline class with a broad-spectrum of activity and an appealing side effect profile. This report describes a 74-year-old female who presented to the hospital with non-ST-elevation myocardial infarction (NSTEMI) requiring coronary artery bypass graft surgery. After surgery, she developed a sternal wound infection that grew multidrug resistant organisms, leading to a much longer than anticipated hospital stay. Eravacycline was eventually added to the antimicrobial regimen for the persistent infection. Shortly after therapy with eravacycline began, the patient started experiencing muscle pain and the creatine phosphokinase (CPK) level was noted to be elevated. Other causes, such as concomitant administration of an HMG-CoA reductase inhibitor, were explored in this case but not thought to be the cause of rhabdomyolysis. The patient’s CPK dropped considerably upon discontinuation of the novel antibiotic, and symptoms resolved. The adverse drug event was reported to the drug manufacturer; however, there are no reports up until this time that address a possible relationship between eravacycline administration and the development of rhabdomyolysis.
Keywords
Introduction
Creatine phosphokinase (CPK) is an enzyme that is associated with muscle damage and acute kidney injury when elevated. 1 This elevation can lead to rhabdomyolysis, a medical emergency comprised of muscle necrosis characterized by myalgias and myoglobinuria. 2 CPK levels can vary based on factors such as race, gender, and physical activity; however, levels usually range from 30 to 200 U/L. 3 Diagnosis of rhabdomyolysis is based on CPK, which, although not standardized, is typically reported as greater than 5-times the upper limit of normal. 4 Aggressive rehydration, management of electrolyte abnormalities, and addressing the precipitating cause are crucial in preventing complications. 3 There are many potential causes of rhabdomyolysis including trauma and exertion, body temperature changes, electrolyte imbalances, and drug-induced causes that have been reported in clinical practice. 4 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitors and daptomycin are 2 of the medications most commonly associated with the development of elevated CPK levels and rhabdomyolysis. 5
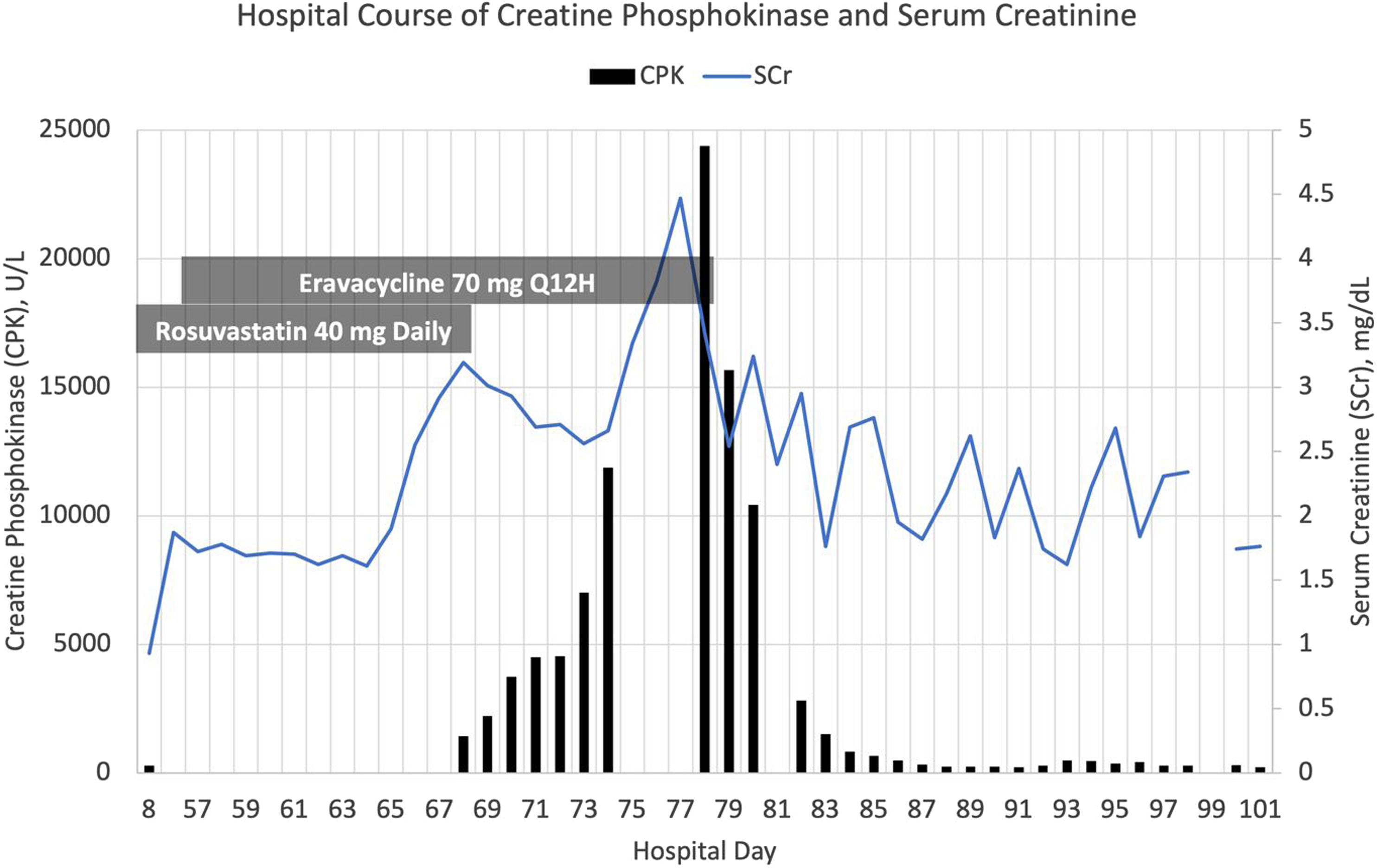
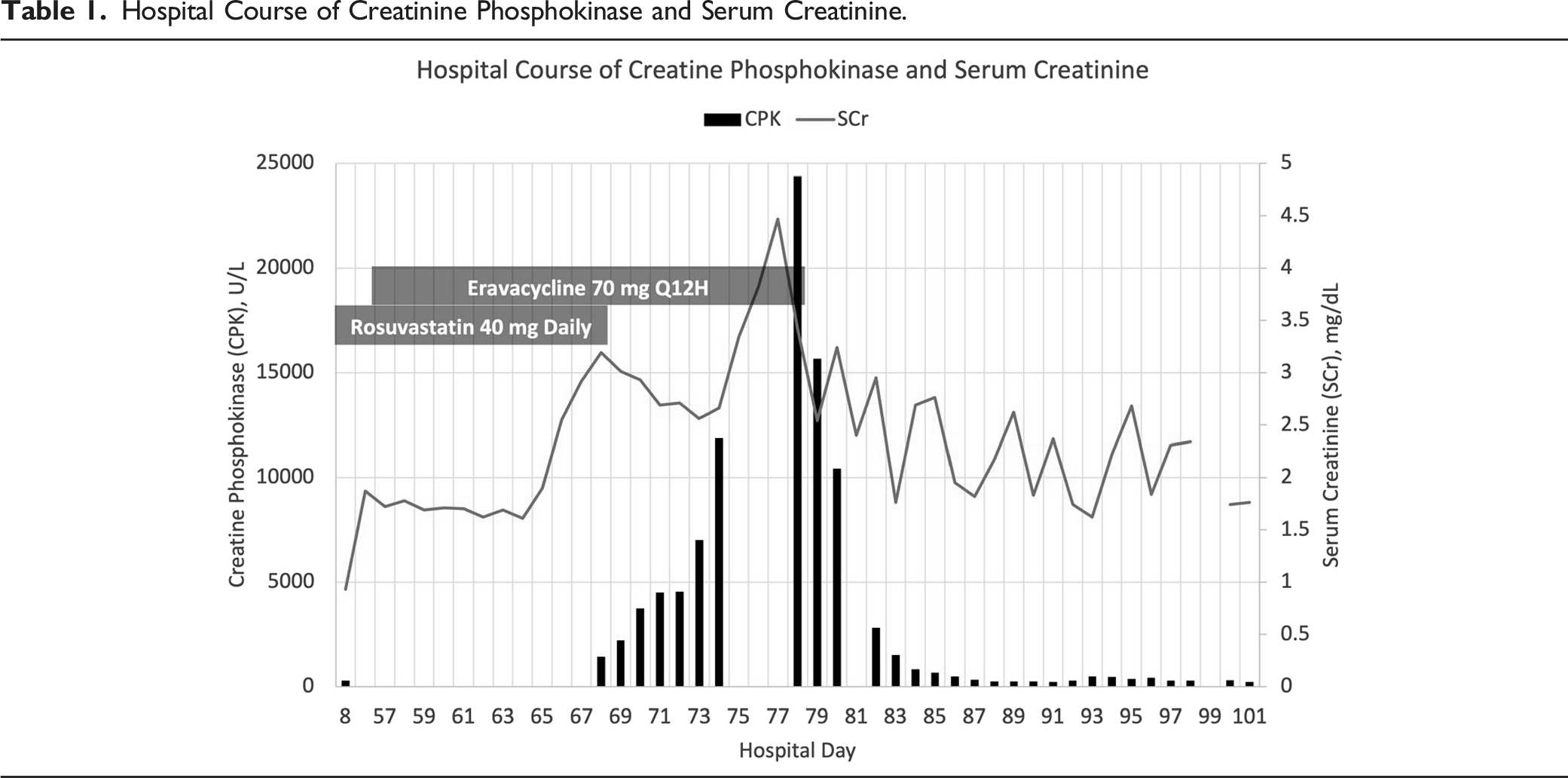
Hospital Course of Creatinine Phosphokinase and Serum Creatinine.
Case Report
A 74-year-old female with a pertinent past medical history significant for heart disease, coronary artery disease (CAD), peripheral artery disease (PAD), and diabetes presented to the emergency department with a non-ST-elevation myocardial infarction (NSTEMI). Given her significant history of CAD and PAD, the patient was taking an HMG-CoA reductase inhibitor, rosuvastatin, at home to prevent further complications of her cardiovascular disease and had tolerated the medication for 5 years without incident. Of note, the patient had no significant history of renal or hepatic impairment at baseline.
The patient required coronary artery bypass graft (CABG) surgery on hospital day 5. The patient’s recovery after surgery was complicated by the development of a sternal wound infection and osteomyelitis on hospital day 44. Wound and bone cultures grew extended spectrum beta-lactamase (ESBL)-producing
The patient developed an acute kidney injury (AKI) on hospital day 56, which continued to progress despite aggressive hydration with intravenous fluids. After approximately a week of persistently worsening renal function, the medical team checked a CPK level to assess for rhabdomyolysis since the patient was also experiencing muscle pain. The CPK resulted at 1436 U/L on hospital day 68 (Graph 1), significantly elevated from the baseline of 279 U/L on hospital day 8. Active medications at that time included amiodarone, famotidine, sertraline, rosuvastatin, and tramadol in addition to the previously mentioned antibiotics. Upon review, daily rosuvastatin 40 mg was deemed to be most likely associated with the CPK elevation and thus was promptly discontinued. However, the CPK continued to rise daily for the next 10 days despite aggressive hydration and discontinuation of rosuvastatin. The CPK peaked at 24 381 U/L on hospital day 78 (Graph 1), at which point eravacycline was discontinued in response. Within 24 hours of discontinuing eravacycline, the CPK level decreased to 15 674 U/L and subsequently continued to decrease gradually, reaching the baseline level on day 88 (10 days after discontinuing eravacycline).
Discussion
Both non-medication and medication-related culprits were evaluated to determine the most likely cause of rhabdomyolysis. Elevated CPK serum concentrations have been reported post-surgery due to muscle damage at the operation site; however, previous literature reports that CPK concentrations generally peak 1-2 days after surgery and gradually decline over the next few days,11,12 with 50% of CPK values back within normal reference range by day 5 post operatively. 12 Given that this patient was approximately 30 days post CABG surgery, it appears unlikely that the rise in CPK was due to surgical complications. Upon initial review of the patient’s medication profile, rosuvastatin was determined to be the most obvious potential inciting agent of this patient’s rhabdomyolysis. Rhabdomyolysis has been reported in patients receiving statin therapy, and available literature suggests the risk of statin-associated muscle toxicity is increased when statin CYP3A4 substrates (simvastatin and atorvastatin) are co-administered with CYP3A4 inhibitors.3,13 Although this patient was taking amiodarone, a moderate CYP3A4 inhibitor, rosuvastatin is not significantly metabolized by CYP3A4 and therefore concomitant use with CYP3A4 inhibitors is less likely to result in rhabdomyolysis. 14 Additionally, the median onset of rhabdomyolysis is approximately 30 days after initiation of an HMG-CoA reductase inhibitor, 15 whereas this patient had been taking rosuvastatin for over 5 years without incident as an outpatient. Rosuvastatin was restarted upon admission to the hospital and continued for weeks without incident prior to the initiation of eravacycline. Furthermore, a 50% reduction in CPK levels is expected within the first 24 hours after discontinuation of an HMG-CoA reductase inhibitor. 16 This patient’s CPK level continued to rise for 10 days after discontinuation, further reducing the likelihood of rosuvastatin being the causative medication.
Despite previous literature of tetracycline-induced rhabdomyolysis being limited to a single case report, eravacycline was determined to be the most likely cause of this patient’s rhabdomyolysis due to the timing of the onset and the quick downtrend of CPK levels after the medication was discontinued. Once eravacycline was discontinued, the CPK level decreased from 24 381 U/L to 15 674 U/L within 24 hours, an approximately 40% reduction in 29 hours. The half-life of serum CPK is 36 hours, 17 so this reduction is consistent with what is expected after discontinuation of the inciting agent. The Naranjo algorithm was used to determine the likelihood of rhabdomyolysis being caused by eravacycline. 18 Overall, with the information available, a Naranjo score of 4 was calculated, which correlates with a possible adverse drug reaction due to eravacycline. The score was calculated based on the adverse event appearing after the suspected drug was administered, and the adverse reaction improving when the drug was discontinued. A re-challenge with eravacycline after resolution of rhabdomyolysis would have provided more information to help further establish causality; however, it was not feasible in this patient.
The exact mechanism by which eravacycline causes rhabdomyolysis is unknown. Drug-induced myopathies can occur through a variety of mechanisms and are classified based on the type of injury to the muscle fiber or organelle. 19 In previously reported cases of minocycline-induced myopathies, the authors’ hypothesized that the patients experienced an immune-mediated inflammatory myopathy since both patients presented with symptoms of hypersensitivity syndrome or autoimmune hepatitis.10,20 The patient in this report did not present with symptoms suggestive of drug-induced hypersensitivity syndrome, making an inflammatory myopathy less likely. Eravacycline causing a direct myotoxic effect cannot be excluded at this time.
To our knowledge, this is the first case to describe the development of rhabdomyolysis likely due to eravacycline. Clinicians should be mindful of this possible adverse effect when a patient receiving eravacycline develops elevated CPK levels along with myalgia and/or myopathy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.