Abstract
Aim
Emesis of oral medications continues to be a problem in the management of acute pediatric asthma exacerbations; therefore, we set out to assess whether smaller volumes of oral dexamethasone resulted in better tolerability.
Introduction
Background: Asthma affects over 12% of Canadian children and acute asthma exacerbations account for the greatest number of Emergency Department (ED) admissions in Canada each year.1,2 These exacerbations often require the use of a corticosteroid as part of treatment. A Cochrane Review found that the use of corticosteroids within 1 hour of presentation to an ED significantly reduces the need for hospital admission in patients with acute asthma. 3 Further, a short course of oral corticosteroids following assessment for an asthma exacerbation significantly reduces the number of relapses to additional care, hospitalizations and use of short-acting beta2-agonist without an apparent increase in side effects. 4 Oral corticosteroids are as effective as intravenous and intramuscular injections in the management of acute asthma.4,5 However, the emesis of oral medications continues to be a problem in the management of acute asthma. Qureshi et al. demonstrated that 2 doses of dexamethasone provided similar efficacy with improved compliance and fewer side effects, including vomiting, than 5 doses of prednisone.6,7 This has influenced the management of acute asthma for emergency departments across Canada. 7 The current standard of care at our tertiary care emergency department (ED) was to receive a 2-day course of dexamethasone (10 mg/mL parenteral formulation) given orally instead of using the liquid oral formulation (1 mg/mL) to decrease the volume of medication delivered. This practice translates to a smaller volume of medication given to the child, but it sacrifices palatability. Some clinicians would argue that smaller volumes of corticosteroids result in less vomiting, regardless of taste. This argument was based on the larger volume of prednisolone that was needed in the treatment of acute asthma exacerbations that resulted in more vomiting compared to the smaller volume of dexamethasone. Yet, there is no documented evidence in the literature that supports this theory that smaller volumes are better tolerated than larger ones.
Importance: Hendeles reported that steroid formulations should be easily swallowed and retained. Palatability was another important factor to consider. 8 The 10 mg/mL formulation was initially intended for parenteral use and it is formulated for this purpose. No flavoring agents have been added and it is bitter because of its isotonic state. The addition of the Ora Sweet aids in masking the bitterness, making the formulation sweet and more pleasant. Because this smaller volume is less palatable and no literature supports the smaller volume theory in asthma management, this investigation set out to provide evidence that smaller volumes result in less vomiting. If this hypothesis is true, then palatability would continue to be sacrificed in order to decrease the emesis rate. However, if volume does not play a role in the emesis rate, palatability should no longer be forfeited. The sooner and longer a patient can keep down the dexamethasone, the faster it will take effect and start to relieve the asthma exacerbation. Every time a child vomits medication, the time to its onset of action is lengthened, as the dose will need to be repeated. The experience of vomiting is also not a pleasant, and if vomiting can be avoided, it will improve the quality of life for the child, and the parent.
Goals of this Investigation: The primary objective of this study was to assess whether smaller volumes of oral dexamethasone resulted in better tolerability, specifically less vomiting, in pediatric patients with an acute exacerbation of asthma. We wished to enroll sufficient subjects that we could be 95% certain that a 50% reduction in the emesis rate from 15 to 7.5% was of the true value (see Primary Data Analysis).
Materials and Methods
Study Design: We designed an open, prospective, randomized controlled trial to compare the tolerability and volumes of 0.3 mg/kg of oral dexamethasone, with and without sweetener, for the treatment of acute asthma in children. This study received full institutional ethics board review at the IWK Health Centre Research Ethics Board, Halifax, Nova Scotia, Protocol 2967 in August 2004. All participants/caregivers provided written, informed consent and the trial was registered at ClinicalTrials.gov, identifier number NCT00973687.
Setting: The participants were recruited from a children’s tertiary care emergency department servicing both urban and rural populations in Eastern Canada. The study took place between September 2004 and April 2006. The total number of ED visits for all ages during that time was 42 405, with 1742 of those visits having asthma as the most responsible diagnosis for children aged 2 and older.
Selection of Participants: The study group consisted of children 2–14 years old presenting with an acute exacerbation of their asthma defined as worsening of their asthmatic symptoms or increased difficulty in breathing, who also required oral systemic corticorsteroids as part of their management. Emergency Department attending physicians were involved in the eligibility criteria because they determined the need for corticosteroid treatment. Children were excluded from the study if they had a preference for pills over liquid formulations, had a history of Nissen fundoplication surgery (decreased the ability to vomit), needed immediate airway intervention, required oral medications to be given via a G or J tube or if patient care would be compromised (as determined by the ED care team). They were excluded if they have been enrolled in the study on a previous visit. Children under the age of 2 were not enrolled to avoid including those patients with bronchiolitis.
Interventions: Participants were then randomized to receive dexamethasone 10 mg/mL or dexamethasone plus a sweetening agent, called Ora Sweet, to produce a formulation of 1 mg/mL, giving a 10-fold larger volume per dose. Ora Sweet contains sorbitol, glycerin and saccharin and is a common pharmaceutical solution in the pediatric setting. Blinding of the treatment group was therefore not feasible. The randomization was computer generated and included dynamic allocation blocking in groups or 2, 4, or 6. This randomization allocation sequence was transferred into individually sealed, opaque envelopes that were placed in the medication room and opened in order, after consent was obtained, thereby concealing the randomization of the formulation. Participants were assigned to their groups based on the allocation specified in the sealed envelope. Participants were stratified, based on history a priori, to a second strata if they have had an episode of emesis 4 hours prior to receiving dexamethasone. A separate set of envelopes was available for that stratum, generated by the same process. The sequence was thus concealed until interventions were assigned.
The corticosteroid used in this study was dexamethasone sodium phosphate injection USP 10 mg/mL, with a weight-based dose of 0.3 mg/kg to a maximum of 16 mg of dexamethasone given orally. If the participant vomited within 45 minutes of taking the dexamethasone, the same randomized formulation was given again. If vomiting occurred again, the emergency attending physician decided if the formulation should be repeated a third time.
Due to the variation in treatment practice amongst the attending emergency physicians in the department, after 45 minutes had elapsed from the time the dexamethasone was given, a second dose of dexamethasone could be ordered. This would bring the total dose of dexamethasone administered to the patient to 0.6 mg/kg, as opposed to the study dose of 0.3 mg/kg. Less than 25%, approximately, of the attending physicians chose to give the second dose, though these numbers were not recorded as part of the study.
When the participant was ready to go home, another dose of dexamethasone, the same formulation as per randomization, was given to take home with him/her, as per the current standard of care. This single dose was to be given to the child the next morning. A follow up phone call was made to the caregiver 7–14 days following the visit to the ED. They were asked 6 “yes or no” questions regarding how well the child’s asthma was presently controlled.
Methods and Measurements: Upon entry into the ED, all children had a set of triage vital signs recorded by the triage nurse. The triage nurse recorded asthma medications used in the preceding 24 hours. Upon placement into the examination room, a registered nurse obtained the child’s weight. The volume of dexamethasone was based on this weight. This information was transferred to the numerically labeled, standardized data collection forms once the participant was enrolled in the study. The registered, research nurse who obtained consent for the study filled in the patient’s data collection form with the following data: volume of medication and dosage, along with the time the dexamethasone was given, whether emesis occurred and how long after giving the dexamethasone did the vomiting occur, and if the dose of dexamethasone was repeated. The number of salbutamol aerosols the child received prior to the dexamethasone was also recorded as well as the total number of salbutamol doses received prior to discharge from the ED. On a separate form, the follow up phone number was recorded. These forms were then separated by the nurse to blind the research associate, making the phone calls, to the formulation the participant received. During the follow up phone call, any unscheduled visits to a physician for asthma along with present symptoms of cough, wheeze, difficulty breathing, night waking and daily activity limitations were recorded in a yes or no format by the research associate. The data was transcribed into a spreadsheet by the research associate for further analysis. Data was then checked by the principal investigator for discordant or missing data and the discordant fields were reconciled by referral to the hardcopy of the data collection form or the patient chart.
Primary data analysis: The emesis rate in the published literature ranges from 3 to 15% for children with a severe asthma exacerbation, requiring systemic corticosteroid treatment.6,9-11 Using an estimate of a 50% reduction in emesis from 15% to 7.5%, with a two-tailed alpha of .05 and power of .80, the number needed in each group was 304, for a total sample size of 608. An interim or preliminary analysis examining the baseline rate of emesis in each group was planned, a priori, to be performed once 200 subjects had been enrolled in the study with the possible intention of adjusting the sample size if the prevalence of emesis was appreciably different from the estimate originally gleaned from the literature. The 2 groups were compared by appropriate tests of association. Chi-square analyses were used to compare the proportions of patients in each study group who exhibited the outcome measure (emesis) and to analyze the secondary outcomes. We used logistic regression to determine whether there would be a relationship between the study groups, the formulation, and the vomiting outcome. Analyses were conducted using SPSS v13.0 (2004) and EpiInfo (CDC 2001).
Results
Characteristics of study subjects: Of the 874 ED patients who had respiratory distress between the ages of 2 and 14 years that were screened for eligibility between September 2004 and April 2006, 430 participants met entry criteria and were enrolled in the study. Reasons for exclusion of the remaining 444 patients are listed in the CONSORT diagram shown in Figure 1. Of the 430 patients randomized, 68 (16%) were stratified to the second strata for prior vomiting. A total of 14 patients were lost to follow up for the secondary outcomes, as they were unable to be reached by phone on multiple occasions. The data was analyzed by intention to treat. As shown in Table 1, demographic and baseline characteristics of the study population were comparable in both groups. In particular, the children had a mean age of 4.79 years (SD 2.65), 142/430 (33%) were females and 154/430 (36%) required admission to hospital. The triage vitals recorded for the 1 mg/ml Ora Sweet group were heart rate (HR): median 130, mode 140, min/max 72/192; respiratory rate (RR): median 34, mode 40, min/max 18/84; pulse oximetry: median 95%, mode 96%, min/max 85/100%. For the 10 mg/mL group, HR median 132, mode 120, min/max 72/186; RR: median 36, mode 40, min/max 18/80; pulse oximetry: median 95, mode 95, min/max 78/100. Consort diagram. Baseline Features by Randomization Group.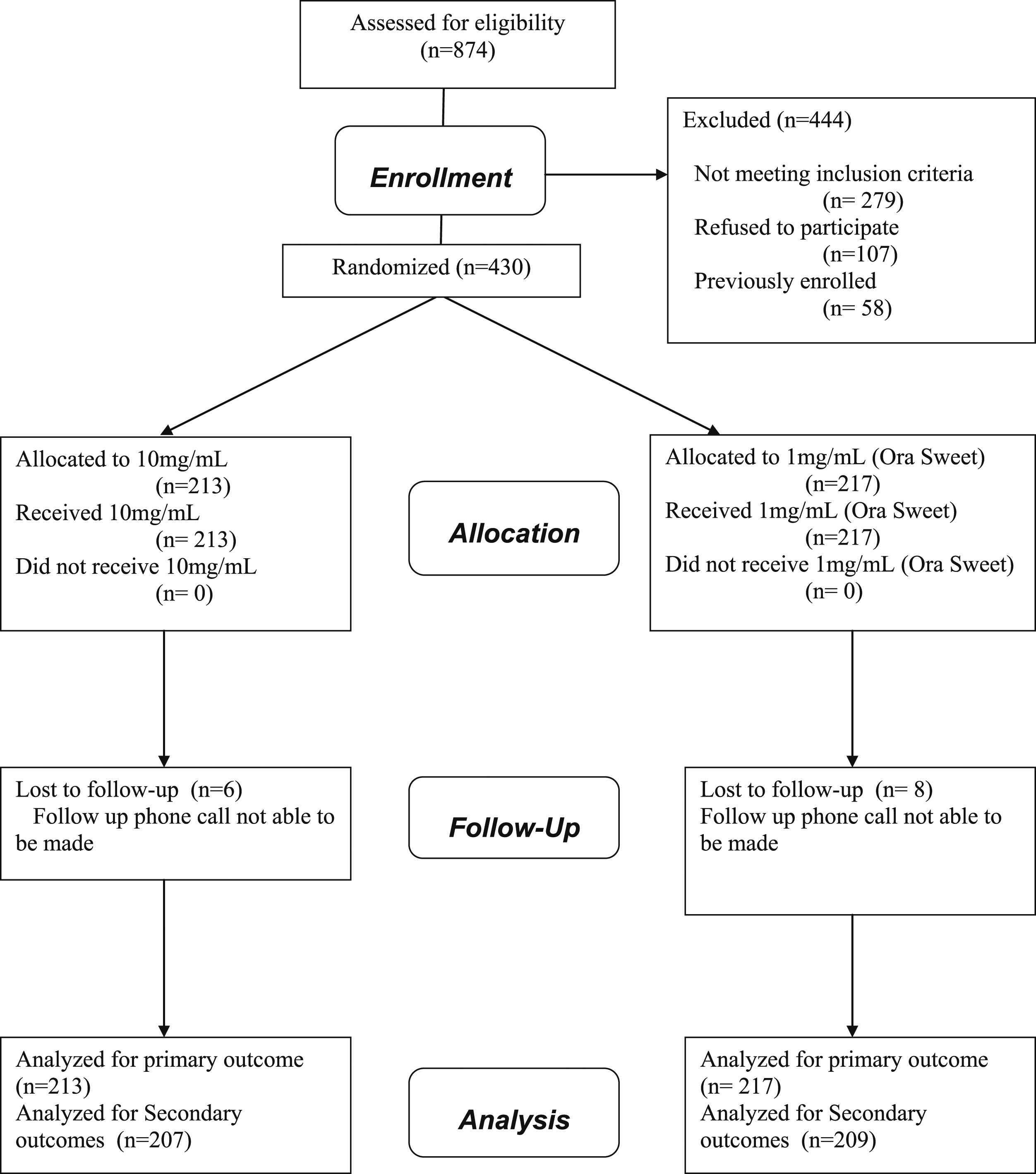
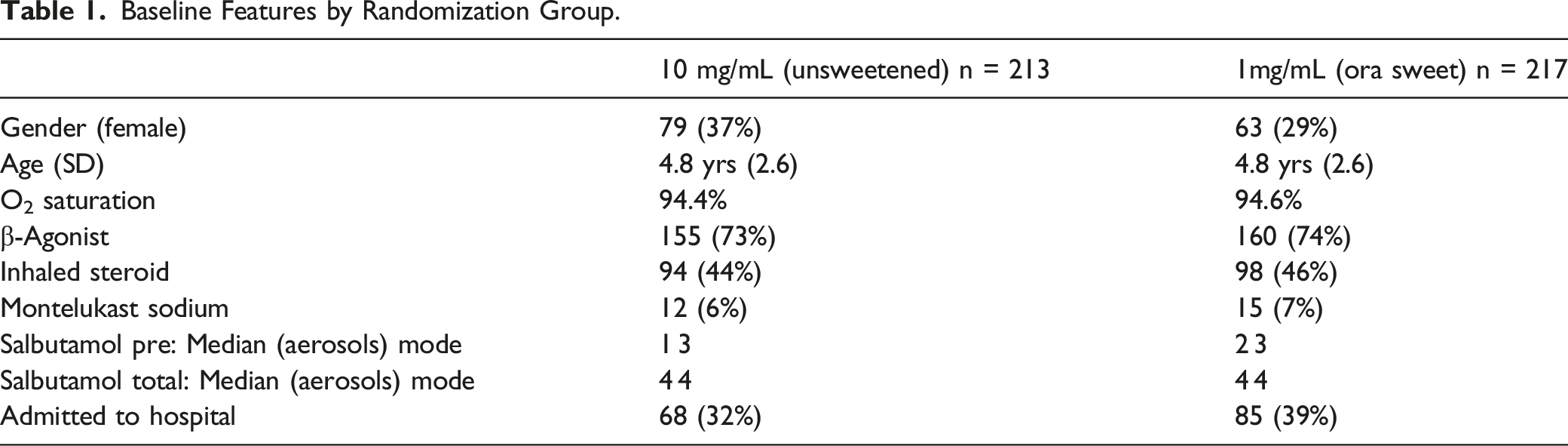
Interim Analysis: A planned interim analysis was conducted in March 2005 after 197 children in total had been randomized, with 38 enrolled in the second strata, to evaluate the rates of emesis. 14/100 (14%) vomited the 10 mg/mL formulation and 9/97 (9%) vomited the 1 mg/mL formulation (RR=.66, P=.42). The rates of emesis were higher in the second strata (those previously vomiting), with 8/20 (40%) in the 10 mg/mL vomiting compared to 4/18 (22%) in the 1 mg/mL group (RR=.56, P=.41). Based on the rates of emesis observed in this study, approximately 772 children would need to be enrolled in each group (for a total of 1544 participants), in order to detect a 50% drop in the rate of emesis.
As a result of this analysis, the advisory board and the principal investigator decided to revise the planned sample size and continue enrollment until April 2006 or until 500 participants had been recruited. We anticipated the children presenting to the ED in respiratory distress would have previously been approached for enrollment in the study should the study be continued further. By continuing enrollment to this number, we could confirm that the current trends were continuing and increase our precision in estimating the tolerability effect.
Primary Outcome: Emesis rates.

There was a significantly greater chance of vomiting after administration of the dexamethasone for those previously vomiting than for those not previously vomiting, regardless of the formulation given. Comparing all patients that received 10 mg/mL formulation, those in the second strata (those previously vomiting prior to receiving dexamethasone), 12/34 (35%) vomited after receiving the dexamethasone compared to 11/179 (6%) who were not previously vomiting (RR:5.74, P < .01). Comparing all patients that received the 1 mg/mL formulation, 8/183 (24%) in the second strata vomited compared to 8/34 (4%) who were not previously vomiting (RR:5.38, P < .01. In a logistic regression model predicting emesis after dexamethasone administration (the dependent variable), we examined 2 explanatory/independent variables, vomiting within 4 hours preceding dexamethasone administration (stratified a priori in the study) and dexamethasone formulation (either sweetened with Ora Sweet or with sweetener). Prior vomiting status was significantly related to the outcome of vomiting (P < .01), while formulation was not (P = .21). No adverse side effects were reported with the Ora Sweet.
Secondary Outcomes: Clinical Equivalency.
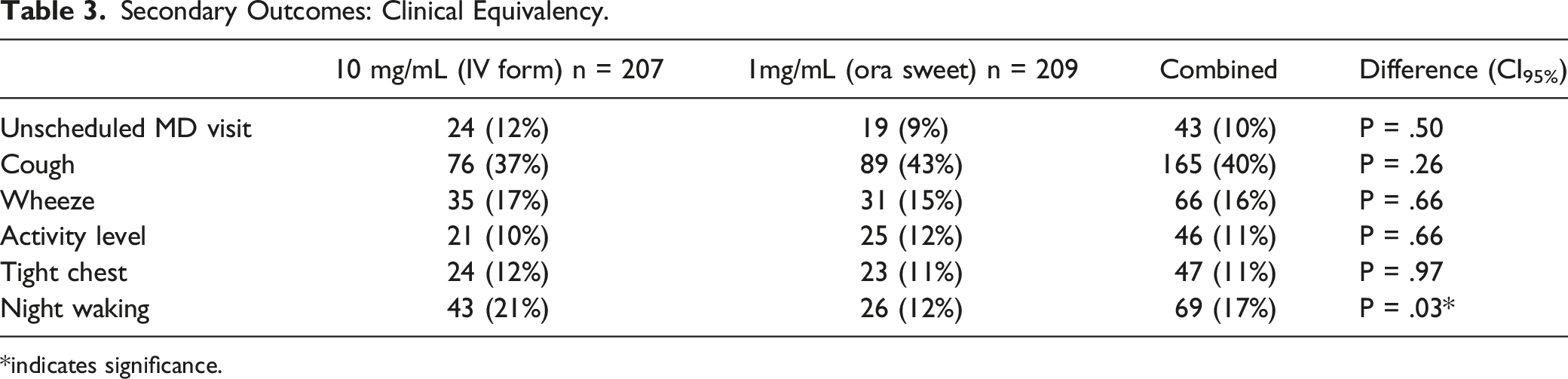
*indicates significance.
Discussion
This prospective, randomized, controlled trial is the first of its kind addressing the tolerability of dexamethasone and the influence of volume. This study found that the rates of emesis and thus tolerability of dexamethasone are not influenced by volume. Since no statistical difference between the high and low volume groups were demonstrated, volume is not an important factor, as previously believed. We did find that if children were vomiting prior to receiving dexamethasone, they were more likely to vomit the medication. Overall, 3–6% of children will have an episode of emesis within 45 minutes of administering dexamethasone and 24–35% of children will vomit if they were previously vomiting within 4 hours prior to receiving dexamethasone. Although dexamethasone is an antiemetic, it acts upon the chemoreceptor zone of the medulla oblogata and requires systemic absorption before exerting this central effect. Emesis of corticosteroids is a faster effect occurring prior to drug absorption and therefore did not play a role in this study.
Tablet crushing is another oral administration practice routinely observed with corticosteroids, giving the crushed pill formulation with a liquid or puree to help mask the taste. Lucas-Bouwman et al. demonstrated that the oral solution of prednisolone had superior tolerability over crushed tablets in the treatment of acute severe asthma in children. In their study, vomiting was observed in 23% of participants using crushed tablets, and in none of the participants taking the oral solution. 11 While the cost of pill crushing is considerably less than using the liquid formulation, it results in the re-administration of the corticosteroid, increasing time both in the emergency department and time to onset of action. This study should dissuade the practice of pill crushing, emphasizing the importance of tolerability in the treatment of childhood asthma.
One point of concern arising from this study is the rate of uncontrolled asthma symptoms 1–2 weeks following an emergency department visit (Table 3). 40% of children are still coughing, 16% are wheezing, 11% have their activity level impacted, and 11% complain of a tight chest. In addition, 17% are waking at night, impacting the sleep of at least 1 caregiver. These findings mirror those found by Benito-Fernandez et al. with 21% of pediatric patients at day 15 after discharge still having persistent respiratory symptoms.12,13 This poor control would suggest the need for stronger follow up with the child’s primary caregiver or asthma specialist following the ED visit, as supported by the literature. 14
Qureshi et al. Comparison to Address Dosage Debate.
Limitations: There are several limitations that pose a threat to internal and external validity. The first threat is sample size. The initial calculations of sample size were based on the estimate of emesis rates gleaned from the existing literature. Although the emesis rates pertained to corticosteroids in the treatment of asthma, nebulized, oral or parenteral formulations were used. Prednisolone/prednisone was the corticosteroid used in several of the studies.5,7-9 The rates of emesis seen in this study are less than the 15% used to calculate the sample size needed to show a reduction of 50% in the emesis rate. Continuing recruitment to over 400 children ensured that we had sufficient precision to estimate the tolerability of dexamethasone. We believe that we are justified in our decision to terminate the study at that point. These adjustments have been made in other studies without compromising outcomes substantially. 10
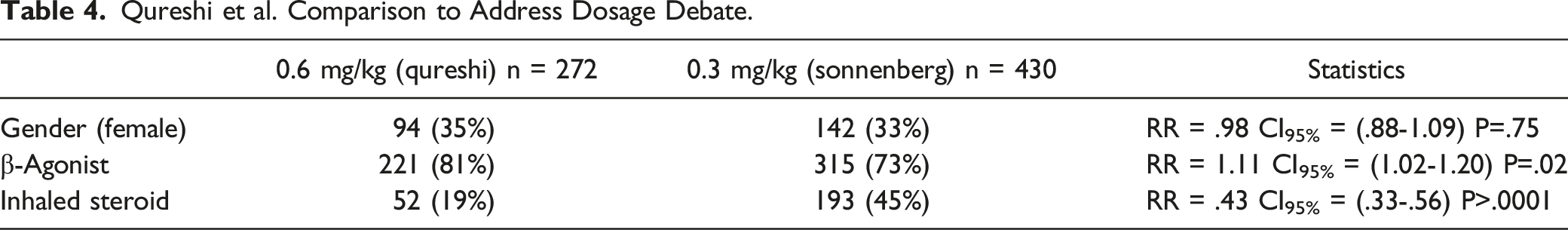
The second threat to validity was the lack of agreement of dosing of dexamethasone in the ED amongst the attending emergency physicians. There is still ongoing debate as to the dosage of oral dexamethasone; however, the 0.3 mg/kg/dose, equivalent to 1 mg/kg of prednisolone, appears to be the more commonly found dosing recommendation. The initial study supported the use of dexamethasone in the treatment of acute asthma used 0.3 mg/kg/dose, once daily, for 2 days. However, some physicians were wanting to use the 0.6 mg/kg over the 0.3 mg/kg dosing and this was impacting enrollment. After 10 patients were enrolled in the study, the protocol was changed to allow the physician to choose either the 0.3 mg/kg or 0.6 mg/kg dose of dexamethasone. This inconsistency amongst final dosing does not impact the primary outcome of emesis, though it does impact on the secondary analysis, as the treatment groups are no longer equal in the dosage of dexamethasone received.
The third threat pertains to the secondary outcome. Six yes or no questions were posed to the caregiver 7–14 days following the child’s visit to the ED. The questions only ask if the child is coughing or wheezing etc., but do not quantify the degree of coughing. It is therefore subjective to parental interpretation. Additionally, it was not always feasible to reach the caregiver by phone during the 7–14 day window. Although this was the goal, it was not always reached. The mean number of days for which the caregiver was reached was 16, with a median of 12 and mode of 10. There were 14 caregivers who were unable to be reached at any time.
The addition of Ora Sweet is a potential confounder since it was not added to both dexamethasone preparations. It is not known whether Ora Sweet has the potential to impact upon emesis rates positively or negatively.
Finally, patient enrollment in the study was influenced by the business of the department, as the nurses were responsible for recruitment. During busier times, recruitment was decreased during these periods. Patients were, however, recruited 7 days a week, around the clock, during the 20-month recruitment period.
Despite these limitations to internal validity, the generalizability of this study remains high due to its large sample size and both urban and rural populations in the hospital’s catchment area. The baseline characteristics for our population are found in Table 1 and are generally in keeping with the published literature.
Conclusions
In conclusion, the volume of dexamethasone does not likely influence its tolerability. Palatability should not be sacrificed to deliver a smaller volume of medication. Mary Poppins may have been right all along, “a spoonful of sugar helps the medicine go down!” 17
Footnotes
Acknowledgments
I would like to thank Dr Paul Pianosi, Dr Brett Taylor, and Diann Nicholson, for their help with study design and encouragement throughout this study, as well as the entire emergency department staff for their help with patient enrollment and data collection. I would also like to thank Dr Colleen O’Connell for her statistical support, Dr Mark Gierl for his recheck of the data, and our research associate, Dr Emily (Zinck) Pelley, for data entry and patient follow up.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the IWK Foundation, Category A, Number 2967.
