Abstract
Purpose
The pharmacology of roflumilast, recent dosing revisions, and the integral roles of pharmacists in effective chronic obstructive pulmonary disease (COPD) management are reviewed here.
Summary
COPD is characterized by progressive airflow limitation and intermittent acute exacerbations of symptoms, which contribute to disease progression, worsening of comorbidities, and reduced health-related quality of life. Patients with COPD may use a variety of pharmacotherapies (in combination with nonpharmacological modalities) to prevent exacerbations, reduce the impact of symptoms, and reduce or prevent COPD progression. Given the complex and multifaceted nature of disease management, pharmacists are uniquely positioned to collaborate with other clinicians to improve treatment adherence and efficacy via a number of diverse avenues in patients with COPD. Central to this endeavor is patient education and counseling regarding their treatment regimen.
Conclusion
Recent findings from a phase 3 clinical trial demonstrate improved tolerability and reduced treatment discontinuation resulting from the use of an uptitration regimen in patients with severe COPD who initiate therapy with roflumilast. Pharmacists have a central role in effective COPD management, especially with respect to patient education about treatments.
Introduction
Current Prevalence, Diagnosis, and Staging of Chronic Obstructive Pulmonary Disease
Chronic obstructive pulmonary disease (COPD) is characterized by fixed airflow limitation, an enhanced airway inflammatory response, and a progressive, largely irreversible decline in lung function. COPD is frequently associated with additional comorbidities and is currently the third leading cause of mortality worldwide. 1 As such, COPD represents a substantial economic burden and is a growing public health concern. 2
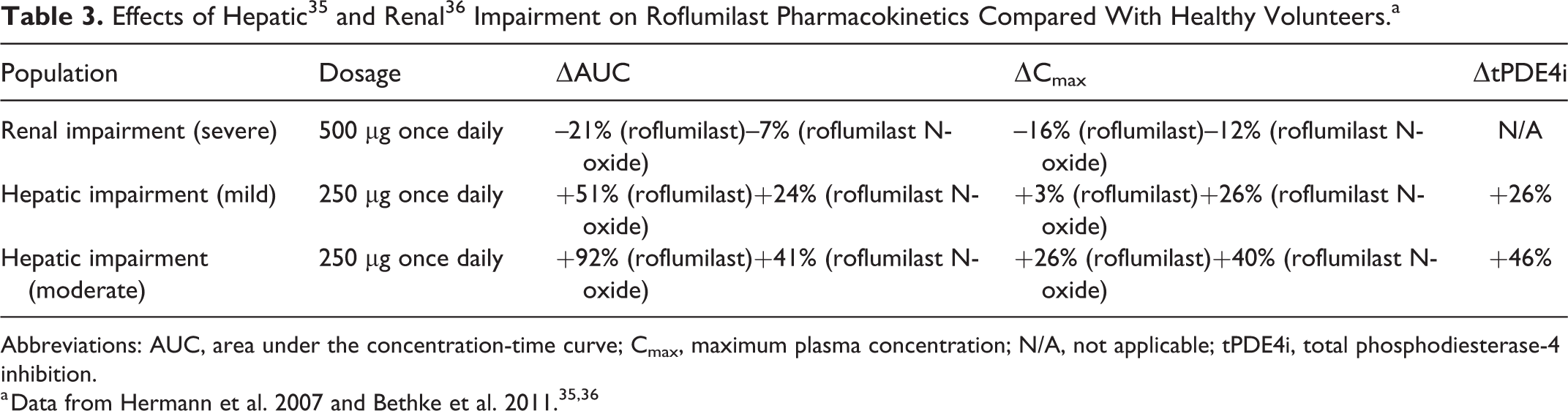
According to the Global Initiative for Chronic Obstructive Lung Disease (GOLD), patients with dyspnea, chronic cough/sputum production, and/or a history of lower respiratory tract infections should be evaluated for COPD. A diagnosis of COPD involves an assessment of patient risk factors (eg, tobacco use, indoor and outdoor air pollution, occupational risks, and host factors), symptoms (eg, chronic cough and shortness of breath), and measurements of lung function. 3 Spirometry is used to measure lung function/airflow limitation; a COPD diagnosis is confirmed by a postbronchodilator forced expiratory volume in 1 second (FEV1)/forced vital capacity (FVC) ratio (FEV1/FVC) of <0.70. 3 Patients with COPD are classified by grade and group. Postbronchodilator spirometry measurements are used to classify COPD according to increasing spirometric grades of severity from GOLD 1 (mild COPD; FEV1 of ≥80% predicted) to GOLD 4 (very severe COPD; FEV1 of <30% predicted). 3 Then the patient is placed in a group based on exacerbation risk and symptom burden indicated by an A to D grouping. Group A corresponds to lower symptom burden and lower exacerbation risk, and group D corresponds to greater symptom burden and greater exacerbation risk (Figure 1). 3 Patient classification based on these factors informs appropriate drug therapy considerations.
The ABCD assessment tool for chronic obstructive pulmonary disease (COPD). A refined version of the combined ABCD assessment tool that separates spirometric grades of COPD and classifies a patient’s individual disease based on symptomatic assessments and risk for exacerbations. Figure reprinted with permission from the Global Initiative for Chronic Obstructive Lung Disease from the Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2020 report). © 2020 Global Initiative for Chronic Obstructive Lung Disease, Inc. CAT, COPD Assessment Test; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; GOLD, Global Initiative for Chronic Obstructive Lung Disease; mMRC, modified British Medical Research Council questionnaire.
COPD Exacerbation Prevention
Exacerbations of COPD are significant events that hasten disease progression and increase overall morbidity and mortality. A major goal of pharmacotherapy is to reduce the frequency and severity of exacerbations. COPD exacerbations are an acute worsening of a patient’s baseline syndrome, beyond the normal day-to-day symptom variation (eg, cough, dyspnea, and sputum production), which requires further therapy, ranging from additional medication to hospitalization. Patients with more severe disease are more likely to have exacerbations, and a history of COPD exacerbations can be indicative of a greater risk for future events.4,5 Furthermore, factors associated with greater exacerbation risk include increased age, duration of COPD, poor treatment adherence, 6 recent antibiotic use or hospitalizations, 7 higher blood eosinophil counts, 8 and the presence of comorbidities (especially cardiac conditions). 9 There is now considerable evidence indicating that prevention of exacerbations in patients with COPD is a critical component in effective disease management. 3
Exacerbations often lead to additional health complications and are associated with worse health-related quality of life, an accelerated decline in lung function, 10 increased hospitalizations, increased health care costs, 11 and increased mortality. 1 Treatment of exacerbations includes the use of short-acting bronchodilators, corticosteroids, and antimicrobials.3,5 A number of treatment options have been reported to either prevent or reduce exacerbations in patients with COPD, including both nonpharmacological (eg, physical exercise, smoking cessation, and pulmonary rehabilitation) and pharmacological interventions. Some clinical guidelines recommend a treatment regimen that leverages a combination of both modalities.3,12
Proper Selection and Use of Currently Available Medications for COPD
A range of therapies are available to manage COPD symptoms and reduce exacerbations and disease progression. The GOLD 2020 guidelines recommend inhaled bronchodilators, such as long-acting β2-agonists (LABAs) or long-acting muscarinic receptor antagonists (LAMAs), as a key component to prevent and reduce symptoms. 3 LABAs and LAMAs can be used alone or in combination and have been shown to relieve symptoms, reduce exacerbation rates, improve lung function and health status, and decrease hospitalizations. In patients who continue to have dyspnea after starting therapy with a LABA or LAMA, a LABA/LAMA combination therapy may provide additional benefits to effective symptom management. 3
For patients with COPD who have a history of exacerbations, the addition of an inhaled corticosteroid (ICS) to a LABA regimen may improve lung function and decrease the frequency of exacerbations, especially in patients with moderate to very severe COPD. ICS monotherapy is not warranted because it does not prevent the decline in FEV1 or reduce mortality. 3 Because regular ICS use can increase the risk for pneumonia, sustained use of an ICS alone is not recommended in patients with COPD. 3 When dual-combination inhaled therapy for COPD does not provide adequate control, triple therapy with an ICS/LABA/LAMA combination is recommended.13,14 Interventions that have been shown to reduce the frequency of exacerbations in patients with COPD are shown in Table 1.
Interventions That Reduce Chronic Obstructive Pulmonary Disease Exacerbation Frequency.a

Abbreviations: ICS, inhaled corticosteroid; LABA, long-acting β2-agonist; LAMA, long-acting muscarinic receptor antagonist.
a Data from the Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2020 report). 3
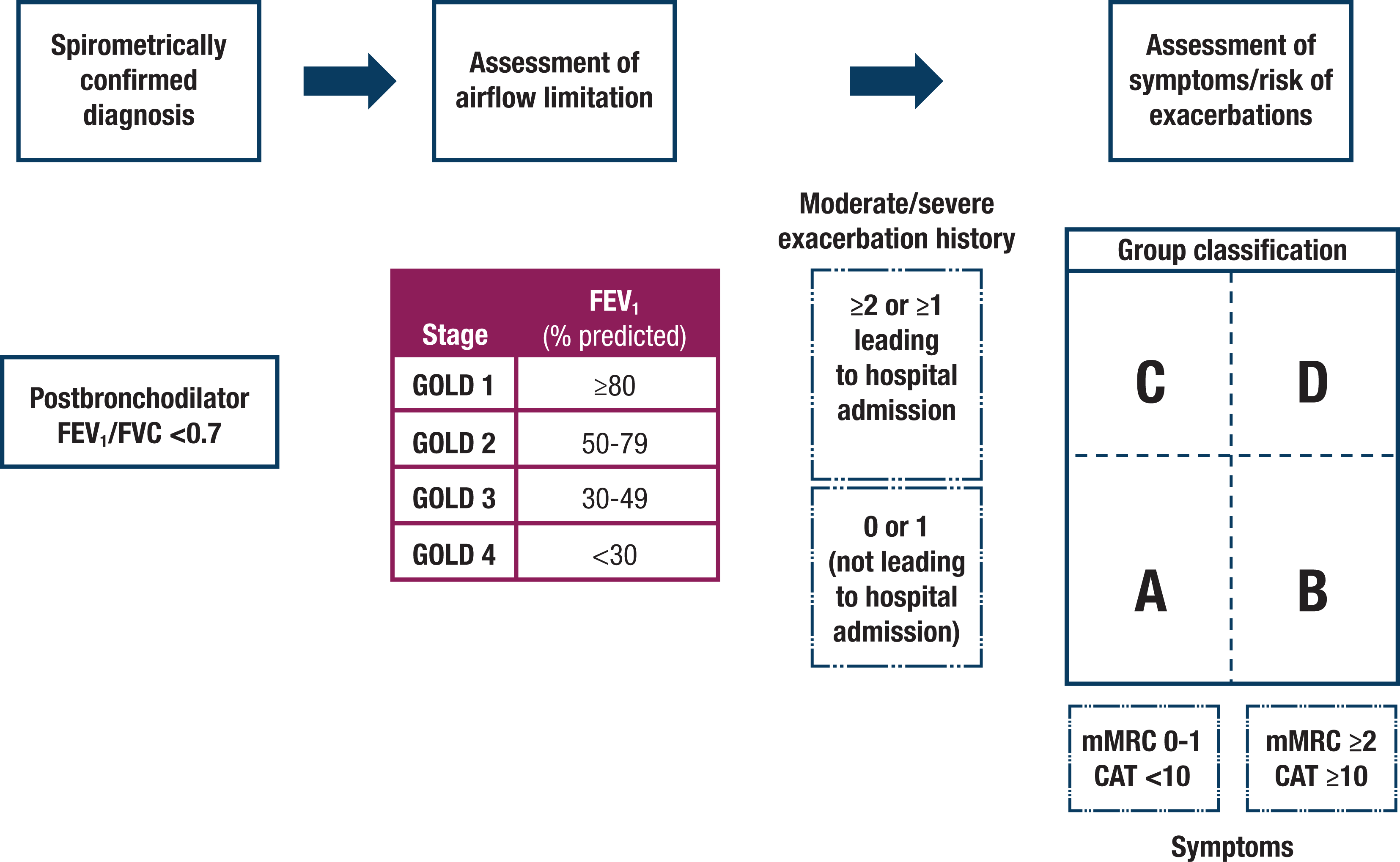
The current GOLD guidelines provide recommendations for initial pharmacotherapy based on disease severity and potential modifications, depending on the control of dyspnea or exacerbation risk. The guidelines include a model for escalation or de-escalation of treatment for each severity group (Figure 2). For patients who continue to have exacerbations, a chronic bronchitis phenotype, and severe to very severe airflow limitations (FEV1 of <50% predicted) while using an ICS/LABA or ICS/LABA/LAMA combination, the GOLD report recommends the use of roflumilast, a phosphodiesterase-4 (PDE4) inhibitor. 3
Suggested algorithm to be used in choosing/refining a patient’s pharmacological regimen for chronic obstructive pulmonary disease (COPD) treatment based on the ABCD assessment. Figure reprinted with permission from the Global Initiative for Chronic Obstructive Lung Disease from the Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2020 report). © 2020 Global Initiative for Chronic Obstructive Lung Disease, Inc. CAT, COPD Assessment Test; eos, blood eosinophil count (cells/µL); FEV1, forced expiratory volume in 1 second; ICS, inhaled corticosteroid; LABA, long-acting β2-agonist; LAMA, long-acting muscarinic receptor antagonist; mMRC, modified British Medical Research Council questionnaire; pred., predicted. *Consider if eos ≥300 or eos ≥100 AND ≥2 moderate exacerbations/1 hospitalization. **Consider de-escalation of ICS or switch if pneumonia, inappropriate original indication or lack of response to ICS.
Overview of Roflumilast
PDE4 Inhibition and the Management of COPD
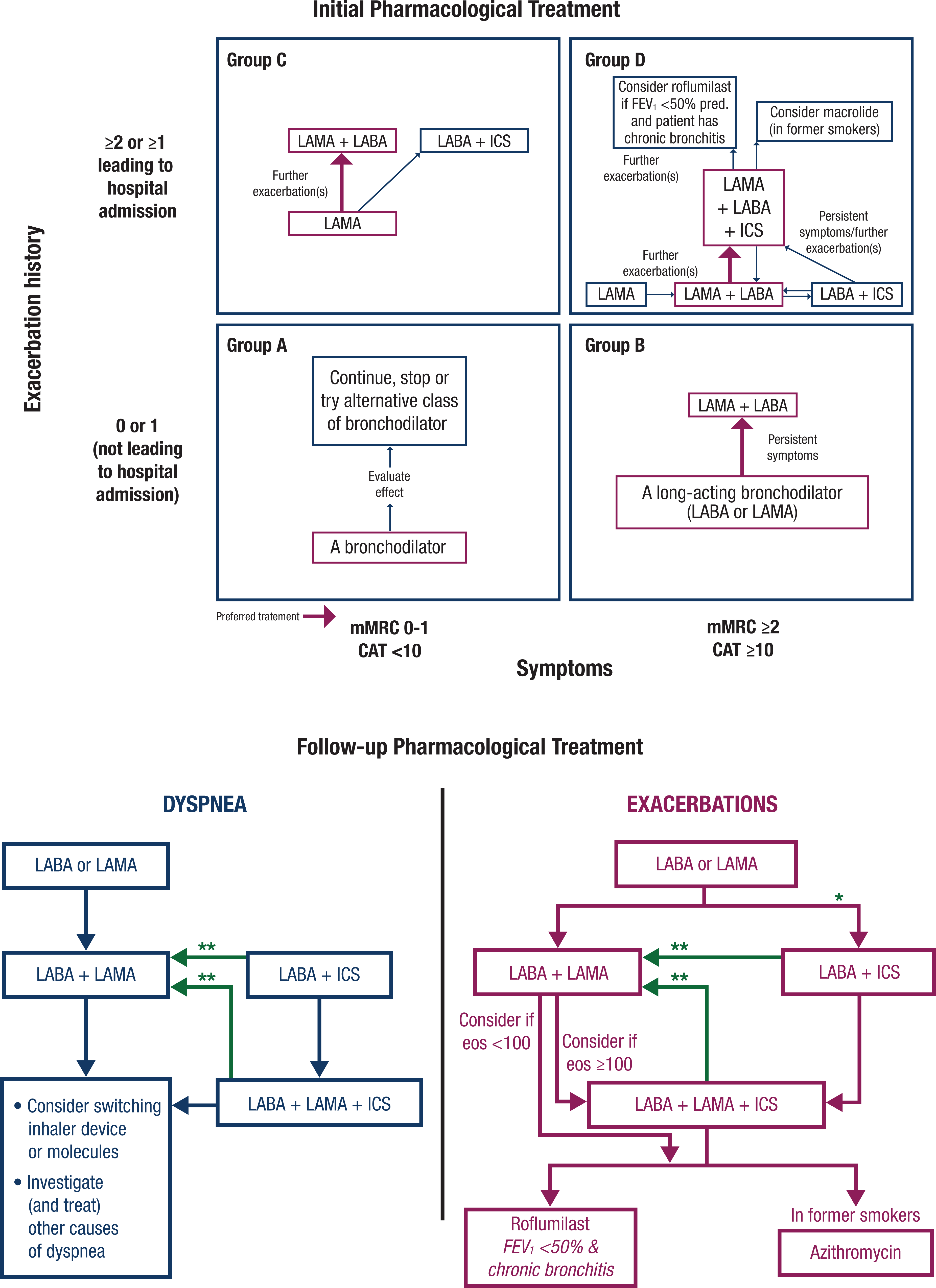
PDE4 is a cyclic-3′,5′-adenosine monophosphate (cAMP)–specific phosphodiesterase expressed in airway smooth muscle, inflammatory cells, immune cells, pulmonary nerves, brain, and cardiovascular tissue that metabolizes cAMP into adenosine monophosphate (AMP). 15 Inhibition of PDE4 leads to increased cAMP in inflammatory cells as well as relaxation of airway smooth muscle, and this is the proposed mechanism by which PDE4 inhibitors help to alleviate COPD symptoms (Figure 3). 16 It has been suggested that PDE4 inhibitors reduce the release of inflammatory mediators (including cytokines) and inhibit cell proliferation and chemotaxis of several inflammatory cell types.17,18 Indeed, PDE inhibitors have been used in several laboratory and clinical studies (eg, PDE5 inhibitors) for treatment of allergic inflammation, although these inhibitors exhibit significantly less efficacy than PDE4 inhibitors. 18 Roflumilast is a selective PDE4 inhibitor indicated by the US Food and Drug Administration as a treatment to reduce the risk for exacerbations in patients who have severe COPD with chronic bronchitis and a history of exacerbations. 16
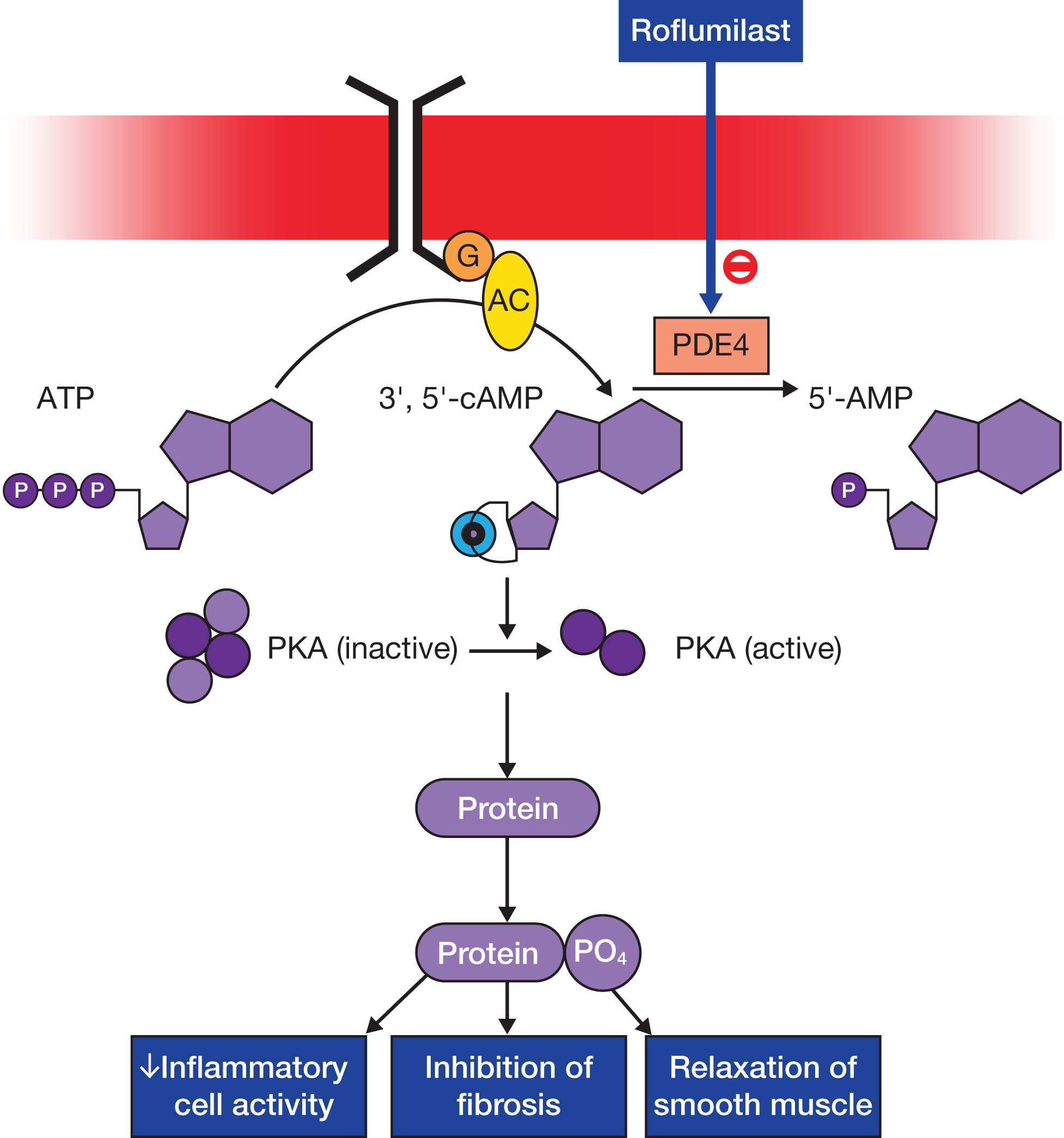
Roflumilast mechanisms of action. Roflumilast inhibition of phosphodiesterase-4 (PDE4) decreases inflammatory cell activity, inhibits fibrosis, and relaxes smooth muscle. Figure reprinted with permission from the British Pharmacological Society from Rabe KF. Update on roflumilast, a phosphodiesterase 4 inhibitor for the treatment of chronic obstructive pulmonary disease. Br J Pharmacol. 2011;163(1):53-67. © 2011 The Author, British Journal of Pharmacology. AC, adenylate cyclase; AMP, adenosine monophosphate; ATP, adenosine triphosphate; cAMP, cyclic adenosine monophosphate; G, G-protein-coupled receptor; PKA, protein kinase A; PO4, phosphate.
Effect of Roflumilast on Lung Function and Exacerbations
A number of randomized, placebo-controlled trials have demonstrated that roflumilast improves lung function and reduces the frequency of exacerbations requiring systemic corticosteroid therapy in patients with severe COPD (Table 2). In the Roflumilast—an Oral Anti-inflammatory Treatment for Chronic Obstructive Pulmonary Disease: a Randomized Controlled Trial (RECORD) and Effect of 1-year Treatment With Roflumilast in Severe Chronic Obstructive Pulmonary Disease (RATIO) trials, patients receiving roflumilast demonstrated significant improvements in pre- or postbronchodilator FEV1 (up to 97 mL difference) and a decreased risk for exacerbation compared with those receiving placebo.20,21 The AURA (M2-124) and HERMES (M2-125) trials, which were identical studies in different patient populations, indicated that roflumilast improved lung function and reduced the frequency of exacerbations in patients with symptoms of bronchitis and severe airflow limitations. 22
Summary of Results From Clinical Trials Investigating Roflumilast.a
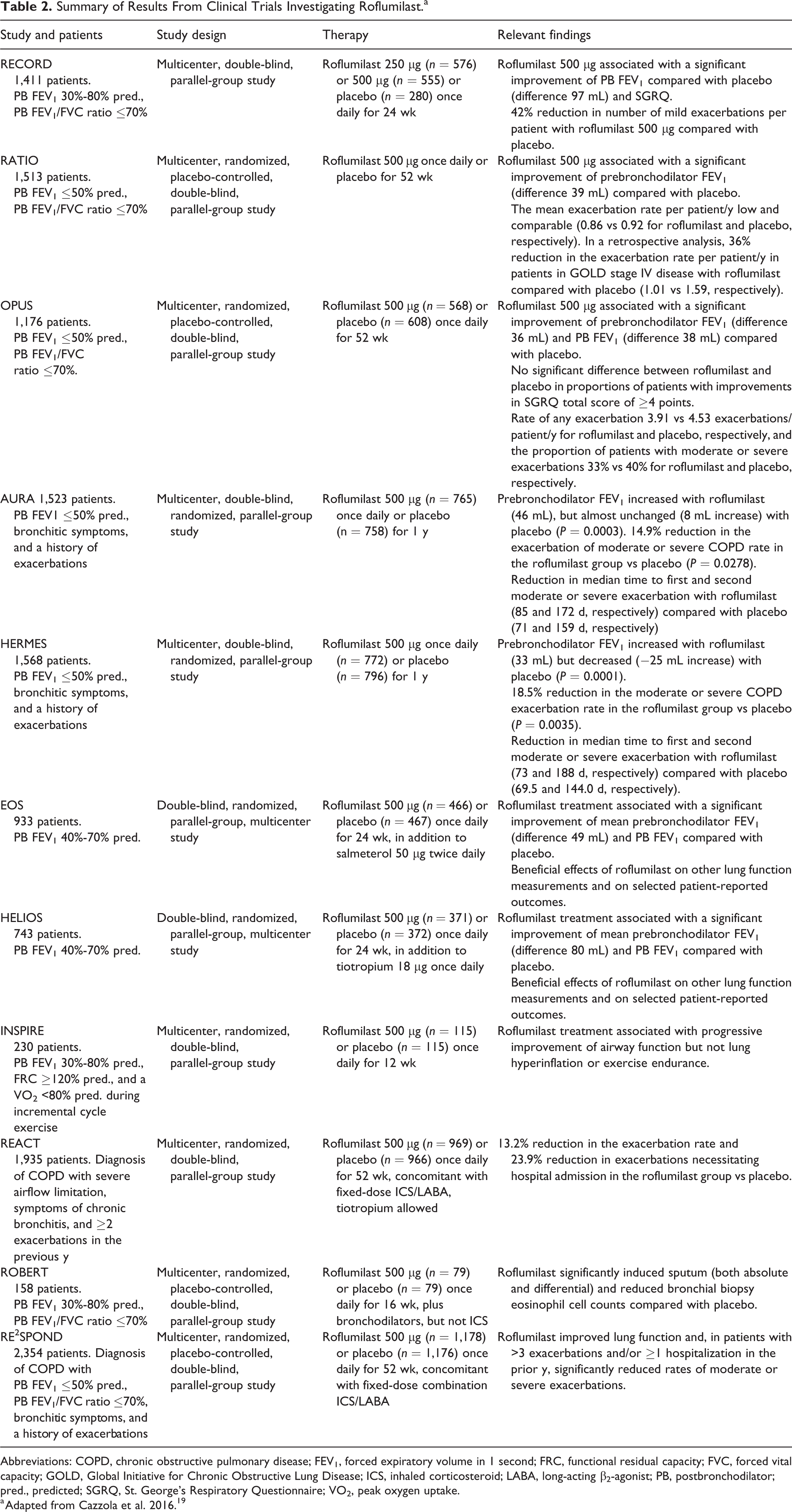
Abbreviations: COPD, chronic obstructive pulmonary disease; FEV1, forced expiratory volume in 1 second; FRC, functional residual capacity; FVC, forced vital capacity; GOLD, Global Initiative for Chronic Obstructive Lung Disease; ICS, inhaled corticosteroid; LABA, long-acting β2-agonist; PB, postbronchodilator; pred., predicted; SGRQ, St. George’s Respiratory Questionnaire; VO2, peak oxygen uptake.
a Adapted from Cazzola et al. 2016. 19
Results from the Roflumilast and Exacerbations in patients receiving Appropriate Combination Therapy (REACT) study showed that roflumilast reduced hospital admissions and exacerbations for patients with chronic bronchitis and severe COPD who were at risk for frequent severe exacerbations despite the use of ICS/LABA or ICS/LABA/LAMA combination therapies. 23 Furthermore, the Effect of Roflumilast and Inhaled Corticosteroid/Long-acting β2-agonist on Chronic Obstructive Pulmonary Disease Exacerbations (RE2SPOND) study also reported that roflumilast improved lung function and reduced exacerbations in patients with a history of frequent exacerbations and/or hospitalization despite using ICS/LABA or ICS/LABA/LAMA combination treatments. 24 Finally, results from a pooled analysis of the REACT and RE2SPOND trials indicated that roflumilast demonstrated a greater treatment effect in those who had a prior hospitalization due to COPD, a high baseline blood eosinophil count (≥150 cells/µL, ≥150 to <300 cells/µL, or ≥300 cells/µL), and a history of frequent exacerbations. 25
Safety Considerations
A recent systematic review and meta-analysis identified diarrhea, headache, nausea, and weight loss as the most common adverse events (AEs) reported among roflumilast trials. 26 There is evidence that roflumilast AEs related to the gastrointestinal (GI) tract (diarrhea, nausea, and weight loss) are dose dependent and also consistent with those expected for PDE4 inhibitors. 19 Rates of early discontinuation from studies of roflumilast were higher for patients taking roflumilast compared with placebo. 27 It is noteworthy that roflumilast has also been associated with psychiatric-related AEs, including insomnia, anxiety, and depression, with suicidal ideation or behavior observed in postmarketing studies, independent of patient history of depression. 16
Pharmacology
Pharmacokinetics
A therapeutic dose of roflumilast is 500 µg, administered orally once daily, and has a mean absolute oral bioavailability of 79% for immediate-release roflumilast.28,29 The time to achieve the maximum plasma concentration (Tmax) of roflumilast is approximately 1 hour in a fasted state, 30 which is delayed by 1 hour in a fed state; the time to achieve plateau-like maximum plasma concentrations of the N-oxide metabolite is approximately 8 hours (range, 4-13 hours, unaffected by food).16,31 Steady-state plasma concentrations of roflumilast and roflumilast N-oxide are achieved after 4 days and 6 days of once-daily dosing, respectively. 30
The single-dose volume of distribution for 500 µg of roflumilast is 2.9 L/kg with 99% and 97% plasma protein binding for roflumilast and roflumilast N-oxide, respectively.16,28,32 Roflumilast is readily distributed to organs and tissues, and data from studies with radiolabeled roflumilast indicate low penetration across the blood-brain barrier.16,27 Roflumilast is metabolized via N-oxidation through the phase I cytochrome P-450 (CYP) enzymes, CYP3A4 and CYP1A2. Further metabolism occurs via phase II reactions. 33
After short-term intravenous (i.v.) infusion, the average plasma clearance of roflumilast was 9.6 L/h.16,27 The terminal plasma half-life is 17 hours for roflumilast and 27 hours for roflumilast N-oxide.27,32 Following both i.v. and oral administration of radiolabeled roflumilast, approximately 70% of roflumilast is detected in urine and 20% in feces (as inactive metabolites).16,27
Pharmacodynamics
Roflumilast is 2 to 3 times more potent than the N-oxide metabolite at inhibiting the PDE4 enzyme. Despite this in vitro finding,33,34 the major metabolite roflumilast N-oxide is responsible for approximately 90% of total PDE4 inhibition (tPDE4i) due to an approximately 10-fold greater plasma area under the concentration-time curve (AUC) compared with the parent roflumilast molecule, which accounts for the remaining 10% of tPDE4i.28,29 As such, coadministration of roflumilast with strong CYP450 inducers or CYP450 inhibitors can affect the systemic exposure and thus the therapeutic efficacy of roflumilast.16,27 Long-term treatment with roflumilast (500 µg once daily) has been shown to reduce sputum neutrophils (31%) and eosinophils (42%). 16
Contraindications and Drug Interactions
The AUC for roflumilast is increased in patients with mild (51%) to moderate (92%) hepatic impairment compared with patients with normal hepatic function. 35 The AUC for the primary metabolite, roflumilast N-oxide, is increased to a lesser extent in patients with mild (24%) or moderate (41%) hepatic impairment (Table 3). It is noteworthy that these changes in the AUCs were associated with concomitant increases in tPDE4i. 35 Although clinicians are advised to consider the risks and benefits of administering roflumilast to patients with mild (Child-Pugh A) hepatic impairment, roflumilast is not recommended for patients with moderate or severe (Child-Pugh B or C) hepatic impairment.16,27,33 Severe renal impairment is reported to moderately decrease the roflumilast and roflumilast N-oxide AUC (21% and 7%, respectively 36 ); however, these changes only moderately reduce tPDE4i (∼9%) compared with healthy control patients, and thus dosage adjustments are not necessary. The changes in AUC in patients with severe renal impairment may be attributed to protein loss because the drug exhibits high protein binding or may be related to reduced absorption in this population.16,27,32 Finally, given the involvement of CYP450 enzymes in roflumilast metabolism and elimination, it should be noted that CYP450 enzyme inducers (eg, rifampicin, phenytoin, and tobacco smoke 37 ) may reduce roflumilast exposure, thereby decreasing the therapeutic efficacy of roflumilast. 16
Abbreviations: AUC, area under the concentration-time curve; Cmax, maximum plasma concentration; N/A, not applicable; tPDE4i, total phosphodiesterase-4 inhibition.
Dosing Revision: Results From the OPTIMIZE Trial
A number of AEs have been reported for patients initiating roflumilast treatment with the approved 500-µg dose, including effects on the GI system (nausea, diarrhea, abdominal pain, loss of appetite, and weight loss), headache, and insomnia.38,39 Although these effects are generally of mild to moderate severity and frequently resolve within weeks of commencing roflumilast treatment, 40 they are nevertheless a common cause of early treatment discontinuation. Overall, discontinuation rates for the recent REACT and RE2SPOND trials were approximately 30%, although rates of roflumilast treatment discontinuation are likely higher in clinical practice.23,24,41
Given the transient nature of these AEs, a recent 12-week, multicenter, double-blind, phase 3 trial evaluated whether an alternative dosing strategy during roflumilast initiation would improve side effect tolerability and increase the likelihood that patients would remain on the treatment. 38 This study enrolled and randomly assigned 1,321 patients with severe COPD, chronic productive cough, and a history of exacerbations (those with ongoing exacerbations or a lower respiratory tract infection unresolved within 4 weeks preceding screening were excluded) taking either a LABA, LAMA, or LABA/LAMA combination approved to treat COPD. Patients in the study were randomized to 1 of 3 regimens (1:1:1): 250 µg of roflumilast once daily for 4 weeks (uptitration), then 500 µg of roflumilast once daily thereafter (roflumilast 250/500 µg once daily); 500 µg of roflumilast once every other day (EOD) for 4 weeks (uptitration), then 500 µg of roflumilast once daily thereafter (roflumilast 500 µg EOD/once daily); or 500 µg of roflumilast once daily for the entire 12 weeks of the study (roflumilast 500 µg once daily). All patients choosing to discontinue the trial were allowed to enter an 8-week downtitration phase, receiving 250 µg of roflumilast once daily. 38
The primary endpoint of the Evaluation of Tolerability and Pharmacokinetics of Roflumilast, 250 µg and 500 μg, As Add-on to Standard COPD Treatment to Treat Severe COPD (OPTIMIZE) trial was the percentage of patients prematurely discontinuing study treatment for any reason. Secondary endpoints were the percentage of patients reporting AEs of interest (also used to evaluate tolerability: headache, insomnia, diarrhea, nausea, decreased appetite, and abdominal pain), the percentage of patients prematurely discontinuing study treatment for any reason in the downtitration phase, and changes in prebronchodilator FEV1 during both the trial and the downtitration phase. 38
The study found that there were significantly fewer study discontinuations in the roflumilast once-daily group who initiated treatment at 250 µg and underwent the 4-week uptitration regimen compared with the group that began treatment with the approved 500 µg of roflumilast once-daily regimen (18.4% vs 24.6%; odds ratio, 0.66; P = 0.017). 38 Although there were numerically fewer discontinuations in the roflumilast EOD/once-daily group undergoing a 4-week uptitration, compared with the roflumilast 500-µg once-daily group, the difference was not significant. 38 Furthermore, of the patients in the roflumilast 250/500-µg once-daily group, significantly fewer experienced AEs of interest compared with those in the roflumilast 500-µg once-daily group (45.4% vs 54.2%; odds ratio, 0.63; P = 0.001). Of the patients with ≥1 AE of interest to evaluate tolerability during the trial, patients in the roflumilast 250/500-µg once-daily group experienced fewer reports of diarrhea (24.5% vs 30.2%), nausea (19.7% vs 24.8%), headache (24.3% vs 26.0%), decreased appetite (22.7% vs 29.1%), insomnia (22.2% vs 24.8%), and abdominal pain (19.3% vs 20.1%) compared with the roflumilast 500-µg once-daily group. 38 The roflumilast 250/500-µg once-daily group also experienced a longer median time to onset of the first AE compared with the roflumilast 500-µg once-daily group (10.0 vs 7.0 days). 38
The results of this study demonstrate that patients initiating treatment with a 4-week uptitration of 250 µg of roflumilast were less likely to discontinue treatment or report AEs of interest compared with patients who began treatment with the approved 500-µg roflumilast dose (odds were 34% and 37% lower, respectively). Indeed, for the patients who began treatment with 500 µg of roflumilast once daily, the rate of study discontinuation in the OPTIMIZE trial was consistent with rates observed in recent roflumilast studies. The findings demonstrate that initiating roflumilast treatment at 250 µg once daily for 4 weeks, before increasing to 500 µg once daily, can improve roflumilast tolerability and reduce roflumilast treatment discontinuation rates. 38
The Pharmacist’s Role in Optimizing Roflumilast Use
Pharmacists represent an excellent avenue for a number of multidisciplinary approaches to improve treatment outcomes for patients with COPD through collaborative efforts. Disease education for patients with or at risk for COPD, in the form of self-management (eg, daily symptom tracking, physical/mental health self-assessments, and action plans to prevent exacerbations) and lifestyle changes (eg, physical activity, nutrition, and weight management), can help to prevent additional health complications and reduce morbidity.2,3,5 Pharmacists can also help to identify patients with poor symptom control or acute worsening of symptoms, refer patients to visit their physician, and play a fundamental role in the effort to improve medication adherence in patients with COPD. 2 Given the hurdles that can arise in beginning treatment and even refilling medications, community pharmacists are uniquely positioned to support patient access to medication, device selection, and account for missed doses. 2 Efforts to leverage the knowledge and experience that community pharmacists have regarding COPD and COPD treatment may lead to significant reductions in the personal, social, and economic costs associated with this disease.5,6,9
Exacerbations of COPD have a significant impact on disease progression and overall costs of care. Preventing COPD exacerbations is a major goal of treatment, and therapies that reduce exacerbation risks—including roflumilast—can provide benefit. Pharmacists are in a unique position to evaluate current control of the disease and adherence to therapy. Therefore, pharmacists are an important resource for counseling and monitoring regarding roflumilast therapy.
Pharmacists should be aware of the role of roflumilast in COPD management according to guidelines and the recent studies involving a titrated dosage regimen. Roflumilast is indicated for patients with severe or very severe COPD with chronic bronchitis and a history of exacerbations and is intended to reduce the risk for exacerbations for these patients. 3 Furthermore, the new uptitration dosing scheme improves tolerability and reduces the frequency of treatment discontinuations for patients beginning roflumilast therapy. 38 Indeed, given the essential role of education when therapies are initiated, pharmacists should be prepared to counsel patients who initiate treatment with roflumilast. Pharmacists can assist patients with education about the role of roflumilast in managing their COPD, proper medication dosing, the importance of medication adherence for treatment effectiveness, the reason for titrating roflumilast, and the anticipated time between treatment initiation and the onset of benefits. Because roflumilast is commonly prescribed for patients using a LABA or fixed-dose ICS/LABA combination, 3 pharmacists should also be prepared to advise patients of the potential drug-drug interactions between roflumilast and other medications they may be taking, as well as the potential side effects of roflumilast and effective strategies by which to minimize them.
Other Considerations
The costs for chronic medications used in COPD management, including inhalation therapies, can be a barrier to optimal care. Roflumilast therapy is indicated in specific situations where a patient with COPD continues to experience exacerbations despite bronchodilator therapy. This medication is covered by most insurance companies but is typically listed as a Tier 4 medication requiring prior authorization. There are discount cards available to support the costs of roflumilast, and the manufacturer sponsors a patient assistance program, which includes stable copayments or savings for up to 12 months of therapy.
There are no specific storage requirements for roflumilast other than a controlled room temperature.
Conclusions
It is well established that COPD exacerbations negatively impact a patient’s health-related quality of life, contribute to disease progression, and represent a significant personal and economic burden. As such, exacerbation prevention is a fundamental component in disease management for patients with COPD. Roflumilast is an effective add-on therapy for patients with severe COPD associated with chronic bronchitis and a history of exacerbations that can reduce the number of exacerbations that patients experience. In several clinical trials, AEs associated with treatment initiation contributed to the number of patients with severe COPD who discontinued roflumilast treatment. Recent results from the OPTIMIZE trial demonstrate that the use of an uptitration phase of 250 µg of roflumilast once daily for 4 weeks, prior to continuing on the prescribed regimen of 500 µg of roflumilast once daily, significantly reduces the odds of early treatment discontinuation by 34% and improves the tolerability of roflumilast. As discussed here, the available evidence demonstrates that community pharmacists can play an important role in COPD management. Based on repeated and consistent interactions with patients, pharmacists can recognize poor symptom control, emphasize adherence, ensure good inhalation technique, and provide vaccinations. Furthermore, education and counseling about the role of the medication and its appropriate use, smoking cessation, and disease management are essential. Improvements in COPD management can reduce exacerbations, disease progression, hospitalizations, and morbidity, as well as improve health-related quality of life for patients with COPD. These observations clearly demonstrate the importance of pharmacists’ understanding of COPD medications and their indications for use, as well as the most up-to-date prescribing guidelines for these treatments.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dennis Williams reports that his spouse owns stock in GlaxoSmithKline.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The primary source of funding was AstraZeneca (Wilmington, DE, USA). Medical writing support was provided by Dan Jackson, PhD, of MedErgy (Yardley, PA, USA) in accordance with Good Publication Practice (GPP3) guidelines and was funded by AstraZeneca (Wilmington, DE, USA).