Abstract
In September 2023, Calgary, Canada experienced a large point-source outbreak of Shiga toxin-producing Escherichia coli (STEC) infection linked to a centralized kitchen serving daycare centers. More than 200 children presented with bloody diarrhea, and 21 developed hemolytic uremic syndrome (HUS). Nine children required acute kidney replacement therapy (KRT). All were managed with urgent-start peritoneal dialysis (PD) initiated on general pediatric wards. Tenckhoff catheters were inserted by pediatric urologists and PD was initiated approximately 12 h after placement using low initial fill volumes that were escalated as tolerated. Mean PD duration was 10 days (range 4–16). No patients required conversion to extracorporeal KRT, and all patients recovered kidney function with normal estimated glomerular filtration rates by 3 months post-discharge. PD effectively managed solute clearance and acid-base balance while minimizing ultrafiltration, supporting maintenance of intravascular volume, a strategy that was linked to better neurological and kidney outcomes in STEC-HUS. One patient developed culture-negative peritonitis treated with intraperitoneal antibiotics, and the same patient subsequently experienced delayed bowel perforation requiring surgical intervention after PD discontinuation and catheter removal. Neurologic outcomes in two children showed substantial recovery at 3 months evaluation. Our findings demonstrate that urgent-start PD can be implemented in general ward settings rather than in pediatric ICUs, conserving critical care resources during periods of high demand. These results underscore the importance of maintaining PD as a viable KRT modality for children with AKI and ensuring ongoing institutional training, readiness, and infrastructure to support effective deployment during surges or outbreaks.
Keywords
Introduction
Hemolytic uremic syndrome caused by Shiga toxin-producing Escherichia coli (STEC-HUS) remains a leading cause of severe acute kidney injury (AKI) in previously healthy children. 1 In large outbreaks, up to half of affected children develop oligo-anuric AKI requiring acute kidney replacement therapy (KRT) within a narrow time window. 2 Early aggressive intravascular volume expansion has been associated with improved neurologic and kidney outcomes, likely through preservation of cerebral and renal perfusion with mitigation of microangiopathic injury.3–6
As such, acute KRT in STEC-HUS must balance solute clearance and acidosis control with the need to avoid net intravascular volume loss and maintain intravascular volume expansion. Acute peritoneal dialysis (PD) in children is well suited to achieve this goal as it provides slow, predominantly diffusive clearance with little or no net ultrafiltration. 7 Current ISPD guidance for pediatric acute kidney injury supports PD as a first-line modality on both physiologic and practical grounds, particularly in younger children and in settings where rapid deployment is needed. 8
In September 2023 a large outbreak of Shiga toxin-producing
Methods and setting
Southern Alberta, Canada, experiences among the highest reported incidences of STEC infections and STEC-HUS in North America.
9
The region is served by a single tertiary pediatric center, the Alberta Children's Hospital, which manages several STEC cases annually, some requiring acute KRT. However, in September 2023 an unprecedented outbreak occurred, affecting almost exclusively children younger than 5 years, representing the most vulnerable population. The point source was traced to a central kitchen supplying multiple daycare centers.
The institutional clinical pathway for STEC-HUS prioritized early PD access placement in anuric patients with central venous access for supportive therapy, liberal administration of packed red blood cells, and avoidance of unnecessary platelet transfusions. Catheter placement was performed by designated pediatric urologists using a standardized technique including routine omentectomy with additional suturing and meticulous fascial and peritoneal sealing to minimize early dialysate leakage. Following each insertion, a structured handover occurred between the surgeon and nephrology team to jointly determine the timing of dialysis initiation, typically within 12–24 h of placement. PD was initiated using low initial dwell volumes (approximately 10 mL/kg) with short dwell times and supine positioning for the first 24–48 h. Dwell volumes were escalated gradually every 1–2 days, to a maximum of approximately 30 mL/kg if clinically indicated; escalation was not automatic, and the protocol emphasized maintaining the lowest effective dwell volume required to achieve adequate metabolic and acid–base control. Ultrafiltration targets were adjusted primarily by modifying dialysate glucose concentration rather than by increasing dwell volumes.
Consistent with institutional protocol informed by ISPD acute PD guidance, perioperative prophylactic intravenous antibiotics were administered in the operating room and subsequently added to the dialysate for 48–72 h, along with intraperitoneal heparin. Peritoneal effluent cell counts and cultures were obtained at PD initiation and then daily, together with serum inflammatory markers, to support early detection of infectious complications. Dialysis is routinely delivered on general pediatric wards to preserve PICU capacity and leverage pre-trained ward nursing staff. Extracorporeal CKRT capability is available and the preferred KRT modality in the PICU. During the outbreak, clinical parameters were recorded prospectively, and renal recovery was assessed 3 months postdischarge using the Schwartz eGFR equation. 10 This report represents a review of institutional outbreak response and is part of quality improvement activities using de-identified aggregate data.
Results
Starting on Day 3 of the outbreak, more than 200 children presented with acute gastroenteritis with hematochezia over a brief period, overwhelming emergency and inpatient services. Twenty-seven children developed findings consistent with thrombotic microangiopathy (TMA), and 21 fulfilled all diagnostic criteria for STEC-HUS (hematocrit <30%, platelet count <150 × 103/mm3, and creatinine greater than the upper limit of normal for age). Nine progressed to oligo-anuria requiring urgent peritoneal catheters placement for acute PD. Four catheters were inserted during a single 24-h period (between outbreak days 8–9) at Alberta Children's Hospital (Figure 1). There were no delays, and no therapy was deferred due to access or staffing limitations. During the peak surge, PD cyclers and dialysate solution bags were mobilized from adult hospitals to meet escalating demand. Pediatric PD swan-neck curled catheter supplies reached critically low levels but coordinated redistribution from other regional centers prevented treatment delays. Mean PD duration was 10 days (range 4–16). Three patients required temporary PICU transfers for nonrenal complications (neurologic monitoring and management of systemic complications) while PD continued uninterrupted. All patients achieved adequate uremic control and acidosis correction, with net ultrafiltration intentionally limited to preserve intravascular expansion. One patient developed culture-negative peritonitis on day 4 of PD, presenting with shortness of breath, bilateral pleural effusions, and cloudy peritoneal effluent. Intraperitoneal antibiotics were initiated promptly. Peritoneal fluid cultures remained negative, and the patient demonstrated progressive clinical improvement with decreasing effluent cell counts and renewed urine output over the subsequent days. PD was discontinued on day 11 and the catheter was removed on day 12, with a plan to complete intravenous antibiotics. Approximately three days after catheter removal, the patient developed persistent abdominal pain and was found to have pneumoperitoneum on imaging. Exploratory laparotomy identified bowel perforation requiring partial colectomy and diverting ileostomy. There were no early dialysate leaks, catheter malfunctions, or tunnel infections observed in the cohort.
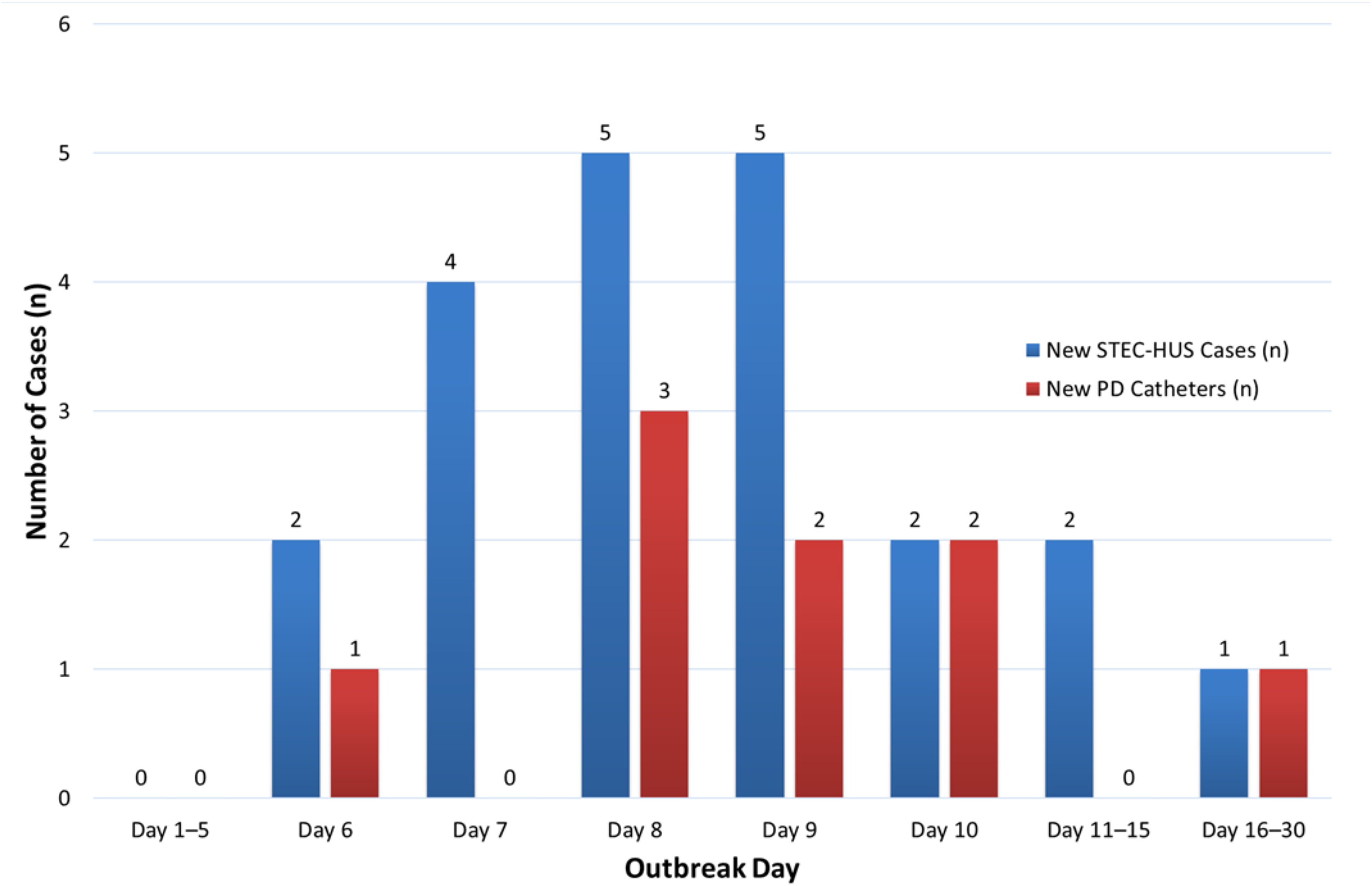
Timeline of new STEC-HUS cases and PD catheter insertions.
Two children experienced significant neurologic events during the acute illness. One developed seizures on day 6 of illness, prior to PD catheter insertion, followed by fluctuating level of consciousness and abnormal neuroimaging (multifocal basal ganglia/thalamic/brainstem involvement) and epileptiform discharges on EEG. At approximately 3 months follow-up, neuro-irritability and hyperkinetic movements had resolved; the child had returned to baseline cognition and social function, with improving mild residual motor asymmetry and a slightly wide-based gait.
A second child experienced a prolonged generalized tonic-clonic seizure requiring a brief intubation/PICU admission while on PD. Brain imaging remained normal and EEG demonstrated rare epileptiform discharges with mild–moderate background slowing. At approximately 3 months follow-up, neurologic examination was normal, no further seizures were reported, and antiseizure medication weaning was initiated. There were no deaths, and all patients recovered kidney function before discharge, with PD catheters removed prior to hospital dismissal. At 3-month follow-up, all had eGFR >90 mL/min/1.73 m2 (range 92–154).
Discussion
This report demonstrates the utility of urgent-start PD during a surge of oligo-anuric AKI in young children with STEC-HUS. In this scenario, PD enabled rapid service scaling to meet unprecedented demand without overwhelming PICU capacity, while providing physiologically appropriate management that maintained protective volume expansion while achieving metabolic and acid-base control.
Although extracorporeal modalities such as continuous kidney replacement therapy and intermittent hemodialysis are increasingly favored in many centers due to improved technology and staff expertise, these approaches have significant limitations during surge conditions.11,12 Requirement of specialized equipment, large-caliber vascular access, and systemic anticoagulation in young thrombocytopenic children may increase risk and pose logistical challenges.7,13 Moreover, rapid intravascular volume shifts associated with extracorporeal therapies may increase vulnerability to cerebral and renal hypoperfusion in children with TMA. 14 In contrast, PD permits solute and acid-base correction with minimal fluid removal, preserving intravascular volume status and potentially reducing risk for perfusion compromise.8,11 Some observational series have reported associations between extracorporeal therapies and adverse outcomes, although these findings are highly susceptible to confounding by illness severity and treatment selection bias. 15
Neurologic involvement remains a key determinant of morbidity in STEC-HUS and has been reported in 17–34% of children with STEC-HUS overall. 16 In our cohort, two children experienced major neurologic events with encouraging early follow-up: one child with imaging-confirmed neuro-HUS demonstrated substantial recovery by 3 months with improving mild residual motor findings, while the second had normal neuroimaging and a normal neurologic examination at follow-up after a prolonged seizure early in the illness. Notably, in the child with neuro-HUS, neurologic manifestations preceded the initiation of KRT, underscoring that neurologic complications may evolve independently of dialysis modality and reinforcing the importance of hemodynamic strategies aimed at preserving perfusion.
The safety profile of urgent-start PD in STEC-HUS must be interpreted cautiously in light of prior reports describing peritonitis rates of 19–24% in pediatric cohorts. 17 In our cohort, one patient developed culture-negative peritonitis that responded to intraperitoneal antibiotics, followed by delayed bowel perforation several days after PD discontinuation and catheter removal. While bowel perforation is an uncommon complication of acute PD in children, it has been reported in large pediatric STEC-HUS cohorts and represents a serious adverse event requiring prompt surgical management. 16 The delayed timing after PD cessation suggests that the perforation may have reflected underlying bowel vulnerability related to systemic illness, ischemic injury, or prior inflammation rather than direct catheter-related injury, although a contributory role of recent intraperitoneal instrumentation cannot be fully excluded. No additional patients experienced infectious or mechanical PD complications, and there were no early leaks, tunnel infections, or catheter malfunctions.
The mean duration of KRT in our cohort was 10 days (range 4–16). Direct comparisons of KRT duration across dialysis modalities in pediatric STEC-HUS are limited, as modality selection is often driven by institutional practice, patient size, and illness severity rather than randomized allocation. 18 However, published pediatric STEC-HUS series typically report dialysis courses on the order of 1–2 weeks in children with oligo-anuric disease, suggesting that the duration observed in our cohort is not atypical. 17 Length of hospitalization in severe STEC-HUS is frequently driven by systemic complications and recovery trajectory rather than dialysis modality alone.
A critical factor in achieving successful short-term outcomes in this outbreak was the logistical framework that enabled rapid and effective deployment of PD. Ward-based safety was supported by a well-established institutional care model for pediatric PD. All nephrology patients were admitted to a designated inpatient unit staffed by core nursing personnel who maintain ongoing competency in manual PD through a structured skills maintenance program, with a subset additionally trained in cycler-based therapies. Each shift included PD-trained nursing staff, and children with acute STEC-HUS receiving PD were routinely assigned 1:1 nursing. Medical care was provided by an in-house hospitalist team with 24/7 coverage, with continuous oversight from an on-call pediatric nephrologist. Standardized protocols, bedside checklists, and established escalation pathways for mechanical, infectious, or hemodynamic concerns supported consistent care delivery during surge conditions and minimized the need for ad hoc retraining.
Placement of seven Tenckhoff catheters within four days followed by prompt initiation of dialysis was made possible by a coordinated, system-level response that included continuous access to pediatric urologists skilled in catheter insertion, established nursing education, protocols for urgent-start PD, and a nephrology team available to safely supervise ward-based dialysis.
Taken together, this experience demonstrates that PD is not only a viable alternative to extracorporeal kidney replacement therapy (KRT) in resource-limited environments but also represents a feasible first-line option in tertiary pediatric centers during surge conditions.
Conclusions
Urgent-start PD, delivered predominantly on general pediatric wards, provided adequate solute and acid–base management for nine consecutive STEC-HUS patients requiring KRT during a large Calgary outbreak. This approach reduced reliance on ICU-based CRRT, need for large-bore vascular access and systemic anticoagulation, while preserving the intravascular expansion strategy associated with improved neurologic and renal outcomes in STEC-HUS. Nevertheless, serious complications may occur, requiring expertise, protocols and vigilant monitoring. Therefore, pediatric centers should maintain operational readiness for urgent-start PD, including trained nursing staff, timely access to skilled surgical catheter insertion, and ward-based PD protocols, as part of outbreak preparedness.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This report represents a review of institutional outbreak response and is part of quality improvement activities using de-identified aggregate data.
Informed consent to participate
Not applicable.
Informed consent to publish
Not applicable.
Acknowledgments
None.
Authorship
All authors made a substantial contribution to data acquisition. Al Dhawi, Samuel, and Grisaru drafted the manuscript. Hamiwka, Wade, Midgley, and Archibald revised it critically for important intellectual content. All authors approved the final version.
Trial registration
Not applicable.
