Abstract
This case report describes a 66-year-old male on continuous cycling peritoneal dialysis (PD) with polycythemia vera and type 2 diabetes. He presented with culture-negative PD-associated peritonitis secondary to splenic infarcts and further accompanied by a splenic vein thrombosis and posterior brain circulation infarcts. His abdominal pain was atypical for peritonitis, being mild and localized to the left side, with an unremitting course despite several treatment attempts with appropriate antimicrobial coverage. An extensive workup for thromboembolic causes was unremarkable. Initially, the patient was started on aspirin and later treated with hydroxyurea and long-term warfarin. His PD catheter was removed due to concerns about an underlying biofilm, and a new one was inserted one month later, while on temporary hemodialysis, without recurrence. This case highlights that non-infectious, culture-negative PD peritonitis related to splenic infarction should be considered in patients with left-sided abdominal pain, poor clinical response to appropriate antibiotics and significant risk factors for thromboembolic events, such as hematologic disorders like polycythemia vera and splenomegaly. Maintaining a high clinical suspicion can prevent unnecessary antibiotic use and reduce repeated exposure to intravenous contrast for imaging studies. Early initiation of long-term anticoagulation might also prevent futile PD catheter removal if subsequent clinical improvement is obtained.
Keywords
Introduction
Peritoneal dialysis-associated peritonitis (PD peritonitis) is a well-recognized complication of PD with significant morbidity and mortality. Most cases are infectious and resolve with an appropriate antimicrobial regimen. However, cultures remain negative in 13.4–40% of cases 1 and some of these cases are non-infectious such as chemical peritonitis, dialysate contamination or reaction to an intraperitoneal (IP) or retroperitoneal process. 2 This case report discusses a rare cause of the latter, namely culture-negative peritonitis caused by splenic infarcts.
Narrative
A 66-year-old man with type 2 diabetes, polycythemia vera with prior phlebotomies, gout, and kidney failure secondary to diabetic nephropathy on continuous cycling PD, was initially admitted to a remote hospital with a two-day history of mild abdominal pain and bloody dialysate. His dialysate cell count showed 1833/μL white blood cells (WBCs) with 86% neutrophils. Mild leukocytosis and chronic mild thrombocytosis with a hematocrit of 0.44 were noted. This first dialysate bacterial culture returned negative. IP tobramycin 1.7 mg/kg and IP vancomycin 30 mg/kg were administered through a 6-h dwell. IP heparin was administered until the hemoperitoneum resolved. An abdominal CT scan with contrast showed massive splenomegaly and splenic infarcts, as depicted in Figure 1. In consultation with hematology, initiation of anticoagulation was deemed unnecessary. His cell count came down to 216/μL with 83% neutrophils within 48 h and continued to improve to 140/μL when checked again on the fifth day of treatment. He was discharged with a plan for 14 days of IP vancomycin and without residual abdominal pain.
He returned to the hospital eight days after the initial presentation with confusion and localized left-sided mild abdominal pain. His dialysate cell count was compatible with peritonitis (5710/μL nucleated cells with 84% neutrophils). Unfortunately, he missed his last dose of outpatient IP vancomycin. In the hospital, he received another course of empiric IP tobramycin and IP vancomycin as well as fluconazole prophylaxis. Both bacterial and fungal dialysate cultures remained negative. A repeat abdominal CT with contrast showed progression of the splenic infarcts and a new splenic vein thrombosis. Multifocal left-sided subacute posterior circulation infarcts were found on brain magnetic resonance imaging. Aspirin 81 mg daily was started. He experienced complete resolution of his abdominal pain within 48 h. He was discharged with IP vancomycin and fluconazole prophylaxis for a total of 14 days with a decreasing cell count to 370/μL with 83% neutrophils ten days later. Unfortunately, no other intercurrent dialysate cell count was available. The patient was advised to follow up through a tertiary care hospital for closer hematologic and renal monitoring, but he was resistant to do so for logistical reasons
One week later, despite completion of a full course of IP vancomycin therapy, he was ultimately admitted to a tertiary hospital with mild ill-defined abdominal pain and a dialysate cell count compatible with peritonitis (1250/μL with 83% neutrophils). He again received an initial dose of both IP vancomycin and IP tobramycin while undergoing investigations. All bacterial, fungal and mycobacterial dialysate cultures remained negative. A CT abdomen with contrast did not identify any new splenic infarcts or progression of his thrombosis. A thromboembolic workup, including an echocardiogram, holter monitor, and thrombophilia testing returned negative. In discussion with hematology, we elected to start the patient on hydroxyurea and anticoagulation with warfarin. His PD catheter was removed due to concerns about an underlying, non-resolving biofilm. After roughly four weeks on hemodialysis, a new PD catheter was inserted and the patient was able to resume PD. He continues on warfarin and has not developed any recurrence of culture-negative peritonitis.
Discussion
Culture-negative peritonitis can be clinically challenging. Most of these episodes are infectious but associated with faulty sampling techniques, ongoing or recent antimicrobial treatment, or unusual organisms. The 2022 International Society for Peritoneal Dialysis (ISPD) guidelines suggest that less than 15% of peritonitis episodes should be culture-negative and superior rates should prompt review of the sampling techniques. 1 Slow-growing fastidious bacteria and yeasts might require subcultures on media with aerobic, anaerobic and microaerophilic incubation conditions for multiple days for identification. Otherwise, non-infectious causes of culture-negative peritonitis should be considered, including include chemical peritonitis (i.e., icodextrin), dialysate contamination (i.e., endotoxins) or an IP or retroperitoneal process such as splenic infarction. 2 In this case, the localized spleen edema and inflammation likely introduced a contiguous IP inflammatory process with the appearance of inflammatory cells with a predominance of neutrophils on dialysate fluid analysis. Similarly, enhanced capillaries permeability from the inflammatory process could favor escape of red blood cells into the dialysate and potentially explain the initial finding of hemoperitoneum in this case as well as some of the reported cases below.
As shown in Table 1, very few cases of culture-negative peritonitis associated with splenic infarction have been reported in the literature.3–6 Four of the six patients were diabetic, but it is unclear if this represents a clinically significant risk factor for splenic infarction although the associated atherosclerosis could have contributed if present in splenic vessels. These cases demonstrated dialysate fluid analyses with a range of WBC counts (244−9089/µL) and neutrophil predominance (70–98%), which was consistent with our case. All patients were initially treated with IP antibiotics covering gram-positive and, in all cases but one, gram-negative coverage while undergoing investigations. After the diagnosis of culture-negative peritonitis secondary to splenic infarction was made, antibiotics were discontinued and anticoagulation with warfarin was initiated. Subsequently, a progressive decrease in dialysate cell counts and resolution of the abdominal pain occurred within a few days in these cases.
Reported cases of culture-negative peritonitis associated with splenic infarction.

This case report describes culture-negative peritonitis associated with splenic infarction in a patient with polycythemia vera and splenomegaly. In our case, expedited management with anticoagulation may have prevented unnecessary exposure to antibiotics and repeated intravenous contrast administration. An important clue was that his abdominal pain was atypical for unremitting infectious peritonitis, being mostly mild and left-sided without rebound tenderness.
The natural history of splenic infarcts typically involves a progressive resolution of the clinical picture over one to two weeks. 7 This correlates to the initially elevated dialysate cell counts and subsequent decreases following the initial bouts of pain and imaging findings of splenic infarction during the first two admissions. In this patient, dialysate cell counts did slightly increase during his last hospitalization despite a stable radiological appearance. Given the patient's hypercoagulable state and splenomegaly, it is possible that he may have developed small infarcts or micro-infarcts, which may not be detectable by a CT scan. Interestingly, fewer than half of all acute splenic infarcts show the typical peripheral wedge-shaped, sharply marginated defect with this imaging modality. 8
Another plausible explanation for the patient's last hospitalization, which cannot be ruled out is the occurrence of an infectious culture-negative peritonitis, considering the significant prior antibiotic exposure. This patient's PD catheter was removed, and if contaminated by biofilm, would have provided adequate source control for an infectious peritonitis. Removing the catheter in such an unremitting course proved to be a safe management decision but perhaps could have been avoided with further observation and repeat dialysate cell counts drawn after anticoagulation initiation, as observed in prior reported cases. Of note, blood cultures were not drawn before admission to the tertiary hospital, considering the absence of fever, though these remained negative as well. It is also worth noting that no vancomycin level was measured during this clinical course. It would have been informative to verify that the patient was not exposed to subtherapeutic dosing, especially given the potential infection hypothesis later on.
Additionally, other hematologic findings of leukocytosis, thrombocytosis and Howell–Joly bodies can be identified with splenic infarction due to functional hyposplenism and represent useful clinical clues.9,10 Our patient had chronically elevated platelets as a confounding factor but no evidence of Howell–Joly bodies. However, he did present with mild peripheral leukocytosis initially. If present, these findings could further justify the decision to avoid additional antibiotic administration.
When faced with splenic infarcts, clinicians should have a systematic approach for major risk factors and causes, which include splenomegaly, hematologic and prothrombotic disorders, septic emboli and thromboembolic cardiac abnormalities such as arrhythmia. 11 In our case, significant risk factors included severe splenomegaly as well as polycythemia vera with a borderline hematocrit level mandating treatment. An extensive diagnostic workup did not reveal any other contributing factors in our patient but should be considered for any patient without an obvious cause. Furthermore, a splenic abscess can also form at the site of a splenic infarct, 12 making the clinical picture even more complex and justifying repeated imaging. Importantly, the presence of a splenic abscess could also cause repeated seeding of the dialysate, leading to a clinical picture of relapsing episodes of acute bacterial peritonitis despite appropriate antimicrobial coverage. 13
Conclusion
In conclusion, culture-negative peritonitis secondary to splenic infarction should be considered in patients presenting with atypical abdominal pain, unremitting clinical course with appropriate antibiotic coverage and significant risk factors for thromboembolic phenomena such as prothrombotic hematologic disorders and splenomegaly. Maintaining a high clinical suspicion could avoid unnecessary exposure to antibiotics and repeated contrast for imaging studies with a risk of residual kidney function loss. Treatment with long-term anticoagulation could also prevent PD catheter removal and its associated morbidity if subsequent clinical improvement is obtained.
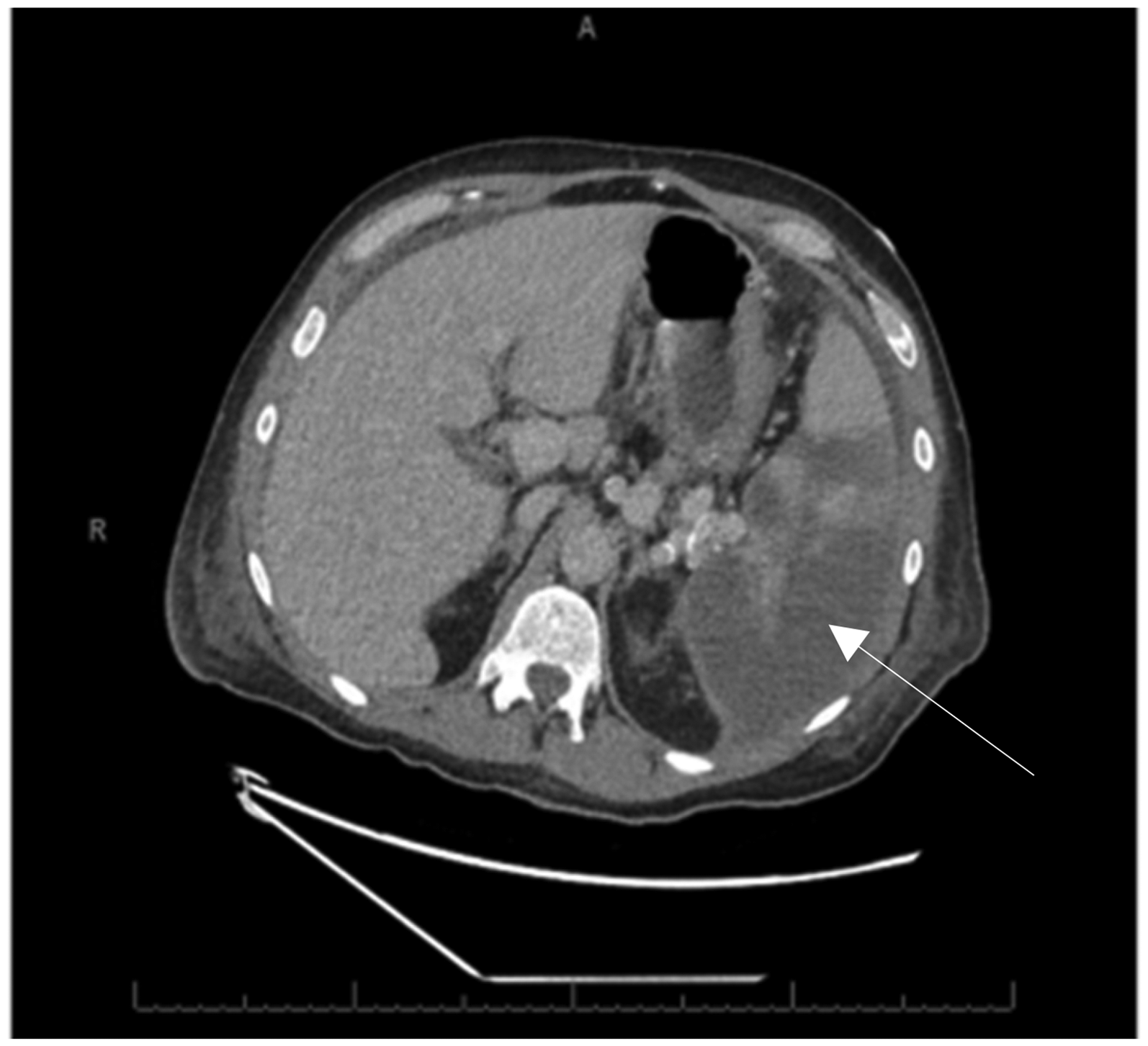
Computed tomography image (axial view) showing a large geographic region of hypoattenuation corresponding to splenic infarcts (white arrow).
Footnotes
Acknowledgments
The author gratefully acknowledges the contribution of Dr Suneet Singh for her mentorship throughout this project and Dr Shannon Wong for her input on content selection and reviewing the manuscript.
Author contributions
RH conceptualized the case report, obtained patient data and authored and reviewed all iterations of the manuscript.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
As this is a single-patient case report, formal ethical approval was not necessary according to our institutional guidelines.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Explicit informed consent for the publication of their details was obtained from the patient.
