Abstract
Background
High-quality and timely peritoneal access is essential for effective peritoneal dialysis (PD). Existing comparisons of medical compared to surgical catheter insertion have focused on the incidence of catheter events, but the cost-effectiveness of providing medical in addition to surgical catheter insertion in a dual pathway, compared to providing surgical insertion alone has not been evaluated.
Methods
Data from the UK Catheter study, exploring how patient, service and insertion technique factors interact was used to estimate the comparative rates of catheter events between medical and surgical catheter insertion. A cost-effectiveness model estimates the health benefits and costs of providing medical in addition to surgical catheter insertion, compared to surgical insertion alone. Parametric modelling estimated time to catheter events, haemodialysis and transplantation to populate the model.
Results
Data on 769 first catheter insertions informs the model (325 medical and 444 surgical). Fewer catheter events were observed with medical insertion. The dual insertion pathway (69% medical, 31% surgical) was therefore associated with lower lifetime catheter events (3.18 vs. 3.34) and longer time on PD (3.07 vs. 3.00 years) than a purely surgical insertion pathway. The lifetime mean differences in quality-adjusted life years (7.12 vs. 7.00) and near identical costs (£226,549 vs. £226,764) meant dual insertion pathway was likely to be cost-effective, a finding robust to a series of sensitivity analyses.
Conclusion
Offering medical in addition to surgical catheter insertion techniques has the potential to improve clinical outcomes and is likely to be highly cost-effective compared to surgical insertion alone.
Keywords
Introduction
High-quality peritoneal access is essential for effective peritoneal dialysis (PD) and in clinical practice it is influenced by multiple factors such as patient and service characteristics, local availability of resources, expertise and experience and other organisational aspects.1–3 Inclusive care pathways require approaches that enable the insertion of timely and effective PD access for individuals who are unsuitable for general anaesthesia, or who have had previous significant abdominal surgery.
Previous research on PD has concentrated on directly comparing the primary function and infective complications between medical techniques (percutaneous, radiological, peritoneoscopic) and surgical approaches (open and laparoscopic) for PD catheter insertion. Meta-analysis of cohort studies in 2018 and 2023 exploring these insertion techniques does not appear to favour a specific technique.4,5 International registries collecting data on catheter insertion techniques and practice patterns have shown wide variations in event rates and catheter survival4–6 potentially due to the use of different endpoints.5–7 However, broader information is required to inform the design of a clinical service in contemporary kidney units. Medical catheter insertion brings potential benefits in addition to surgical insertion: it does not require a general anaesthetic, potentially widening the population suitable for PD and increasing responsiveness as insertion is less reliant on other resources (pre-operative assessment, operating theatres and hospital bed days). However, in cases where medical insertion is unsuccessful or where patient-specific factors require advanced surgical techniques during the insertion procedure, provision of medical catheter insertion alone may not be suitable. Provision of both medical and surgical insertion methods is therefore commonplace. 8 Factors for designing the best PD catheter insertion pathway in the UK Cath study have previously been reported. 9 Out of 784 first catheter insertions, 59% had adverse catheter–related events (catheter removal, catheter-related hospitalisation and infection) in the first year. Following adjustment for potential confounders, medical insertion had a 0.70 hazard ratio versus surgical for catheter events after insertion (95% confidence interval [CI] 0.43–1.13). Once established on PD the hazard ratio became statistically significant and was 0.77 (95% CI: 0.62–0.96). 6
Advocates for person-centered care, policy makers and as a consequence guidelines, emphasise the promotion of home dialysis, including PD.10,11 This is supported by studies showing that PD can offer health outcomes that are as good as or even better than other forms of dialysis in terms of quality of life and patient survival, while also being cost-effective.11–14 However, the cost-effectiveness of offering medical in addition to surgical insertion options as part of a PD service compared to a surgical-only service is unknown. This study develops a cost-effectiveness model and uses data from the UK Cath study to fill this evidence gap. Estimates of the lifetime health benefits and costs could assist policy makers, mangers and clinicians in the decision to invest in a dual catheter service.
Methods
This cost-effectiveness study is reported in accordance with the Consolidated Health Economic Evaluation Reporting Standards 2022 (CHEERS 2022) statement. 15
Target population and setting
UK Cath was a prospective multi-centre cohort study of incident PD patients at first catheter insertion conducted in 44 of the 72 dialysis centres in the UK, 20 of which were simultaneously participating in the Peritoneal Dialysis Outcomes and Practice Patterns Study (PDOPPS).9,16 The PDOPPS is an international prospective cohort study designed to identify optimal practices for people treated with maintenance PD. The UK Cath protocol was published in 2017, prior to study closure and analysis. 9 Individuals aged 18 or older with irreversible kidney failure who intended to initiate PD as their initial treatment modality and had plans for PD catheter insertion within the following 30 days were given the opportunity to participate in the study by providing consent. Exclusions were made for patients who had already initiated PD, had their PD catheter inserted at a different healthcare facility, or were incapable of providing informed consent. Recruitment began in July 2015 and concluded at the end of December 2017, with data collection ending in April 2019. All subjects received a patient information sheet and gave written consent. Patient partners were involved at each stage of the research process including the design of patient-facing questionnaires.
Cost-effectiveness model
We constructed a decision model to evaluate the costs and health benefits associated with two distinct service approaches for providing PD. One approach (dual pathway) involves two possible PD catheter insertion techniques: medical (requiring percutaneous, radiological, peritoneoscopic techniques) or surgical (requiring open surgical, laparoscopic techniques) while the other approach (surgical pathway) exclusively insert PD catheters surgically. Health benefits are expressed in terms of Quality Adjusted Life Years (QALYs). Costs focus on those that are incurred by the health care system.
The model was developed in Simul8 (Simul8 Corporation, Boston, US) and is summarised in Figure 1. A summary of the key features is provided here with a full account of assumptions made provided in Supplemental Table 1. Patients, representative of those observed in UK Cath, are tracked, one at a time, through the simplified treatment pathway represented by the model. The characteristics of each patient on entering the model and their subsequent treatment experience impact the route and time taken through the various health states the model describes. Patients’ characteristics are age (in 5-year increments from 20 to 90), sex and comorbidities (diabetes, atherosclerotic cardiovascular disease, other cardiovascular disease and cancer). Each patient is simulated having their PD catheter inserted in a service with a dual pathway and again in the service which offers only surgical insertion of PD catheters. A portion of patients (31%) undergo surgical catheter insertion in the dual-pathway simulation. The outcomes for an individual receiving a surgically inserted catheter in the dual-pathway simulation are identical to the outcome for that individual in the simulation of the service exclusively offering surgical insertion for PD catheters. A large number of patients are tracked over a lifetime and the mean costs and health benefits then calculated. The characteristics that inform each patient's pathway through model health states are sampled from incident dialysis patients in the renal registry.
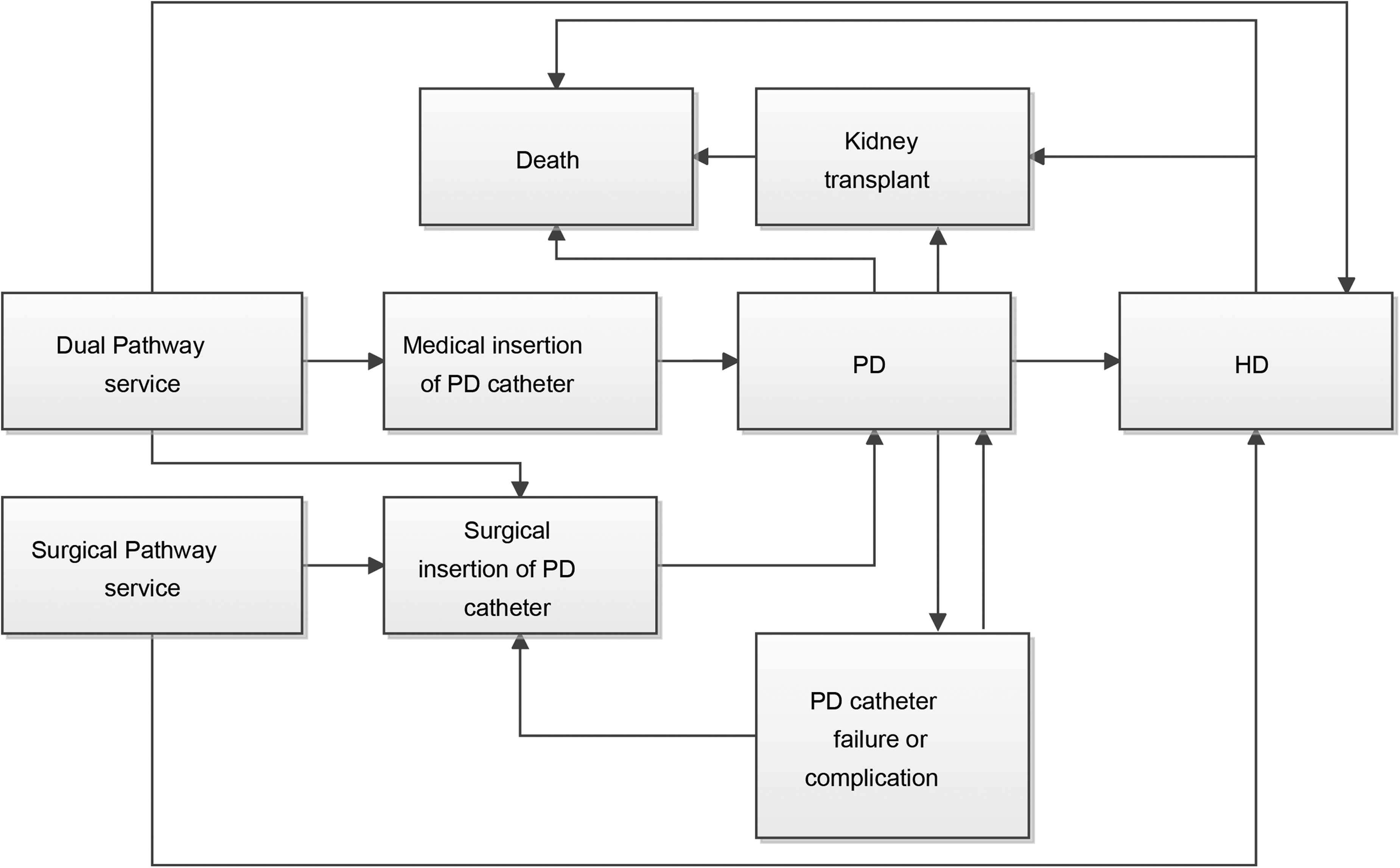
Health states and possible transitions in the health economic model. All transitions informed by UK Cath study data, with the exception of those into death (informed by age/sex life-tables from USRDS data).
The model first assigns the sampled patient to a PD catheter insertion modality, conditional on the service type, with the patient in the surgical pathway always receiving their PD catheter surgically. At the time of PD catheter insertion, the model then checks if there will be a catheter failure or a catheter-related event in the first 6 weeks based on the probability of failure in the study data. If there is a catheter event, the time at which it occurred is set based on the mean time to a catheter event in the study data conditional on patient characteristics. The model makes a distinction between catheter-related event occurring in the first 6 weeks (primary failure) and those occurring after 6 weeks (secondary failure) to allow for any differences in types of events or the management of events in the early compared to later post-catheter insertion period (Supplemental Table 1).
Patients who do not encounter primary catheter failure remain on PD until they experience a catheter event beyond 6 weeks, transition to haemodialysis (HD) without experiencing a catheter event, receive a kidney transplant or die. Informed by the study data, transition to HD also occurs if the patient experiences three catheter failures, three instances of PD-associated peritonitis (PD peritonitis), five exit-site infections requiring antibiotics or three hospitalisations due to complications (informed by clinical opinion). Once patients initiate HD, they cannot move back to PD, therefore, any subsequent catheter events (failure or catheter complications) will lead to reestablishing the patient on haemodialysis. The UK Cath Study did not provide a means to reliably estimate this specific probability. All catheter reinsertions are performed surgically. Catheter-related events incur both health service costs and reductions in quality of life, based on the UK Cath study data.
The times at which each individual patient will receive a transplant (if at all), or die, are both calculated at the beginning of the model. For those patients that receive a transplant, time to mortality is adjusted to reflect its mortality benefit. The times for entry into these health states (catheter failure, death, PD, HD, kidney transplant) were estimated using parametric survival models including patient characteristics as explanatory variable. The time to catheter events is also a function of the insertion technique (medical or surgical). Separate models were fitted for the duration of PD following second and subsequent catheters, reflecting that these catheters have worse outcomes. We present adjusted Cox model results using the same predictors as our previously published analysis of UK Cath, to allow for a direct comparison between analyses.
Health utilities were assigned to specific health states. The SF-12, patient-reported outcome, was administered as part of UK Cath. Patients in different modality states: PD, HD, transplant (received within one year) and transplant (received one year or longer) – responded to this quality-of-life instrument. These states align with the health states in the model and their responses were transformed into EQ5D-3L utility scores. 17 Both costs and benefits were discounted at a 3.5% annual rate. These costs are calculated for each patient based on the average annual cost of each event state, adjusted for the duration of an event. NHS Reference Costs were used as the source for the cost of dialysis treatment, hospitalisation for catheter events and catheter insertion costs, stratified based on whether the insertion was medical or surgical. NHS reference costs are published by the NHS and represent the average expenses incurred by the NHS for specific services within a fiscal year. This encompasses all aspects of NHS expenditure associated with service delivery, covering direct treatment costs, facility overheads, staffing expenses and patients’ hospital stays. It does not include patient-borne costs. Therefore, from the cost perspective, we consider only the expenses borne by the health service and exclude patient out-of-pocket expenditures and indirect costs, such as loss of earnings when travelling to a hospital appointment. When unit costs were not available in the 2018/2019 NHS reference costs, all expenses were adjusted to 2018/2019 prices using the NHS Cost Inflation Index Pay and Prices from the Unit Costs of Health and Social Care 2020 publication. 18 A detailed list of parameters, assumptions and information sources can be found in Supplemental Table 2. All events have EQ5D-3L utility scores, with sex and age also influencing health utility.
Uncertainty in the inputs to the model is reflected using standard methods. Probabilistic sensitivity analysis is used to demonstrate the impact of the combined uncertainty in model inputs. We present the probability that each service delivery type is cost-effective, across a range of values for a QALY that decision makers may find acceptable. The graphical representation is called the Cost-Effectiveness Acceptability Curve (CEAC). We also conducted a series of sensitivity analyses, varying inputs within their 95% CIs.
Results
Following consent of 837 patients, there were 784 first catheter insertions. Catheter insertion technique was missing for 15 participants, leaving 769 individuals to inform the model. The characteristics of this population, taken from the UK Cath study, are displayed in Table 1. A total of 325 (42%) patients received medical insertions and 444 (58%) received surgical insertions; 399 (61%) white patients had surgical insertions compared to 40 (39%) those of non-white groups 116 (70.1%) of those with a history of abdominal, genitourinary or gastrointestinal surgery had a surgical insertion compared to 328 (54.7%) of those who had not. Twenty-four participants had congestive heart failure (8.1%) in medical catheter insertions and nineteen (4.7%) of those receiving surgical insertions, but other co-morbidities including diabetes were equally distributed between medical and surgical insertion techniques.
Patient baseline characteristics by catheter insertion technique.
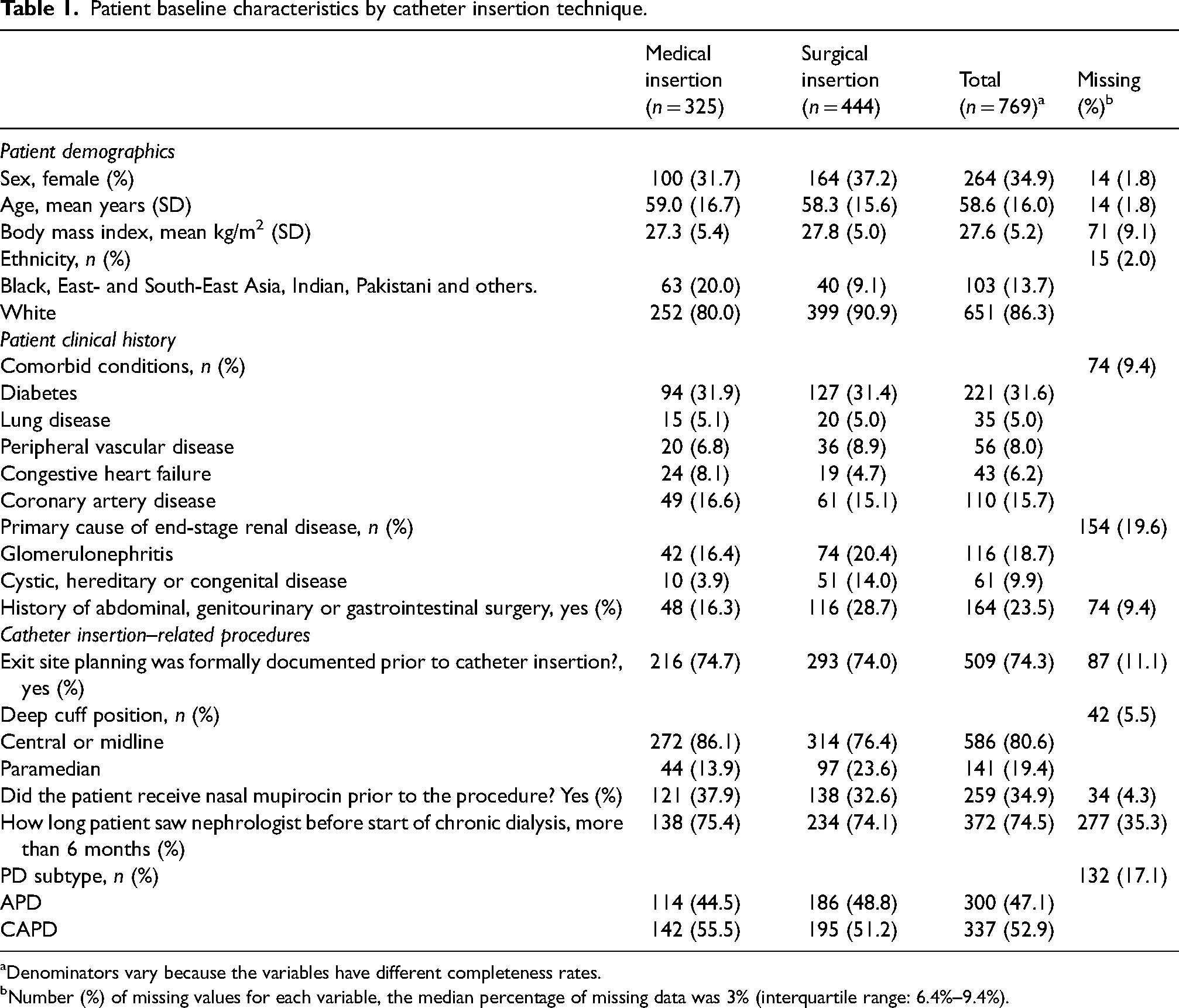
Denominators vary because the variables have different completeness rates.
Number (%) of missing values for each variable, the median percentage of missing data was 3% (interquartile range: 6.4%–9.4%).
The time from catheter insertion to catheter event was shorter in those receiving surgical catheter insertions, illustrated without adjustment in Figure 2. For comparison to our primary endpoint paper, the adjusted hazard ratio for experiencing a catheter-related event for medical insertion compared to surgical catheter insertion estimated from the Cox model of 0.796 (95% CI: 0.648–0.978, p = 0.03). When sampling from the distributions of medical and surgical catheter insertion for the population recruited into the study, time to catheter event was a mean of 2.6 months shorter with surgical insertion compared to medical insertion and was shorter with surgical insertion in 65% of samples.
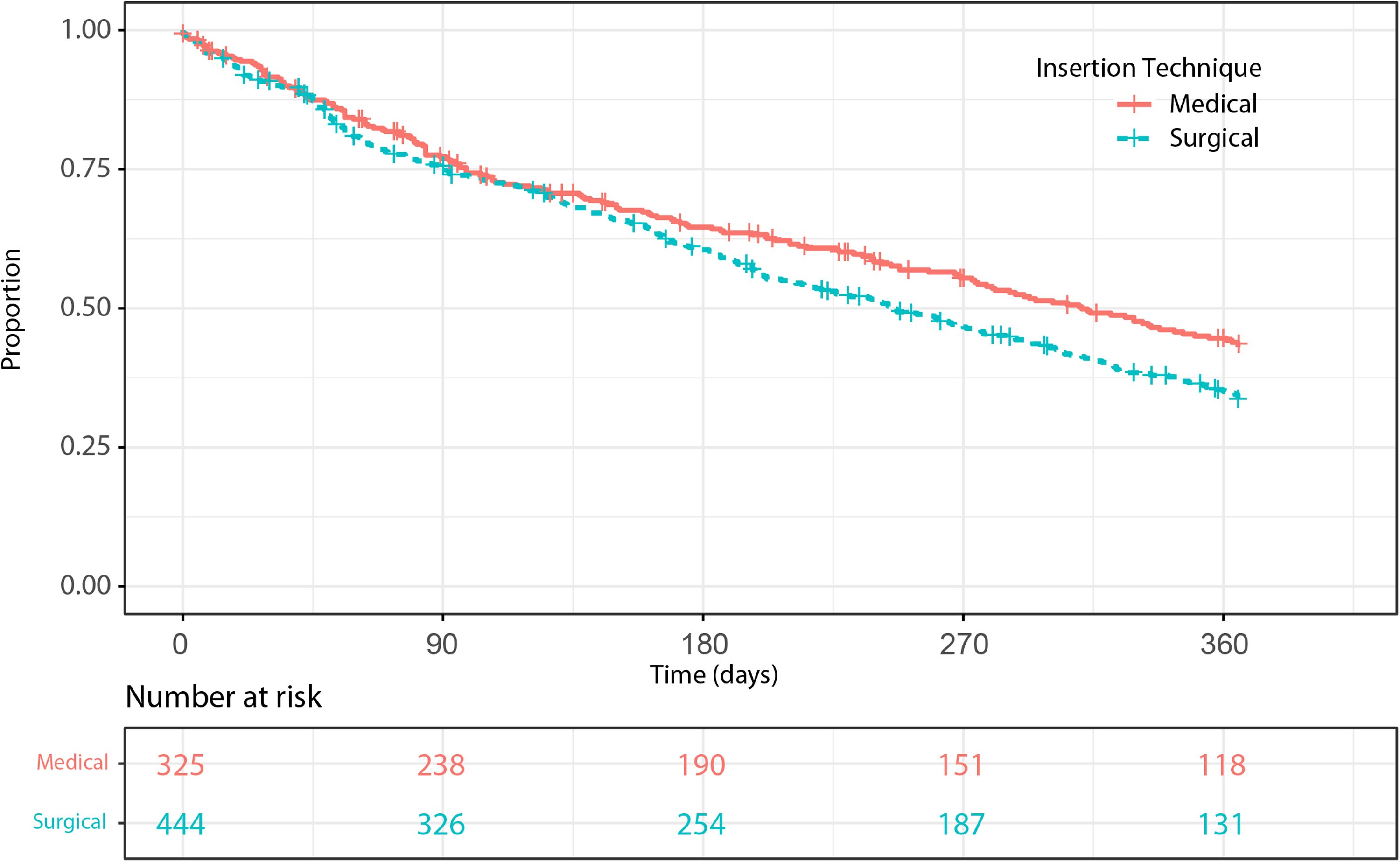
Kaplan–Meier graph of catheter event-free survival, stratified by medical and surgical catheter insertion. Catheter event defined as further operative procedures or catheter removal (as a consequence of dialysis fluid leak, hernia, poor or absence of flow, catheter displacement, bleeding, catheter-related pain, exit-site infection, tunnel infection, wound infection, PD peritonitis – supplemental item Catheter Event Worksheet), catheter-related infections not requiring a procedure or hospital admission for catheter-related problems.
Duration of treatments and the patient journal through the health states, by catheter insertion pathway
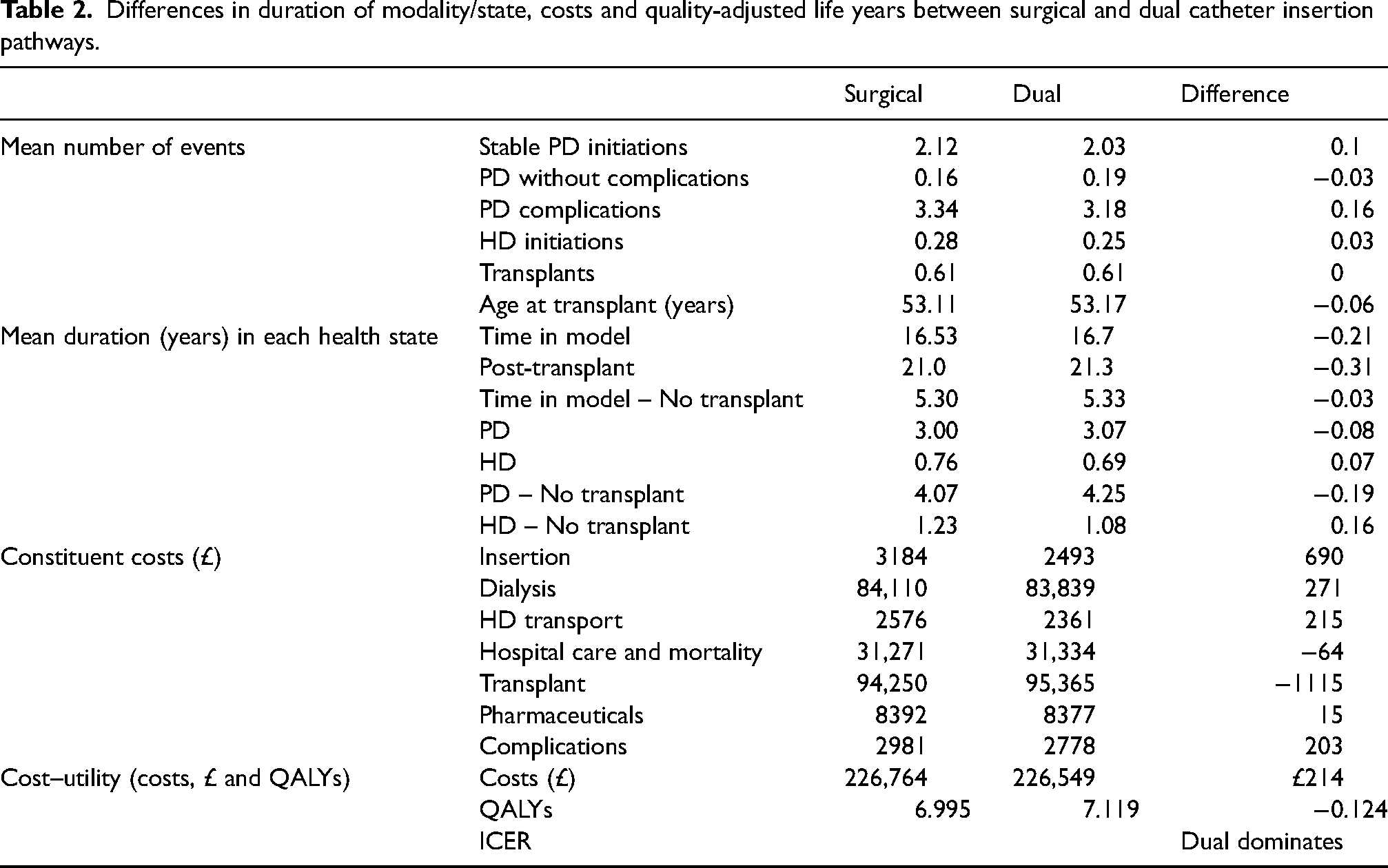
In services offering a dual insertion pathway, 69% of catheters were inserted medically and 31% surgically. Due to the lower catheter event rate associated with the medical catheter insertion technique, the patient journey though the model and the associated number of events and duration of treatment modalities differed between catheter insertion pathways (Table 2). Dual pathways (medical in addition to surgical) had fewer PD complications and initiations on HD, and longer time on PD (in those transplanted and those not transplanted).
Differences in duration of modality/state, costs and quality-adjusted life years between surgical and dual catheter insertion pathways.
Cost-effectiveness of medical in addition to surgical catheter insertion (dual pathway) compared to surgical-only catheter insertion (surgical pathway)
Results, reported in Table 2, show that, in the base case analysis the dual (medical and surgical) catheter pathway set-up dominates the surgical approach (i.e., is both cost-saving and QALY gaining). The availability of both insertion techniques results in an average gain of 0.12 QALYs over a lifetime and marginally lower mean lifetime costs of £214. This difference is approximately 0.1% of the mean overall costs borne by patients throughout their kidney care journey. More detail on the source of these cost differences is provided in the constituent costs in Table 2.
The CEAC is shown in Supplemental Figure 1 for values of the maximum acceptable cost per QALY gained between £0 and £50,000. The CEAC reveals that when the maximum willingness to pay for a QALY reaches or exceeds £10,000, the probability of the dual pathway option being cost-effective surpasses 99%. The dual pathway can be considered cost effective at all conventional decision maker thresholds for the value of a QALY.
There is a 60% probability that services offering only surgical insertion are the least costly option. However, there is little uncertainty that services offering dual catheter insertion pathway generate greater health gains. Varying parameters within the ranges of their upper and lower 95% CIs in a series of one-way sensitivity analyses did not change these general conclusions.
Discussion
This cost-effectiveness analysis, informed by a prospective multi-service cohort study, estimated increased QALYs and reduced costs associated with the dialysis unit practice of offering both medical and surgical PD catheter insertion techniques in a dual pathway when compared with surgical insertion alone. This dual pathway approach recognises that a service which leads with medical insertion still requires surgical insertions for individuals with complex intra-abdominal circumstances or where medical catheters have not been successful. In performing this lifetime assessment of QALYs, we also report contemporary data on the patient journey through kidney replacement therapy. These results complement the findings of the comparative clinical outcome effectiveness analyses published from this study, which demonstrate similar hazard ratios. This alignment between the economic model and the comparative effectiveness analyses adds credibility to the overall study findings. 7
Many authors compare the relative cost-effectiveness of PD to HD, but to our knowledge analyses of medical compared to surgical catheter insertion techniques are limited to individual procedure costs and do not recognise the use of both pathways when providing medical alongside surgical insertion. There were not sufficient observed catheter insertion subtypes (e.g., laparoscopic insertion) to assess the cost-effectiveness of these specific techniques in our model. However, there is a potential decrease in the cost-effectiveness of the dual pathway if all surgical services transitioned to a 100% laparoscopic technique: two studies have shown that laparoscopic PD catheter insertion was a more cost-effective option than open insertion, despite higher initial costs.19,20 Our primary data analyses on utilities (health-related quality of life) fall within the range identified in recent systematic reviews,14,21 with relative differences in health-related quality of life between modalities aligning with other studies using the same or similar instruments.
Our study compares real-world service types rather than insertion techniques per se. It draws on primary data collection to inform the pathways patients take and the impact of these differing journeys on health outcomes and resource use. Limitations of our study include the lack of randomised controlled data to support our comparative effectiveness assessments. Our assessment properly adopts a lifetime horizon for costs and health benefits but, of course, no study can fully inform this timescale. Our estimates still required extrapolation and incorporation of external evidence, for example, relating to mortality rates. Our assessment is based on evidence from the UK NHS healthcare system and caution is required in extrapolating results to other settings. One limitation of the model is that kidney transplants last the lifetime of the patient, so patients cannot return to dialysis if the transplant fails. This likely favours scenarios where transplants are more common (dual pathway) but does not accurately reflect reality.
This study indicates that a service design that incorporates the capability for medical insertion is cost-effective, which is a finding that builds upon the clinical efficacy demonstrated in our previous study. 6 Our evidence suggests a dual pathway will assist services in achieving home therapy rate targets set by national policies in a range of geographies, and the size of this impact may be underestimated in our model as it does not explicitly model broader access to PD enabled by reduced dependence on pre-operative assessment, operative lists and general anaesthetics or enabling greater access to PD for those who present in an unplanned fashion. No insertion technique is perfect, with medical and surgical having their own cause-specific complication rates driving our composite catheter event definition, and understanding the drivers of these to further maximise clinical and cost-effectiveness should be a priority for the PD access community.
Our analysis contributes to the accumulating evidence that the proportion of people with kidney failure already accessing PD can stay on this therapy for longer in services offering both insertion techniques. This dual pathway approach also enables sites to offer PD catheter insertion that is tailored to the patient's specific requirements, while achieving similar or better outcomes and as similar or less cost.
Authors’ note
Methods: Study Sponsorship and Ethical Approval: This study was sponsored by Sheffield Teaching Hospitals NHS Foundation Trust and Ethical Committee approval for the study was granted by the National Research Ethics Committee London – City and East in December 2013 (Ref: 13/LO/1943).
Supplemental Material
sj-docx-1-ptd-10.1177_08968608251314976 - Supplemental material for The clinical implications and cost-effectiveness of the provision of medical in addition to surgical catheter insertion for peritoneal dialysis in people with kidney failure
Supplemental material, sj-docx-1-ptd-10.1177_08968608251314976 for The clinical implications and cost-effectiveness of the provision of medical in addition to surgical catheter insertion for peritoneal dialysis in people with kidney failure by Harry Hill, Andrew Rawdin, Allan Wailoo, Victoria Briggs, Mark Lambie, Keith McCullough, Louese Dunn, Simon Davies, Martin Wilkie and James Fotheringham in Peritoneal Dialysis International
Footnotes
Acknowledgements
The authors would like to acknowledge the support of the National Institute for Health Research Clinical Research Network research staff who supported the study, and the members of the steering group: Mr Badri Shrestha, Dr Sarah Jenkins, Yvonne Jackson, Dr Lin Wang, Stephanie MacNeill, Prof Fergus Caskey, Professor Helen Hurst, Dr Nana Theodorou, Jodi Paget and Diane Palframan. The authors would like to thank Cheryl Bailey, Jean Winterbottom, Alina Andras, Dr John Belcher, Dr Ivonne Solis-Trapala and Dr Louise Philips-Derby for support with the conduct of the research and management of the associated data, and Karen Chambers and Jennifer King who coordinated DOPPS for the UK
Author contributions
A.W., J.F., M.W., S.D., K.M., L.D. and M.L. designed the study and secured funding, V.B., L.D. and M.W. designed data collection instruments, L.D. and M.W. coordinated data collection, J.F. curated the study data. A.R., H.H., A.W. and J.F. performed the analyses. All authors drafted and revised the manuscript and approved the final manuscript.
Disclosures
James Fotheringham and Martin Wilkie have received speaker honorarium from Fresenius Medical Care and have conducted research funded by Baxter. Mark Lambie has received speaker honoraria from Baxter Healthcare and Fresenius Medical Care and a research grant from Baxter Healthcare in 2013. Simon Davies has received speaker honoraria from Fresenius Medical Care, research funding from Baxter Healthcare and advisory board fees from Baxter HealthCare and Ellen Medical.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by UK NIHR Research for Patient Benefit PB-PG-0613-31028, with ancillary funding from PDOPPS (Arbor Research Collaborative for Health); VB received funding from a Baxter Clinical Evidence Council award. The views expressed are those of the author(s) and not necessarily those of the NIHR or the Department of Health and Social Care. The Dialysis Outcomes and Practice Patterns Study (DOPPS) Program is funded by a consortium of private industry, public funders and professional societies. More information on DOPPS funding can be found here: ![]() . Funding for PDOPPS has been provided by: National Health and Medical Research Council (Australia); National Institute for Health Research (UK); National Institute of Diabetes and Digestive and Kidney Diseases, (USA); Patient-Centered Outcomes Research Institute, (USA); Japanese Society of Peritoneal Dialysis; Canadian Institute for Health Research (Canada); Baxter International Inc. (USA); The National Research Council of Thailand (2558-113); Rachadaphiseksompot Endorcement Fund (GCURS_59_12_30_03), Chulalongkorn University, Thailand; and the National Science and Technology Development Agency (NSTDA), Thailand.
. Funding for PDOPPS has been provided by: National Health and Medical Research Council (Australia); National Institute for Health Research (UK); National Institute of Diabetes and Digestive and Kidney Diseases, (USA); Patient-Centered Outcomes Research Institute, (USA); Japanese Society of Peritoneal Dialysis; Canadian Institute for Health Research (Canada); Baxter International Inc. (USA); The National Research Council of Thailand (2558-113); Rachadaphiseksompot Endorcement Fund (GCURS_59_12_30_03), Chulalongkorn University, Thailand; and the National Science and Technology Development Agency (NSTDA), Thailand.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
