Abstract
Background
The stability of antimicrobials in peritonitis during peritoneal dialysis (PD) solutions is a critical factor influencing treatment success. This study investigated the stability of daptomycin (DAP) when combined with icodextrin-based PD solution, by measuring DAP concentrations and observing any structural changes.
Methods
A dose of DAP (350 mg) was dissolved in 7 mL of saline in a clean bench. The solution was then injected into the large compartment (1,260 mL) of NICOPELIQ® Peritoneal Dialysis Solution and thoroughly mixed. Samples were collected at intervals ranging from 0 to 336 h (7 points in total). The concentration of DAP was quantified using high-performance liquid chromatography (HPLC). The structure of any unidentified peaks was determined using HPLC coupled with electrospray ionization tandem mass spectrometry.
Results
DAP maintained 90% of the initial concentration in NICOPELIC® for 72 h at room temperature and 12 h at 37 °C. Unidentified peaks, distinct from DAP, were detected during analysis. Further investigation indicated that these peaks corresponded to anhydrated DAP.
Conclusions
The findings from this stability study are expected to enhance the effectiveness of outpatient management and preparation for treating peritonitis using DAP.
Background
Patients undergoing peritoneal dialysis (PD) face a heightened risk of peritonitis, often stemming from infections at the peritoneal catheter insertion site.1–3 Peritonitis represents a significant complication in PD, necessitating proactive prevention and treatment to reduce morbidity and mortality. This priority is emphasized in the 2022 International Society for Peritoneal Dialysis (ISPD) guidelines for peritonitis prevention and treatment.4–6 Commonly, peritonitis in PD patients is managed by administering intra-abdominal antibacterial drugs in an outpatient setting. This treatment involves combining the antimicrobial agent with the PD fluid. The ISPD guidelines recommend the immediate initiation of empiric antimicrobial treatment, either intra-abdominally or systemically, following proper microbiological sampling. The antibiotics used include aminoglycosides, cephalosporins, penicillins, and antifungals, with PD solutions divided into glucose-based and icodextrin-based categories. NICOPELIQ® Peritoneal Dialysis Solution, an icodextrin-based PD solution, is designed for prolonged efficacy, typically 8–12 h. Approved in Japan in 2014 as a generic to the same icodextrin formulation, Extraneal®, NICOPELIQ® comprises a dual-chamber system to separate the ingredients, servicing approximately 1,500 annually in Japan as of January 2024 (information provided by the manufacturer). Icodextrin stability in low pH range 7 is maintained in a dual-compartment polypropylene bag; the large compartment maintains a low pH to preserve the stability of icodextrin. To prevent organismal harm from the low pH, 8 a pH adjuster is added, and the pH is neutralized (pH 6.2–6.8) before use by opening the partition wall. The stability of antimicrobials in PD solutions is crucial for treatment success. For instance, gentamicin remains stable for 14 days at room temperature 9 and under refrigeration in glucose-based PD solutions, whereas shows similar stability in icodextrin-based solutions.9–11
Data on the stability of new antimicrobials in PD solutions are important for clinical readiness but remain scarce. Daptomycin (DAP), a cyclic lipopeptide antimicrobial, is recommended for both intermittent (300 mg/day)12–16 and continuous (100 mg/L loading dose, 20 mg/L maintenance dose) use; 17 yet, its stability in PD solutions, particularly at concentrations used for intermittent administration or over extended periods like 2 weeks, remains underexplored. Furthermore, there have been no reports on the structural changes in DAP as its concentration decreases. Although a systematic review discusses DAP's stability in glucose and Nutrineal solutions, its stability in icodextrin-based PD remains understudied. 18 The concentrations of icodextrin, pH, and osmotic ratio in the large chamber of NICOPELIQ® differ from those in Extraneal® (Table 1). Consequently, there are limitations in applying Extraneal® data directly to NICOPELIQ®. This study, therefore, aims to examine the stability of DAP when mixed with NICOPELIQ® by measuring its concentration and observing any structural changes.
Comparison of icodextrin concentration, pH, and osmotic ratio between NICOPELIQ® and Extraneal®.

Materials and methods
Materials
NICOPELIQ® (lot numbers 171204SF, 170925SF, and 171225SF) was sourced from Terumo Co., Ltd, Tokyo, Japan, along with a 10-mL syringe and an 18G needle. Cubicin® (350 mg DAP preparation for intravenous injection, lot number N022064) was purchased from MSD, Kenilworth, NJ, USA. Acetonitrile and formic acid, utilized for high-performance liquid chromatography (HPLC) and liquid chromatography–mass spectrometry (LC–MS), respectively, were obtained from FUJIFILM Wako Pure Chemical Co., Osaka, Japan. Purified water was produced using the Elix (RO+EDI®) purified water production system (Elix Essential UV3, MERCK MILLIPORE, Billerica, MA, USA).
Methods
Preparation of a standard solution
A standard stock solution was prepared by weighing 20.0 mg of DAP, transferring it into a 20-mL volumetric flask, and diluting with ultrapure water to a concentration of 1,000 μg/mL. The stock solution was further diluted with ultrapure water to create standard solutions, which were stored at 4 °C and in dark conditions to ensure stability.
Preparation and recovery of DAP in NICOPELIQ®
NICOPELIQ® was utilized with the septum remaining unopened. DAP (350 mg) was first dissolved in 7 mL of saline on a clean bench. This solution was then injected into the large compartment (1,260 mL) of NICOPELIQ® and thoroughly mixed. Samples of 5 mL were collected at 0, 12, 24, 72, 168, and 336 h post-injection.
Storage conditions
The study utilized two storage conditions: in a PD fluid heater assumed to be used just before administration and at room temperature with light protection. A specific PD fluid heater (Terumo Co., Ltd) was utilized for this purpose. The mixed solution was stored in boxes at room temperature under blackout conditions to prevent light exposure. Storage conditions (temperature, humidity, and light exposure) were controlled using a TR-72wf rooster temperature and humidity data logger (T&D, Nagano, Japan), which recorded temperatures of 22.3 ± 3 °C and humidity at 61.8 ± 15%. Complete light exclusion was maintained to ensure darkness.
Measurement of DAP concentration in NICOPELIQ®
HPLC analysis was conducted using a Nexera XR system (Shimadzu Corporation, Kyoto, Japan), comprising a liquid transfer unit (LC-20ADXR), an autosampler (SIL-20AXR), and a column oven (CTO-20AC) equipped with a UV detector (SPD-M20A). An Inertsil® ODS-3 column (2.1 mm × 100 mm i.d., 5 μm, GL Science Inc., Tokyo, Japan) was used, with the column oven temperature set at 40 °C. The mobile phase included water with 0.1% formic acid (A-liquid) and acetonitrile (B-liquid), with a flow rate of 0.4 mL/min. The injection volume was 1 µL, using UV light absorption at 370 nm for detection. LabSolutions software (Shimadzu Corporation) facilitated system control and data processing. Calibration curves were generated using six DAP concentrations: 10, 30, 50, 100, 300, and 500 µg/mL. Validation was performed to ensure the reliability of the assay. The calibration curve exhibited perfect linearity (R2 = 1.000). Inter-day and intra-day reproducibility were assessed at 30.0, 100, and 500 µg/mL, with accuracy ranging from -2.87% to 4.56% and precision within 6.99%.
HPLC/ESI-MS/MS analysis
The mass spectrometry was performed using a triple-quadrupole mass spectrometer (LCMS-8040) equipped with an ESI probe in the ionization section. This was connected to a Nexera X2 system for ultrafast liquid chromatography, which included a liquid transfer unit (LC-30AD), an autosampler (SIL-30AC), and a column oven (CTO-20A) (Shimadzu Corporation). The nebulizer gas flow rate was set at 3 L/min, desolvation line temperature at 250 °C, heat block at 400 °C, and drying gas flow rate at 15 L/min. System control and data processing were performed using the LabSolutions software from Shimadzu Corporation.
The mobile phase was a 70/30 mix of 0.1% formic acid in water and acetonitrile, at a flow rate of 0.4 mL/min. Analyses used 1-μL injections of DAP and its impurities on an Inertsil® C8-3 column (2.1 mm × 100 mm, i.d., 5 μm, GL Science Inc.). Mass spectrum parameters ranged from m/z 100 to m/z 2,000, with collision energy (CE) set at 25 V. The product ion spectrum was set in the range of m/z 100 to m/z 1,000, and the CE was set at 25 V. Measurements were conducted in positive ion mode, and the detected fragments were analyzed as reported by Liu et al. 19
Measurement items for stability tests
For stability testing, six samples were used. The key parameters measured for stability tests included concentration and pH. Samples retaining at least 90% of the initial DAP concentration were deemed compliant.
Statistical processing
Differences in the data were evaluated using Dunnett's test, with a significance threshold set at p < 0.05. Statistical analysis was conducted using JMP® (version 13.2.1) software.
Results
Examination of stability at 37 °C under dark conditions
Given that the samples are intended to be heated to 37 °C before use, we decided to examine their stability at room temperature, as well as at 37 °C utilizing a dialysate heater set to 37 °C. We combined DAP with NICOPELIQ® PD fluid and stored it in a dialysis fluid heater, with the heating initiation time set at zero. The temperature within the NICOPELIQ® stabilized at approximately 37 ± 1 °C approximately 4 h after heating commenced (data not shown). The DAP concentration decreased to below 90% of its initial concentration level 24 h after heating started. Specifically, from an initial concentration considered 100% at time zero, the retention rate was 95.5% at 12 h, dropped to 87.0% at 24 h, and further declined to 25.9% at 336 h (Table 2). The pH levels remained stable between 4.3 and 4.6, with no significant fluctuations observed. No obvious color change was observed (data not shown).
Time required for DAP concentration in NICOPELIQ® to reach 90% at 37 °C and RT.
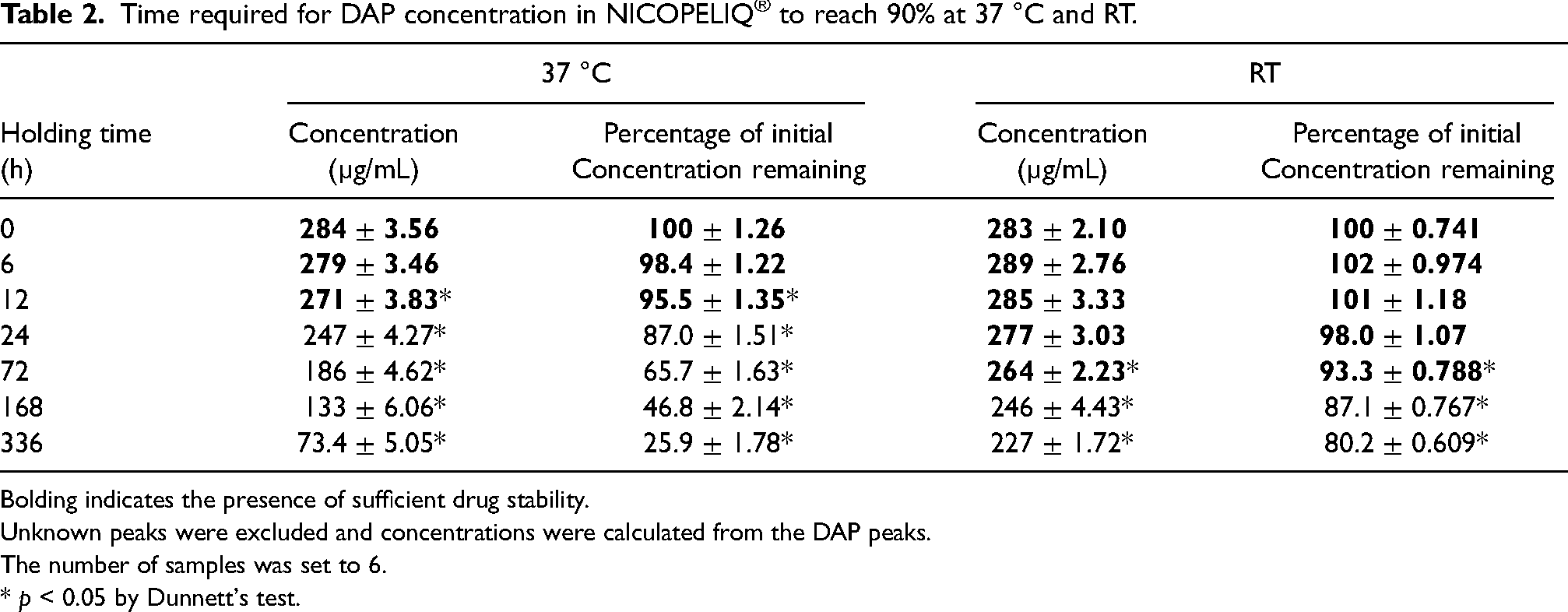
Bolding indicates the presence of sufficient drug stability.
Unknown peaks were excluded and concentrations were calculated from the DAP peaks.
The number of samples was set to 6.
* p < 0.05 by Dunnett's test.
Examination of stability under room temperature and dark conditions
DAP was combined with NICOPELIQ® PD solution and stored at room temperature in a dark environment immediately after mixing. The concentration of DAP significantly decreased after 72 h. From an initial concentration of 100% at 0 h, the retention rate was 93.3% at 72 h, 87.2% at 168 h, and further declined to 80.2% at 336 h (Table 2). The pH remained consistent between 4.3 and 4.6, with no notable changes. No obvious color change was observed (data not shown).
Identification of an unknown peak
Initially, only the DAP peak was observed at 0 h. However, as the measurements progressed, a new peak emerged, appearing later than the DAP peak at 6.14 min. This peak, not initially present in the sample, surfaced at 6.81 min after 2 h and gradually increased in intensity, eventually surpassing that of DAP by 336 h (Figure 1). The peak's intensity increased steadily, plateauing after 168 h. Under room temperature and light-protected conditions, this increase was less pronounced, with the peak area only one-third of that under heated conditions.
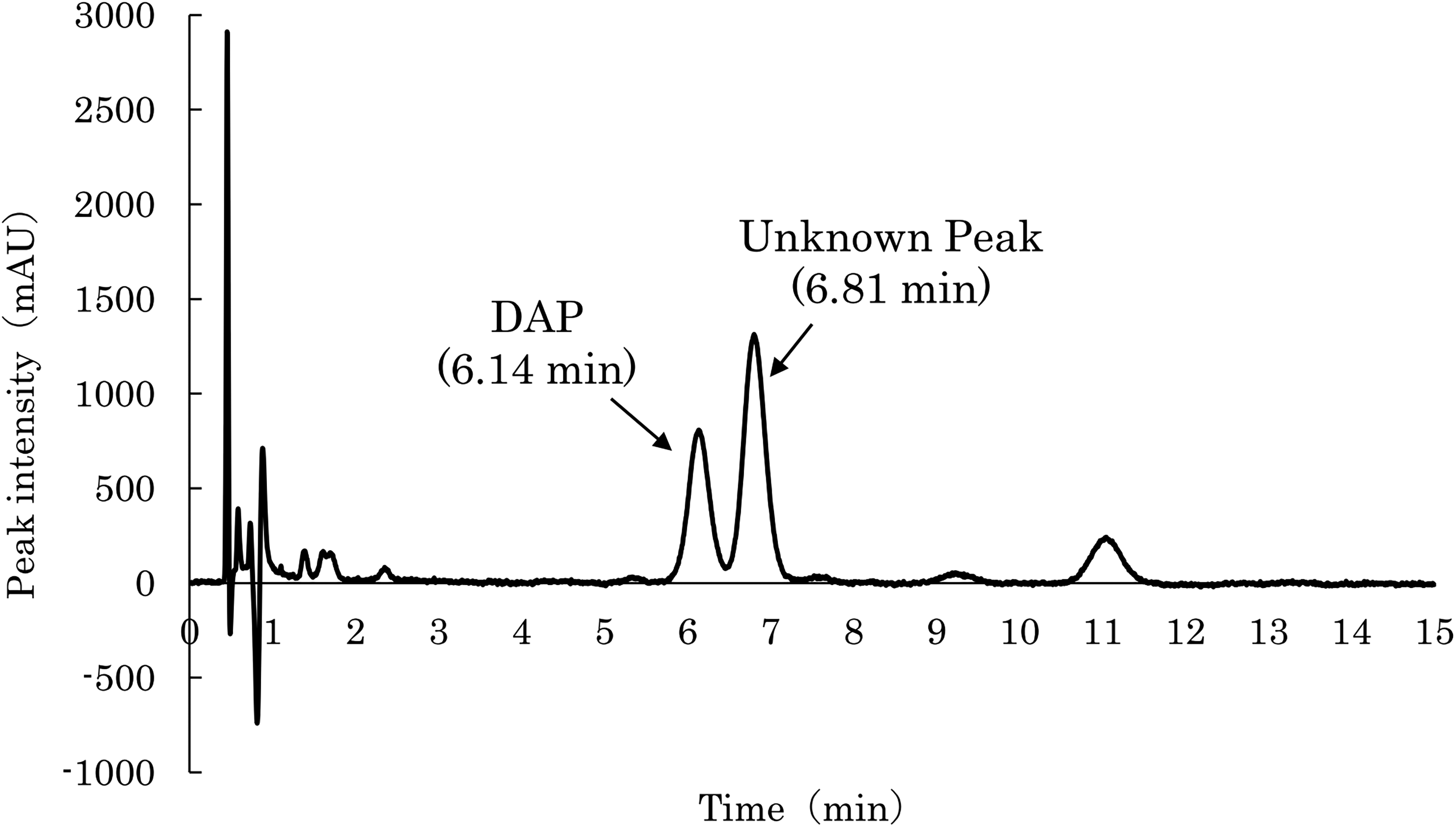
Chromatogram of DAP and unknown peaks after storage at 37 °C for 336 h.
To identify the impurities, we conducted a precursor ion scan using HPLC/ESI-MS/MS on a sample collected 336 h after heating commenced. Given that DAP's molecular weight is m/z 1,620, it was presumed to be detected as [2M+H]+. However, an impurity peak at m/z 802 was observed. The UV spectrum of this impurity resembled that of DAP. Given the similarity, we hypothesized that this peak may represent [2M+H]+, indicating an impurity with a molecular weight of 12. Subsequently, HPLC-ESI-MS/MS was used for product ion scanning of the protonated molecules [2M+H]+ of DAP and the impurities, with the spectrum range set from m/z 100 to m/z 1,000. For DAP, strong peaks were noted at m/z 641, m/z 341, and m/z 313. The unknown impurity displayed peaks at m/z 632, m/z 341, and m/z 313 (Figure 2). Since the peaks at m/z 341 and m/z 313 matched, and the mass difference between m/z 641 and m/z 632 was 18, we concluded that the unknown peak corresponds to the fragment ion of anhydrous DAP.
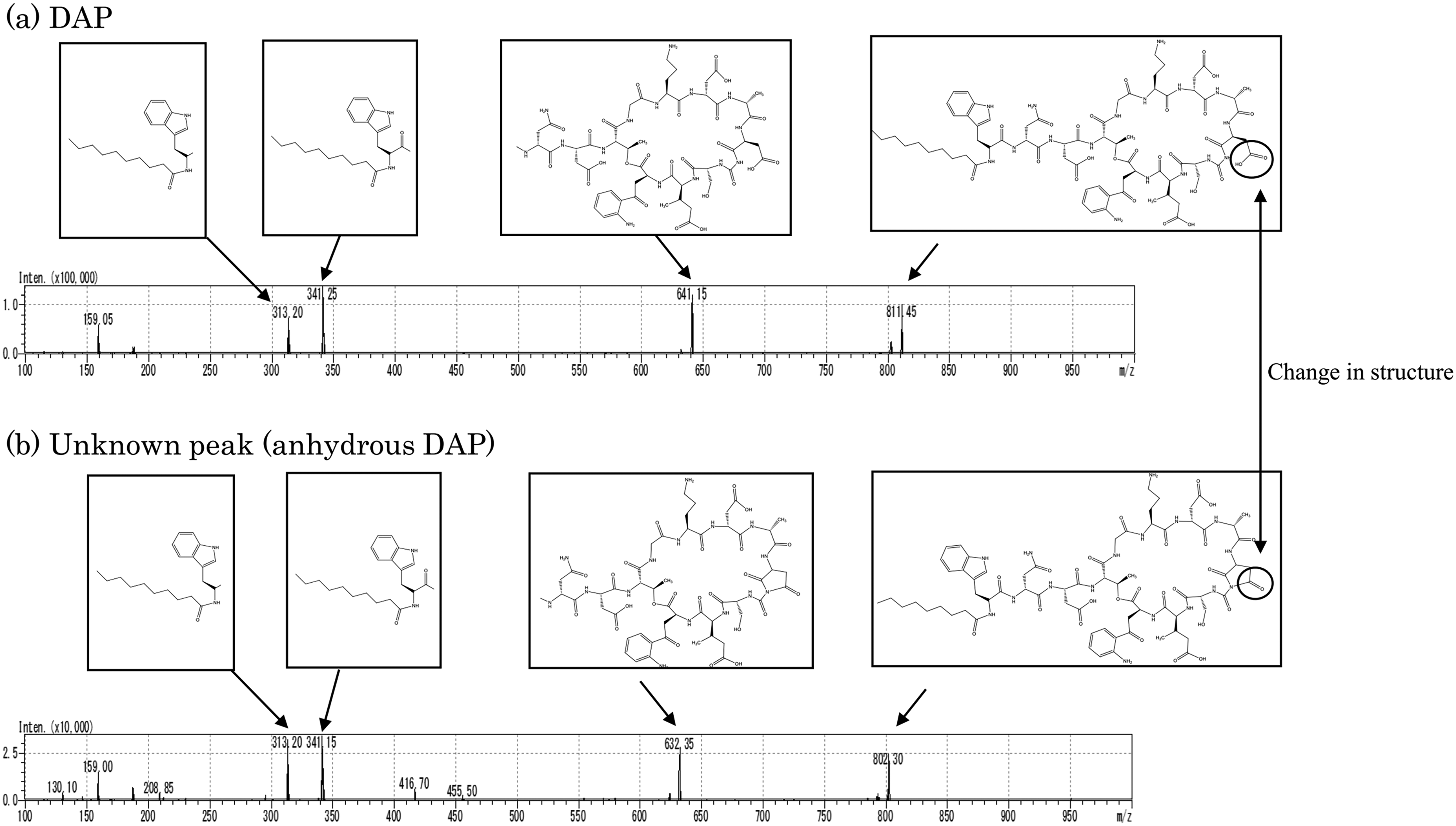
Product ion scan of (a) DAP and (b) unknown peak identified as anhydrous DAP.
Discussion
While previous studies have explored the stability of DAP in PD fluid,20–22 data on the stability of NICOPELIQ® PD fluid combined with antimicrobial agents remain scarce. This study aimed to assess the stability of an antibacterial drug in NICOPELIQ® by monitoring changes in concentration and pH over time. For this purpose, DAP was mixed in the large chamber of NICOPELIQ®, and stored under two conditions room temperature with light shielding and in a PD solution heater, specifically in the dark to examine the effect of temperature. Considering outpatient treatment, we set the measurement duration at 336 h (2 weeks).9–11
The findings indicate that the DAP-PD mixture maintained a concentration above 90% for 12 h at 37°C in light-shielded conditions.
Previous research suggests DAP undergoes degradation through aspartyl transpeptide conversion at the Asp-9 residue in weakly acidic solutions (pH 3–6), 20 leading to dehydration. Two study limitations are noted. First, while we examined the long-term stability of the product over 336 h, short-term results are crucial in actual clinical practice. Measurements at 36 and 48 h could provide additional pertinent data for clinical settings. Second, since DAP was mixed with NICOPELIQ®, which has a pH range of 4.0–6.0, dehydration may have occurred. 20 However, research into whether anhydrous DAP affects drug efficacy is lacking. Therefore, this study assessed the stability of DAP excluding the anhydrous form, which appeared as an unknown peak. The peak at 11 min was predicted to have a structure similar to that of DAP based on the UV spectrum. However, its structure could not be analyzed due to the low expression levels. DAP exhibits concentration-dependent activity against nearly all gram-positive pathogens. 22 In vivo condition simulations at high clinical concentrations after incubation for 4 h and 8 h show that DAP achieves more rapid activity than other tested antibiotics. 22 This report provides valuable data, and similar studies on NICOPELIQ® are recommended for the future.
Conclusions
Our research provides a comprehensive analysis of DAP stability and structural integrity when mixed with NICOPELIQ®, an integral component of PD treatment. These results will help to improve the efficiency of outpatient and preparation work in the treatment of peritonitis using DAP.
Footnotes
Acknowledgements
The authors are grateful to Yui Mukai (Division of Clinical Pharmaceutics, Faculty of Pharmaceutical Sciences, Tohoku Medical and Pharmaceutical University, Sendai, Japan) for cooperation in the research.
Author contributions
HS, KK, TU, YA, and HN designed the study; HS, KK, TU, TK, HN and YM contributed to the analysis and interpretation of data; HS, KK, and TU drafted the manuscript; HS, KK, KO, HN, and YM revised it critically for important intellectual content; Each author provided final approval of the version to be published.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent to participate
Not applicable.
Informed consent to publish
Not applicable.
Trial registration
Not applicable.
