Abstract
Background
Frailty is associated with poor outcomes for patients on dialysis; however, dedicated studies among incident peritoneal dialysis (PD) patients are lacking. This study aims to determine the association between frailty and mortality/transfer to hemodialysis (HD) among incident PD patients and identify whether dialysis modality (PD vs. HD) modifies the association of frailty and mortality following dialysis initiation.
Methods
A single center retrospective cohort of incident PD and HD patients was analyzed from 2009 to 2020 (last follow-up December 2021). The first version of the clinical frailty scale (CFS) ranging from 1, very fit, to 7, severely frail, was used to characterize patient frailty at dialysis initiation. Time to death/transfer to HD was analyzed using multivariable Cox Survival analyses. Patients were censored at transplant/last follow-up. Dialysis modality was evaluated as a potential effect modifier on the impact of frailty on mortality.
Results
Of 253 patients who initiated PD, 182 had completed CFS scores. Mean age at dialysis initiation was 63 ± 13 years and mean CFS score was 3 ± 1. There were 42 deaths and 69 instances of transfer to HD over 379 patient-years at risk. Vulnerable/frail patients (CFS ≥ 4) had a two-fold increase in the adjusted relative hazard for death/transfer to HD (HR 2.04, 95% CI [1.10–3.77]). Incident dialysis modality did not modify the association between frailty and mortality.
Conclusions
A higher severity of frailty at PD initiation is associated with both mortality/transfer to HD and mortality alone, and the association between frailty and mortality is not modified by dialysis modality.
Introduction
Frailty is a state of increased vulnerability due to a loss of reserves in energy, physical ability, cognition, and health. 1 Individuals who are frail experience higher rates of several adverse health outcomes including hospitalization and mortality. 2 Specific to kidney disease, frailty is highly prevalent in dialysis 3 and associated with a number of negative outcomes including falls, hospitalization, and mortality. 4
Many tools have been developed and validated for the assessment of frailty. In dialysis, the fried frailty phenotype has most frequently been used for the evaluation of frailty.4,5 More recently, the clinical frailty scale (CFS) has been used in dialysis.6,7 The CFS (a tool that uses clinical judgment) correlates well with other measures of frailty, 1 and has predictive validity in dialysis. 8 We previously showed that the CFS had good interrater reliability and that a higher severity of frailty among incident maintenance dialysis patients and was associated with an increased risk of mortality. 6 In a follow-up study, a higher frailty severity was associated with both an increased risk of death and hospitalization when jointly modeled. 7
Peritoneal dialysis (PD) is most often delivered at home and thus, patients (especially those who are independent) tend to be healthier compared to in-center hemodialysis (HD). 9 To our knowledge, there are a paucity of studies evaluating whether frailty is associated with poor health outcomes among incident PD patients. In a study of Chinese PD patients, frailty (calculated using a self-reported questionnaire) was shown to be an independent risk factor for hospitalization, however no significant relationship was found between frailty and mortality or peritonitis. 10 In another study involving prevalent home dialysis patients (home HD and PD) both objective and subjective measures of frailty associated with transfer to HD and death. 11 Other studies have also demonstrated an association between frailty and adverse outcomes in PD, particularly when combined with other comorbidities.12,13
Further study is needed to identify the association between frailty and important health outcomes in PD patients such as mortality and transfer to HD. In this cohort study of PD patients, we aimed to (1) describe the proportion of patients who were frail (and the severity of frailty), (2) to determine the impact of frailty on outcomes after PD initiation including transfer to HD and death, and (3) to determine if dialysis modality modified the risk of frailty on mortality following dialysis initiation.
Methods
Study population
We analyzed a cohort of consecutive, adult patients who initiated PD from 2009 to 2020 at a large tertiary care center in Nova Scotia, Canada. This center services all incident dialysis patients from a catchment area of 750,000 individuals. Maintenance dialysis patients were identified from a local electronic database as those for whom the treating physician diagnosed as having kidney failure requiring dialysis without possibility of recovery. The subgroups of incident PD patients and in-center HD comparator group (objective 3) were subsequently identified from this cohort. We included patients who initiated dialysis after a failed kidney transplant. Incident PD patients were those who initiated dialysis with PD (either CAPD or APD); in a sensitivity analysis, we also defined incident PD patients as those who initiated PD within three months of starting dialysis. Patients were followed for outcomes until December 31, 2021.
Exposure assessment
The CFS 1 was used to characterize the level of frailty for all patients. We previously identified that the CFS has moderate agreement with the frailty index (an established objective tool to identify frailty), 14 and the CFS has high interrater reliability (ICC > 0.97). 1 The original seven-point version CFS was used, consisting of the categories of 1 = very fit; 2 = well without active disease; 3 = well with treated comorbid disease; 4 = apparently vulnerable; 5 = mildly frail; 6 = moderately frail; and 7 = severely frail (Supplemental Table 1). The CFS was primarily treated as a continuous variable and a three-level categorical variable using categories of 1–2, 3, and 4–7.15–17 CFS scores were assigned prospectively at the time of dialysis initiation by the primary nephrologist or nurse-practitioner caring for the patient prior to dialysis initiation based on clinical judgment. For patients who started as an inpatient without prior follow-up, the CFS score was assigned by the physician managing the inpatient care of that patient at the time of dialysis initiation. Characteristics and outcomes of patients who had missing frailty scale scores were captured for comparison. For patients initiating with HD but transferring to PD within three months, frailty was measured using the score ascertained at HD start.
Outcome assessment
Outcomes of interest included (A) death and (B) the composite of transfer to HD or death. Date of death and cause of death as well as date of and reason for transfer to HD were ascertained from documentation in electronic records and patient charts.
Death was defined as all-cause mortality. Time to death from date of dialysis initiation was examined with patient survival being censored at time of transplantation, transfer to HD, loss to follow-up or study end. In a sensitivity analysis, we examined time to death without censoring at transfer to HD. Transfer to HD was defined as any switch from PD to HD irrespective of the duration. The composite of time to transfer to HD or death was examined with patients censored at time of transplantation, loss to follow-up or study end. In a sensitivity analysis, we included only switches to HD that lasted for ≥ three months. In another sensitivity analysis we examined time to transfer to HD while accounting for the competing risk of death.
For study objective 3, the outcome of death was compared for frail versus non-frail patients with dialysis type (HD vs. PD) as an effect modifier.
Baseline characteristics/covariates
Baseline characteristics were captured at the start of dialysis by the patient's primary nephrologist and included demographics, body mass index, comorbidities (diabetes, coronary artery disease, peripheral vascular disease, congestive heart failure, cerebral vascular disease, chronic pulmonary disease, malignancy, liver disease), cause of kidney failure, and laboratory data (hemoglobin, albumin, phosphorus, creatinine). Estimated glomerular filtration rate (eGFR) using the 2021 CKD-EPI equation was calculated for all patients at the start of dialysis based on serum creatinine values on the day of dialysis initiation. If unavailable, the most recent values within the preceding month were used.
Statistical analysis
Descriptive statistics were used for baseline characteristics and described for the overall cohort and by CFS categories of <3, = 3, and >3. Means and standard deviations and medians and interquartile range were used for continuous normal and continuous nonnormally distributed variables. Categorical variables were expressed as counts and percentages. CFS scores were displayed graphically using a histogram. For analyses, frailty was treated as a continuous ordinal, and categorical variable as has been done in prior study.6,7 For objective 2, time to death and the composite of time to death or transfer to HD was analyzed using adjusted Cox proportional hazards models. Results were reported using relative hazards (HRs) and 95% confidence intervals. Anticipating a small number of outcome events, we used sequential adjusted Cox survival models to determine the adjusted association between frailty and outcome. Our initial model included demographics and diabetes as a comorbidity.18,19 Each additional sequential model added other factors associated with death or transfer to HD including coronary artery disease, congestive heart failure, albumin, kidney failure etiology, and BMI.18,19 Variables with missing data were added to the final model to avoid excluding patients at earlier levels. We did not include interaction terms in the adjusted models to avoid overfitting, however, in a secondary analysis, one-way interactions between frailty (treated as a binary variable using a threshold of ≥4 vs. <4) were evaluated for albumin (using a threshold of ≥30 vs. <30 g/L), age (using a threshold of ≥70 years vs. <70 years), diabetes, and congestive heart failure. For objective 3, the population included both HD and PD patients. Time to death was evaluated in an adjusted Cox survival analysis inclusive of modality (PD and HD), frailty and the interaction of frailty and modality. For all statistical comparisons, a two-sided p-value of <.05 was deemed the threshold for statistical significance. All analyses were performed using Stata IC version 17.0 (StataCorp, Tx, USA).
Results
Overall, 1318 patients started maintenance dialysis, of whom 956 (73%) had an assigned CFS score (Figure 1). Of those with completed scores, 182 (19%) initiated on PD and 763 (80%) initiated in-center HD. Eleven patients (1%) started on home HD and were excluded from this analysis. A flow chart of all patients is detailed in Figure 1. A summary of outcomes where a switch in dialysis modality was not considered a censoring event (for the outcome of mortality) is in Supplemental Figure 1.
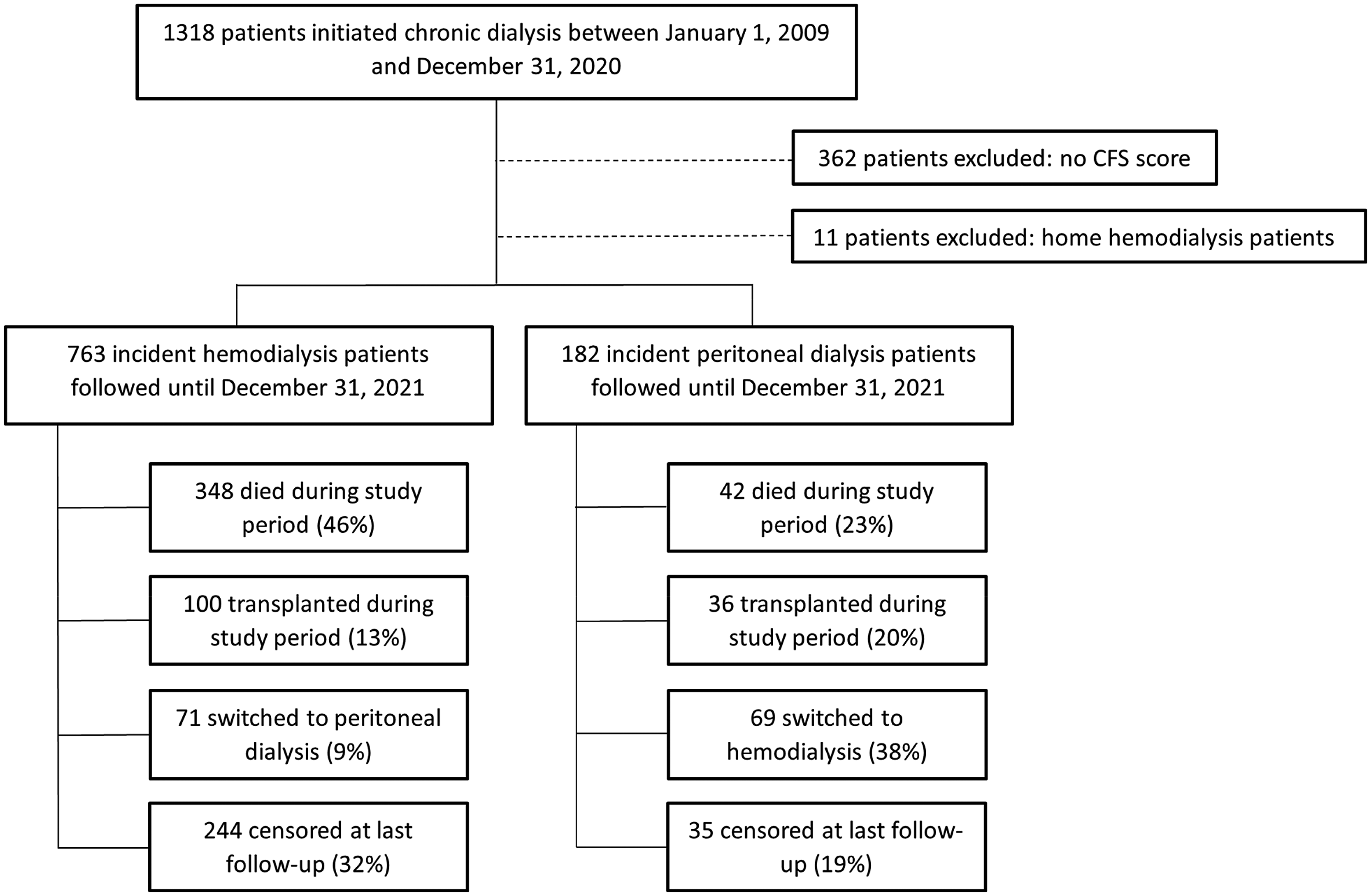
Cohort selection.
Characteristics of PD patients stratified by CFS score category are shown in Table 1. Mean age was 63 ± 13 years, 90% were white, 62% were male, and 35% had kidney failure caused by diabetic nephropathy (Table 1). Vulnerable/frail patients (CFS > 3; N = 65) were older (68 ± 11 years), had a lower serum albumin (33 ± 5), had a lower hemoglobin (99 ± 12), and had a higher frequency of comorbidities compared to those with lower CFS scores. Baseline characteristics were similar when patients who transitioned from HD to PD within three months of dialysis initiation were included in the incident PD cohort (Supplemental Table 2). PD patients with/without CFS scores had similar baseline characteristics (Supplemental Table 3).
Baseline characteristics for the incident PD cohort stratified by CFS category.
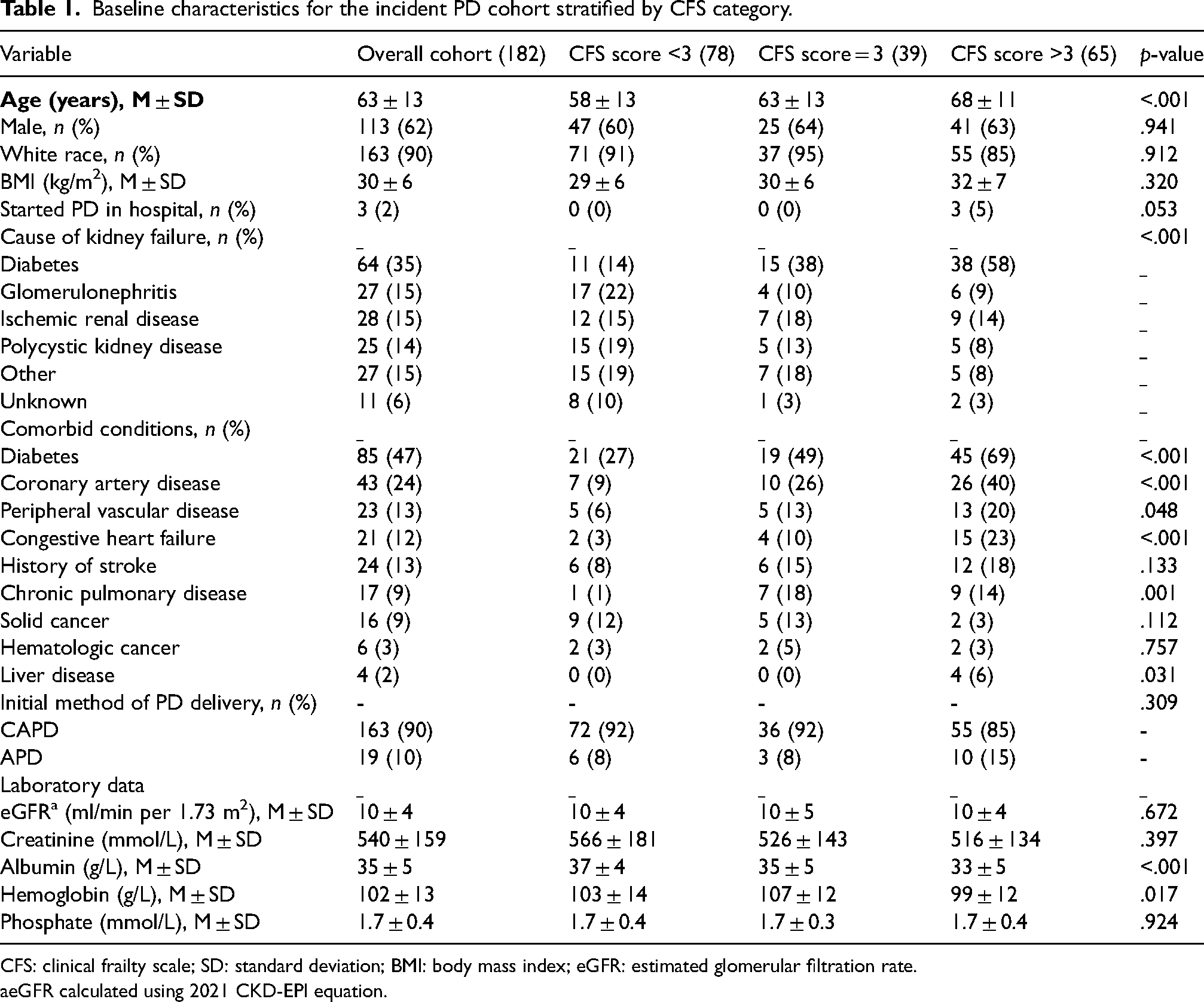
CFS: clinical frailty scale; SD: standard deviation; BMI: body mass index; eGFR: estimated glomerular filtration rate.
eGFR calculated using 2021 CKD-EPI equation.
Characteristics of incident HD patients by CFS category are noted in Supplemental Table 4. In general, incident PD patients were younger, had more favorable laboratory data, and had a lower rate of comorbid conditions (Supplemental Table 5).
The distribution of incident PD patients by CFS score is displayed in Figure 2. The mean CFS score was 3 ± 1 compared to a mean CFS score of 4 ± 1 among the HD comparator group.
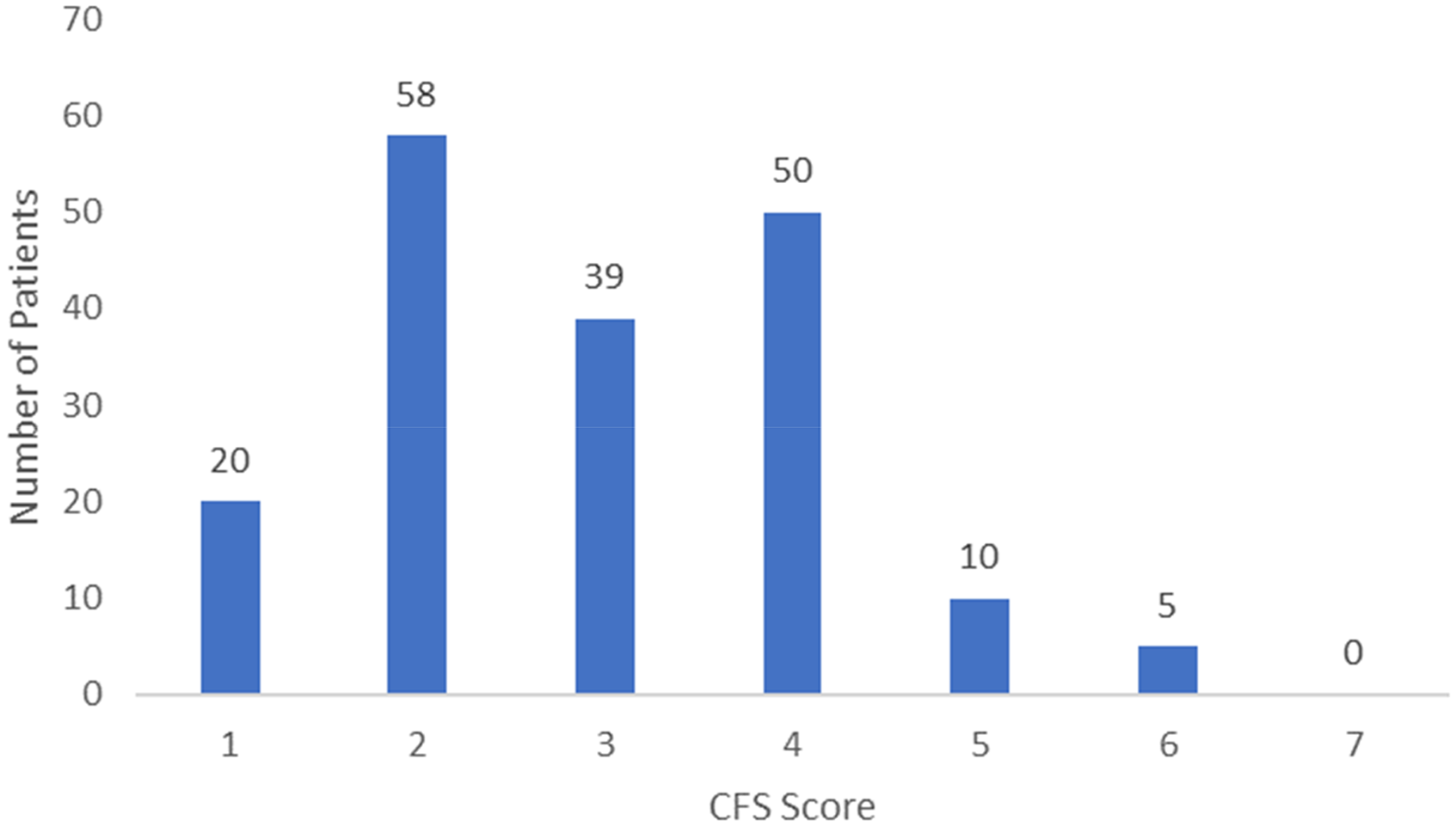
Distribution of CFS scores for incident PD patients.
Among the incident PD cohort, there were 42 deaths and 69 instances of transfer to HD over 379 patient-years at risk (median follow-up of 1.6 years, interquartile range = 0.8–3.1). The proportion of patients who experienced transfer to HD, died, were transplanted or were alive at the end of the follow-up period (without transplant or transfer to HD) stratified by CFS score is displayed in Supplemental Figure 2. Causes of death and transfer to HD are noted in Supplemental Table 6. Death was most commonly due to withdrawal from dialysis and cardiac causes while peritonitis, dialysate leak or hernia was the most common reason for HD transfer. The proportion of patients with each cause of death and each cause of treatment failure were similar, regardless of CFS score.
Time to death and death or transfer to HD among incident PD patients in unadjusted and adjusted Cox survival analyses are summarized in Figure 3 (C, D). In adjusted models, point estimates for the outcome of death were highest for those with CFS scores of >3 (HR ranging from 2.75 to 3.31 relative to a CFS score <3), although this finding was not statistically significant in all models. Vulnerable/frail PD patients had a two-fold increase in the HR for death or transfer to HD (HR 2.04, 95% CI [1.10–3.77], model 4) after adjustment.
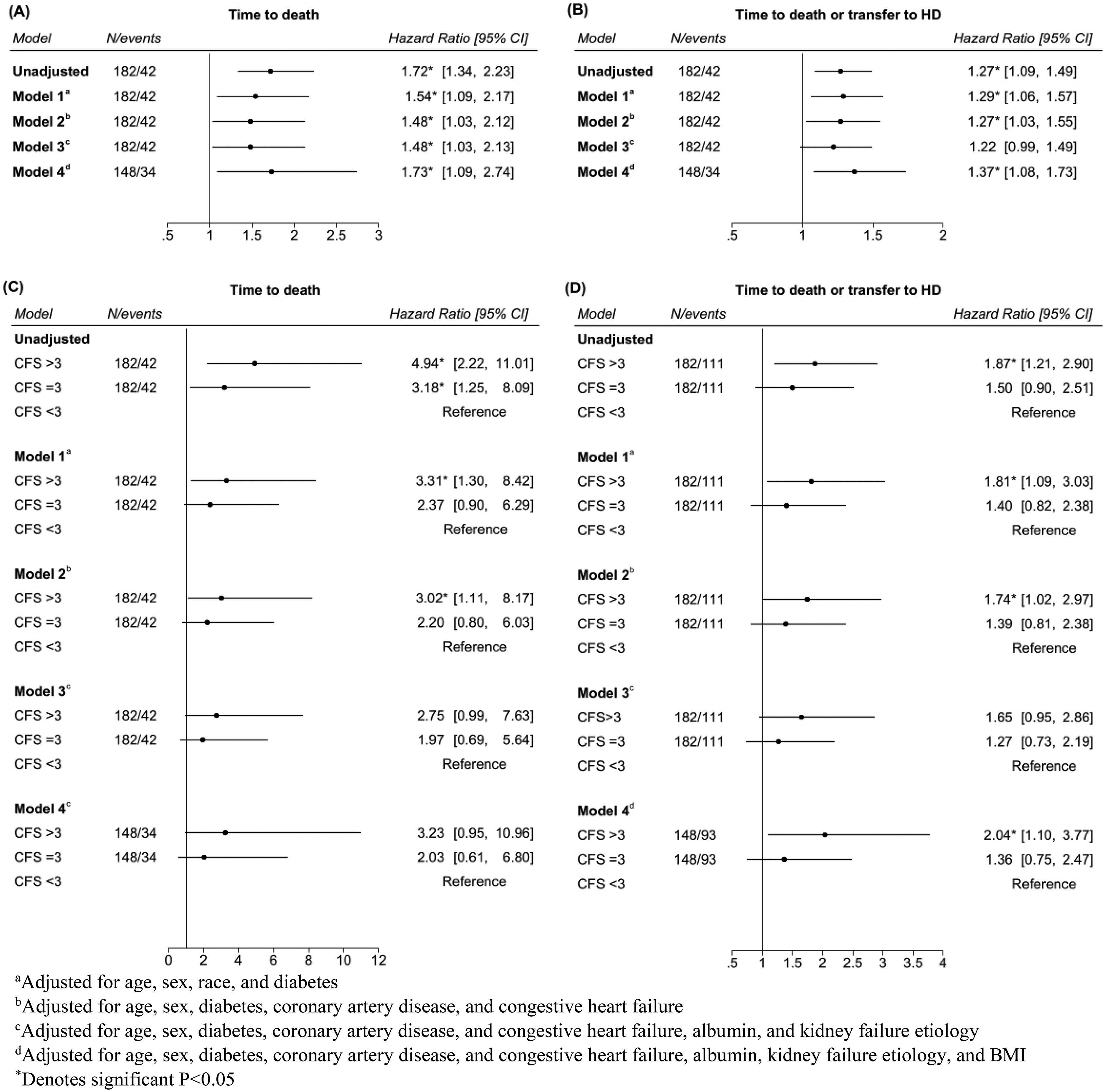
(A) Time to death in cox proportional hazards models for each one-point increase in CFS score for incident PD patients. (B) Time to death or transfer to HD in Cox proportional hazards models for each one-point increase in CFS score for incident PD patients. (C) Time to death in Cox proportional hazards models by CFS category for incident PD patients. (D) Time to death or transfer to HD in Cox proportional hazards models by CFS category for incident PD patients.
Similar trends for survival and transfer to HD were seen when treating frailty as a continuous variable (Figure 3 (A, B)). Each 1-point increase in the CFS score was associated with a 73% increase in the HR for mortality (HR 1.73, 95% CI [1.09–2.74], model 4) and a 37% increase in the HR for mortality or transfer to HD (HR 1.37, 95% CI [1.08–1.73], model 4) after adjustment.
Time to death and time to death or transfer to HD in various sensitivity analyses can be found in Figure 4 (A, B). When treating the CFS as a categorical variable, the inclusion of patients who transitioned from HD to PD within three months of dialysis initiation as members of the incident PD cohort did not significantly change the results for time to death (HR 3.23, 95% CI [0.95–11.01] for CFS > 3) or time to death or transfer to HD (HR 1.87, 95% CI [1.03–3.39] for CFS > 3). Similarly, when treating the CFS as a continuous variable, including patients who transitioned to PD within three months did not significantly change the results (HR for death 1.77, 95% CI [1.26–2.48], HR for death or transfer to HD 1.32, 95% CI [1.10–1.59]). In all other sensitivity analyses, the point estimates were either lower or not always statistically significant but in a similar direction as the primary analysis. Point estimates were in a similar direction when evaluating the association between frailty and outcomes while accounting for the competing risk of death (Figure 4 (C)) although not statistically significant. Additionally, there were no significant one-way interactions for those in the highest frailty group (CFS ≥ 4) versus those with lower scores (CFS < 4) across albumin, age, diabetes and heart failure (Supplemental Table 7).
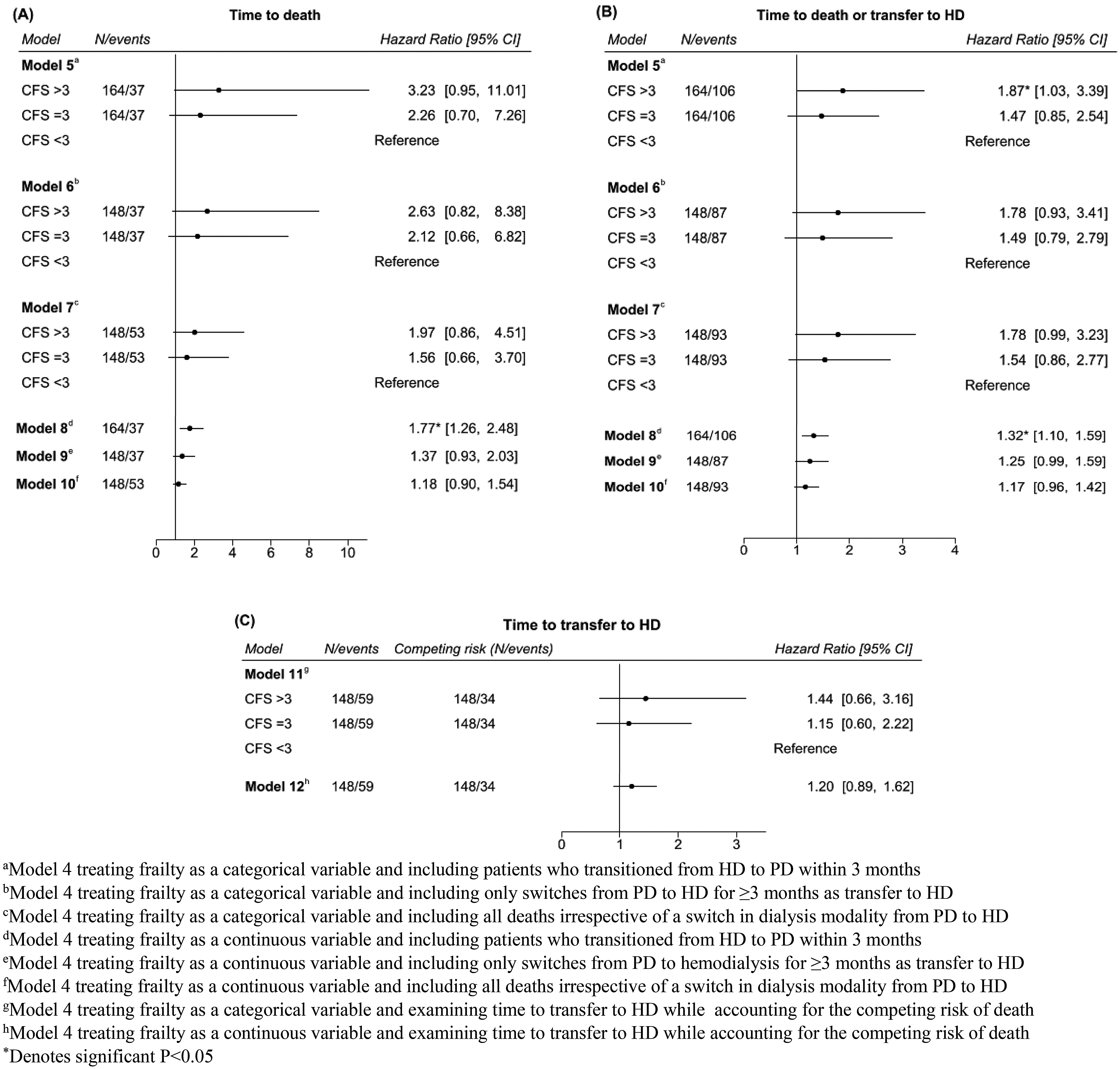
(A) Time to death in additional sensitivity analyses. (B) Time to death or transfer to HD in additional sensitivity analyses. (C) Time to transfer to HD while account for the competing risk of death.
In an analysis examining the interaction between frailty and incident dialysis modality (HD vs. PD) on the outcome of mortality, p-values for the interaction terms of CFS = 3 with modality (p = .706) and CFS > 3 with modality (p = .746) were not significant (Supplemental Table 8).
Discussion
In this study, we identified an association between frailty (as defined by the CFS) and mortality among incident PD patients. Similarly, an association was found between frailty and the composite of mortality and transfer to HD both when treating the CFS score as a categorical and continuous variable. These associations persisted after covariable adjustment and in several sensitivity analyses. In addition, we also demonstrated that incident dialysis modality (HD vs. PD) did not modify the association between frailty and mortality.
The prevalence of frailty among our PD cohort was lower compared to our HD comparator group. Only 36% of the PD patients had CFS scores >3 (corresponding to vulnerable/frail) and 8% had CFS scores >4 (corresponding to frail) compared to 64% of the HD patients with CFS scores >3 and 34% with CFS scores >4. These findings align with the existing literature, namely, that HD patients are more likely to be frail than those on PD. 20 The lower prevalence of frailty among PD patients is not surprising given the fact that PD patients tend to be healthier and younger compared to those receiving in-center HD,9,21 a finding that was also present in our study. These differences at baseline emphasize the need for concerted efforts to study and explore the relationship between frailty and outcomes specific to this population. In other PD cohorts, the prevalence of frailty has been reported to vary between 8.4% 13 and 72.7% 22 depending on the criteria used to define it. In particular, studies using the CFS score have reported rates of frailty from 8.4% to 27.6% which is more comparable to the prevalence in our study.12,13,23
Our current findings regarding frailty and mortality provide an extension to those reported from a previous study completed at our center which demonstrated that higher levels of frailty, as defined by the CFS, were associated with an increased mortality risk in incident dialysis patients. 6 The extension of these findings to the PD subgroup of chronic dialysis patients as well as our findings that other factors (albumin, age, and comorbidity) do not modify the association between frailty and outcomes suggests that routine measurement of the CFS, in addition to other established mortality predictors in PD, has the potential to add prognostic information in PD. The observed association is also consistent with the results of prior studies in PD patients.11,13 In one study examining the impact of sarcopenia and frailty on mortality in a cohort of PD patients, frailty as defined by the CFS was found to be an independent predictor of mortality. 13 Similarly, in another study involving prevalent home dialysis patients, both objective and subjective measures of frailty associated with an increased risk of death. 11 However, in another study using a 30-question dichotomous questionnaire to assess frailty in a cohort of prevalent PD patients, frailty was not an independent predictor of mortality. 10 Once again, the variability in literature may relate to the type of tool being used, size of the cohort or whether other important confounders were included.
The need for transfer to HD remains a burdensome and common complication that is unique to home dialysis patients.24–26 There are many known risk factors such as increasing age and comorbidity,27,28 and our study suggests that frailty may also associate with the outcome of HD transfer. However, we found that the strength of this association was not as high as with mortality, and this may point to variability in practice when frail PD patients are having difficulty at home. The results of previous studies have been contradictory when it comes to the impact of frailty on transfer from PD to HD. In a study of home dialysis patients (PD and home HD), physician impression of frailty and frailty using the Fried criteria were both associated with a > two-fold increase in the risk of transfer to HD in adjusted analyses. 11 However, in a cohort of PD patients from China, frailty was not an independent predictor of transfer to HD, 10 and in a study looking at the prognosis of coexisting frailty and cognitive impairment in patients on continuous ambulatory PD, there was no significant difference in time on PD therapy among those classified as frail versus not frail. 12 We hypothesize that frail patients may prefer to remain on PD despite the risks to health. Consistent with this notion, a recent study identified that older PD patients prioritized multiple other factors including flexibility with time and ability to work above mortality. 29 Therefore, faced with a decision to transition to a modality that is not within one's preference, patients may choose to forgo this decision and remain on home dialysis.30,31 Additionally, the availability of assisted PD impacts rates of transfer to HD with data from France showing that those with assisted PD had a lower risk of transfer to HD compared to those performing PD autonomously.32,33 Our finding of an association between transfer to HD and frailty could therefore highlights the importance of assisted PD, particularly for those with the highest frailty severity.
Although we found differences in the prevalence of frailty comparing HD to PD, incident dialysis modality did not modify the association between frailty and mortality. This emphasizes that there should be no modality preference when faced with a decision about what treatment to use in a frail patient based on survival. Once again, this is consistent with existing literature that emphasizes the importance of considering other factors when choosing a dialysis modality, particularly for older patients where the focus should be on quality of life and patient priorities. 34 That is not to say that frailty should not be an important consideration when managing a patient on PD, rather, the ascertainment of patient frailty at baseline using the CFS could be used to identify patients who might benefit from interventions to support frailty. 35 Various interventions to support frailty have been found to be beneficial among the general population including: exercise, nutritional interventions, and patient-centered geriatric care models with multicomponent interventions.35–37 Though there are unique challenges when applying these treatments to individuals receiving dialysis, there is some evidence that they can be effective and research is ongoing in this area. 38
The potential prognostic information provided by frailty severity, defined using the CFS, could also be used to guide shared decision-making for patients who experience ambivalence toward initiating dialysis and for whom a palliative or incremental approach may be more appropriate for maximizing quality of life. 39 In these patients, an incremental dialysis prescription (more days off, fewer exchanges or smaller exchange volumes) could be considered to improved patient centered outcomes such as symptomatic improvement while minimizing treatment burden and thereby enabling the individual to continue engaging in activities that are important to them. 40 A cohort study of adults with end-stage kidney disease demonstrated that frail patients are at increased risk of having poor health related quality of life, 41 however there are a paucity of studies focusing on the association between frailty and patient reported outcome measures among PD patients, highlighting an ongoing need for further study in this area. Another potential future direction of research would be developing and validating a risk prediction tool with frailty in addition to other parameters to evaluate mortality among incident PD patients.
This study has several strengths. It is one of only a few studies examining the association between frailty and health outcomes among incident PD patients. The prospective acquisition of CFS scores limited the possibility of misclassification of patients and improved the accuracy of data collection. We had a reasonable representation of patients with completed CFS scores and no difference in the baseline characteristics between those with and without CFS scores assigned, suggesting our included population was representative of the dialysis cohort at this center.
Despite these strengths, there are limitations. As this study was observational, there may have been unknown or unmeasured confounders that affected the association between frailty and mortality. We did not have information about caregiver dependency for either dialysis related care or other needs or utilization of nurse-assistance. However, nurse-assisted PD was available within our catchment area for the entirety of the study duration, suggesting that any patient who required assistance in performing PD would have had access. We also did not have access to patient's socioeconomic characteristics. Nevertheless, we were able to adjust for several important variables that have been shown to be associated with mortality in other studies of patients on dialysis. We do acknowledge that the CFS is subjective and different clinicians may grade severity differently or misclassify patients, although we previously demonstrated good interrater reliability, and moderate correlation comparing the CFS with objective measures. Furthermore, there are guidelines to enhance scale reliability42,43 and the CFS is a more realistic reflection of routine clinical practice which is another reason why it has emerged as a common metric for frailty across diverse populations, including dialysis patients. 8 We acknowledge that the CFS was only measured at the time of dialysis initiation and not followed longitudinally limiting our ability to examine the impact of changes in a patient's frailty status over time on outcomes. One study looking at progression of frailty in PD patients over time found that frailty transitions were common among this patient population. 22 Further study is therefore needed to examine whether changes in CFS score over the course of treatment impacts outcomes in PD patients. There was a low rate of frailty among PD patients and therefore when treating frailty as a three-level categorical variable, a cut-off of CFS > 3 (which corresponds to vulnerable and frail patients) was used for the highest category rather than a cut-off of CFS > 4 (corresponding to frail patients) to maintain an adequate sample size in this category. However, it is unlikely that inclusion of only frail patients in this highest category would have changed the results of our study and if anything, this may have strengthened the association between frailty and mortality. Finally, we acknowledge that this was a single-center study and our multivariable adjustment was limited by the small sample size. We acknowledge that our models were likely overfit for the outcome of mortality alone and our study would need to be replicated for a larger sample size to truly ascertain whether there is an independent association between frailty and mortality alone in PD.
Conclusion
In summary, frailty as defined by the CFS, is associated with an increased risk of mortality and mortality, or transfer to HD among incident PD patients and incident dialysis modality does not modify the association between frailty and mortality. Future studies are needed to evaluate whether an association exists between frailty and other short and long-term outcomes among PD patients such as hospitalizations, infectious complications (PD peritonitis and PD catheter related infections), and mechanical complications (dialysate leak, and PD catheter dysfunction).
Supplemental Material
sj-docx-1-ptd-10.1177_08968608241274095 - Supplemental material for Impact of frailty on mortality and transfer to hemodialysis after peritoneal dialysis initiation
Supplemental material, sj-docx-1-ptd-10.1177_08968608241274095 for Impact of frailty on mortality and transfer to hemodialysis after peritoneal dialysis initiation by Sophie Gaube, David Clark, Dylan Cooper, Annie-Claire Nadeau-Fredette, Amanda Vinson and Karthik Tennankore in Peritoneal Dialysis International
Footnotes
Acknowledgements
None
Author contributions
KT and DCl conceived the study. SG and KT researched the literature and were involved in protocol development, gaining ethical approval and data analysis. SG and DCo were involved in data collection. SG wrote the first draft of the manuscript. All authors reviewed and edited the manuscript, provided intellectual content and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was obtained from the Nova Scotia Health Authority Research Ethics Board (REB#1028066).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent to participate
Informed consent was not sought for the present study because this was a retrospective study for which it would be impracticable to obtain consent. A waiver of consent was obtained from the Nova Scotia Health Authority Research Ethics Board.
Informed consent to publish
Informed consent was not sought for the present study because this was a retrospective study for which it would be impracticable to obtain consent.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
