Abstract
Background:
Despite several advantages compared to haemodialysis (HD), peritoneal dialysis (PD) remains an underused dialysis technique due to its high technique failure rate related to membrane fibrosis and peritonitis events. Previous work has suggested a harmful role for the complement system in these processes, highlighting the need for a more comprehensive examination in PD.
Methods:
Plasma levels of C1q, mannose-binding lectin (MBL), Properdin, Factor D, C3d/C3-ratio and soluble membrane attack complex (sC5b-9) were determined in PD patients (n = 55), HD patients (n = 41), non-dialysis chronic kidney disease (CKD) patients (n = 15) and healthy controls (n = 14). Additionally, C1q, MBL, Properdin, Factor D and sC5b-9 levels were assessed in the peritoneal dialysis fluid (PDF). In a subgroup, interleukin-6, matrix metalloproteinase-2 (MMP-2), myeloperoxidase (MPO) and elastase were measured in the PDF.
Results:
PD patients had significantly higher systemic levels of sC5b-9 compared to healthy controls, CKD and HD patients (p < 0.001). Plasma levels of C1q and C3d/C3-ratios were significantly associated with systemic sC5b-9 levels (p < 0.001). Locally, sC5b-9 was detected in the PDF of all PD patients, and levels were approximately 33% of those in matched plasma, but they did not correlate. In the PDF, only Properdin levels remained significantly associated with PDF sC5b-9 levels in multivariate analysis (p < 0.001). Additionally, PDF levels of sC5b-9 positively correlated with elastase, MPO and MMP-2 levels in the PDF (p < 0.01).
Conclusions:
Our data reveal both systemic and local complement activation in PD patients. Furthermore, these two processes seem independent considering the involvement of different pathways and the lack of correlation.
Introduction
As a major part of innate immunity, the complement system is essential in the defence against pathogens, while dysregulation can lead to inapprioate activation thereby inducing tissue damage. 1 In peritoneal dialysis (PD), this system has been proposed to be an important mediator of peritoneal membrane damage and inflammation. 2,3 Furthermore, complement activation in dialysis has also been linked to relevant clinical implications such as cardiovascular disease, vasculopathy, peritoneal fibrosis and peritonitis. 2,3 However, only a limited number of studies have been conducted in PD. The complement system consists of a complex network of proteins that can be activated through distinct pathways: the classical pathway (CP), the lectin pathway (LP) and the alternative pathway (AP). 2,4,5 The CP is activated by C1q binding to immune complexes or other proteins, such as C-reactive protein (CRP). The LP is activated by mannose-binding lectin (MBL), ficolins and/or collectins binding to sugars or acetylated compounds. 6 Activation of the AP occurs via spontaneous hydrolysis of C3 or by binding of Properdin to altered surfaces. 7 Factor D is a serine protease that cleaves Factor B and thereby amplifies AP activation. All pathways converge at the level of C3 and ultimately lead to the formation of C5b-9. 8
Both systemic and local complement activation have been suggested in PD. Paediatric patients on PD have enhanced systemic complement activation in comparison with healthy controls, but plasma levels are not significantly higher compared to children with non-dialysis chronic kidney disease (CKD).
9
Evidence for local complement activation in the peritoneal cavity in children on PD was provided by a higher than expected dialysate/serum ratio for C3 activation products.
10
One of the proposed mechanisms of local complement activation in PD is the loss of complement regulators on the peritoneum.
The current study aims to investigate whether PD is associated with systemic and/or local complement activation in stable adult patients without peritonitis. In addition, we wanted to dissect the pathway(s) involved in PD-induced complement activation by measuring various complement components and initiators. Furthermore, we determined elastase, myeloperoxidase (MPO), interleukin-6 (IL-6) and matrix metalloproteinase-2 (MMP-2) to further elucidate the link between the complement system, leukocyte activation, inflammation and tissue remodelling in PD.
Materials and methods
Study design and population
Adult patients (≥18 years) being treated with chronic PD at the Peritoneal Dialysis unit of Hospital São Teotónio, Portugal, were recruited for this study. Exclusion criteria were signs of active inflammation, acute liver disease, infection and allergic reactions. Plasma ethylenediaminetetraacetic acid (EDTA) samples and peritoneal dialysis fluid (PDF) were collected after an overnight dwell with 1.36% glucose solution which was prescribed the day before the peritoneal equilibration test (PET), irrespective of their standard dialysis prescription. The overnight dwell duration was not standardised for this study and varied between 8 and 10 h. In a subgroup of 16 PD patients, two additional time-points, besides baseline, were collected with intervals between 6 and 12 months.
Plasma EDTA samples were also collected from control groups: (1) patients on maintenance haemodialysis (HD), (2) patients with non-dialysis CKD and (3) age- and sex-matched healthy controls. For 35 HD patients, plasma EDTA samples were collected both immediately before and after HD. These patients were recruited at the Hospital de Braga, Portugal, when they fulfilled the following criteria: ≥18 years, dialysis vintage of more than 3 months and no signs of inflammatory conditions as described above for the PD patients.
All patients and controls were Caucasian, and samples were uniformly collected and handled. Samples were centrifuged within 30 min of collection at 3500 r/min for 15 min at +4°C. Next, samples were stored in aliquots at −80°C until further analysis. Before analysis, samples were thawed and cleared by centrifugation.
Clinical data and laboratory procedures
Patient data were retrieved at the time of first sample collection and included age, sex, diabetes, blood pressure, dialysis vintage and type of technique (continuous ambulatory PD vs. automated PD), type of dialysis solution, residual kidney function, ultrafiltration volume, peritoneal membrane transport status, protein loss to the dialysate (estimated from the PDF protein concentrations at the end of the 4-h-dwell from the PET), body mass index (BMI) and body fluid volume and nutritional status using body composition monitor (Fresenius Medical Care, Bad Homburg, Germany) to determine relative overhydration, fat tissue index and lean tissue index.
Complement measurements
Plasma and PDF levels of C1q, MBL, Properdin, Factor D and C3d were measured using in-house developed sandwich enzyme-linked immunosorbent assay (ELISA), as previously described. 12 –14 Measurements of C3 were performed through radial immunodiffusion technique with monospecific anti-serum. 12 –14 Levels of C3 were only found in the PDF of 19 patients out of the 55, most likely due to the detection limit of our assay (0.18 mg/mL). Consequently, PDF C3d/C3-ratios were not used for further analysis. Plasma and PDF levels of soluble C5b-9 (sC5b-9) were assessed by ELISA, as described previously. 12 –14
Inflammatory and pro-fibrotic makers
IL-6 and MMP-2 were measured in PDF samples using a human magnetic Luminex assay (R&D Systems Inc., Minneapolis, Minnesota, USA) according to the manufacturer’s instructions. Levels of MPO and elastase in the PDF were measured using a commercial ELISA kit according to the manufacturers’ instructions (Hycult Biotech, Uden, The Netherlands).
Statistical analysis
Statistical analysis was performed using IBM SPSS 25.0 (IBM Corporation, Chicago, Illinois, USA). Results are presented as mean ± standard deviation for normally distributed data, median [interquartile range] for non-normally distributed data and total number of patients with percentage [n (%)] for nominal data. Differences between groups were assessed with the Student’s t test or the Mann–Whitney U test for normally and non-normally distributed variables, respectively, and the χ 2 test for categorical variables. The paired sample t test and the Wilcoxon signed-rank test were used to compare the values of consecutive samples. Correlations were assessed by using Spearman’s correlation for non-normally distributed data and Pearson’s correlation for normally distributed data. Differences between multiple groups were assessed with the Kruskal–Wallis test for non-normally distributed data and one-way analysis of variance (ANOVA) for normally distributed followed by a Dunnett post hoc test. Univariable and subsequent multivariable linear regression analyses, including all variables that were significantly (p < 0.05) associated with the outcome in univariable analysis, were used to identify independent determinants. p Values < 0.05 were considered significant.
Ethics
The study was approved by the local ethical committee and performed according to the principles of the declaration of Helsinki. All participants gave written informed consent.
Results
Study population
A total of 55 PD patients were included in the study. The primary kidney disease was diabetic kidney disease in 11, hypertensive kidney disease in 11, chronic glomerulonephritis in 10, tubulointerstitial disease/congenital anomalies of the kidney and urinary tract in 7, autosomal dominant polycystic kidney disease in 6 and unknown in 10. All patients were dialysed with neutral pH, low-GDP solutions (Physioneal®, Baxter or Balance®, Fresenius Medical Care), and 73% had a night exchange with Icodextrin®, Baxter, solution (Online Supplemental Table S1). The average age was 57 ± 13 years and 60% was male. In the control groups, the average age of the healthy controls (n = 14), non-dialysis CKD patients (n = 15) and maintenance HD patients (n = 41) were 56 ± 4, 77 ± 11 and 66 ± 16 years, and 64%, 60% and 71% were male, respectively (Online Supplemental Table S2). The non-dialysis CKD group included six CKD stage 3, six CKD stage 4 and three CKD stage 5. Their median estimated glomerular filtration rate was 22 [9–45] mL/min/1.73 m2. All maintenance HD patients received online-haemodiafiltration three times per week for 4 h using high-flux polysulphone dialysers (Fx80, Helixone; Fresenius Medical Care, St Wendel, Germany), except for three patients, who were dialysed with low-flux HD (Fx10, Helixone; Fresenius Medical Care, St Wendel, Germany; or cellulose triacetate filters CT190G; Baxter, McGaw Park, Illinois, USA).
Systemic complement activation in PD patients
Plasma levels of sC5b-9 were significantly higher (p < 0.001) in PD patients compared to healthy controls, non-dialysis CKD patients and HD patients (Figure 1(a)). Mean sC5b-9 levels in PD patients were 219 ± 147 ng/mL, whereas levels in healthy controls, CKD patients and HD patients were 24 ± 20, 45 ± 23, 60 ± 37 ng/mL (pre-HD) and 76 ± 34 (post-HD), respectively. In PD patients, plasma sC5b-9 levels were not influenced by age, and no significant sex-related differences were found (Online Supplemental Figure S1). A trend was seen for a positive correlation between plasma C5b-9 levels and BMI in PD patients (r = 0.23, p = 0.09), while dialysis vintage did not seem to impact systemic complement activation in the cross-sectional samples (Online Supplemental Figure S1). To discriminate between vintage time-related changes and a survival-related bias, sC5b-9 levels were also assessed in three consecutive samples in a subgroup of PD patients (n = 16). A significant increase over time was found for plasma C5b-9 levels in PD patients (Figure 1(b), p = 0.047). Among the baseline clinical data, no major determinants were found for plasma C5b-9 levels in PD patients (Online Supplemental Table S1). Hence, altogether these results suggest that systemic complement activation in PD patients cannot be attributed to CKD-related factors or dialysis in general. In addition, systemic complement activation seems to increase over time providing further indication that PD might induce systemic complement activation.
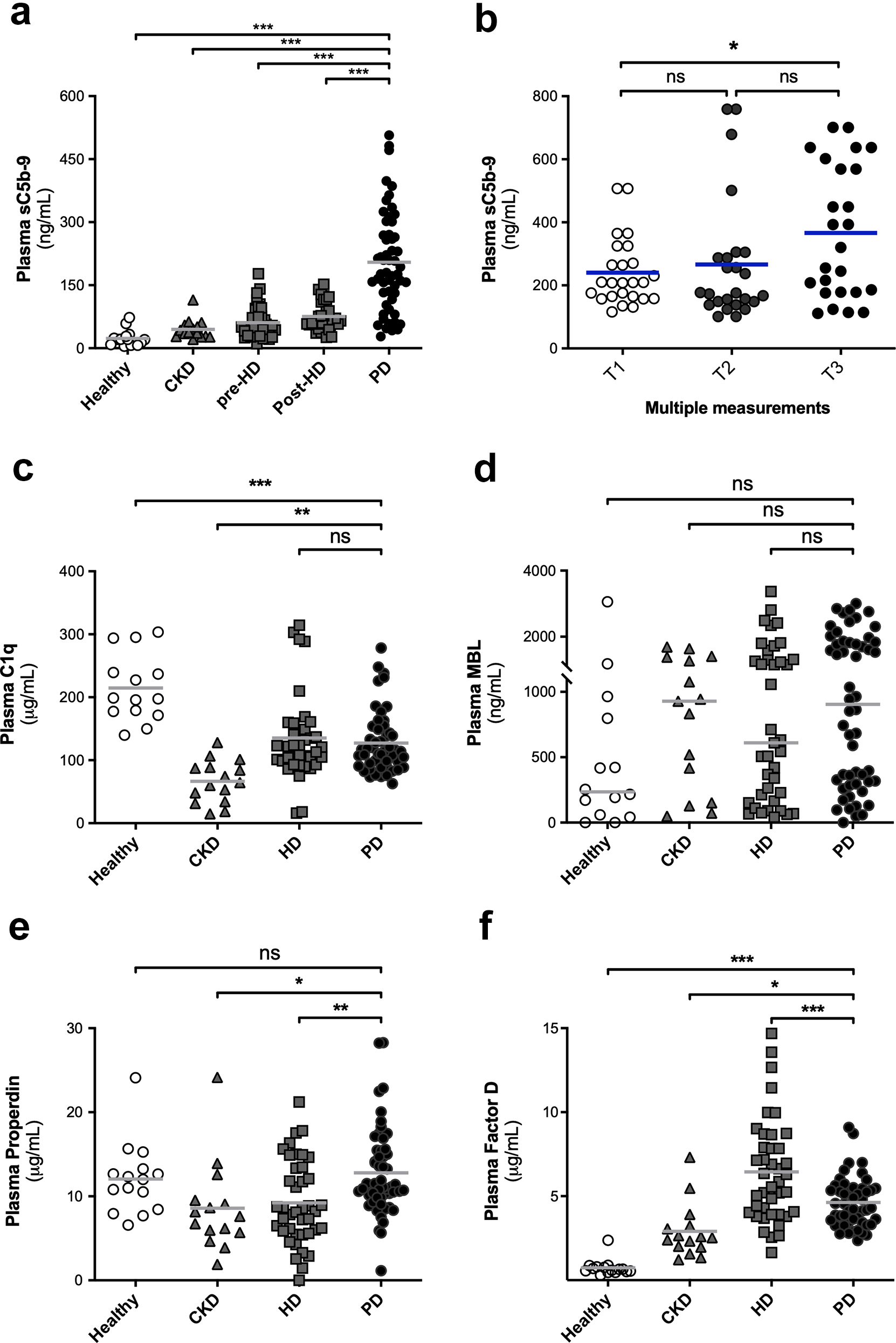
Plasma levels of (a) soluble C5b-9 (sC5b-9) in sex- and age-matched healthy controls (n = 14), non-dialysis dependent CKD patients (n = 15), HD patients prior to dialysis (n = 41) and PD patients (n = 55). (b) Scatter plot is shown for plasma sC5-9 levels in PD patients in three consecutive samples. Plasma levels of (c) C1q, (d) MBL, (e) Properdin and (f) Factor D in PD patients and controls. Data are presented as mean (for sC5b-9, C1q, Properdin and Factor D) or median (for MBL) and were analysed by one-way ANOVA with the Dunnett post hoc test or Kruskal–Wallis test with an option for multiple comparisons using PD as the control group (***p < 0.001). Significant differences were found for sC5b-9, C1q, Properdin and Factor D levels between PD patients and controls. sC5b-9: soluble membrane attack complex; CKD: chronic kidney disease; HD: haemodialysis; PD: peritoneal dialysis; MBL: mannose-binding lectin.
PD-associated systemic complement activation is linked to the classical pathway
To assess the pathway responsible for complement activation in PD, plasma levels of C1q, MBL, Properdin and Factor D were measured and compared to controls. Plasma levels of C1q were similar between PD and HD patients (p = 0.87) but significantly lower than healthy controls (Figure 1(c), p < 0.001). No differences were found in plasma MBL levels among the different groups (Figure 1(d)). Interestingly, plasma levels of Properdin were not significantly different between PD patients and healthy controls (p = 0.95), whereas levels were significantly higher compared to HD and CKD patients (Figure 1(e), p = 0.003 and p = 0.02, respectively). Plasma Factor D levels were the highest in the HD group, but levels in PD patients were significantly higher compared to CKD patients and healthy controls (Figure 1(f)).
In PD patients, systemic complement activation positively correlated with C1q levels (Figure 2(a), r = 0.55, p < 0.001) and C3d/C3-ratio (Figure 2(b), r = 0.56, p < 0.001). Accordingly, plasma C1q and C3d/C3-ratio were independent determinants of plasma sC5b-9 in our multivariable linear regression model (Table 1). This model explained 44% of the variation in plasma C5b-9 levels in PD patients. Taken together, our data indicate that the CP is possibly the pathway responsible for systemic complement activation in PD.
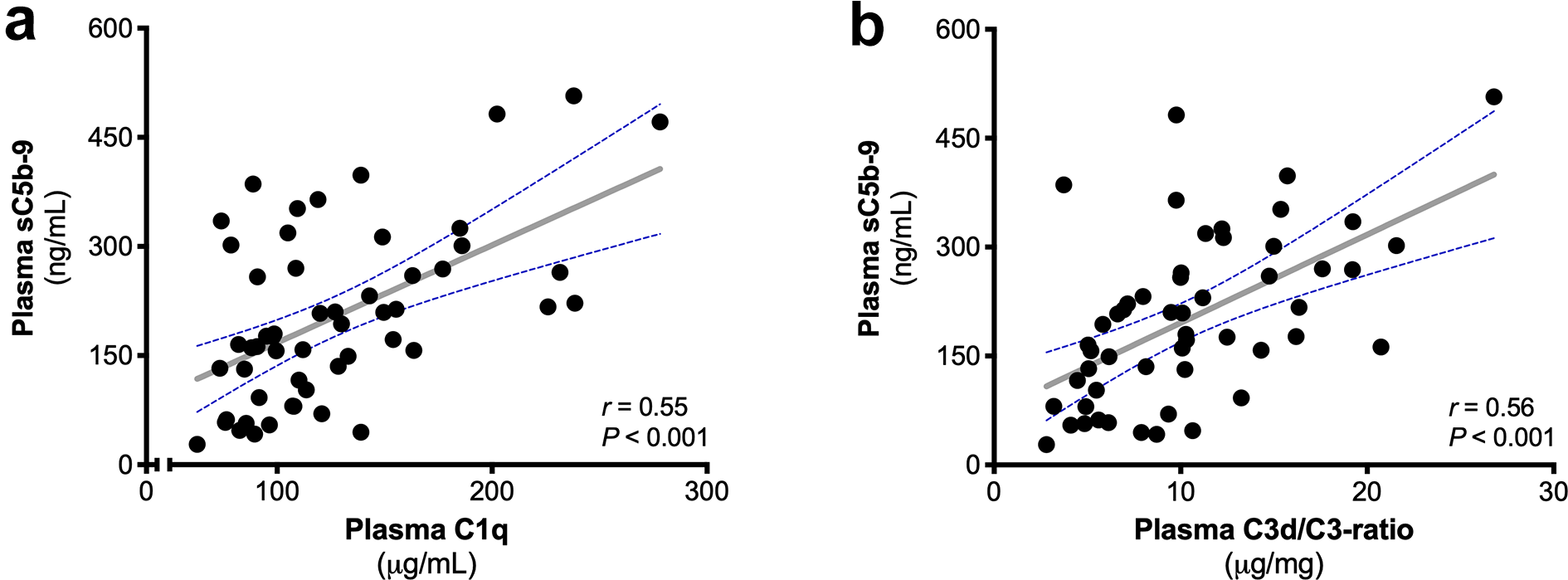
Classical pathway and C3 activation are correlated with systemic complement activation. The correlation between plasma soluble C5b-9 (sC5b-9) and (a) plasma C1q as well as (b) the C3d/C3-ratio using the Pearson correlation coefficient (r represents the Pearson’s rho). The dashed blue lines show the 95% confidence interval for the regression line (grey). A significant correlation was found between the plasma levels of sC5b-9 and C1q as well as between plasma levels of sC5b-9 and C3d/C3-ratio.
The association of complement components with soluble C5b-9 in plasma of peritoneal dialysis patients.a
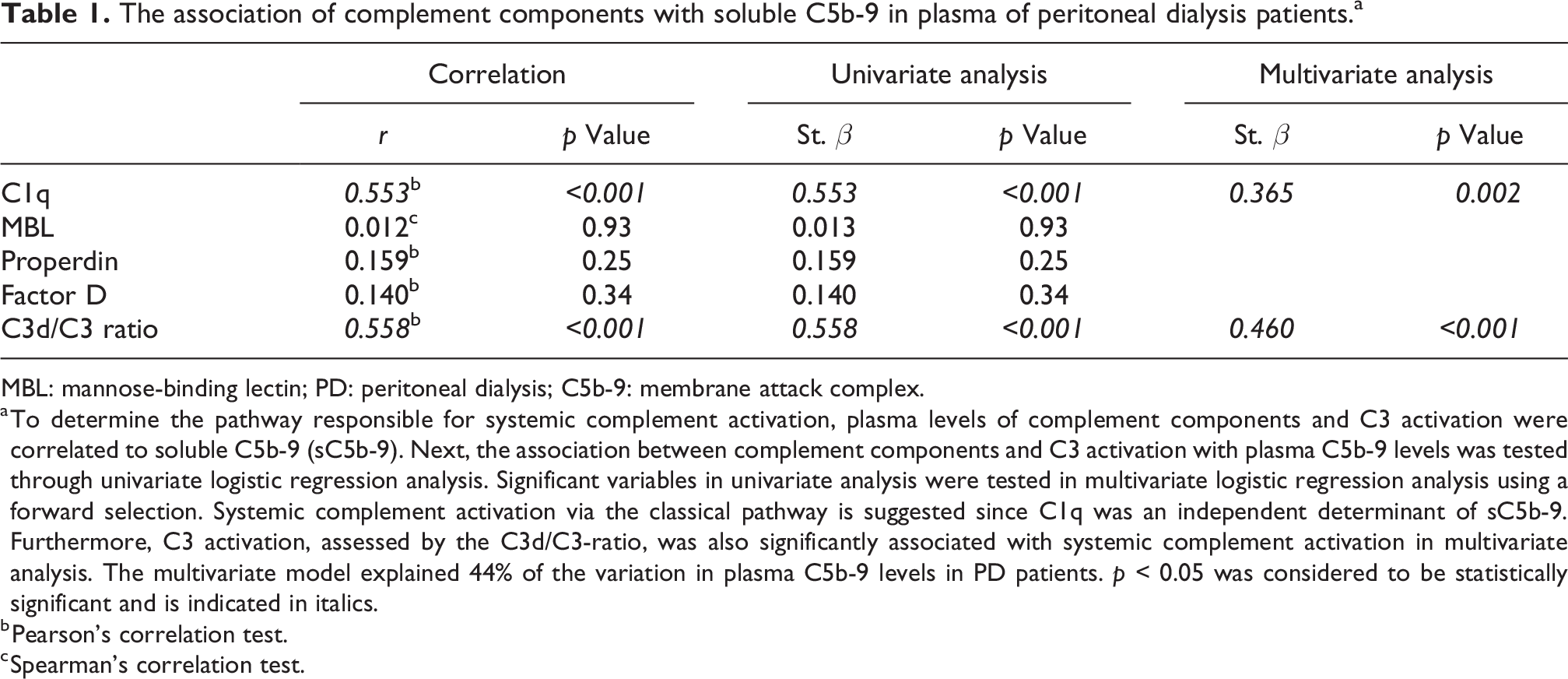
MBL: mannose-binding lectin; PD: peritoneal dialysis; C5b-9: membrane attack complex.
a To determine the pathway responsible for systemic complement activation, plasma levels of complement components and C3 activation were correlated to soluble C5b-9 (sC5b-9). Next, the association between complement components and C3 activation with plasma C5b-9 levels was tested through univariate logistic regression analysis. Significant variables in univariate analysis were tested in multivariate logistic regression analysis using a forward selection. Systemic complement activation via the classical pathway is suggested since C1q was an independent determinant of sC5b-9. Furthermore, C3 activation, assessed by the C3d/C3-ratio, was also significantly associated with systemic complement activation in multivariate analysis. The multivariate model explained 44% of the variation in plasma C5b-9 levels in PD patients. p < 0.05 was considered to be statistically significant and is indicated in italics.
b Pearson’s correlation test.
c Spearman’s correlation test.
Local complement activation in PD patients seems independent from systemic activation
The presence of sC5b-9 was detected in the PDF of all patients with median levels of 69 ng/mL [35–89] (Figure 3(a)), and the median value was 33% of the levels in matched plasma (Figure 3(b)). Considering the high molecular weight of sC5b-9 (>1000 kDa), the levels detected in the dialysate, therefore, arise from local generation. In conformity, plasma levels of sC5b-9 did not correlate with sC5b-9 levels in the PDF (Figure 3(c), r = 0.03, p = 0.83). PDF sC5b-9 levels weakly, but significantly, correlated with age (Online Supplemental Figure S2, r = 0.28, p = 0.04). No significant sex-related differences were found for sC5b-9 in the PDF (p = 0.64), and PDF sC5b-9 levels were also not influenced by BMI (Online Supplemental Figure S2, p = 0.81). A negative correlation was seen between PDF C5b-9 levels and dialysis vintage in PD patients (r = −0.30, p = 0.03), and consecutive samples in a subgroup of PD patients showed a similar trend towards lower levels over time (Online Supplemental Figure S2). Among the baseline clinical variables analysed, no significant determinants were found for PDF C5b-9 levels (Online Supplemental Table S3). These data indicate that PD might lead to local complement activation. Furthermore, it appears that this process might be separate from the systemic complement activation seen in PD.
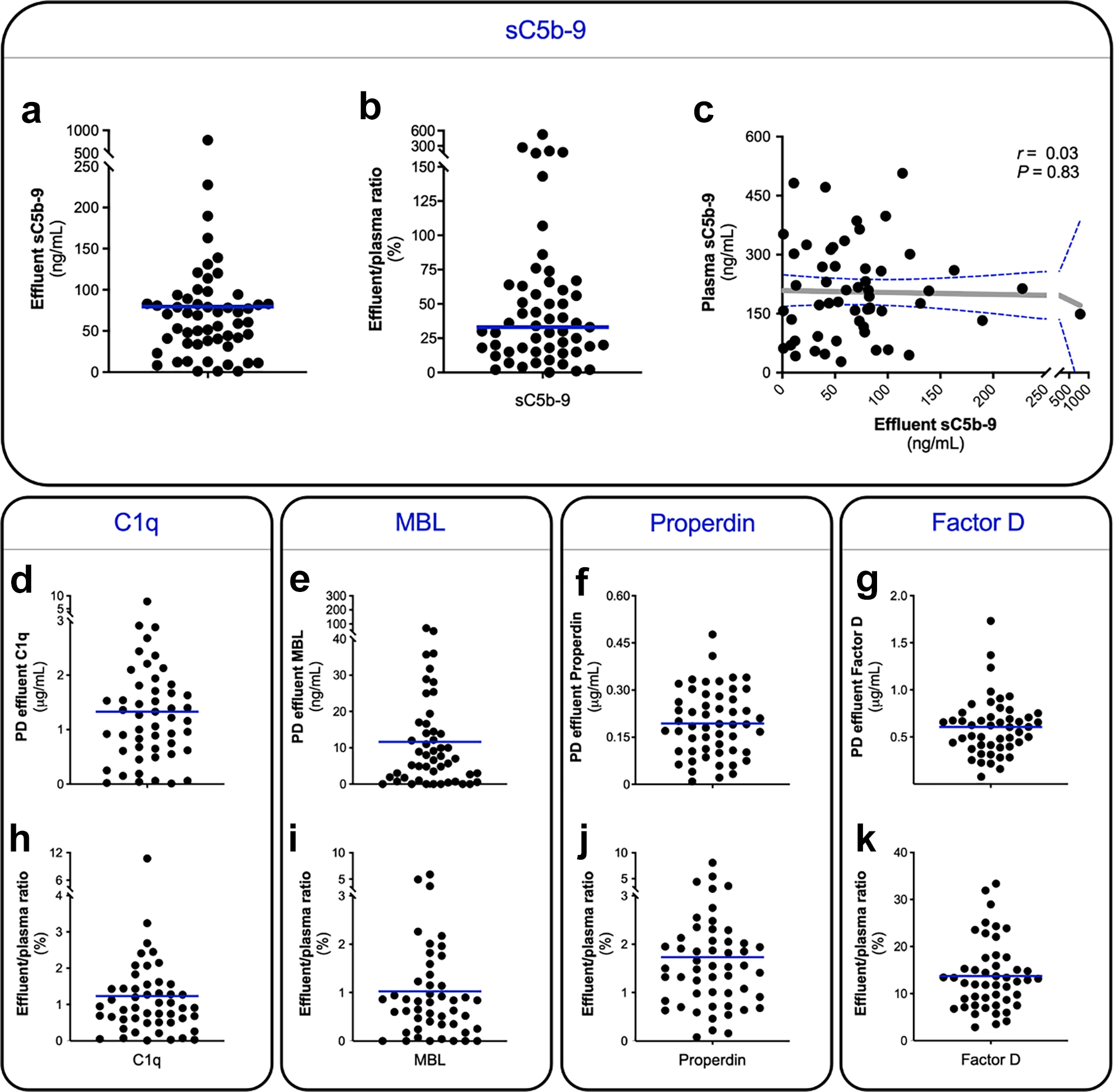
Scatter plot is shown for (a) soluble C5b-9 (sC5b-9) levels in the PDF and (b) the dialysate-to-plasma ratio of sC5b-9 in percentage. The dialysate–plasma ratio was calculated per patient by dividing the PDF level by the plasma level and then multiplied by 100%. The blue horizontal lines represent the median. (c) The correlation between plasma and PDF levels sC5b-9 using the spearman correlation rank coefficient (r represents the spearman’s rho). The dashed blue lines show the 95% confidence interval for the regression line (grey). Scatter plots are shown for PDF levels of (d) C1q, (e) MBL, (f) Properdin and (g) Factor D, as well as for the dialysate-to-plasma ratio of (h) C1q, (i) MBL, (j) Properdin and (k) Factor D.
Local complement activation in PD is linked to the alternative pathway
We next investigated the contribution of the different complement pathways to local complement activation. C1q, MBL and Properdin were all detected in the PDF, although at low levels (Figure 3(d) to (f)). In contrast, PDF Factor D levels were the highest among all complement components (Figure 3(g)). The mean, or median, PDF/plasma ratio for C1q, MBL and Properdin were below 2% (Figure 3(h) to (j)), whereas the average PDF/plasma ratio for Factor D was 14% (Figure 3(k)). To disentangle local production from protein leakage, complement components were correlated to their respective plasma levels and peritoneal protein loss (Table 2). MBL was the only complement component in which PDF levels significantly correlated with plasma levels (r = 0.61, p < 0.001). However, PDF levels of all the other complement components significantly correlated with protein loss. Furthermore, all the complement components significantly correlated with sC5b-9 levels in the PDF (Table 2), with Properdin showing the strongest correlation (Figure 4(a), r = 0.63, p < 0.001). In univariate linear regression analysis, C1q, Factor D and Properdin were shown to be significantly associated with sC5b-9 levels in PDF (Online Supplemental Table S4). However, in the multivariate regression analysis, only Properdin was an independent determinant of PDF sC5b-9 levels (St. β = 0.597, p < 0.001). In conclusion, our data suggest that PD results in the leakage of plasma complement components to the PDF, which subsequently could trigger local complement activation via the AP.
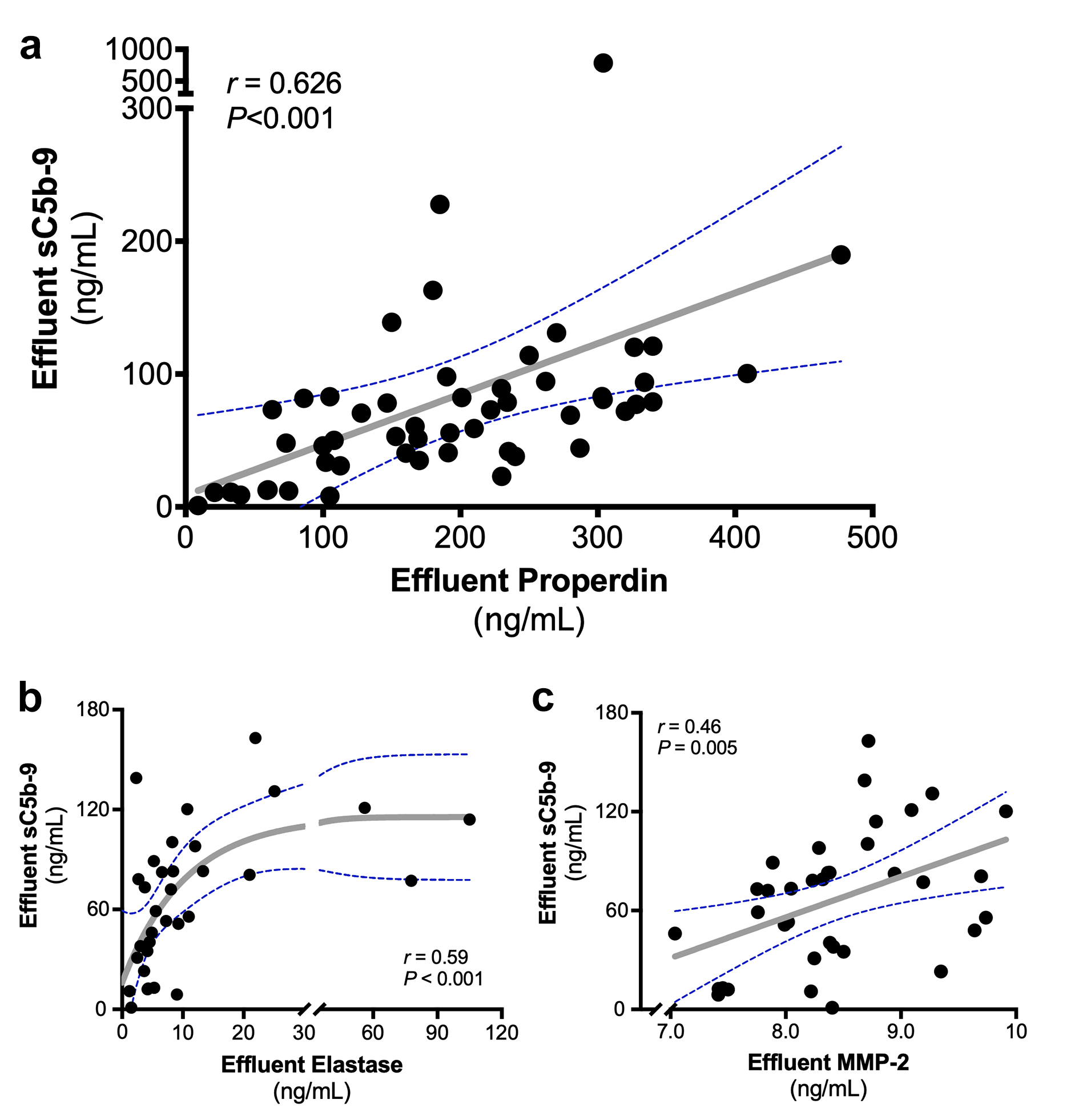
(a) The correlation between PDF levels of soluble C5b-9 (sC5b-9) and Properdin using the Spearman Rank correlation coefficient (r represents the spearman’s rho). The correlation between PDF levels of sC5b-9 with (b) elastase and (c) MMP-2 using the Spearman correlation coefficient (r represents the Spearman’s rho). The dashed blue lines show the 95% confidence interval for the regression line (grey).
The correlation between complement component levels in the peritoneal dialysate fluid and corresponding plasma levels and peritoneal protein loss.a
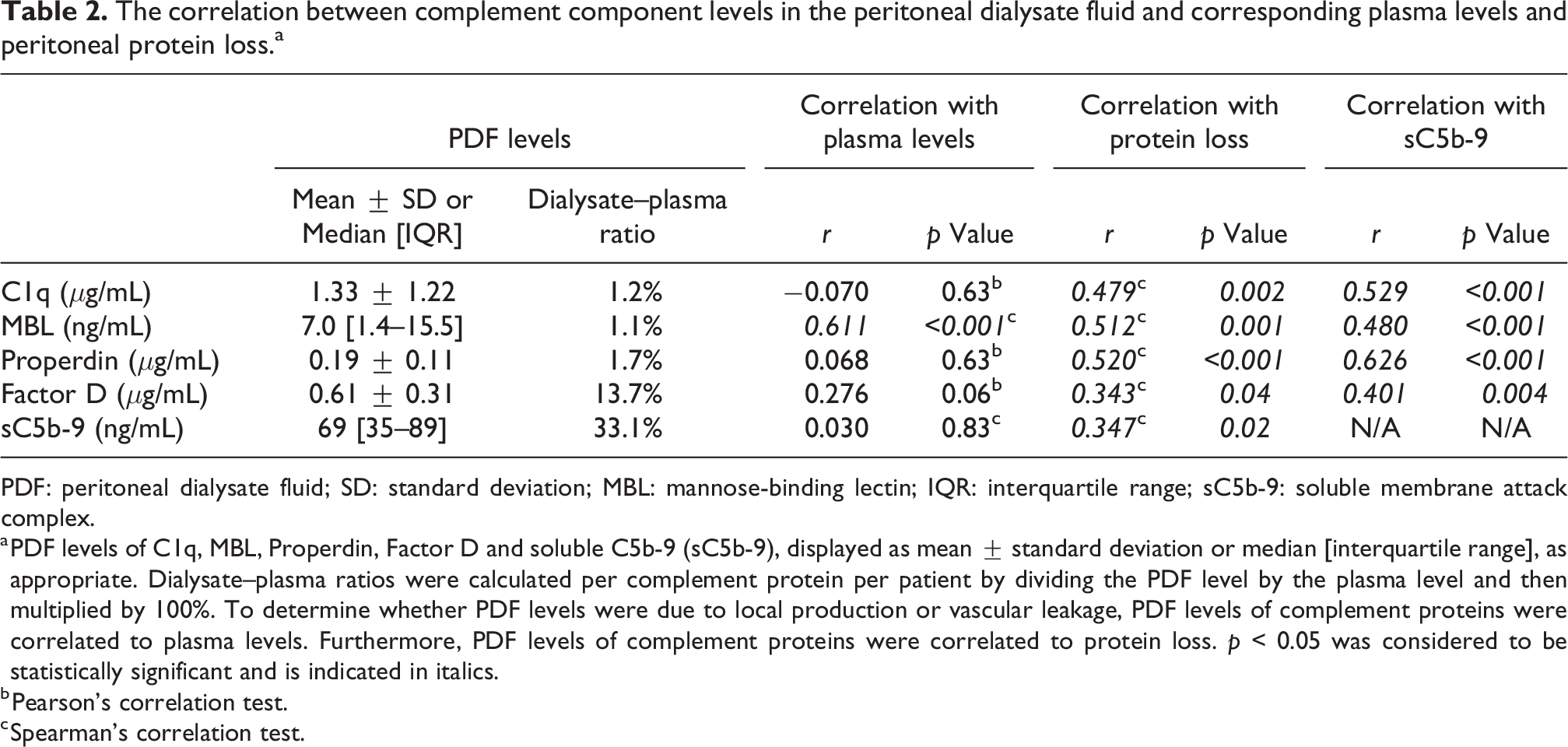
PDF: peritoneal dialysate fluid; SD: standard deviation; MBL: mannose-binding lectin; IQR: interquartile range; sC5b-9: soluble membrane attack complex.
a PDF levels of C1q, MBL, Properdin, Factor D and soluble C5b-9 (sC5b-9), displayed as mean ± standard deviation or median [interquartile range], as appropriate. Dialysate–plasma ratios were calculated per complement protein per patient by dividing the PDF level by the plasma level and then multiplied by 100%. To determine whether PDF levels were due to local production or vascular leakage, PDF levels of complement proteins were correlated to plasma levels. Furthermore, PDF levels of complement proteins were correlated to protein loss. p < 0.05 was considered to be statistically significant and is indicated in italics.
b Pearson’s correlation test.
c Spearman’s correlation test.
Local complement activation is linked to leukocyte activation and tissue remodelling
To evaluate the effect of local complement activation on inflammation and tissue remodelling, we determined elastase, MPO, IL-6 and MMP-2 PDF levels in a subgroup of PD patients due to limited sample availability (Online Supplemental Table S1, n = 35). PDF levels of sC5b-9 correlated with PDF levels of elastase (r = 0.59, p < 0.001) and MPO levels (Figure 4(b), r = 0.45, p = 0.009). Interestingly, a non-linear correlation was observed. Furthermore, sC5b-9 in the PDF significantly correlated with PDF levels of MMP-2 (Figure 4(c), r = 0.46, p = 0.005). No significant correlation was found between PDF levels of IL-6 and sC5b-9 (r = 0.25, p = 0.14). To summarise, local complement activation is associated with markers of leukocyte activation and tissue remodelling.
Discussion
Several modalities of kidney replacement therapy such as kidney transplantation and HD are known to induce complement activation. Yet, our understanding of the complement system in PD remains limited. Previous studies suggest that PD is characterised by local, intraperitoneal, complement activation in response to continuous exposure to bioincompatible dialysis solutions. In the current study, we report that PD patients have both systemic and local complement activation. We observed substantial and extensive complement activation in plasma of PD patients, which was significantly higher than that seen in healthy controls, CKD and HD patients. In addition, all PD patients had significant levels of local complement activation in the PDF. Further analysis suggested that systemic activation was linked to the CP, while local activation was linked to the AP. We, therefore, propose that the PD procedure itself aggravates systemic complement activation, while inducing local complement activation, and that these processes are uncoupled and triggered via distinct pathways.
More than 20 years ago, Young et al. already showed that PD patients had higher systemic levels of sC5b-9 compared to healthy controls. 15 However, it was unknown whether the systemic complement activation seen in PD patients was due to CKD-related factors or caused by the PD procedure itself. Subsequently, Reddingius et al. compared levels of complement activation products between paediatric patients on PD, paediatric CKD patients as well as healthy controls and concluded that the uremic state itself and not the dialysis technique was responsible for complement activation. 9 Our data do not support this assumption, since we found that plasma sC5b-9 levels in PD patients were significantly higher than those in CKD patients and HD patients. However, it is important to note that the study by Reddingius et al. studied paediatric patients (median age: 8 years), whereas our study was performed in an adult population (mean age: 57 years). Age has previously been shown to influence both complement activity and levels. 16 Secondly, these children had been on PD much longer than our adult population. Thirdly, the CKD stage of the patients could influence the results, since Reddingius et al. only included paediatric patients with an advanced CKD stage, whereas in our study a broader range of CKD patients were included. 9 Thus, our CKD population might be less severely ill and, therefore, have lower plasma sC5b-9 levels. Yet, the higher systemic complement activation seen in PD compared to HD in our study contradicts this argument.
Previously, systemic complement activation was assumed to occur via AP, since systemic Bb levels were higher. 15 However, considering the role of the AP as an amplification route in CP-induced complement activation, this conclusion might have been preliminary. 17 Later studies suggested a role for the CP since PD patients had significantly higher levels of C1q, C4 and C3d compared to healthy controls. 9 Recently, Bartosova et al. showed C1q, C3d and C5b-9 deposition in the peritoneal arterioles of PD patients. 18 Moreover, the deposition of C1q and C5b-9 correlated significantly with each other, confirming a possible role for the CP. Although our data suggest that systemic complement activation in PD could be due to CP activation, yet the trigger responsible for this activation is unknown. We speculate that CP-mediated complement activation in PD could be induced by circulating immune complexes, lipoproteins, apoptotic cells or advanced glucose end products that are reabsorbed into the systemic circulation. 19 –21
The paradigm of PD inducing local complement activation was first established by Young et al. who demonstrated high dialysate levels of sC5b-9 (approximately 30%). 15 Other studies have confirmed this. 10,18,22 As observed in our cohort, AP activation was also suggested by Young et al. 15 Previously, Properdin has been described to bind to kidney tubular epithelium and lead to the AP activation 7 , a similar mechanism might therefore exist at the peritoneal mesothelial surface, where Properdin would bind and induce membrane injury. One could argue whether complement components found in the PDF are the result of local production or mainly result from vascular leakage through the peritoneal membrane. We were unable to predict PDF levels based on the three-pore model, due to the varying molecular weight of C1q, MBL and Properdin (Online Supplemental Table S5). However, the PDF/plasma ratio of Factor D (24 kDa) was similar to those predicted by the three-pore model (14.5%) and regression lines (14.6%), 10,23 as well as to ratios found for other molecules of similar size (16% for sCD59 [18–21 kDa]). 11 Furthermore, considering that levels of C1q, MBL and Properdin correlated with protein loss and generally corresponded to 1–2% of plasma levels, vascular leakage seems the most likely source for complement in the PDF. 15 In addition, complement activation has previously been suggested to further enhance protein loss, which may partially explain the correlations between complement levels and protein loss to the PDF. 24 Taken together, complement activation in stable PD patients could be the trigger of a vicious cycle of inflammation and peritoneal damage.
Chronic inflammation in PD patients is linked to clinical outcomes. 25,26 Lambie et al. demonstrated that systemic and local inflammation are independent processes and impact clinical outcome differently. 23 Systemic IL-6 is associated with patient survival, while local IL-6 predicts the peritoneal solute transport rate. 27 Taking into account the differences we found in this study between systemic and local complement activation, we speculate that like IL-6, local and systemic complement activation have different clinical implications. Recently, Bartosova et al. showed that systemic PD-induced complement activation correlated with the severity of vascular damage. 18 Furthermore, complement activation in the vessels was associated with the degree of vasculopathy in these PD patients. 18 Locally, the continuous complement activation in PDF could contribute to peritoneal tissue damage. 22 In line with these findings, we observed that local complement was associated with tissue remodelling enzymes.
The link between tissue remodelling and complement is relatively new; nonetheless, recent data demonstrated that complement activation can stimulate the progression to fibrosis after injury. 28 In certain PD patients, chronic peritoneal inflammation is believed to promote epithelial-to-mesenchymal transition of the mesothelial cells, leading to peritoneal fibrosis and sclerosis. 29 In other diseases, it has been demonstrated that complement induces epithelial-to-mesenchymal transition, predominantly via C5a/C5a-receptor interactions. 30 –32 We found a correlation of local complement activation with elastase, MPO and MMP-2. In PD, intra-peritoneal elastase is a specific marker of neutrophil activation and peritoneal leukocyte count, whereas MPO levels have been associated with long-term outcome. 33 –35 Furthermore, neutrophils have been proposed to play an important role in the pathogenesis of peritoneal fibrosis. 36 MMP-2 is involved in peritoneal membrane injury in PD and is associated with a fast transport status, peritonitis, as well as encapsulating peritoneal sclerosis. 37,38 In accordance with our study, Zavvos et al. identified complement components of the AP as biomarkers of encapsulating peritoneal sclerosis in their prospective proteomics study. 39 Altogether, the results from our study strengthen the hypothesis that the complement system could be involved in the development of peritoneal fibrosis in PD.
The findings in the current study, while interesting and potentially very important, should be viewed as preliminary and require further corroboration as well as mechanistic studies. Additionally, our results should be considered in the light of certain limitations. Firstly, while we believe the associations presented here to be causal, but the observational nature of our study does not allow for definitive establishment of causality. Secondly, our overall population size is relatively small, and the relationship between complement activation and clinical outcome (such as peritoneal membrane failure) was not assessed. Thirdly, although we sought to unravel the impact of hemodynamic factors on complement activation in PD patients, such as vascular leakage and reduced kidney clearance, we must acknowledge that they cannot be entirely ruled out as potential confounding variables. Nevertheless, according to the knowledge of the authors, the current study has the largest sample of adult PD patients in whom a comprehensive study of the complement system has been performed. However, other biomarkers were only measured in a subgroup and did not include other established markers such as transforming growth factor beta (TGF-β). Alternatively, strengths include the multiple control groups (HD, CKD and healthy subjects), the extensive complement analysis, the combination of local and systemic measurements as well as the longitudinal data.
In conclusion, despite significant improvements in the biocompatibility of PD solutions during recent years, the current study indicates both systemic and local complement activation in PD patients through seemingly distinct pathways, suggesting that these processes might be independent.
Supplemental material
Supplemental Material, sj-docx-1-ptd-10.1177_08968608231198984 - Systemic and local complement activation in peritoneal dialysis patients via conceivably distinct pathways
Supplemental Material, sj-docx-1-ptd-10.1177_08968608231198984 for Systemic and local complement activation in peritoneal dialysis patients via conceivably distinct pathways by Bernardo Faria, Mariana Gaya da Costa, Anita H Meter-Arkema, Stefan P Berger, Carla Lima, Catia Pêgo, Jacob van den Born, Casper FM Franssen, Mohamed R Daha, Manuel Pestana, Marc A Seelen and Felix Poppelaars in Peritoneal Dialysis International
Footnotes
Authors’ note
BF and MGC are shared first authors.
Acknowledgements
None.
Author contributions
BF, MAS, and FP researched the literature, conceived the study, and were involved in protocol development. BF was involved in gaining ethical approval and patient recruitment. All authors were involved in the data analysis. BF, MGC and FP wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FP currently owns or owned stock in ChemoCentryx, Apellis Pharmaceuticals and Omeros Corporation. The other authors declared no conflict of interest.
Ethical approval
The study was approved by the local ethical committee and performed according to the principles of the declaration of Helsinki.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent to participate
Written informed consent was obtained from all subjects before the study.
Informed consent to publish
None.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
