Abstract
Children with kidney failure who receive maintenance peritoneal dialysis (PD) are at increased risk for thyroid dysfunction. A poorly appreciated cause of hypothyroidism related to PD is iodine overload from exposure to iodine-containing cleaning solutions, iodinated contrast agents or povidone–iodine-containing PD caps, particularly in infants and small children. An international survey was conducted to understand current practices regarding iodine exposure in PD patients, the frequency of iodine-induced hypothyroidism (IIH) in patients receiving PD, and to assess awareness of this issue among paediatric nephrologists. Eighty-nine paediatric nephrology centres responded to the survey. Hypothyroidism in PD patients was diagnosed in 64% (n = 57) of responding centres, although only 19 of these centres (33%) suspected or diagnosed IIH. Aetiologies of IIH included exposure to povidone–iodine-containing PD caps (53%), cleaning solutions with iodine (37%) and iodinated contrast (10%). While most centres (58%, n = 52) routinely evaluate thyroid function, only 34% (n = 30) specifically aim to limit iodine exposure. Of centres not routinely evaluating for or utilising methods to prevent iodine exposure and hypothyroidism, 81% reported being unaware of the risk of IIH in PD patients. Hypothyroidism is diagnosed in a substantial percentage of paediatric PD programmes internationally. Increased education on the risk of iodine exposure in children receiving PD may decrease the incidence of IIH as an aetiology of hypothyroidism.
Introduction
Iodine is an essential micro-nutrient required for thyroid hormone synthesis. Both deficiency and excess (Wolff–Chaikoff effect) can cause hypothyroidism. 1 Hypothyroidism is underrecognised in children with chronic kidney disease (CKD) and kidney failure. It can be due to various aetiologies such as decreased response to thyrotropin-releasing hormone, the reduced affinity of thyroid hormones to thyroid-binding proteins (TBPs), loss of TBPs in the dialysis effluent, inflammation, malnutrition, mineral deficiencies and metabolic acidosis. 2 –6 Patients with CKD have a high prevalence of hypothyroidism, which increases with declining kidney function. 7 Similarly, the prevalence of subclinical hypothyroidism can range from 15% to 27% in adult patients receiving automated peritoneal dialysis (APD), a frequency that is at least three times higher than in the general population. 3,8
An underappreciated cause of hypothyroidism is iodine-induced hypothyroidism (IIH) in children receiving APD. The exposure to iodine may arise from povidone–iodine-containing PD caps, iodine-containing cleaning solutions 9 or iodinated contrast agents. 10 In paediatric patients receiving APD, IIH is particularly prominent during infancy and early childhood. These children are more likely to have a low initial drain volume due to a small last fill volume and residual kidney function, the combination of which can lead to povidone–iodine from the cap remaining in the tubing and being pushed back into the peritoneal cavity with the first fill. 11 After identifying four cases of IIH in children due to iodine exposure from PD caps in our institutions, 12 we conducted an international survey to gather information on current practices regarding potential iodine exposure in PD patients, the frequency of IIH, and to assess awareness of this issue among paediatric nephrologists.
Methods
A survey (Supplementary Table 1) was disseminated to paediatric nephrology centres worldwide through the International Pediatric Nephrology Association (IPNA) membership using Research Electronic Data Capture (REDCap) electronic data capture tools hosted at Children’s Mercy Kansas City after IRB approval. 13,14 The collected data included the size of the PD programme, the ages of the patient population, diagnosis of hypothyroidism, specifically IIH, diagnostic tests performed, treatment and what preventive measures, if any, are used to decrease the risk of IIH in children receiving PD. The survey was distributed in October 2020. Reminder emails were sent to members every 2 weeks, and the survey was redacted in December 2020. The results were descriptive, and thus no formal statistical analysis was conducted. Only surveys with 90% completion were included. Duplicate surveys from the same centre were excluded.
Results
There were 128 responses in total. Thirty-nine survey results were excluded due to incomplete responses (n = 33) and multiple responses from a single centre (n = 6), resulting in data from 89 centres included in the study. The largest percentage of centres were from Europe (30%, n = 27), followed by North America (27%, n = 23), Asia (25%, n = 22), South America (13%, n = 12), Africa (3%, n = 2) and Australia (3%, n = 2) (Table 1). Most centres (62%, n = 55) had small PD programmes with less than 10 patients at the time of the survey. APD was the predominant PD modality in 71% (n = 63) of the centres.
Demographics and characteristics of patients on peritoneal dialysis – all participating centres.
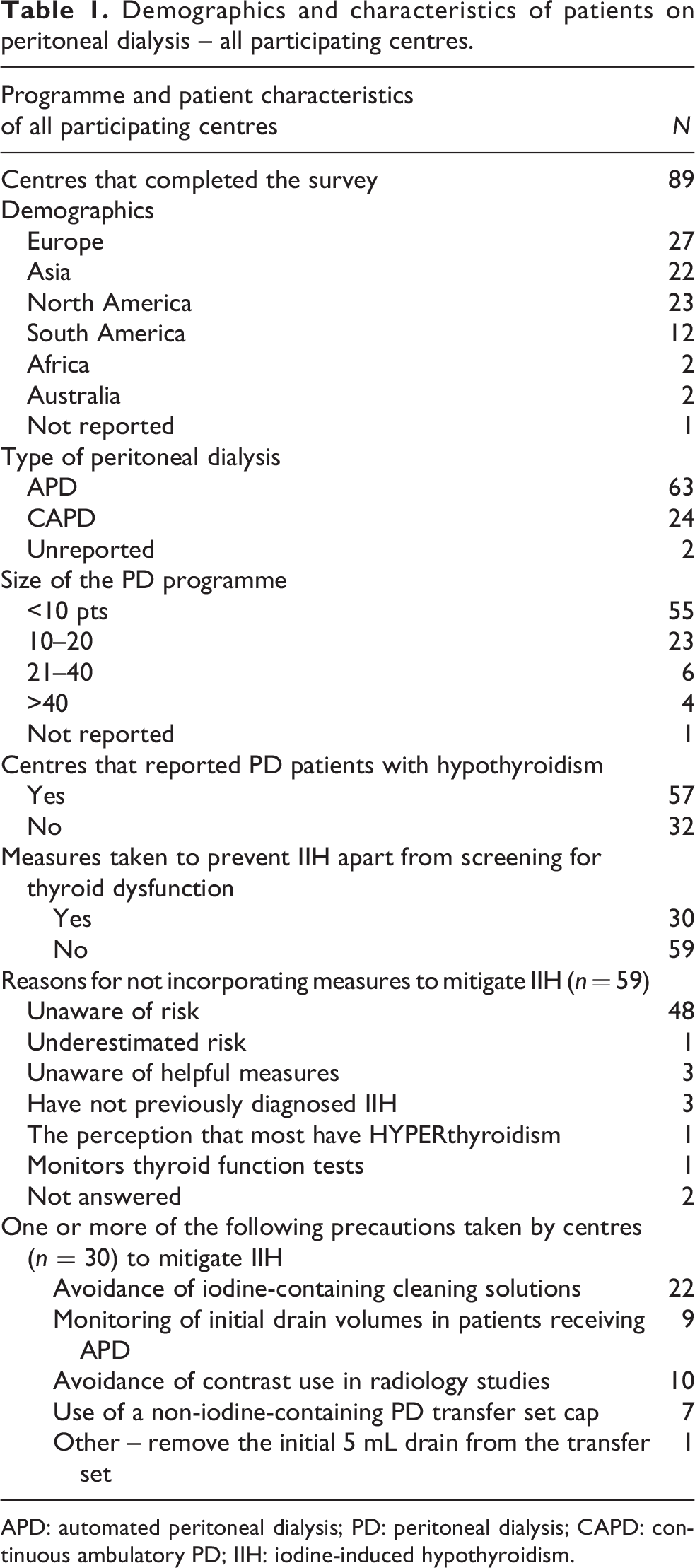
APD: automated peritoneal dialysis; PD: peritoneal dialysis; CAPD: continuous ambulatory PD; IIH: iodine-induced hypothyroidism.
Although hypothyroidism in PD patients had previously been diagnosed in 64% (n = 57) of the responding centres, only one-third of those centres (n = 19) had ever considered or diagnosed IIH. Most centres (58%, n = 52) reported routinely evaluating thyroid function in their PD patients every 6–12 months or based on the clinical suspicion of hypothyroidism. In contrast, only 29 of the 52 centres routinely aim to limit iodine exposure by following one or more of the following measures: avoidance of iodine-containing cleaning solutions (n = 22) and radiology contrast agents (n = 10), monitoring initial PD drain volume (n = 9), use of a non-iodine-containing PD transfer set cap (n = 7) or removal of 5 mL effluent drain volume prior to cycler connection (n = 1). Of centres not routinely evaluating for or utilising methods to prevent iodine exposure, 81% reported being unaware of the risk of IIH in paediatric PD patients.
In the 19 centres with IIH (Table 2), aetiologies included exposure to povidone–iodine-containing PD caps (53%, n = 10), cleaning solutions with iodine (37%, n = 7) and iodinated contrast agents (10%, n = 2). Twelve of the 19 centres reported that they had measured iodine levels in the blood, urine, PD fluid or a combination thereof, and 7 centres were unsure what specimens were used for diagnosis. Twelve of 19 centres reported that their patients received thyroxine supplementation for variable periods. Seven of the 12 patients were subsequently able to discontinue thyroxine at the time of this survey. Sixteen of 19 centres reported subsequently instituting preventive measures to limit patient exposure to iodine.
Diagnosis and management of iodine-induced hypothyroidism in the reporting centres.

APD: automated peritoneal dialysis; PD: peritoneal dialysis; IIH: iodine-induced hypothyroidism.
Discussion
Hypothyroidism in PD patients was diagnosed in two-thirds of responding centres, and less than one-third suspected or diagnosed IIH. In addition, less than 60% of centres that responded to our survey reported routine screening for hypothyroidism in their children receiving PD. This highlights the need for more awareness of this complication, despite previously published paediatric data (case reports) on the subject. 3,5,12,15 The lack of a clear-cut definition of the at-risk population and the absence of screening guidelines also probably contributes to the variable practice. Whereas a literature search identified a prior case series of 10 children receiving PD with IIH in which it was hypothesised that the hypothyroidism could be due to pituitary dysfunction, 16 given our survey data, it is possible that a few patients in that study could have had IIH secondary to the use of povidone–iodine PD caps. 11,15 Although ingesting iodised salt typically does not cause IIH, excessive consumption of foods like seaweed or betadine gargling solutions can cause IIH in vulnerable populations. 17
Although 19 centres in our study considered IIH the cause of hypothyroidism in their patients, only 12 measured iodine in various specimens. This is not surprising as many laboratories in non-academic centres or centres with limited resources (e.g. absence of laboratory availability, cost) may be unable to measure iodine levels. In all children with hypothyroidism who are on APD, the diagnosis of IIH should be considered. Serial serum and urine levels (for the non-anuric patient) of iodine should be measured for confirmation, particularly if no other aetiology has been identified. A presumed diagnosis can be made if there is evidence of exposure and laboratory confirmation is not possible. Where available, commercial labs such as Quest and LabCorp can determine the concentration of iodine in urine and serum within several days. Cut-off values for normal levels of iodine in serum and urine are <100 μg/L and <300 μg/L, respectively, with values that exceed these levels supportive of exposure. The urinary clearance of iodine is decreased in patients with severe CKD, so alone may not accurately reflect the exposure and systemic burden of iodine. 18,19 Thus, both serum and urine iodine levels should always be obtained whenever possible.
IIH is reversible, with the reversal dependent on identifying and removing the source of iodine exposure. Temporary use of thyroid replacement therapy may also be indicated upon diagnosis. Nearly two-thirds of the 19 centres that responded to our survey and that experienced IIH provided thyroxine supplementation to their affected patients. In many cases, treatment with thyroxine was discontinued during follow-up. Restricting iodine exposure can revert thyroid dysfunction in many patients and either avoid the need for thyroxine supplementation or minimise treatment duration. 12,20
As noted in our previously published patient experience, IIH is primarily a problem in infants with low fill volumes and low initial drains; measures such as increasing the last fill volume of the PD prescription in infants and young children to at least 60 mL (tubing volume) for low recirculation volume cassettes and 100 mL for larger volume cassettes, 12 removing the iodine-tinged 5–10 mL of initial drain effluent prior to before connecting to the cycler, avoiding iodine-containing antiseptic solutions and reducing the dose and use of iodinated contrast agents, may prevent or resolve IIH until non-iodinated PD caps are widely available.
This is the largest collection of data to date regarding the diagnosis of IIH in infants and young children on APD, a strength made possible by including responses from centres around the globe through their IPNA membership. Similar participation in the number of centres from Europe, North America and Asia and moderate participation from South America suggest that these data could be generalised to much of the population worldwide. The nature of a survey study and the possibility of recall bias are potential weaknesses of the study.
Conclusion
Hypothyroidism secondary to iodine exposure is a complication to which infants and young children receiving PD are particularly vulnerable. Awareness of the disorder and the application of effective mitigation strategies is poor. Education of the paediatric nephrology community about IIH and the development and institution of standardised screening recommendations in the high-risk patient population is likely to decrease the incidence of the complication.
Supplemental material
Supplemental Material, sj-docx-1-ptd-10.1177_08968608231183853 - Iodine-induced hypothyroidism (IIH) in paediatric patients receiving peritoneal dialysis: Is risk mitigation possible?
Supplemental Material, sj-docx-1-ptd-10.1177_08968608231183853 for Iodine-induced hypothyroidism (IIH) in paediatric patients receiving peritoneal dialysis: Is risk mitigation possible? by Sai Sudha Mannemuddhu, Heather A Morgans and Bradley A Warady in Peritoneal Dialysis International
Footnotes
Acknowledgements
The authors sincerely thank IPNA for distributing the survey and all the PD centres that completed the survey.
Author contributions
All the authors (SSM, HAM, BAW) made a substantial contribution to the concept or design of the work, analysis and interpretation of data. SSM drafted the first draft of the manuscript, and all the authors (SSM, HAM, BAW) revised it critically for important intellectual content and approved the version to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Institutional Review Board (IRB)
The East Tennessee Children’s Hospital (ETCH) IRB – Exempt ETCH#269 – approval1020.
The Children’s Mercy (CM) IRB – Exempt for the study (STUDY00001528).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent to participate
This is not applicable as this is a survey study.
Informed consent to publish
There is no identifiable patient information as this is a survey study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
