Abstract
Background:
The most used PD fluids contain glucose as a primary osmotic agent. Glucose peritoneal absorption during dwell decreases the osmotic gradient of peritoneal fluids and causes undesirable metabolic consequences. Inhibitors of sodium–glucose co-transporter (SGLT) type 2 are wildly used for the treatment of diabetes, heart and kidney failure. Previous attempts to use SGLT2 blockers in experimental peritoneal dialysis yielded contrasting results. We studied whether peritoneal SGLTs blockade may improve ultrafiltration (UF) via partial inhibition of glucose uptake from dialysis fluids.
Methods:
Kidney failure was induced in mice and rats by bilateral ureteral ligation, and dwell was performed by injection of glucose-containing dialysis fluids. The effect of SGLT inhibitors on glucose absorption during fluid dwell and UF was measured in vivo.
Results:
Diffusion of glucose from dialysis fluid into the blood appeared to be sodium-dependent, and blockade of SGLTs by phlorizin and sotagliflozin attenuated blood glucose increment thereby decreasing fluid absorption. Specific SGLT2 inhibitors failed to reduce glucose and fluid absorption from the peritoneal cavity in a rodent kidney failure model.
Conclusions:
Our study suggests that peritoneal non-type 2 SGLTs facilitate glucose diffusion from dialysis solutions, and we propose that limiting glucose reabsorption by specific SGLT inhibitors may emerge as a novel strategy in PD treatment to enhance UF and mitigate the deleterious effects of hyperglycaemia.
Introduction
Peritoneal dialysis (PD) is one of the treatment modalities available for end-stage kidney disease. The advantages of PD include ease-of-access achievement and training, better preservation of residual kidney function, a wide range of regimens available for individualising prescriptions, minimal interference with family/school/social life, physical and mental well-being and others, providing a better impact on PD patients compared to haemodialysis patients. 1,2
PD is typically performed using a commercial peritoneal dialysis fluid (PDF) containing glucose as an osmotic agent and lactate as a buffer. 1 The main limitation of glucose use in PD is its substantial absorption into the blood resulting in mitigation of the serum/dialysate osmotic gradient and, subsequently, decreasing the ultrafiltration (UF) rate. In addition, a high glucose load in PD patients is associated with an accelerated decrease in residual kidney function. 3 Moreover, in most patients, and particularly in diabetic patients, glucose absorption during PD has undesirable metabolic consequences, including hyperosmolar stress, hyperlipidaemia, obesity, nutritional alterations 4 and a high prevalence of metabolic syndrome worsening patient survival. 5 In diabetic patients, glucose load causes further instability of blood glucose levels and a higher insulin demand, leading to the development or progression of diabetic complications. 6
In the last decades, several types of glucose transporters, including sodium–glucose co-transporters (SGLTs), have been discovered in various tissues. 7 Inhibiting glucose reabsorption in the kidney 8 by an SGLT2 inhibitor became a target for diabetes therapy and broadened the spectrum of glucose-lowering agents. There is growing evidence of SGLT2 inhibitor’s role in the prevention of cardiovascular mortality and kidney function deterioration which may be of particular benefit in patients on dialysis. 9
Reports suggest that mechanisms other than simple paracellular diffusion are involved in glucose leakage during PD, namely SGLTs and glucose transporters (GLUTs) responsible for transcellular transport via mesothelial cells. 10,11 Schroppel et al. found that SGLT1 is located on the apical surface of peritoneal mesothelial cells, while GLUTs – on the basolateral surface of these cells. 12 In contrast to a broad knowledge of SGLTs functions in the kidney, little is known about their role in the peritoneum especially in kidney dysfunction and during PD treatment. Recently, Schricker et al. described an increased SGLT2 expression with PD duration, in particular in EPS patients, 13 which demonstrates the contribution of glucose transport to pathological changes in the peritoneal membrane.
We hypothesised that peritoneal SGLTs facilitate the transition of glucose from PDFs into the blood thereby rendering their blockade beneficial for the performance of PD and mitigating mesothelial cell toxicity, as previously shown. 10
In the current study, we used a PD model in uraemic rodents and applied bilateral ureteric ligation. Kidney dysfunction is critical in this experimental setup for two main reasons: the uraemic milieu may impair molecular transport, including the reduction of insulin-stimulated glucose transport in patients with kidney failure. Second, ureteric ligation mitigates the bias of glucosuria on serum glucose levels in the presence of SGLT inhibition.
Methods
Animals
Experiments were conducted after obtaining permission from the Israel Committee for Animal Experiments (IL-42-06-2015). One-month-old CD1 female mice (weight ∼20 g) and male Sprague-Dawley rats weighing 200–250 g were purchased from Envigo (Jerusalem, Israel). Animals were allowed to acclimatise for 1 week prior to the experiments under standard laboratory conditions and were allowed food and water ad libitum with a 12 h fast before operation.
SGLT inhibitors
Phlorizin, a non-selective SGLT inhibitor naturally found in apple tree bark, 14 was purchased from Sigma (Rehovot, Israel) and was administered by subcutaneous (SC) or intraperitoneal (IP) injection 20 mg/kg in mice or 100 µg/ml IP in rats (added to PDF).
Dapagliflozin (Forxiga), a selective SGLT2 inhibitor (Bristol-Myers Squibb with AstraZeneca, UK), was administered by SC injection (10 mg/kg).
Empagliflozin (Jardiance), a selective SGLT2 inhibitor (Ingelheim, Germany), was administered by gavage in mice (10 mg/kg) 15 and in rats (2 mg/kg).
Sotagliflozin (Zynquista), a dual SGLT1 and 2 inhibitor (LX4211, Selleck Chemicals Houston, USA), was administered to rats by gavage or IP (4 and 8 mg/kg)(Table 1).
Design of experimental setup (animals, solutions and SGLT blockers used).

SC: subcutaneous, injected 15 minutes before dialysis; IP: intraperitoneal in dialysis fluid; GV: gavage, 15 min before dialysis.
Model of kidney failure: Bilateral ureteric ligation
Mice: An acute kidney failure model was induced by bilateral ureteric ligation. 16 Briefly, after anaesthesia (intramuscular administration in a hind leg of a mixture of ketamine and xylazine), a 1.5-cm long skin incision was made at the dorsal midline. The psoas muscle was exposed and cut 0.5 cm in length. Through this incision, the kidneys were exposed, and the ureters were identified near the lower pole and ligated 0.3 cm below the kidney pelvis. The kidneys were returned to the place, and the incision of the psoas muscle was sutured. The animals were placed in separate cages and received analgesics per protocol.
Rats: The procedure was similar to mice except for the skin incision of 2.5 cm, psoas muscle cut 0.7 cm in length and ureters were ligated 0.5 cm below the kidney pelvis. In preliminary experiments, bilateral ureter ligation in rats after 24 h caused a significant elevation of blood urea levels to 589.3 ± 13.9 mg/dL (n = 3), consistent with uraemia.
PD model, glucose uptake test and UF assessment
Mice PD model: In most experiments, at 24 h following bilateral ureteric ligation, mice were anesthetised and injected IP with 0.2 ml phlorizin or saline followed by IP injection of 2 ml of PDF (1.5% Dianeal, Baxter Healthcare Corporation Deerfield, IL) diluted 1:4 with normal saline to obtain 0.375% glucose (PDF 0.375 glucose). In a preliminary experiment, we found that the original Dianeal 1.5% solution overwhelmed the effect of SGLT inhibition thereby requiring dilution of Dianeal to reduce the paracellular diffusion fraction and enable measuring the impact of SGLT inhibition on transcellular transport. Blood glucose levels were followed using a standard glucometer Accu-Chek® (Performa, Roche, Indianapolis, USA) by tail bleeding every 15 min for 90–120 min. Glucose was measured, and the area under the curve (AUCs) of blood glucose levels was compared between the experimental groups. In additional experiments, we used SGLT inhibitors (dapagliflozin, empagliflozin and sotagliflozin) instead of phlorizin. To demonstrate sodium-dependent glucose transport, diluted PDF was replaced with 2 ml of a sodium-free solution containing 0.375% glucose in water.
Rats PD model: At 24 h following ureteric ligation, rats were anesthetised, and a peritoneal catheter was inserted IP. Thirty millilitres of preheated to 37°C PDF (1.5% Dianeal, Baxter) with or without phlorizin (100 μg/ml) was injected via the catheter. In the experiments with selective SGLT inhibitors, empagliflozin (2 mg/kg) and sotagliflozin (4 and 8 mg/kg) were given by gavage 15 min before PDF insertion. Additionally, we used sotagliflozin (4 mg/kg) by IP route (in PDF).
The activity of phlorizin, dapagliflozin, empagliflozin and sotagliflozin at the doses and administration modes was confirmed by their ability to induce glucosuria in healthy rodents (not shown).
Dialysate samples were taken at 15, 30, 60, 90 and 120 min after completion of inflow. Inflow, sampling and outflow of the effluent during the experiment were performed via a catheter (Bard Navarre™ Opti-Drain™ Multi-Use Drainage Catheter, Becton, Dickinson, and Company, Franklin Lakes, USA), which was inserted intraperitoneally just behind the xiphoid process of the median line of the abdomen by trocar insertion with the tip located in the pelvis. Peritoneal fluid (0.5 ml) was obtained for each intermediate sampling by a syringe flushed twice to avoid a dead space effect. Tail blood glucose levels were measured by standard glucometer Accu-Chek® (Performa) every 15 min for 120 min. Dialysate samples were tested for glucose concentration in our hospital biochemistry laboratory. At the end of dialysis, fluid was drained, rats were euthanized, dissected and the residual peritoneal dialysate was absorbed with pre-weighed lignin pads. For UF assessment, rats were weighed before dialysis and after drainage of the abdominal fluids.
Statistical analysis
All comparisons between groups were carried out by t-test or by one-way ANOVA using Prism 9 software (GraphPad; San Diego, CA). p Values below 0.05 were considered significant. Data are presented as means ± SD.
Results
The effect of SGLT inhibition on blood glucose levels and osmotic gradient
Mice PD model
As shown in Figure 1(a), SC administration of the non-selective SGLT inhibitor phlorizin attenuated hyperglycaemia during dwell with PDF (0.375% glucose). The mean AUC of phlorizin-treated mice was 30% lower than saline-treated control mice (p < 0.05).
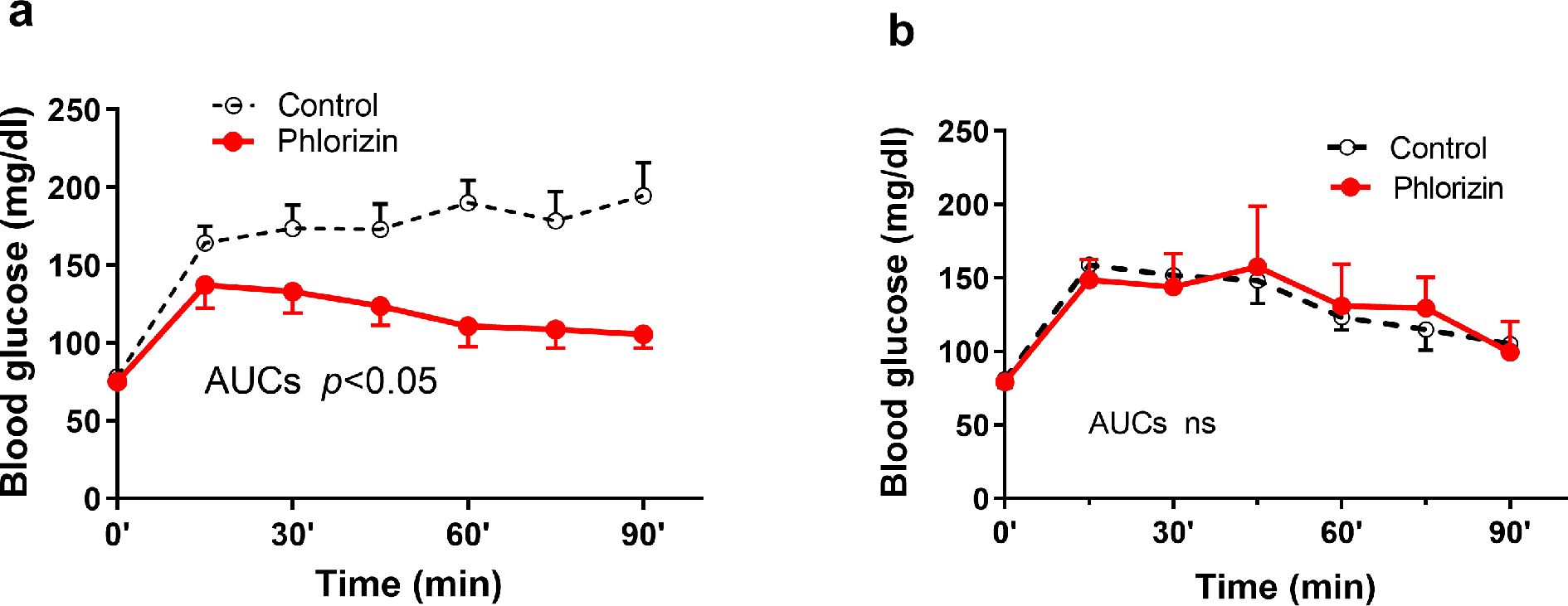
In mice: blockade of peritoneal SGLTs reduces blood glucose transport from peritoneal dialysis solution. (a) Uraemic mice (n = 5 in each group) were injected SC with saline (control) or phlorizin and then exposed to 2 ml of diluted commercial peritoneal dialysis solution (PDF 0.375% glucose). Blood glucose AUCs comparison (p < 0.05). (b) Uraemic mice (n = 5 in each group) were injected SC with saline (control) or phlorizin (SGLT inhibitor) and then exposed to 2 ml of sodium-free solution (0.375% glucose in water) (AUCs not significant (ns)). SGLT: sodium–glucose co-transporter; AUC: area under the curve; PDF: peritoneal dialysis fluid; SC: subcutaneous.
To further confirm that a sodium-dependent glucose transporter contributes to glucose transport in control animals, we prepared a sodium-free solution with an identical glucose concentration as the diluted PDF (0.375%). As shown in Figure 1(b), in the absence of sodium, blood glucose levels during dwell were similar in the control and phlorizin groups (differences in AUCs ns). In contrast to phlorizin, in the mice model, treatments with the selective SGLT2 inhibitors empagliflozin (SC) and dapagliflozin (orally) did not affect blood glucose loading AUCs compared to control mice (Supplementary Figure 1).
Rat PD model
Mice model had several limitations in our experiments: the SGLT blocker effect on glucose absorption was demonstrated in diluted solutions only; furthermore, the small filling volume makes challenging fluid sampling during dwell, and the UF measurement was inaccurate. In the rat model, we proved the influence of SGLT blockers both in diluted and commercial solutions (Figure 2) and fluid samples were taken during the experiment.
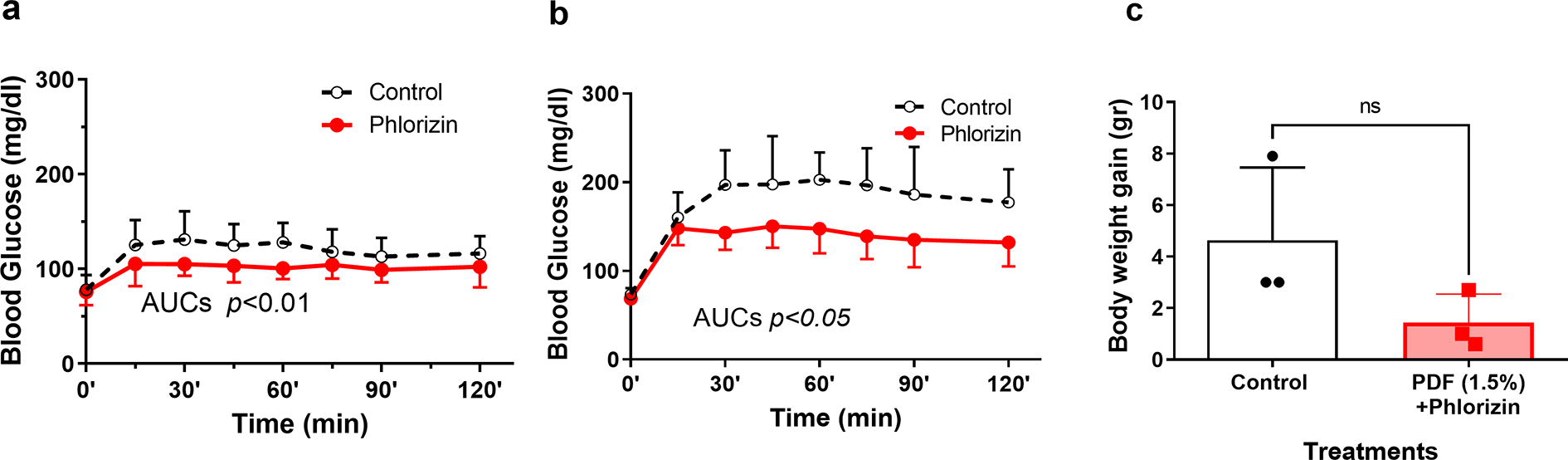
In rats: phlorizin diminished blood glucose levels. (a) Glucose containing diluted PDF, or (b) standard PDF (1.5% Dianeal) were injected IP to uraemic rats with (n = 8 and 7) or without (n = 7 and 6) phlorizin (100 μg/ml). (c) We compared dialysis weight gain in a few animals treated with standard PDF (n = 3 in each group). Glucose AUCs comparisons for diluted PDF and standard PDF p < 0.01 and p < 0.05 accordingly. Weight gain was in the control group 4.63 ± 2.83 g and in the phlorizin group 1.43 ± 1.12 g (ns). AUC: area under the curve; PDF: peritoneal dialysis fluid; IP: intraperitoneal; ns: not significant.
Using a uraemic rat PD model, the increment in blood glucose level was more evident in animals treated with 1.5% glucose (Figure 2(b)) than diluted Dianeal (Figure 2(a)). Phlorizin attenuated hyperglycaemia in both diluted and standard PDF (Figure 2(a) and (b), p < 0.01 and p < 0.05). As shown in Supplementary Figure 2(A), using dialysate with 0.375% glucose, in the phlorizin group, we observed a decline in dialysate glucose levels compared to control starting (p < 0.05) which along with reduced blood glucose levels may be interpreted as increased UF. In rats dialysed with 1.5% glucose, the difference in glucose dialysate concentrations between phlorizin and the control group did not reach significance (Supplementary Figure 2(B)).
For assessment of UF, we measured the weight changes in rats following dialysis (Figure 2(c)). In our experiments, UF was mostly negative, which may be explained by high peritoneal transport in rats and elevated serum osmolality due to high urea levels. In the situation of partial absorption of PD fluids, we supposed that weight gain should better characterise the final fluid balance during PD considering, that there was no fluid addition or loss during the experiment aside from PD dwell.
In control rats, we observed negative UF with a weight gain of 4.63 ± 2.83 g, while in the presence of phlorizin in PDF the tendency of less weight gain 1.43 ± 1.12 g (ns) was noted, probably representing decreased fluid absorption and accordingly favourable UF compared to controls.
In the last set of experiments, we tested the effects of sotagliflozin, a dual SGLT1and 2 inhibitor. As shown in Figure 3(a) and (b), blood glucose AUCs were significantly lower in rats dialysed with PDF containing sotagliflozin compared to other groups. Remarkably, the same and even increased dose of sotagliflozin and empagliflozin, a specific SGLT2 inhibitor, given orally by gavage, did not reduce glucose AUC during PDF dwell compared to control (Figure 3(b)). Baseline glucose level elevation before PDF injection measured 15 min after SGLT inhibitor administration in orally treated animals was noted only in empagliflozin-treated animals (p < 0.01) (Figure 3(c)).
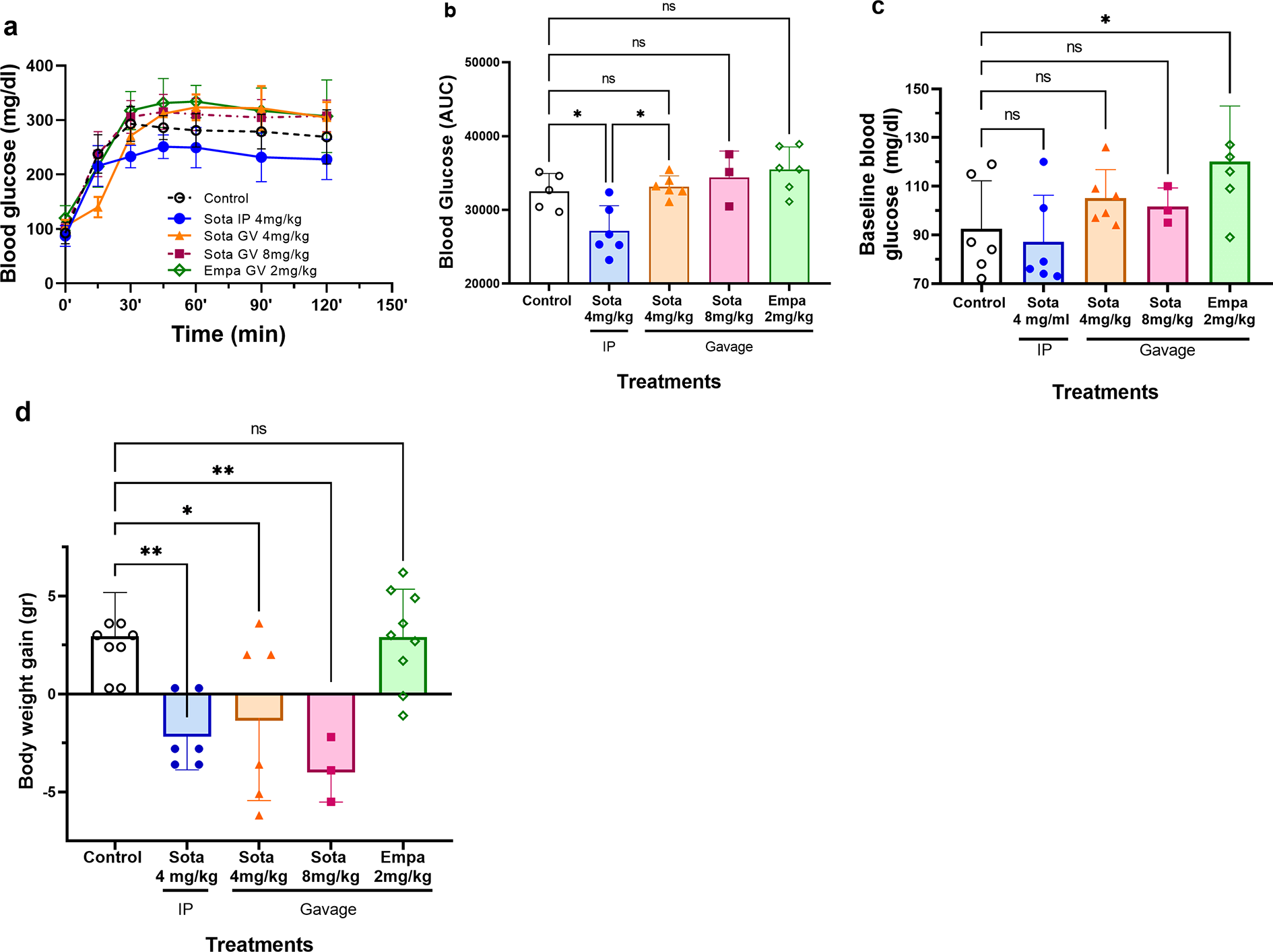
Blood glucose levels during PDF dwell and ultrafiltration in uraemic rats treated by SGLT inhibitors. Animals were treated by PDF (Dianeal 1.5%) (control n = 9), IP sotagliflozin in PDF (n = 6), sotagliflozin in gavage (n = 6) in two doses and empagliflozin in gavage (n = 9). (a) Blood glucose levels during dialysis. (b) Blood glucose AUCs. (c) Baseline blood glucose following treatments with SGLTs and (d) body weight gain following dialysis (ultrafiltration). *p < 0.05, **p < 0.01. SGLT: sodium–glucose co-transporter; AUC: area under the curve; PDF: peritoneal dialysis fluid.
Sotagliflozin treatments by gavage or IP improved UF as illustrated by the significant loss of weight during dialysis (−2.03 ± 0.75 g, p < 0.01 for IP and −1.22 ± 1.73 g, p < 0.05 for gavage). In contrast, blocking SGLT2 by oral administration of empagliflozin did not affect UF (Figure 3(d)).
Discussion
To the best of our knowledge, this study is the first to report the role of SGLT-mediated glucose absorption in a uraemic animal PD model. The effects of SGLT inhibitor on peritoneal glucose transport suggest that this pathway can be a therapeutic target to improve PDF performance and reduce the local and systemic adverse effects of glucose absorption in PD.
We found that administration of a non-specific SGLT blocker (phlorizin) significantly attenuated hyperglycaemia, suggesting slower glucose absorption from the peritoneal cavity, as evidenced by the lower AUC of blood glucose levels in phlorizin-treated animals. Blood glucose elevation during PDF dwell we used as an index of the SGLT effect. High PDF glucose concentration increased glucose absorption by paracellular mechanism and obscure the SGLT inhibitor impact on glucose transport, hence in mice, we could not show the decrease in glucose absorption after SGLT blockade with standard PDFs. In experiments with diluted PDFs, we found significantly reduced glucose levels after non-specific SGLT inhibition.
Further evidence that glucose transport is sodium-dependent was attained in animals exposed to sodium-free glucose-based solutions. Under the latter conditions, in control animals, blood glucose was not elevated compared to phlorizin groups, suggesting as previously described that part of peritoneal glucose transport requires the presence of a sodium-driven electrochemical gradient. 8,17 In rats, some beneficial effects of SGLT blocking, that is, attenuated hyperglycaemia and improved UF, were perceived even using standard undiluted PDF. However, in rats, dialysate glucose decline as a surrogate for UF, was significant only with diluted PDF. Of note, under these non-physiologically high glucose levels, the differences in glucose concentrations between solutions were probably below the detection level in small animals.
Schroppel et al. 12 and Debray-García et al. identified SGLT1 in peritoneal mesothelial cells, and Debray-García et al. and Zhou et al. demonstrated presence of SGLT2 on these cells. 10,18 In our studies, phlorizin, a non-specific SGLT inhibitor, and sotagliflozin, a blocker of SGLT1, 2 reduced blood glucose AUCs compared to untreated control. However, in the studies of Zhou et al. and Martus et al., 18,19 as well as in the current study where we mechanically blocked kidney glucose excretion, empagliflozin, a selective SGLT2 inhibitor, did not reduce blood glucose elevation during PD. In our model, we observed that gavage with empagliflozin increases basal glucose levels. It is possible that this elevation is due to the increase in glucose production by the liver, a known effect of SGLT2 inhibitor drugs (reviewed in Bonner et al. 20 ). This phenomenon has been described in the literature as a result of increased hepatic gluconeogenesis or increase endogenous glucagon production described in diabetic patients. 21 –23 The exact mechanism responsible for this effect of empagliflozin is not completely elucidated. We consider that in a clinical setting it generally is overlooked because emergence of glycosuria.
Reasonable to assume with the same explanation, in animals that were treated with gavage of sotagliflozin (SGLT1, 2 inhibitor), increased glucose liver production masks blood levels reduction by peritoneum SGLT blocking. SGLT inhibitors given IP reach the peritoneum and diffuse directly to circulation in contrast to orally administered inhibitors given before dialysis that are absorbed and get to the liver via portal vein. The induction of glucose production, seen in SGLT2-treated patients, might explain the elevation of basal blood glucose in empagliflozin-treated rats, and the elimination of AUCs reduction in rats treated with oral sotagliflozin compared to rats treated IP.
In our experimental rat model, only sotagliflozin given in PDF or by gavage before dialysis improved UF while treatment with empagliflozin was not beneficial, suggesting that SGLT1, and not type 2 SGLT, is a primary facilitator of peritoneal glucose transport. Unlike our study, Zhou et al. reported that 3 days of treatment of non-uraemic rats with empagliflozin improved UF compared to baseline before treatment. 18 Since we have not performed experiments with intraperitoneally SGLT2 inhibitors, we cannot exclude different results in rats injected IP with specific SGLT2 inhibitors. More studies are required to settle these contrasting findings.
Of note, orally administered sotagliflozin did not lower significantly blood glucose levels in rats but decreased weight gain during PD dwell. We suppose that during PDF dwell blood glucose level is influenced by absorption occurring in a transcellular and paracellular way thus blood glucose elevation can’t be fully abolished by SGLT blockade. IP sotagliflozin administration probably caused more effective local SGLT inhibition than systemic administration, demonstrated by a decrease in blood glucose AUC and fluid absorption.
Our study provides proof of principle for the potential mitigation of hyperglycaemia and enhancement of UF by IP SGLT blocker. It is possible blockade of peritoneal transcellular glucose transport will reduce the deleterious effects of high glucose-containing PD solutions on mesothelial cells integrity 24 –27 preserving peritoneal membrane, 28 reducing peritoneal fibrosis and sclerosis, 27,29 –31 technique failure and even the mortality in PD patients. 32 –34 The inhibition of SGLT in peritoneal membrane may bring long-term benefits in decreasing proinflammatory cytokines release, microvascular changes which may contribute to pathogenesis of EPS 13 – harmful complication of PD treatment, which remains unresolved problem even after cessation of PD.
A limitation in interpreting the results obtained in the rat model is the negative UF (fluid absorption) in animals treated with standard 1.5% glucose PDF reported in the current study and by Zhou et al. 18 probably explained by significant difference from human subjects in the ratio of PDF volume to peritoneal membrane surface area and blood volume.
This study is the first to report SGLT-mediated inhibition of peritoneal glucose absorption in uraemic PD animal models to the best of our knowledge. It suggests that transcellular peritoneal glucose transport plays an essential role in glucose absorption in PD. Thus, attenuation of PD-related glucose load in kidney failure may help prevent a broad spectrum of metabolic complications and may confer a positive effect on UF during PD. Taken together, these experiments suggest the presence of peritoneal SGLTs and illustrate their role in glucose transport from peritoneal cavity to blood. Limiting glucose reabsorption by SGLT inhibitors supports their potential role as novel agents in PD treatment to enhance UF and mitigate deleterious effects of glucose load.
Supplemental material
Supplemental Material, sj-tif-1-ptd-10.1177_08968608231165865 - Blockade of sodium–glucose co-transporters improves peritoneal ultrafiltration in uraemic rodent models
Supplemental Material, sj-tif-1-ptd-10.1177_08968608231165865 for Blockade of sodium–glucose co-transporters improves peritoneal ultrafiltration in uraemic rodent models by Marina Vorobiov, Boris Rogachev, Reut Riff, Cidio Chaimowitz, Endre Z Neulander, Anna Basok, Alla Shnaider, Amos Douvdevani and Yosef-Shmuel Haviv in Peritoneal Dialysis International
Supplemental material
Supplemental Material, sj-tif-2-ptd-10.1177_08968608231165865 - Blockade of sodium–glucose co-transporters improves peritoneal ultrafiltration in uraemic rodent models
Supplemental Material, sj-tif-2-ptd-10.1177_08968608231165865 for Blockade of sodium–glucose co-transporters improves peritoneal ultrafiltration in uraemic rodent models by Marina Vorobiov, Boris Rogachev, Reut Riff, Cidio Chaimowitz, Endre Z Neulander, Anna Basok, Alla Shnaider, Amos Douvdevani and Yosef-Shmuel Haviv in Peritoneal Dialysis International
Footnotes
Acknowledgements
The authors thank Valeria Frishman for her excellent technical assistance.
Author contributions
MV, BR, CC, AD and Y-SH designed the study. MV, BR, EN and RR collected the data. MV, EN and AD analysed and interpreted the data. MV, AB, AS and AD made the tables and figures. MV, AD and Y-SH drafted and revised the article. All authors critically edited the manuscript and approved the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Experiments were conducted after obtaining permission from the Israel Committee for Animal Experiments (IL-42-06-2015).
Funding
The author(s) received financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Israeli Society of Nephrology and Hypertension and the Dr Montague Robin Fleisher Kidney Transplant Unit Fund (UK).
Informed consent
Not applicable.
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
