Abstract
Calciphylaxis is an uncommon but life-threatening syndrome in end-stage kidney disease, characterised by painful medial and intimal calcification of the arterioles in the deep dermis and subcutaneous tissues. Intravenous sodium thiosulfate serves as an off-label but effective treatment in haemodialysis patients. However, this approach confers considerable logistical challenges for affected peritoneal dialysis patients. In this case series, we demonstrate that intraperitoneal administration can be a safe, convenient and long-term alternative.
Introduction
Calciphylaxis is a rare, but life-threatening syndrome that is characterised by painful medial and intimal calcification of arterioles supplying the deep dermis and subcutaneous adipose tissue. It has poor outcomes with approximately 40% succumbing to it within 1 year. 1 Classically it manifests either peripherally or centrally as tender plaques or nodules with violaceous discoloration in early disease and dusky discoloration and necrosis in later stages. 2 Its exact pathogenesis is unclear but cited risk factors include end-stage kidney disease (ESKD), female gender, obesity, diabetes mellitus, hypercalcaemia, hyperphosphataemia, hyperparathyroidism and use of drugs like warfarin, calcium-based phosphate binders and vitamin D analogues. 2 Intravenous (IV) sodium thiosulfate (STS) has been the mainstay treatment, although off-label, over nearly 2 decades. 3 –5 Treatment is usually prolonged and administered conveniently during haemodialysis. 4 The IV route is also preferred as it is associated with fast pain relief. 5 However in peritoneal dialysis (PD) this approach is fraught with logistical challenges. 6 –8 Intraperitoneal (IP) STS administration can therefore serve as a convenient self-administered alternative but this is widely discouraged and little supporting literature exists. 6,8,9 Here, we report our experience on the safe and prolonged use of IP STS in three consecutive PD patients with calciphylaxis. Based on local institutional review board mandates informed consent was waived.
Case description
Case 1
A 64-year-old Chinese female, who had ESKD secondary to diabetic kidney disease (DKD), had been on continuous ambulatory peritoneal dialysis (CAPD) for 3 years. Other past medical history included hypertension, hyperlipidaemia and ischaemic heart disease (IHD). Her long-term medications were lanthanum carbonate 1000 mg twice a day and calcitriol 0.5 mcg twice a week. She was not on warfarin. She presented with left lateral calf skin lesion of 4-months duration. On examination, she had a well demarcated 3 × 3 cm erythematous plaque with overlying dry eschar on her left lateral calf. Blood investigations are shown in Table 1. Histology of a skin biopsy demonstrated ulceration with small vessel microcalcification in the subcutis, suggestive of calciphylaxis. She was started on IP STS 25 g added to her night Extraneal® dwell, three times a week. The STS dose was reduced to 12.5 g three times a week for a few weeks after she complained of nausea and vomiting. Once symptoms resolved it was increased back to 25 g without further issues. She was treated for 6 months with partial resolution of the lesion. Her calcitriol was stopped and she was placed on cinacalcet 25 mg daily. Monitoring of electrocardiogram did not show peri-treatment QTc prolongation. Pain control was achieved with subcutaneous fentanyl.
Clinical characteristics and outcomes of patients treated with intraperitoneal sodium thiosulphate.
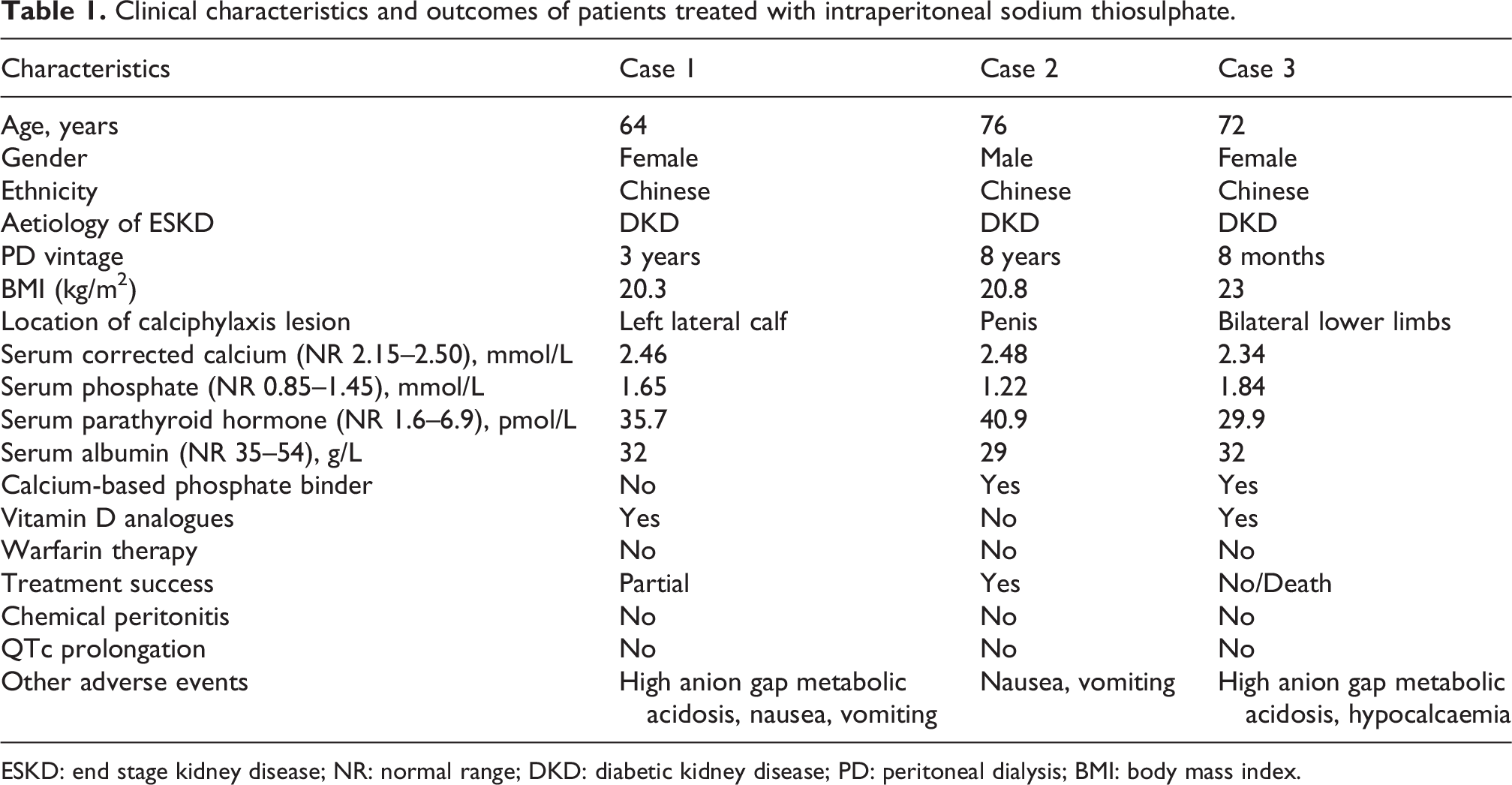
ESKD: end stage kidney disease; NR: normal range; DKD: diabetic kidney disease; PD: peritoneal dialysis; BMI: body mass index.
Case 2
A 76-year-old Chinese male, with ESKD secondary to DKD, had been on CAPD for 8 years. His past medical history included hypertension, right eye blindness from diabetic retinopathy, peripheral vascular disease with right toe gangrene amputated 6 years before. He was on calcium acetate 1334 mg morning only. He was not on any warfarin or vitamin D analogues. He presented with a 2-week history of painful penile ulcer. Physical examination revealed a superficial erosion on the glans penis with overlying greenish slough. Blood investigations are shown in Table 1. Histology of a punch biopsy of the lesion showed microcalcification of small blood vessels, consistent with calciphylaxis. He was started on IV STS 25 g three times a week via a peripherally inserted central catheter (PICC) which unfortunately fell off after 3 weeks. As he was not keen on reinsertion, he was converted to IP STS 25 g added to a Dianeal® 2.5% night dwell three times a week. The patient completed a total of 3 months treatment with resolution of the penile ulcer. Additionally, calcium acetate was switched to lanthanum and cinacalcet. There was no QTc prolongation during treatment and pain could be controlled with non-opioids.
Case 3
A 72-year-old Chinese female, who had ESKD secondary to DKD had been on CAPD for 8 months. Other past medical history included hypertension, hyperlipidaemia and IHD. She was on calcium acetate 667 mg daily, nutritional cholecalciferol 1000 IU 1 tablet twice a day and calcitriol 0.25mcg daily. She was not taking warfarin. She presented with a 2-weeks history of ulcers on both legs. Examination of her lower limbs revealed bilateral ulcers with overlying eschars, worse on the right side. Blood investigations are shown in Table 1. Histology of skin biopsy confirmed calciphylaxis. She was started on IP 12.5 g STS which was added to the night Dianeal® 2.5% dwell, three times a week. Calcium acetate and vitamin D were switched to lanthanum and cinacalcet. Monitoring of QTc did not show any prolongation. Unfortunately, after about 5 weeks into treatment, she died from a myocardial infarction without improvement in the skin lesions.
Discussion
These three cases illustrate that IP STS is convenient and well tolerated in PD patients in the long term but has variable efficacy. It certainly obviates the need for costly PICC insertion and in-centre administration which is associated with higher adverse outcomes like sepsis and metabolic issues. 6,7 Although, ESKD patients on PD are traditionally switched to haemodialysis, the basis for that has more than often been the convenience it confers with regards to IV administration as opposed to its greater ability at clearing phosphate. Furthermore it also still a matter of debate if PD results in more calciphylaxis risk compared to haemodialysis based on published literature. 7,10,11 We therefore opted to keep those patients on PD. The thrice-weekly of 25 mg IP dosing was adapted from the recommended IV dose formulation of 25 mg thrice weekly for 45 doses. 4 The patients could easily be taught how to withdraw STS which comes in 12.5g/50 mL vial using a 50 mL syringe and inject aseptically once or twice for the 12.5 g or 25 g dose respectively into a 2 L PD bag after removing an equivalent 50 to 100 mL of dialysate. The balance of vials were safely stored in home refrigerators.
So far IP STS use has been reported in only five cases as concerns abound regarding the risk of chemical peritonitis. 6 However 2 confirmed cases have attributed this to contaminated particulates in used STS. 8,12,13 Contrastingly, based on a case series, use of IV STS in PD patients led to 41% conversion to haemodialysis and the rest having PICCs resulting in a high mortality rate of 71% mainly from sepsis. 7 Interestingly, lower fractionated but more frequent IV doses of 3.2–6.4 g have been shown to mitigate some of these adverse effects which are usually more pronounced in Asian patients. 14 We therefore opted for a lower dose of 12.5 g in case 3 over such concerns and indeed she did not experience any nausea or vomiting.
In our series, none of the 3 patients had sepsis or prolonged QTc although they did experience occasional nausea and vomiting and mild high anion gap metabolic acidosis from the released thiosulfuric anion (Table 1). Moreso, besides the conversion to non-calcium based phosphate binders and cinacalcet, there was no need for adjunct treatment with bisphosphonates or hyperbaric oxygen therapy. Our patients were surprisingly of low body mass index (BMI). This could be due to the fact that obesity is usually associated with central calciphylaxis while all our patients had the peripheral type which is more seen in malnourished hypo-albuminaemic patients. 1,10,11 Intuitively, low BMI also compels lower STS dosing which hence mitigates adverse effects and reduces costs.
The benefits of STS are through chelation of calcium from calcific tissue beds to form the highly dialysable, calcium thiosulfate, and its vasolidating and antioxidant effects. 6 Given its small molecular size of 158 dalton, STS can easily cross biomembranes, explaining its systemic effect even when administered IP, whereby it has been shown to be capable of removing 0.65 mmol/L of extra calcium compared to STS-free dialysate. 14,15 However what is unclear about the IP route is its impact on pain control given two patients required prolonged opioid administration. Unknown also is its long-term effect on peritoneal membrane status. Furthermore, patients on automated nocturnal PD do require an additional day dwell of at least 6 hours for effective administration.
Conclusion
Intraperioteneal STS for calciphylaxis treatment in PD is convenient and variably effective. Our findings are however not conclusive but suggest it is unlikely to cause adverse events that warrant its discontinuation. Larger prospective and pharmacokinetic studies are needed to further validate its long-term effects.
Footnotes
Acknowledgements
None.
Author contributions
Made a substantial contribution to the concept or design of the work; or acquisition, analysis or interpretation of data – YKT/RCJ. Drafted the article or revised it critically for important intellectual content – YKT/RCJ. Approved the version to be published – RCJ.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Based on the number of patients and retrospective nature of study, ethical approval was waived (DSRB 2022/00986).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Not applicable.
