Abstract
Secondary embedding of a peritoneal dialysis (PD) catheter has been performed for patients whose kidney function has improved enough to stop dialysis but recovery is not expected to be long term. In addition, we have also performed the procedure for patients who have poor general condition due to severe cerebrovascular and/or cardiac disease or who wish to have PD again at the end of life. Here, we report the case of the first terminal haemodialysis (HD) patient who resumed PD using a secondarily embedded catheter as an end-of-life choice. The patient had undergone secondary embedding of a PD catheter and had been transferred to HD, during which time multiple pulmonary metastases of thyroid cancer were observed. She hoped to resume PD in the end-of-life period, and the catheter was subsequently externalised. The catheter was used immediately, and the patient has continued on PD over the past 1 month without infectious or mechanical complications. For elderly end-stage kidney disease patients with progressive disease and cancer, secondary embedding of the PD catheter may be an option to permit them to live the remainder of their lives at home.
Introduction
Secondary embedding of peritoneal dialysis (PD) catheters was first reported by Crabtree, and the procedure has been performed for patients whose kidney function has improved enough to stop dialysis, but recovery is not expected to be long term. 1 In addition, we have also performed the procedure as an alternative to removing the catheter to reduce the surgical burden in high-risk patients with severe cerebrovascular and/or cardiac disease or those who may wish to resume PD in the end-of-life period. 2
However, none of the patients who underwent this procedure, which we have been performing since 2011, resumed PD in the end-of-life period. The case of the first terminal haemodialysis (HD) patient who resumed PD using a secondarily embedded catheter as an end-of-life choice is reported.
Case presentation
An 80-year-old woman with end-stage kidney disease due to chronic interstitial nephritis had been on HD. She chose PD as kidney replacement therapy 14 years earlier and had a catheter placed in an open operation. Thereafter, the patient had progressed smoothly without any peritonitis or exit-site or tunnel infection, but a tumour shadow was found in the right upper lung field on a chest X-ray 3 years later. After a detailed examination, she was diagnosed with bone metastases from thyroid cancer, and in the same year, she underwent rib tumour resection and total thyroidectomy. She had been able to continue PD after surgery, but her residual kidney function decreased gradually. An arteriovenous fistula was created, and combination therapy with PD and HD (once a week HD, 5 days PD and one day off PD) was started 5 years after starting PD.
The combination therapy was continued for 3 years, and transition to HD alone became necessary due to difficulty in fluid management (on PD for a total of 8 years). At that time, there were two options (removal of the catheter and secondary embedding of the catheter), and since she hoped to resume PD in the end-of-life period, secondary embedding of the catheter was performed under local anaesthesia. The PD catheter was cut in the abdominal cavity side of the subcutaneous cuff. The residual catheter was capped with a BioHole Plug (Nipro Co. Ltd., Osaka, Japan). The detail of the secondary embedding procedure has been presented in previous reports. 2
Although the embedded catheter showed no abnormalities after HD alone, previously undetected, multiple pulmonary nodules were noted 2 years later. The surgeon determined that anticancer drugs and radiotherapy were not indicated for her, and best supportive care was suggested. After that, following shared decision-making among her, her family and the medical staff regarding future kidney replacement therapy, she had not changed her intention to resume PD in the end-of-life period. It was explained to the patient that the catheter might be obstructed because the catheter had been embedded for approximately 6 years. However, she had a strong desire to resume PD, and, therefore, an operation was performed under spinal anaesthesia with the possibility of removing the obstruction with laparotomy in mind.
The procedure for externalisation of the embedded catheter is shown in Figure 1. Under antibiotic prophylaxis and aseptic conditions, a small skin incision was made to expose the catheter. The catheter was flushed with saline to confirm its patency. Fortunately, despite the prolonged period that the catheter had remained embedded, it was still patent. A titanium extender (Hayashidera Co. Ltd., Ishikawa, Japan) was used to connect the embedded and new catheters. The new catheter was externalised at the right upper abdomen through a new subcutaneous tunnel using a tunnelling tool. The catheter was used immediately, and the patient has continued on PD for the past month without infectious or mechanical complications.
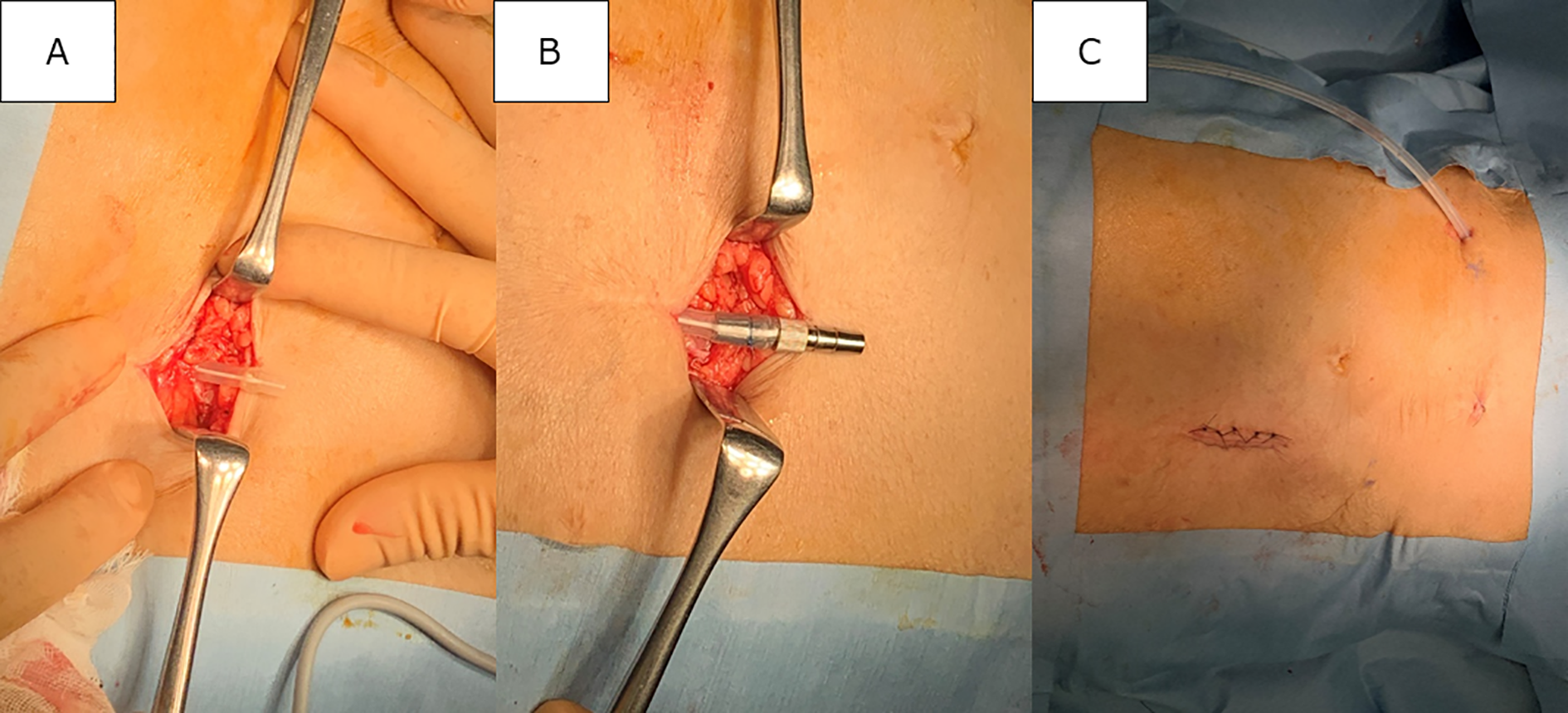
The procedure for externalisation of the embedded catheter. A small skin incision was made to expose the catheter (a); a titanium extender (Hayashidera Co. Ltd., Ishikawa, Japan) was used to connect the embedded and new catheters (b); and the new catheter was externalised at the right upper abdomen through a new subcutaneous tunnel using a tunnelling tool (c).
Discussion
To the best of our knowledge, this is the first case report in which a secondary embedded catheter was externalised and PD was resumed in the end-of-life period. We have previously reported the long-term prognosis of 10 patients with secondary embedding of the catheter. 3 One patient who recovered kidney function and was able to discontinue PD underwent re-externalisation of the embedded catheter after 4 years. All other patients were elderly, six patients died within 40 months postoperatively from causes unrelated to the embedded catheter. Thus, the patients who will be undergoing re-externalisation of embedded catheters are not that frequently encountered.
Korzets et al. reported that some specific complications (retained cuff, peritoneal fistula and pseudo aneurysm in the inferior epigastric artery) occur after catheter removal by open surgery. 4 In addition, abdominal wall hernia has been reported as a delayed complication. 5 Secondary embedding does not cause the above complications, but if the embedded catheter becomes infected, tunnel infection may develop into peritonitis. Secondary embedding should not be performed in the presence of a tunnel infection. Also, it may develop subcutaneous abscess formation. However, we believe that infection can be avoided with sufficient evaluation by ultrasonography. Bowel perforation has also been reported in unused PD catheter. 6 However, it seems to be a very rare occurrence and in many cases the issue was bowel injury during catheter placement and then delayed recognition on exteriorisation.
Catheter obstruction is also the concern in the re-externalisation of secondarily embedded catheters. The procedure for embedding a catheter was first described by Moncrief and Popovich. 7 Whereas the procedure is used before the start of PD, secondary embedding is performed at the end of PD. These embedding procedures always carry the risk of omentum, fibrin or thrombus occlusion. Davenport et al. reported that only 25 of 47 embedded catheters (53.2%) were functional at the time of externalisation. 8 On the other hand, Crabtree and Burchette reported that 85.7% of 84 embedded catheters was exhibited immediate function. 9 The prerequisite for secondary embedding is normal catheter flow function. 10 Therefore, the risk of flow failure might even be less with secondary embedding. If the externalised catheter is obstructed, it can be removed by a small laparotomy and returned to the abdominal cavity again. 11 However, not only does it require open surgery, but it also increases the risk of leakage. In the future, it is necessary to develop a method to prevent occlusion during embedding to increase the use of secondary embedding of PD catheters.
In conclusion, re-externalisation of embedded catheters may be an option for elderly end-stage kidney disease patients with progressive disease (e.g. neurodegenerative disease, dilated cardiomyopathy) and cancer to spend the rest of their lives at home.
Footnotes
Acknowledgements
None.
Author contributions
TS wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
St. Marianna University School of Medicine Hospital does not require ethical approval for reporting an individual case.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent to participate
Not applicable.
Informed consent to publish
Informed consent was obtained from the patient for their anonymised information to be published in this article.
