Abstract
Introduction:
Peritonitis remains a potentially serious complication of peritoneal dialysis (PD) treatment. It is therefore important to identify risk factors in order to reduce the incidence of peritonitis. The aim of the present analysis was to identify factors associated with time to first peritonitis episode.
Methods:
Incident PD patients from 57 centres in Europe participated in the prospective randomised controlled Peritonitis Prevention Study (PEPS) from 2010 to 2015. Peritonitis-free, self-care PD patients ≥18 years were randomised to a retraining or a control group and followed for 1–36 months after PD initiation. The association of biochemical, clinical and prescription data with time to first peritonitis episode was studied.
Results:
A first peritonitis episode was experienced by 33% (223/671) of participants. Univariable Cox proportional hazard regression showed a strong association between the time-updated number of PD bags connected per 24 h (PD bags/24 h) and time to first peritonitis episode (HR 1.35; 95% confidence interval (CI) 1.17–1.57), even after inclusion of PD modalities in the same model. Multivariable Cox regression revealed that the factors independently associated with time to first peritonitis episode included age (HR 1.16 per 10 years; 95% CI 1.05–1.28), PD bags/24 h (HR 1.32; 95% CI 1.13–1.54), serum albumin <35 versus
Conclusion:
This study of incident PD patients indicates that older age, greater number of PD bags connected/24 h, higher body weight and hypoalbuminaemia are independently associated with a shorter time to first peritonitis episode.
Introduction
Peritonitis remains a major complication of peritoneal dialysis (PD) and is associated with transfer to in-centre haemodialysis, 1,2 morbidity and mortality. 3 –5 Several risk factors for PD-related peritonitis have been described. 6 –9 Some are not modifiable, such as higher age, 10 –14 male gender, 10,12,14 –16 ethnicity, 13,17 lower socio-economic status, 18 diabetes mellitus, 19,20 coronary artery disease and chronic lung disease. 13 Among modifiable risk factors for peritonitis are lapses in aseptic technique when performing PD, 21,22 obesity 13,15,23 –25 and reduced serum potassium. 26 Several studies have also found that patients on continuous ambulatory PD (CAPD) are at higher risk of peritonitis than those on automated PD (APD). 15,24 However, conflicting results have been reported regarding several of these risk factors. Some studies found no association between older age and peritonitis risk based on registry data, 18,27 even including elderly patients on assisted PD, 19 and that female gender 12 and underweight 28 were risk factors while diabetes was not. 10,18 Most investigations of risk factors associated with peritonitis are single-centre observational studies conducted in prevalent PD patients and few include incident patients.
It is important to identify risk factors for peritonitis in order to develop strategies to reduce this complication. The aim of this study was to examine factors associated with time to first peritonitis episode in patients who participated in the randomised controlled Peritonitis Prevention Study (PEPS). 29
Subjects and methods
Study design and subjects
The PEPS has previously been described in detail. 29 Briefly, PEPS was an investigator-initiated, non-commercial, randomised, controlled, open label, parallel group multicentre trial that evaluated the effect of regular targeted testing and retraining of new PD patients about the risk of a first peritonitis episode compared with control PD patients. The trial was conducted from January 2010 to December 2015 and included 671 patients at 57 centres; Sweden (25), Norway (11), Finland (7), Denmark (5), Estonia (1), Latvia (1), the Netherlands (5) and the United Kingdom (2).
The trial recruited peritonitis-free incident PD patients aged 18 years or over, who were able to perform CAPD or APD with or without daytime exchanges and with no assistance. Help with the handling of PD bags and managing exit-site care was allowed. The patients who remained eligible at 1 month after commencing PD were randomised either to a control group (
The trial was conducted in accordance with the principles of the Helsinki Declaration and the International Conference on Harmonization Good Clinical Practice guidelines. It was approved by ethics committees in the participating countries. All participants provided written informed consent before participation. The trial was registered with CinicalTrials.gov number NCT01293799.
Procedures
All participating centres confirmed that their baseline PD training followed the recommendations by the International Society for Peritoneal Dialysis (ISPD). 30
Data collected at baseline included sex, age, body weight, body mass index (BMI), main cause of kidney failure, previous kidney replacement therapy, functional status (Karnofsky performance scale 31 ), social factors (visual impairment with reading problems, hand function impairment (unspecified), working status, living alone, need of translation/interpreter and current smoking), routine biochemical characteristics before PD start (serum creatinine, urea, albumin, blood haemoglobin), type of PD start (acute/planned), whether prophylactic antibiotics were used before PD catheter insertion, if prophylactic antibiotic cream or ointment on the catheter exit site was used, PD catheter characteristics, and date of PD commencement at home. At each study visit we recorded the number of PD bags connected to the PD catheter per 24 h in CAPD patients, while in APD patients we recorded the number of PD bags connected to the tubing set per 24 h adding the daily number of PD bags connected to the PD catheter if additional manual daytime exchanges were used. Data on the time-varying variables including number of PD bags connected per day, comorbidity (the Stoke comorbidity score 32 ), treatment with corticosteroids or cytotoxic drugs, PD modality, volume of dialysis fluid used per day, and if help was needed with exit-site care were recorded both at baseline and at all study visits. The onset time of all peritonitis episodes was recorded. The diagnoses of peritonitis and exit-site infection were based on the ISPD recommendations. 33
Primary and secondary outcome
The primary outcome of the PEPS study, which has been reported previously, 29 was time from randomisation (at one month after PD initiation) to first peritonitis episode. In the present analysis of factors associated with peritonitis, which was a secondary outcome of the trial, our object was to identify factors associated with time to first peritonitis episode (peritonitis-free survival time).
Statistical analysis
The results were described as frequencies, percentages for categorical variables, mean and standard deviation (SD) for continuous normally distributed variables, and median values with the interquartile range (IQR) for continuous non-normally distributed variables. The association of factors associated with time to first peritonitis episode was analysed using the Cox proportional hazard regression model. This model is recommended for investigation of causal relationships between risk factors and a given outcome when there are competing risks,
34
the main competing events in the present study being kidney transplantation, transfer to haemodialysis or death. Subjects were censored at death, kidney transplantation, transfer to haemodialysis or assisted PD, cessation of PD due to spontaneous recovery of kidney function, withdrawal of consent, other causes and on the closing date of the study. The studied factors were collected at baseline except for medication, PD modality, number of PD bags used per day, volume of dialysis fluid used per day, and if help was needed with exit-site care. Data for the latter variables were collected at the study visits, as the exact date of changes in these variables was not recorded in the study. These variables were included as time-varying covariates in a time-dependent Cox regression. Univariable Cox regression was used to identify variables significantly associated with time to first peritonitis episode. The interaction between the number of PD bags per day and PD modalities (CAPD and APD) was also studied. Multivariable Cox regression was performed using the backward elimination procedure, stopping when all remaining factors were significant at
Results
Study population
Of the 671 participants in the trial, 77% (518/671) discontinued the study, 74% in the control group and 80% in the retaining group due to kidney transplantation in 40% (205/518), transfer to haemodialysis in 31% (160/518), death in 11% (56/518), transfer to assisted PD in 8% (42/518), withdrawal of consent in 5% (26/518), partial recovery of kidney function without further need for dialysis in 2% (13/518), termination of kidney replacement therapy in 1% (5/518), exclusion due to non-compliance in 1% (5/518), transfer to other hospital in 0.6% (3/518) and ceased participation of study centre in 0.6% (3/518). The study visits at 6, 12, 24 and 36 months after PD commencement were made by 81% (
Baseline characteristics.a
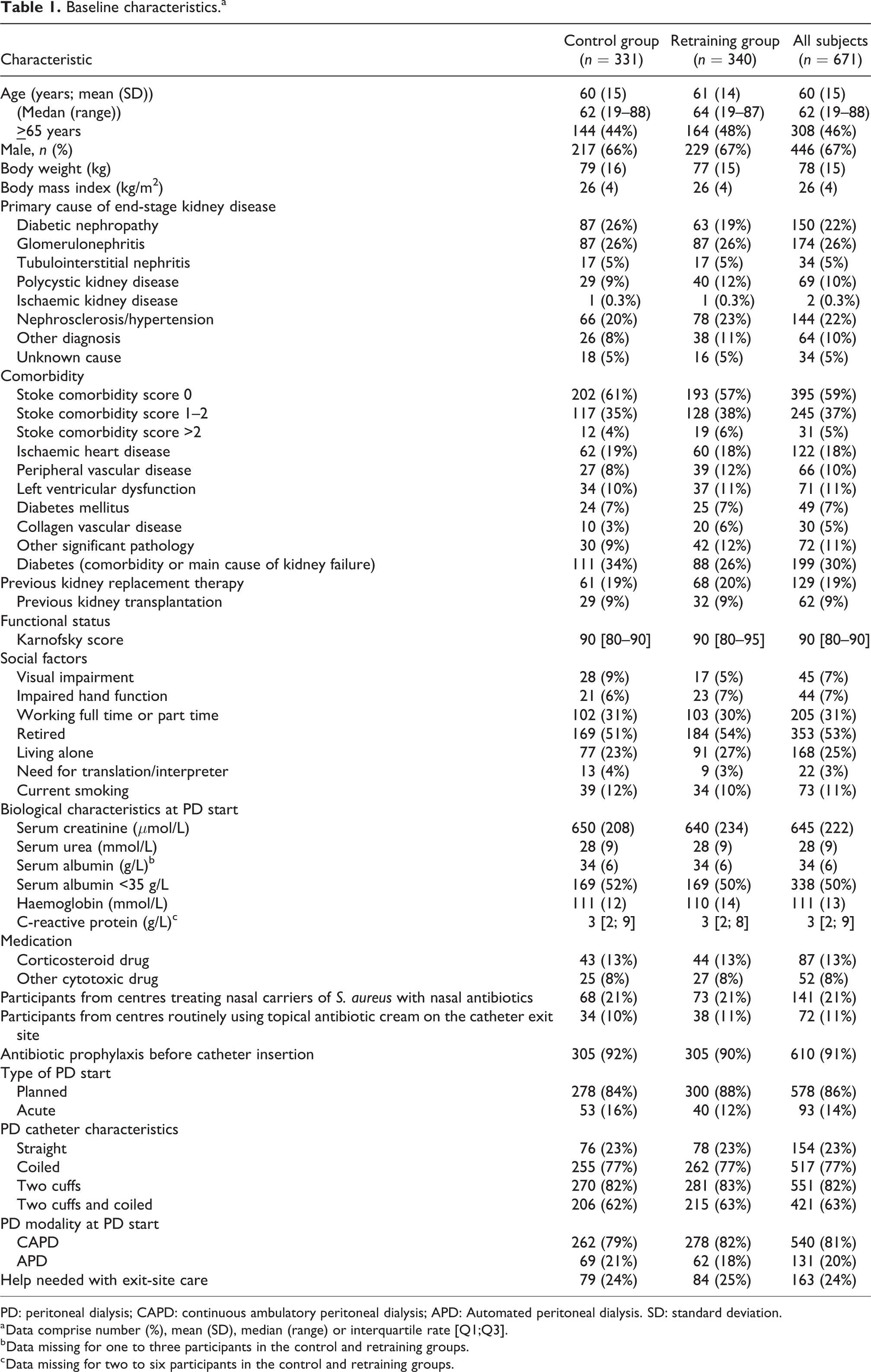
PD: peritoneal dialysis; CAPD: continuous ambulatory peritoneal dialysis; APD: Automated peritoneal dialysis. SD: standard deviation.
a Data comprise number (%), mean (SD), median (range) or interquartile rate [Q1;Q3].
b Data missing for one to three participants in the control and retraining groups.
c Data missing for two to six participants in the control and retraining groups.
Number of dialysis bags used per 24 h by participants at various study visits after start of peritoneal dialysis.
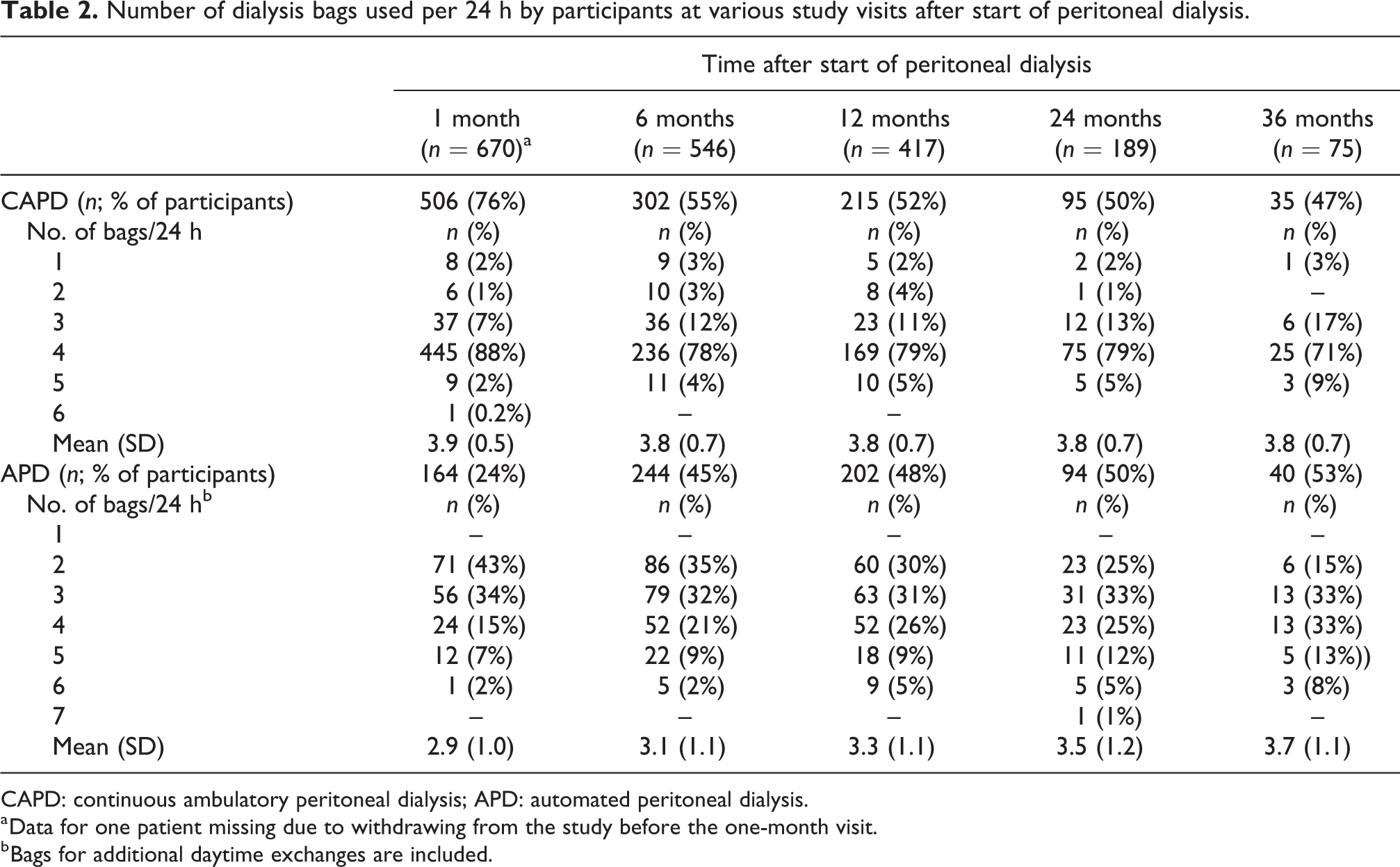
CAPD: continuous ambulatory peritoneal dialysis; APD: automated peritoneal dialysis.
a Data for one patient missing due to withdrawing from the study before the one-month visit.
b Bags for additional daytime exchanges are included.
Characteristics of the study centres
The median peritonitis rate at the participating centres the year before taking part in the study was 0.46 (range 0.23–0.96; IQR 0.34–0.65) episodes per patient year.
Nasal screening and treatment of nasal carriers of
Incidence of peritonitis and exit-site infection
As previously reported, a total of 320 episodes of peritonitis occurred during the study period 29 and the peritonitis rate was 0.35 episodes/patient-year. A first peritonitis episode was experienced by 33% (223/671) of participants; 37% (121/331) in the control group and 30% (102/340) in the retraining group. The mean incidence of a first peritonitis episode/patient-year was 0.31 (95% CI 0.26–0.37) and 0.29 (95% CI 0.23–0.35) in the control and retraining groups, respectively. Hence there was no significant difference between the groups (hazard ratio (HR) 0.92; 95% CI 0.71–1.19).
Of the 223 first peritonitis episodes, 184 (83%) were culture positive. The different types of microorganisms in the first peritonitis episode were equally distributed across the groups, except for ‘Other Gram-negative microorganisms’, which were more common in the control group (Table 3). The incidence of exit-site infection was 0.29 events/patient-year in the controls, 0.28 in the retraining group and 0.28 in the whole study group. Concomitant exit-site or tunnel infection was seen in 10% (24/233) of the peritonitis episodes. The cure rate, defined as resolution of evidence of peritonitis with antibiotic therapy and without the need for catheter removal, 35 was 74%. The overall peritonitis-related mortality, defined as death within 4 weeks of the onset of peritonitis, 36 was 2.7% (6/223) (Table 4).
Microorganisms in the first peritonitis episode.a

NS: Not significant.
a Microorganism proportions are calculated for patients with culture-positive peritonitis.
Outcome of first peritonitis episode.
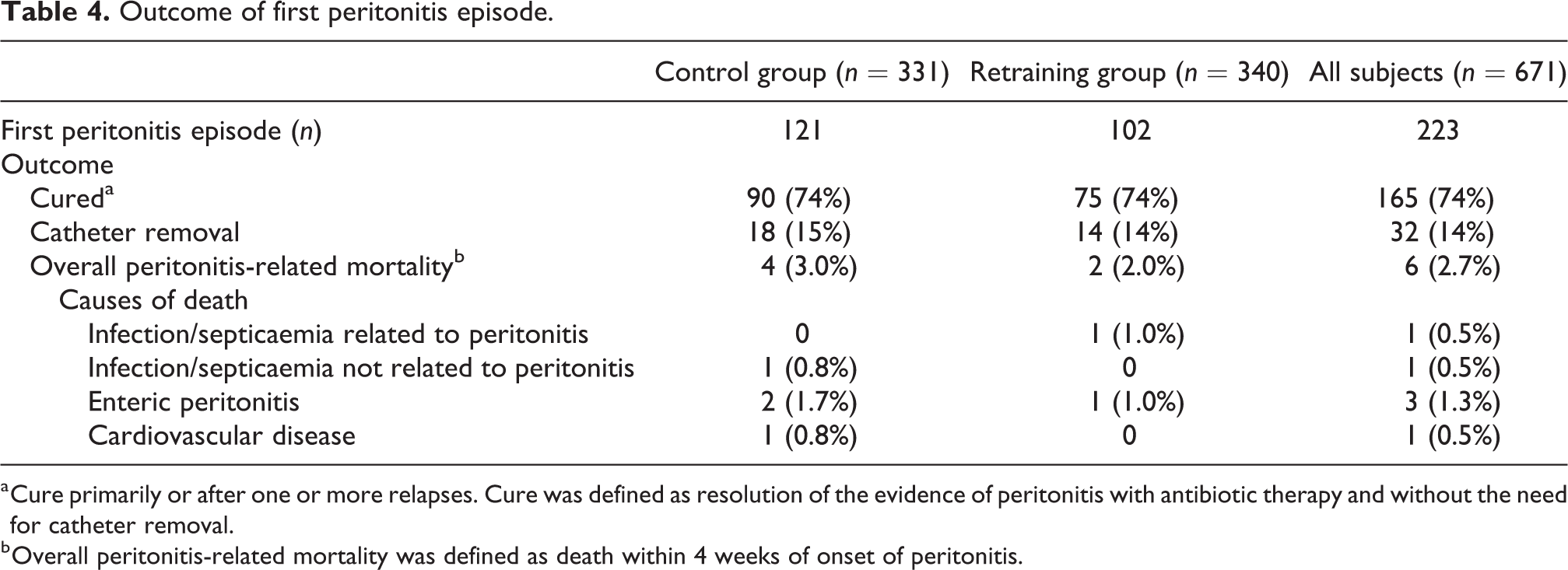
a Cure primarily or after one or more relapses. Cure was defined as resolution of the evidence of peritonitis with antibiotic therapy and without the need for catheter removal.
b Overall peritonitis-related mortality was defined as death within 4 weeks of onset of peritonitis.
Factors associated with time to first peritonitis episode
The associations of baseline and time-updated factors with time to first peritonitis episode based on Cox regression analysis are presented in Table 5. Factors significantly associated with shorter time to first peritonitis episode according to univariable Cox regression included age, body weight, BMI, being retired, time-updated number of PD bags used per day (HR 1.35; 95% CI 1.17–1.57) and serum albumin <35 g/L versus higher values. The time-updated volume of dialysis fluid used per day was not associated with an increased risk of peritonitis.
Association of baseline characteristics or time-updated variables with time to first peritonitis episode analysed by univariable and multivariable Cox proportional hazard regression.
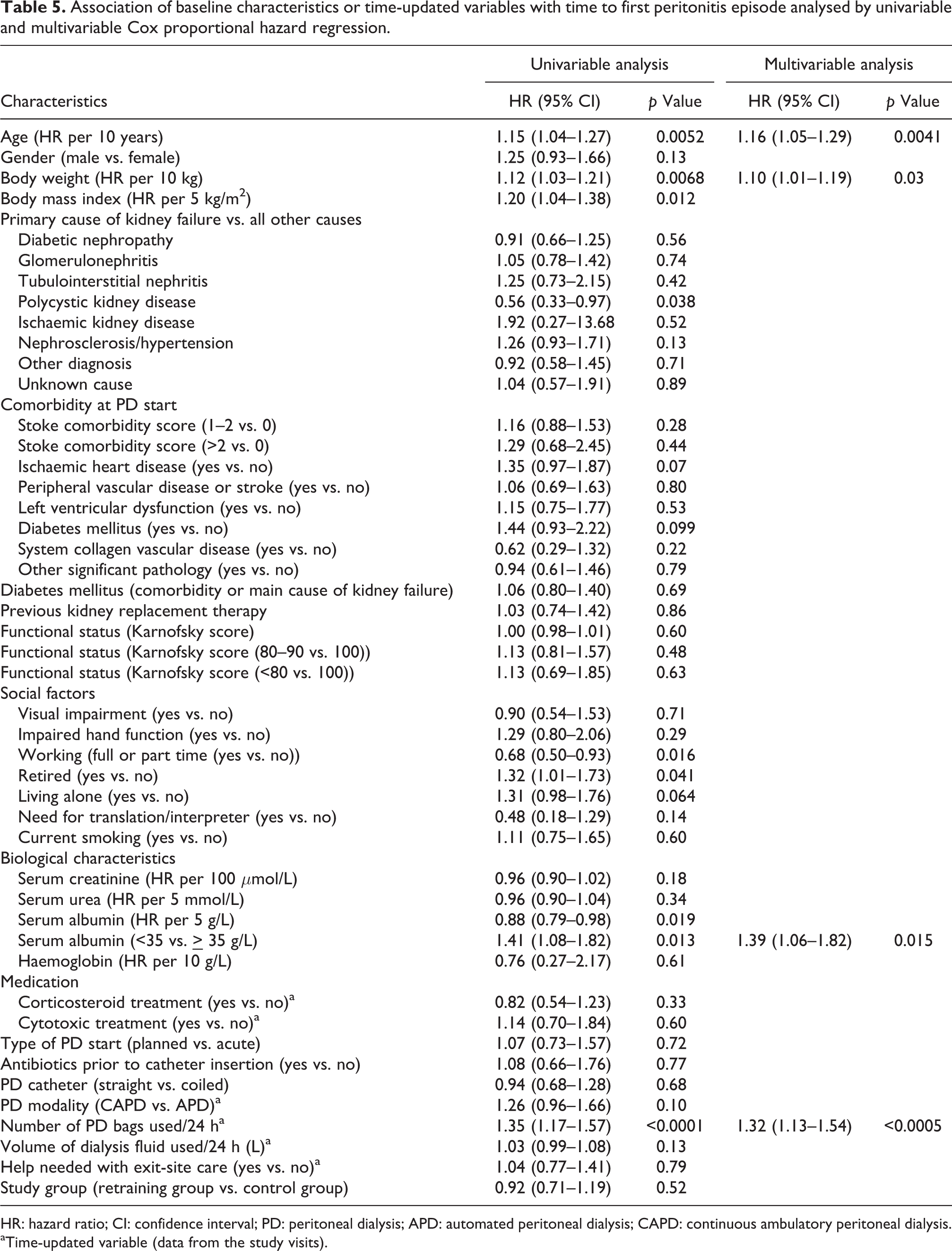
HR: hazard ratio; CI: confidence interval; PD: peritoneal dialysis; APD: automated peritoneal dialysis; CAPD: continuous ambulatory peritoneal dialysis.
aTime-updated variable (data from the study visits).
After country indicators were included in our analysis model to control for possible differences in PD routines in the participating countries, the association between the number of PD bags per day and time to first peritonitis episode remained significant (HR 1.34; 95% CI 1.14–1.56).
The risk of peritonitis developed approximately linearly with the number of daily PD bags used in all participants as well as in the CAPD and APD subgroups, and no clear cut-off point was identified, as demonstrated by univariable Cox regression analyses of 1 to 5 PD bags per day versus 4 PD bags per day. There was no significant association between PD modality (CAPD vs. APD) and time to first peritonitis episode (HR 1.27; 95% CI 0.95–1.66). When the number of PD bags per day and the two PD modalities were included in the same model, which was thus adjusted for CAPD and APD, the association between the number of PD bags per day and time to first peritonitis episode remained significant (HR 1.34; 95% CI 1.15–1.56). In addition, we investigated whether the effect of the number of PD bags per day differed in APD and CAPD by including an interaction term (with a
Factors associated with a significantly reduced risk of a first peritonitis episode included working full or part time versus unemployment, higher serum albumin levels and polycystic kidney disease versus all other main causes of kidney disease (Table 5). Although the median age of the participants with polycystic kidney disease was 2 years lower than that of the whole study group, an interaction analysis demonstrated that their lower peritonitis risk was not influenced by age (
Multivariable Cox regression analysis of the studied covariates identified four factors independently associated with time to first peritonitis episode (Table 5). One factor was age (HR 1.16; 95% CI 1.05–1.29). For each 10-year increase in age, there was a 16% increased likelihood of a first peritonitis episode. Another independent factor was the time-updated number of PD bags used per day (HR 1.32; 95% CI 1.13–1.54). With each additional PD bag used per day, the average risk of a first peritonitis episode increased by 32% in the whole study group. The other independent factors were body weight (HR per 10 kg 1.10; 95% CI 1.01–1.19) and serum albumin, <35 versus
Discussion
In this study of 671 incident, initially peritonitis-free PD patients, four independent factors associated with time to first peritonitis episode were identified: older age, higher time-updated number of PD bags used per day, higher body weight and serum albumin below 35 g/L.
Our finding that older age was an independent predictor of shorter time to first peritonitis episode is in line with previous retrospective single-centre reports. 10,16,37 In contrast, time to first peritonitis episode was not associated with age in the large prospective, multicentre BRAZPD study. 18 Nor was time to first peritonitis associated with age in a retrospective study from the Australia and New Zealand Dialysis and Transplant Registry (ANZDATA registry), 27 which found that patients aged 65 years or older had a similar peritonitis-free survival as younger patients. The conflicting results between our findings and the results of the latter studies can probably be explained by differences in study design, length of follow-up, as well as ethnic, cultural and social differences in the studied populations. Both the above-mentioned studies included patients who had remained on PD for at least 90 days. Thus, peritonitis episodes occurring during the first three months after PD initiation were not included. In the present study, we registered peritonitis episodes occurring after one month of PD treatment. In addition, the proportion of participants aged 65 years or older in the two above-mentioned studies was somewhat lower (36% and 40%, respectively) than that in the present study (46%).
In addition to age, the time-updated number of PD bags used per day was independently associated with time to first peritonitis episode (HR 1.32; 95% CI 1.13–1.54), the average increase in risk per additional bag being 32% in the whole group. This association, which has not been previously reported in a prospectively studied large group of incident PD patients, was independent of age, body weight and the presence of hypoalbuminaemia.
The risk of peritonitis increased in the whole group as well as in the APD and CAPD subgroups approximately linearly with the number PD bags and no clear cut-off point was identified. When we looked at APD and CAPD separately, we found to our surprise that the association between the number of PD bags connected per day and time to first peritonitis episode seemed to be stronger for APD than for CAPD, which warrants further investigation. Around 30% of the APD patients used three PD bags daily and 24–54% utilised four or more PD bags daily as shown in Table 2. However, the mean daily number of PD bags used at the study visits was lower in APD (2.9–3.7) than in CAPD (3.8–3.9; Table 2). In addition, the APD patients connected the tubing set to the PD catheter and thus made one further daily PD-related connection, which was not included in the data. As the number of PD bags used in APD patients includes PD bags for additional manual daytime exchanges, it may be difficult to separate the influence of strictly APD and CAPD on the association between the number of PD bags per day and risk of peritonitis.
Our main finding that the number of PD bags connected per day was independently associated with time to first peritonitis episode suggests a possible benefit of incremental PD, 38 where patients with significant residual function start on a lower PD dose, thus implying fewer PD connections. The present findings are supported by a recent study based on data of 620 incident CAPD patients from the French peritoneal dialysis registry, which demonstrated that 22 or more exchanges per week were associated with a significantly shorter time to first peritonitis episode compared to less than 22 exchanges per week. 39 That study, however, differs from the present one, as most of the studied patients were more than 75 years old and had nurse-assisted CAPD, which is reimbursed in France. Nurse-assisted PD thus appears to be a useful method for reducing the peritonitis risk in older patients in need of PD assistance. A more frequent use of nurse-assisted PD in this group of PD patients can therefore be recommended.
In contrast to our results, a retrospective single-centre study of CAPD patients from Taiwan found no association between the baseline number of daily PD exchanges and the risk of a first peritonitis episode. 14 Unlike that study, which analysed the number of PD bags used at the start of PD therapy, we studied the number of PD bags at the last study visit preceding the first peritonitis episode. Furthermore, a Chinese prospective controlled single-centre study, in which 139 new CAPD patients were randomised to either three or four PD exchanges per day, 40 found a tendency towards a longer time to first peritonitis episode in the three-exchange group, but no significant difference between groups. Both the Taiwanese and Chinese studies contained only CAPD patients.
Our study included both CAPD and APD patients, and APD was used by around 50% of the participants. In addition, 30% of the participants changed their PD modality during the study. As APD and CAPD are different submodalities of PD, it may be questioned whether they can be analysed together. However, an APD regime can also include manual day-time exchanges. Furthermore, as demonstrated in the present study, the number of PD bags connected per day can vary as much for APD patients (from 1 to 7) as for CAPD patients (from 1 to 6). Based on the results of the present study, it is important that APD patients are not excluded from studies analysing the risk of peritonitis, because APD is the dominant modality in many countries. 9
According to univariable Cox regression, we found that the PD modality was not significantly associated with time to first peritonitis episode, which is in line with the results of the multicentre observational BRAZPD study, 18 as well as a retrospective single-centre study from Uruguay, 10 and an analysis from the ANZDATA registry. 41
A likely explanation for the association between the number of PD bags used per day and the risk of a first peritonitis episode is that a higher number of PD bag connections per day increases the risk of contamination of the PD catheter, PD bag connections or the APD tubing set with pathogenic skin bacteria, especially
We also found that higher baseline body weight was independently associated with shorter time to first peritonitis episode, which is in agreement with previous reports. 13,23,24 Moreover, BMI was significantly associated with increased risk of a first peritonitis episode, although not independent of other risk factors, as in studies from the ANZDATA registry. 13,23,25,27
The observed association between low baseline serum albumin levels and a shorter time to first peritonitis episode in our study is also consistent with several earlier studies. 16,25,43 –45 As hypoalbuminaemia can be a marker of malnutrition, inflammation and the comorbidity burden, 7,16 this variable may be difficult to modify.
The strengths of this study were its prospective design, that it included incident, peritonitis-free patients, and the fact that the patients had either CAPD or APD similar to the general PD population. Data were available for all participants, and updated values of time-varying variables were employed in some of the analyses of factors associated with time to first peritonitis episode.
The main limitation of this study was the high proportion of patients who discontinued the study mainly due to kidney transplantation, transfer to haemodialysis or death. This could have limited the possibility to find factors associated with peritonitis and reduce the power to detect true relationships (type II error). Only self-care patients were included, which limits the generalisability to other PD populations, where assistance may be common. In addition, a possible cluster effect cannot be ruled out, although this was less likely, as individual randomisation was performed and each participant was independent of the others. Furthermore, no significant centre effect on the PD treatment outcome was found.
Differences in the use of prophylactic antibiotics may also influence the generalisability of the results, as previously pointed out.
29
The ISPD recommendation to administer prophylactic antibiotics prior to PD catheter insertion for the prevention of peritonitis
33
was followed in the majority of patients (91%) in this study, as in many other countries.
9,46
The guidelines also recommend the use of prophylactic antibiotic cream or ointment on the PD catheter exit-site, which was applied by only 11% of the participants in the present study. Adherence to this recommendation is known to vary both between and within different countries.
9,46
The low use of topical antibiotic exit-site prophylaxis in this study, which was likely a result of the fact that the health authorities in the Scandinavian and Baltic countries do not encourage long-term antibiotic prophylaxis, due to the risk of development of resistant bacterial strains, may also reduce the generalisability to other PD populations, in which antibiotic prophylaxis is more common. The incidence of exit-site infection, which is reported to vary broadly between studies from 0.1 to 1.2 episodes/patient-year,
47
was 0.28 episodes/patient-year in this study. The proportion of peritonitis episodes with a concomitant exit-site or tunnel infection of 10% in this study of appears to be low, as it has been estimated that 20% of all peritonitis episodes and up to 50% of
In conclusion, the result of the present prospective study of incident CAPD and APD patients demonstrated that older age, higher number of PD bags connected per day, higher body weight and low serum albumin levels at baseline were independently associated with early peritonitis onset. This indicates a possible benefit of incremental PD, which starts with a lower PD dose, but such a strategy should be tested in further studies. By taking these risk factors into account in the individualisation of the training, follow-up and PD prescription for new PD patients, it may be possible to prolong the time to the first peritonitis episode and lower the peritonitis rate.
Footnotes
Acknowledgements
The authors thank the PD nursing staff and the investigators at the participating hospitals for their contribution and Mattias Molin from the Statistical Consulting Group, Gothenburg, Sweden, for statistical analyses.
Author contributions
SL, DP and JEJ conceived the study. SL obtained funding, was the principal investigator and managed the trial: She participated in the design, data analysis and interpretation, and drafting of the first manuscript. DP, JEJ, OH, AP, MO-R, A-C J, TEJ, BS and MR participated in the design of the study. SL, DP, JEJ, AP, HS, MO-R, DS and MW were chief investigators in their respective countries. SL, JEJ, DP, AP, MO-R, HS, DS, MW, OH, BS, TE-J, A-CJ, MR and HG approved the protocol, supervised the study, participated in the data analysis and interpretation of the results. The biostatistician LH-A advised on and participated in the data analysis. All authors reviewed and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declare the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SL received a research grant from Baxter Healthcare Corporation that partly funded this trial. OH declared having served on advisory boards of AstraZeneca, Vifor, Opterion and Gilead and has received speaker’s honoraria from AstraZeneca, Vifor, Baxter Healthcare Corporation and Fresenius Medical Care, which has no bearing on the submitted work. MW is Advisory Editor and OH is a member of the Editorial board of Peritoneal Dialysis International. The handling of and decision-making regarding this manuscript was taken care of by other members of the editorial board. MW has received a research grant from Baxter Healthcare Corporation and speaker’s honoraria from Baxter Healthcare Corporation and Fresenius Medical Care, and has served as a consultant to Triomed AB, which has no bearing on the submitted work. All the other authors declared no competing interests.
Ethical approval
The study protocol and its attachments were reviewed and approved by independent ethics committees in the participating countries.
Funding
The author(s) received financial support for the research, authorship, and/or publication of this article: The trial was supported by The Health & Medical Care Committee of The Regional Executive Board, Region West of Sweden (VGFOUREG-78061, 226521 and 383641), Baxter Healthcare Corporation (McGaw Park, IL, USA; Clinical Evidence Council grant number 10CECEU1004), Swedish Society of Nephrology, The Swedish Kidney Association, The Society of Kidney Patients in West of Sweden, The John and Brit Wennerström Foundation, The Bertil and Berit Adström Foundation, The Foundation for Kidney Failure (Sweden), The Swedish Kidney Foundation and Norwegian Society of Kidney Patients. The funders did not have any role in the study design, data collection, analysis, reporting, or the decision to submit the manuscript for publication.
Informed consent to participate
Free and informed consent was given in writing by each patient before the start of the study.
Informed consent to publish
Written informed consent was obtained from all patients for their anonymised and aggregated information to be published.
Trial registration
The trial was registered with ClinicalTrials.gov number NCT01293799.
