Abstract
Background:
The clinical course of COVID-19 in peritoneal dialysis (PD) patients has so far only been analysed in relatively small, often single-centre case series. Therefore, we studied patient- and disease-related characteristics and outcomes of COVID-19 in a larger European cohort of PD patients.
Methods:
We used data from the European Renal Association COVID-19 Database (ERACODA) on PD and haemodialysis (HD) patients with COVID-19 (presentation between February 2020 and April 2021). Hazard ratios (HR) for mortality at 3 months were calculated using Cox proportional-hazards regression. In addition, we examined functional and mental health status among survivors at this time point as determined by their treating physician.
Results:
Of 216 PD patients with COVID-19, 80 (37%) were not hospitalised and 136 (63%) were hospitalised, of whom 19 (8.8%) were admitted to an intensive care unit. Mortality at 3 months for these subgroups was 18%, 40%, and 37%, respectively (p = 0.0031). Compared with HD patients, PD patients had higher mortality (crude HR: 1.49; 95% CI: 1.33–1.66), even when adjusted for patient characteristics and disease severity (adjusted HR: 1.56; 95% CI: 1.39–1.75). Follow-up data on 67 of 146 patients who survived COVID-19 showed functional recovery to pre-COVID-19 levels in 52 (78%) and mental recovery in 58 patients (87%) at 3 months after the COVID-19 diagnosis.
Conclusion:
The mortality rate in the first 3 months after presentation with COVID-19 is high, especially among PD patients who were hospitalised. PD patients with COVID-19 had a higher mortality risk than HD patients. The majority of surviving patients recovered both functionally and mentally from COVID-19 within 3 months.
Introduction
Previous studies have shown that dialysis patients are a vulnerable population at high risk for a severe course of COVID-19 and for COVID-19-related mortality. 1 –3 Because only about 11% of all dialysis patients worldwide are treated with peritoneal dialysis (PD), 4 it is not surprising that most of the published reports on COVID-19 include only or mainly haemodialysis (HD) patients. As a result, less is known about the course, treatment, and outcomes of COVID-19 in PD patients.
However, there are important differences between PD and HD patients which might have impact not only on the risk to get infected with SARS-CoV-2, but also on the clinical course and outcome of COVID-19. First, PD is a home-based treatment which makes self-isolation easier because travelling is not necessary. Second, PD patients are less frequently seen by their nephrologist, especially during the COVID-19 pandemic. As a result, PD patients may present at a later stage of COVID-19. Third, the presence of PD fluid in the peritoneal cavity increases intra-abdominal pressure which might lead to impaired diaphragm mobilisation and reduction in lung volume. 5,6
To date only few and relatively small series of PD patients with COVID-19 have been described, 7,8 some as part of articles that also describe other dialysis patient groups. 2,9 –12 An European Renal Association (ERA) Registry report compared 125 PD patients with 3126 HD patients and showed that there was no statistically significant difference between PD and HD patients, although the adjusted probability of death at day 28 was higher in PD patients (21.6%) than in HD patients (18.0%). 2 Unfortunately, details of the disease course and treatment were not available in this cohort. Another retrospective, single-centre study performed in the Philippines, that included 103 PD patients with COVID-19 reported higher (in-hospital) mortality on day 30 for HD (38%) than for PD patients (20%). 7 In this study, somewhat more clinical details were presented, but the number of included patients was still relatively small and the follow-up was limited to 1 month.
Thus, to our knowledge, a comprehensive analysis of the clinical course and long-term outcomes of COVID-19 in a large cohort of PD patients is lacking. We therefore aim to provide an overview of patient and disease characteristics and to describe the outcomes of COVID-19 in 216 PD patients from across Europe, based on a large multicentre study with granular data collection. These characteristics and outcomes were compared to those of HD patients from the same cohort.
Materials and methods
Study population
This study is based on data from the European Renal Association COVID-19 Database (ERACODA), which contains granular data on adult patients (aged >18 years) with kidney failure who were treated with dialysis or living with a functioning kidney allograft and who developed COVID-19. 13 The COVID-19 diagnosis was based on a positive result on a real-time polymerase chain reaction (PCR) assay or a rapid antigen test of nasal and/or pharyngeal swab specimens, and/or compatible findings on CT scan or chest X-ray of the lungs.
The database uses REDCap software, a secure web application for building and managing online databases (Research Electronic Data Capture, Vanderbilt University Medical Center, Nashville, TN, USA), 14 and is hosted at the University Medical Center Groningen (UMCG), the Netherlands. Patient information is stored pseudonymised. The study was approved by the Institutional Review Board of the UMCG, who deemed the collection and analysis of data exempt from ethics review regarding the Medical Research Involving Human Subjects Act (WMO). This study was reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline. 15
Data on outpatients and hospitalised patients were voluntarily reported by physicians responsible for their care. A total of 225 physicians from 141 centres in 33 countries, mostly in Europe and bordering the Mediterranean Sea, entered data in ERACODA. For the current study, we included all PD patients who presented for the first time with COVID-19 between February 1, 2020 and April 30, 2021 and for whom information on age, sex and hospitalisation admission was available. These patients were compared to HD patients from the same cohort with the same inclusion criteria. The inclusion started at the start of the COVID-19 pandemic in Europe with the predominant original SARS-CoV-2 genotype (wild type or alpha-variant of SARS-CoV-2), whereas it ended when the effect of the vaccination campaign in Europe became of influence.
Data collection
We collected detailed information on patient characteristics, including age, sex, ethnicity, frailty, comorbidities, hospitalisation and medication use, and on COVID-19-related characteristics such as symptoms, vital signs and laboratory test results. Frailty was assessed using the Clinical Frailty Score, which ranges from 1, representing very fit, to 9, representing terminally ill. 16 Comorbidities were recorded at presentation from patient charts.
Information on functional and mental health outcomes was collected 3 months after the first presentation with COVID-19. Due to the study design, we could not invite individual patients to report on their functional and mental outcome and we therefore asked nephrologists to report on these outcomes. Although they do not meet their PD patients very often (frequency varying between once per 4 to 12 weeks), they know most of their patients well. We composed a standardised questionnaire for this purpose (Supplementary material). If patients had not recovered at 3 months, we asked which limiting factors were judged to be responsible for not reaching the pre-COVID-19 functional and mental health status and how long they estimated the time interval to reach the pre-COVID status will be.
Statistical analysis
All baseline patient and disease characteristics are summarised using standard descriptive statistics and presented for the total population, and separately for the categories: not hospitalised, hospitalised but not admitted to an intensive care unit (ICU) and hospitalised and ICU admitted.
Unadjusted cumulative survival probability curves by hospitalisation and ICU admission status were created using the Kaplan–Meier method and compared using a Log-rank test. Unadjusted and adjusted hazard ratios (HRs) with 95% confidence interval (CI) were estimated using Cox proportional-hazards regression to compare the risk of mortality in the three subgroups according to hospitalisation status among PD patients. The multivariable model included the potential confounders age, sex and frailty score.
Because the numbers were too small in the subgroups based on hospitalisation status, we tested in the total group of PD patients the strength of age and sex adjusted associations of patient- and disease characteristics with 28-day mortality. Then a multivariable regression model was built using a backward elimination procedure on the set of variables associated with 28-day mortality after age and sex adjustment with a p value <0.1.
We also compared mortality at 28 days and 3 months between PD and HD patients using the Kaplan–Meier method and Cox regression models. We estimated unadjusted and adjusted HRs for the total group of patients and in subgroups for the presence of symptoms (yes/no) and for hospitalisation (yes/no). In the multivariate models we adjusted for age, sex, frailty score, diabetes mellitus (yes/no), temperature (°C), respiration rate (per minute) and oxygen saturation (%).
To account for missing values for frailty score (missing in 89 patients, 41.2%), we imputed missing values using multiple imputation by chained equations using the variables age, sex, hospital admission status and dialysis modality. In total, 10 imputed datasets were created with 100 iterations. Estimated coefficients and corresponding standard errors across imputed datasets were pooled as per Rubin’s Rules. 17,18
Among those who were alive at 3 months after COVID-19 diagnosis, we analysed the residence status (still in the hospital/nursing home/at home) and the functional and mental health outcome using descriptive statistics. All analyses were performed using Stata version 14.0 (Stata Corp LP, College Station, TX, USA). A two-sided p value <0.05 indicated statistical significance.
Results
Study population and patient characteristics
A total of 3380 dialysis patients with COVID-19 were included in the ERACODA database, consisting of 3041 patients on HD, 231 on PD and 108 patients with unknown dialysis modality. Information on age, sex and hospitalisation was available for 216 of the 231 PD patients included in the analyses. Eighty PD patients (37%) were not hospitalised, 117 patients (54%) were hospitalised but not admitted to an ICU and 19 patients (8.8%) were admitted to an ICU.
Table 1 shows the baseline patient and disease characteristics for the total group and the subgroups. Patients who were ICU-admitted were significantly younger and more often had abnormal X-ray and/or CT-scan findings. COVID-19-related symptoms were most severe in patients admitted to an ICU when compared to those not ICU-admitted, with more often shortness of breath, diarrhoea and myalgia/arthralgia. Patients admitted to the hospital and/or ICU also had a higher respiration rate, diastolic blood pressure, and C-reactive protein (CRP) level, and a lower oxygen saturation and lymphocyte count.
Baseline demographic and clinical characteristics of PD patients with COVID-19 by hospitalisation and intensive care unit (ICU) admission status.a
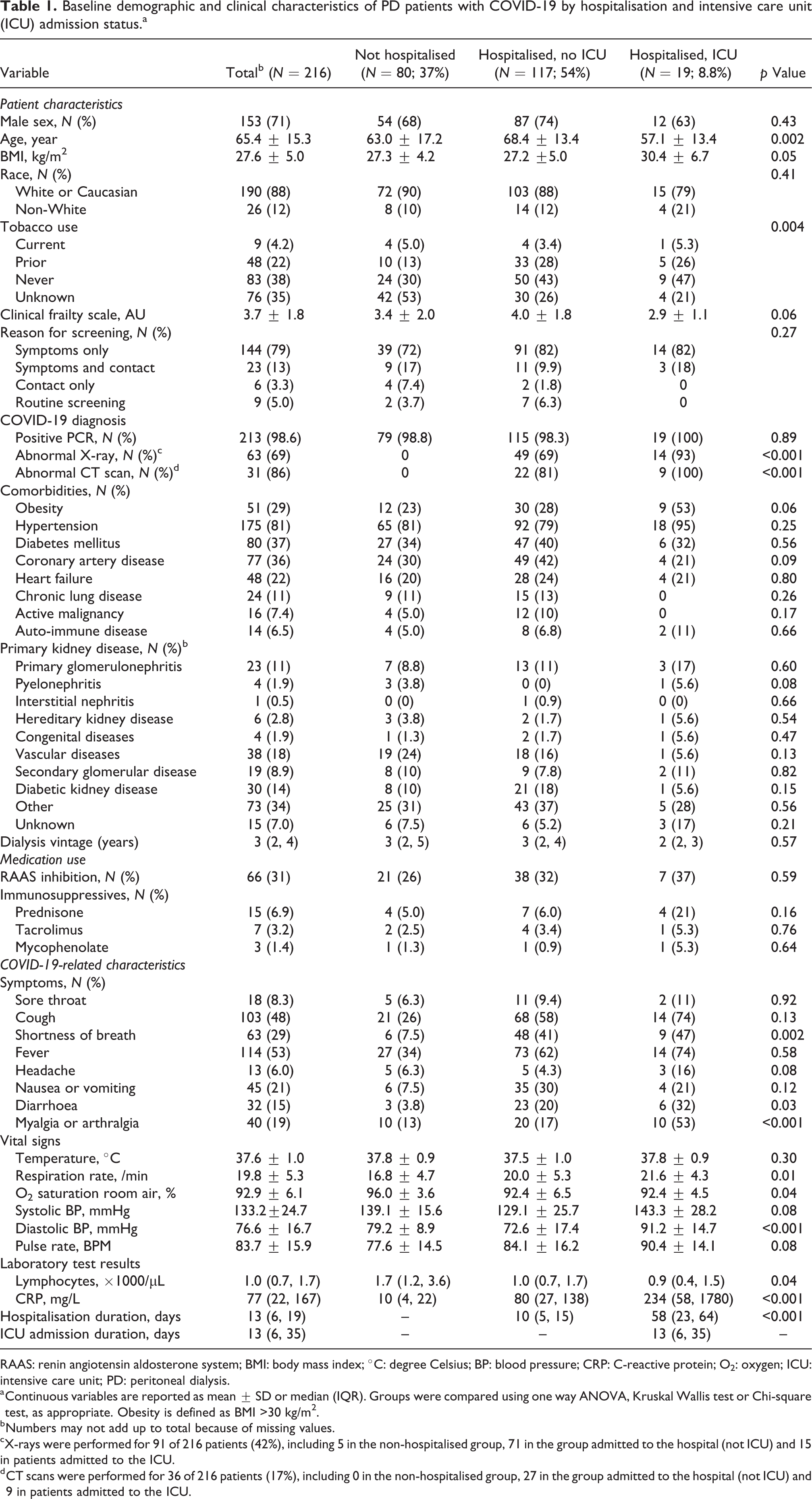
RAAS: renin angiotensin aldosterone system; BMI: body mass index; °C: degree Celsius; BP: blood pressure; CRP: C-reactive protein; O2: oxygen; ICU: intensive care unit; PD: peritoneal dialysis.
a Continuous variables are reported as mean ± SD or median (IQR). Groups were compared using one way ANOVA, Kruskal Wallis test or Chi-square test, as appropriate. Obesity is defined as BMI >30 kg/m2.
b Numbers may not add up to total because of missing values.
cX-rays were performed for 91 of 216 patients (42%), including 5 in the non-hospitalised group, 71 in the group admitted to the hospital (not ICU) and 15 in patients admitted to the ICU.
d CT scans were performed for 36 of 216 patients (17%), including 0 in the non-hospitalised group, 27 in the group admitted to the hospital (not ICU) and 9 in patients admitted to the ICU.
Patient characteristics for 2990 of the 3041 HD patients whose information on age, sex and hospitalisation was available are compared with those of PD patients in Table S1. In Table S2, characteristics are presented for HD patients in categories based on hospitalisation status; 1276 HD patients (43%) were not hospitalised, 1474 (49%) were hospitalised but not admitted to an ICU and 240 (8%) were admitted to an ICU. Overall, patients presented with COVID-19 at the hospital 1.60 days after their symptoms started; this was 1.46 days for HD patients and 2.78 days for PD patients (p = 0.002).
Mortality at day 28 and at 3 months
At day 28, the overall mortality was 28% in all PD patients, 14% in the PD patients who were not hospitalised, 36% in those who were hospitalised but not admitted to the ICU and 37% in those who were admitted to the ICU (p value Log-rank test 0.0017). Mortality at 3 months overall was 32% and for the subgroups 18%, 40% and 37%, respectively (p value Log-rank test 0.0031). Kaplan–Meier curves for the three groups are presented in Figure 1 and indicate that most of the patients died within 28 days of the COVID-19 diagnosis and that mortality between 28 days and 3 months after diagnosis was relatively low.
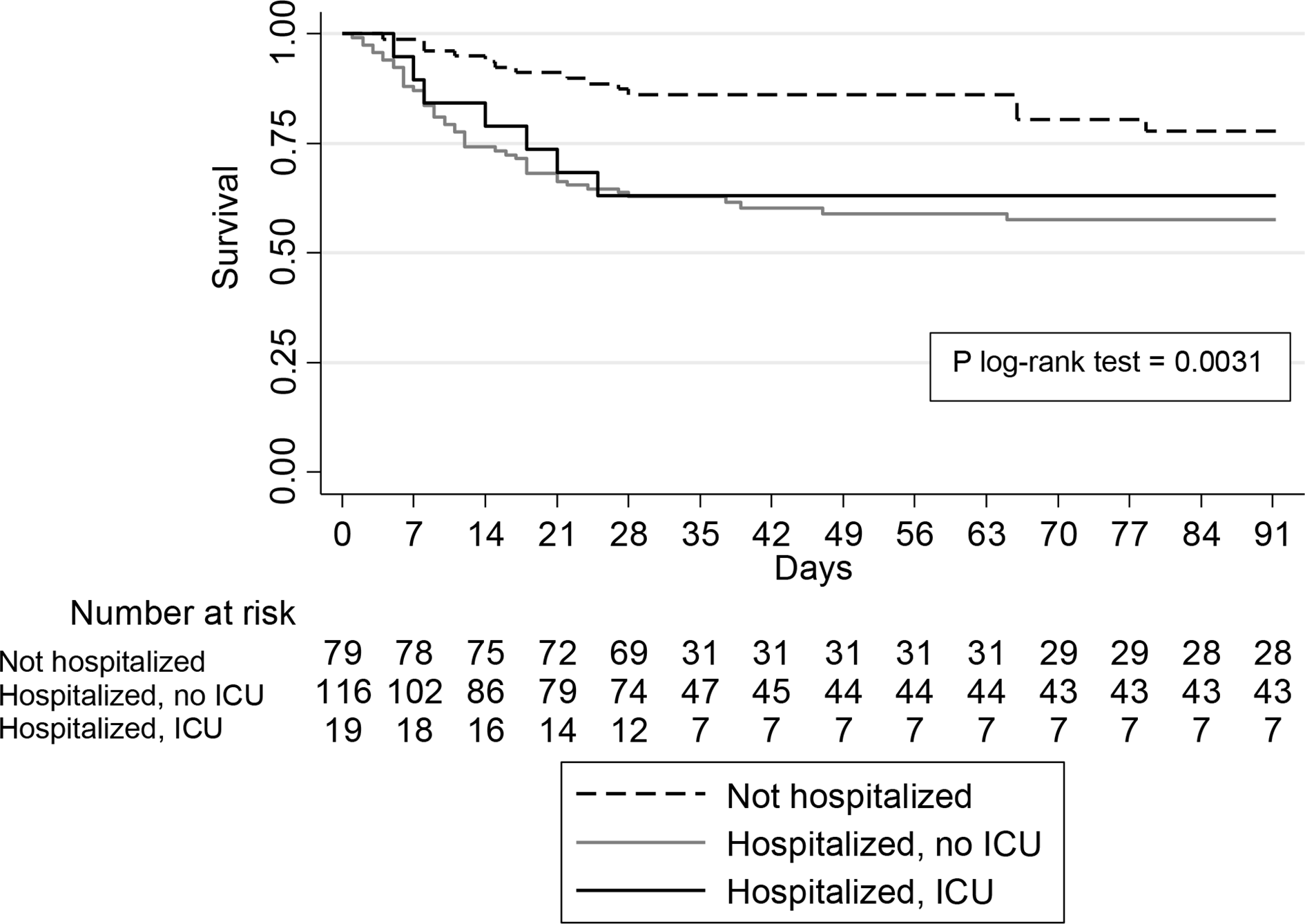
Kaplan–Meier curves presenting cumulative 3-month survival probability (in days) among PD patients with COVID-19. PD: peritoneal dialysis.
Unadjusted and adjusted HRs for mortality with 95% CIs were estimated using Cox proportional-hazards regression and are presented in Table S3. Compared with the non-hospitalised patients, the HR for mortality at day 28 adjusted for age, sex and frailty score was significantly higher in patients who were hospitalised but not admitted to an ICU (HR: 4.62; 95% CI: 3.43–6.22) and in those admitted to an ICU (HR: 6.19; 95% CI: 3.79–10.11). Mortality at 3 months was also higher for these subgroups.
When we assessed which patient and disease characteristics were associated with 28-day mortality in (all) PD patients, we found that a higher frailty score and the presence of shortness of breath as a symptom of COVID-19 had the strongest association with mortality in the fully adjusted model (Table 2).
Univariable and multivariable Cox regression analysis of patient- and disease-related factors associated with 28-day mortality in PD patients with COVID-19 (n = 216).
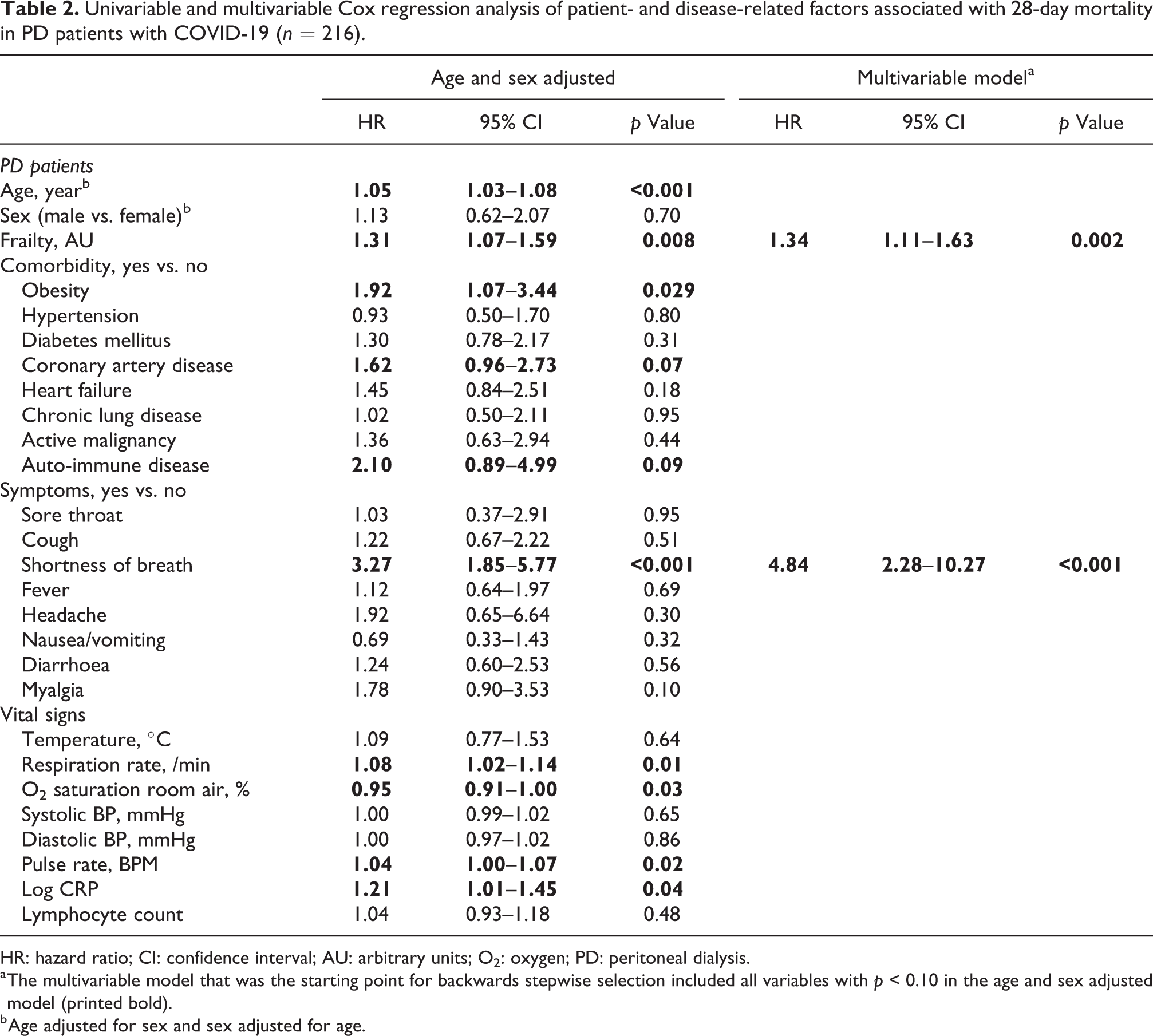
HR: hazard ratio; CI: confidence interval; AU: arbitrary units; O2: oxygen; PD: peritoneal dialysis.
a The multivariable model that was the starting point for backwards stepwise selection included all variables with p < 0.10 in the age and sex adjusted model (printed bold).
b Age adjusted for sex and sex adjusted for age.
In addition, we compared the mortality between PD and HD patients. The overall mortality at 3 months was 31.8% in PD patients and 24.9% in HD patients (Figure S1; p value Log-rank test 0.02). HRs for mortality risk in PD patients versus HD patients are presented in Table 3. In the unadjusted analysis, the risk of dying was 37% higher in PD than in HD patients (HR: 1.37; 95% CI: 1.21–1.54) and after adjusting for age, sex, frailty, diabetes mellitus, temperature, respiration rate and oxygen saturation this increased to 51% (HR: 1.51; 95% CI: 1.34–1.74). The adjusted mortality risks at 3 months was 1.56 (95% CI: 1.39–1.75).
Crude and adjusteda hazard ratios (HRs) with 95% confidence interval (CI) for mortality at day 28 and 3 months in PD patients versus haemodialysis patients.
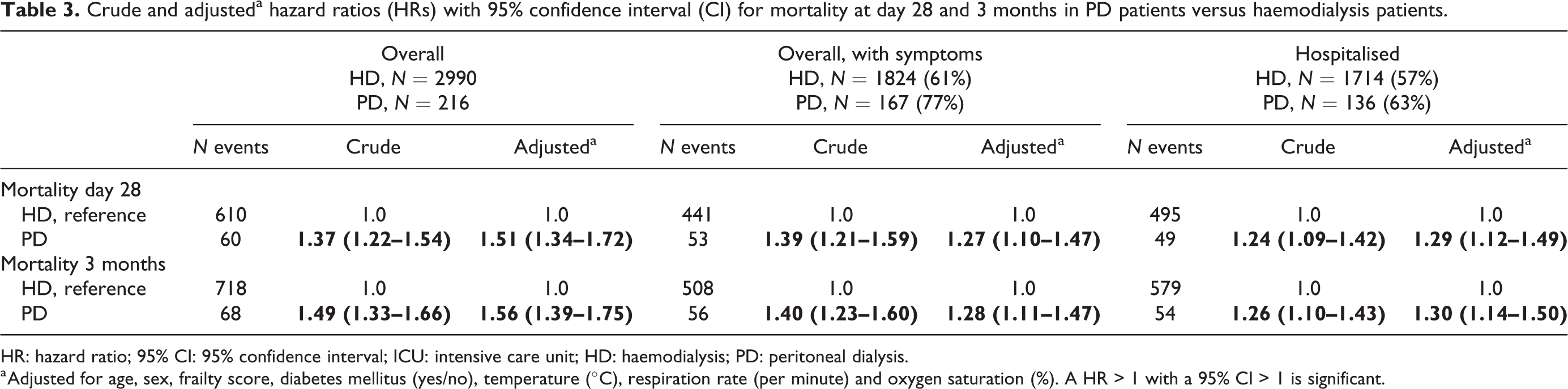
HR: hazard ratio; 95% CI: 95% confidence interval; ICU: intensive care unit; HD: haemodialysis; PD: peritoneal dialysis.
a Adjusted for age, sex, frailty score, diabetes mellitus (yes/no), temperature (°C), respiration rate (per minute) and oxygen saturation (%). A HR > 1 with a 95% CI > 1 is significant.
When we repeated the analysis only in those patients who were screened for COVID-19 because they presented with symptoms, the unadjusted HRs were very similar. The adjusted mortality risk was also significantly higher for PD than for HD, although the difference was slightly reduced to a HR of 1.27 (95% CI: 1.10–1.47) at day 28 and 1.28 (95% CI: 1.11–1.47) at 3 months. When the analysis was restricted to the patients hospitalised because of COVID-19, the mortality risks were 29% higher at day 28 (HR 1.29; 95% CI: 1.12–1.49) and 30% higher at 3 months (HR: 1.30; 95% CI: 1.14–1.50) in PD than in HD patients. The small number of PD patients admitted to an ICU (N = 19) precluded a meaningful comparison between ICU-admitted PD and HD patients.
In the far majority of cases, COVID-19 was reported to be the cause of death, with a similar proportion (p value 0.75) in PD (N = 47, 92%) and HD patients (N = 474, 91%).
Residency, functional and mental health status 3 months after COVID-19 diagnosis
Among those patients who were still alive at 3 months after the COVID-19 diagnosis (N = 146), 1 patient was still in the hospital, 4 patients were living in a nursing home, 66 patients were at home and for 75 patients (51%) the residency status was unknown.
Data on functional and mental health status were collected at the time point 3 months after COVID-19 diagnosis and were available for 67 patients who were alive at this time point and had follow-up data available. According to their nephrologist functional status had recovered to the pre-COVID-19 level in 52 (78%) of the patients, whereas this was not the case in 15 patients (22%) (Table 4). Among those who had not yet recovered, the most important limiting factors were reduced muscle strength (40%), impaired lung function (33%), reduced mobility (33%), tiredness (33%) and disturbed mental status (33%). For 74% of these patients, their nephrologist estimated that they would reach their pre-COVID-19 functional status within 1 year after diagnosis, while they estimated that 6.7% of patients would not fully functionally recover.
Functional and mental health status 3 months after COVID-19 diagnosis in PD patients (N = 67).
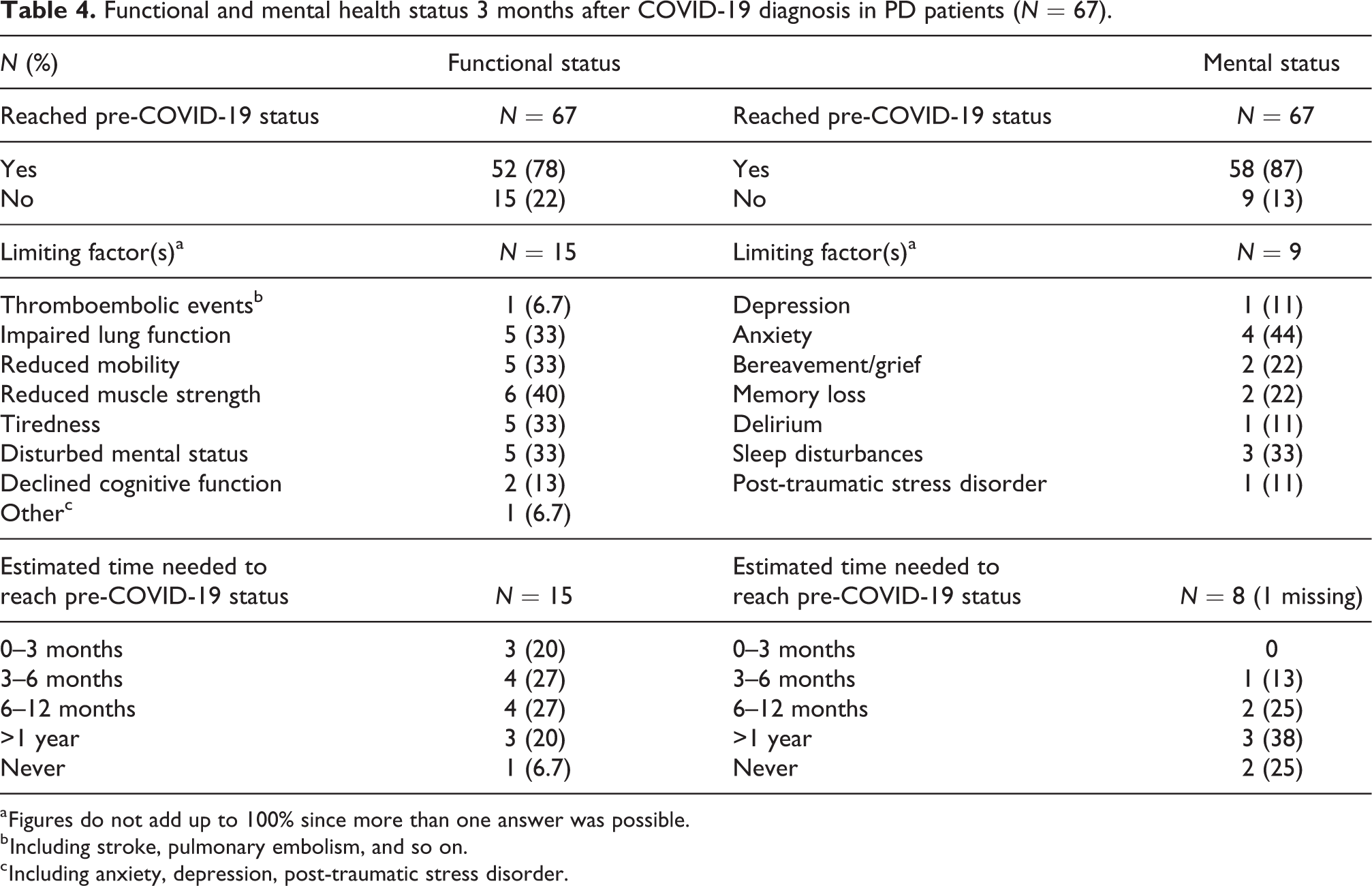
a Figures do not add up to 100% since more than one answer was possible.
b Including stroke, pulmonary embolism, and so on.
c Including anxiety, depression, post-traumatic stress disorder.
Treating nephrologists indicated that 58 (87%) of the patients had reached their pre-COVID-19 mental health status at 3 months after COVID-19 diagnosis (Table 4). Among the nine patients (13%) who had not reached their pre-COVID-19 mental health status, the most commonly mentioned limiting factors were anxiety (44%) and sleep disturbances (33%). For 38% of them, their nephrologist expected that the pre-COVID-19 mental status would be reached within the upcoming year, whereas 25% were expected to never fully recover their mental health state.
Discussion
This is the first study to show in a relatively large cohort of PD patients with detailed information on patient and COVID-19 characteristics that the risk of death in the first 3 months after COVID-19 is high. In fact, mortality was even higher than in HD patients, who were already known to have very high COVID-19-related mortality. As in HD patients, the increased risk of mortality was most pronounced in patients who were hospitalised. Furthermore, we found that the most prominent risk factors for mortality in PD patients with COVID-19 were a higher frailty score and the presence of shortness of breath as a symptom of COVID-19. Our study also shows that, although the short-term mortality of COVID-19 in PD patients is high, surviving patients have a good prognosis.
An important finding of this study was the higher risk of death for patients on PD compared to their counterparts on HD, even if we adjusted for important patient characteristics and disease severity and if we analysed only those subgroups of patients with the most severe course of COVID-19 (i.e. those with symptoms and those hospitalised). There are several factors that could potentially explain this difference. For example, the fact that PD patients presented at the hospital significantly later in the course of the disease than HD patients (2.78 days for PD vs. 1.46 days for HD patients) could play a role. Another option may be that the immune system of PD patients responds differently to SARS-CoV-2. It is known that patients with end-stage kidney disease have a weakened immune system, making them more susceptible to infection than the general population. In addition, it has been shown that they characteristically have delayed viral clearance after the resolution of clinical SARS-CoV-2 infection. To the best of our knowledge there is, however, no evidence for a difference in the immunological function between PD and HD patients.
Several other studies have also compared COVID-19-related mortality between PD and HD patients, showing mixed results. A study reporting on 103 PD patients with COVID-19 reported a higher in-hospital mortality at day 30 for HD (38%) than for PD patients (20%). 7 In contrast, some earlier reports indicated that mortality in these patient groups was not significantly different, possibly due to the small sample sizes. 7 –10 For example, according to a report from Japan, there was no difference in prognosis between the two groups when propensity score matching was performed on age and oxygen saturation. However, this study included only 25 PD patients. 19 Also in a report from the ERA Registry including data on 125 PD patients in the first pandemic wave, there was no statistically significant difference between HD and PD patients, although the adjusted probability of death at day 28 was higher in PD patients (21.6%) than in HD patients (18.0%; HR: 1.24; 95% CI 0.85–1.80). 2
Frailty and the presence of shortness of breath at presentation were the most prominent risk factors for mortality in PD patients with COVID-19. In the total group of dialysis patients in the ERACODA cohort (predominantly on HD), age and frailty were identified as the most important risk factors for mortality. 3 So, these findings are only in part in line with each other and with other previous studies. In a previous study in 106 PD patients, the most important risk factors was also older age (over 60 years). In addition, they found that diabetes mellitus, being on PD treatment for 5 years or longer, need for hospitalisation and hospital-acquired infection were important risk factors. 20 All these factors could help to identify high-risk patients. We found no difference in the HR for mortality at day 28 and at 3 months between patients who were hospitalised but not admitted to an ICU and in those admitted to an ICU. This could probably be explained by the fact that older, frail patients were less likely to be admitted to the ICU.
According to their nephrologists, surviving patients had a good prognosis; the functional and mental health had recovered to the pre-existent level in the vast majority (78% and 87%, respectively) of PD patients at 3 months after COVID-19 diagnosis. These percentages were somewhat lower than reported for HD patients in ERACODA, in whom 87% recovered functionally and 94% mentally. 21 This difference could potentially be explained by the fact that nephrologists see their PD patients less often – generally once every 4–12 weeks – when compared with HD patients, who visit the dialysis unit thrice weekly. As a result, nephrologists may have a better perception and understanding of how their HD patients feel and how well they recover.
The data that were analysed for this study were collected in the time period before the influence of vaccinations against SARS-CoV-2 played a role in the course of disease (for 4 PD patients (1.85%) and 44 HD patients (1.47%) it was known that they were vaccinated). This could be considered both as a strength and as a limitation of the study. We cannot exclude the possibility that the disease severity and mortality would have been attenuated after patients were vaccinated, but we believe that the analysis of risk factors for mortality and the comparison between HD and PD was probably not influenced by the vaccination status. Both after a natural infection and after vaccination the large majority of dialysis patients develop a robust antibody response. 22 Based on previous studies we cannot draw any firm conclusions about differences in the effect of the vaccines on the disease course of patients on PD versus HD, because two studies found a similar effect, 23,24 while two others found higher antibody levels after vaccination in PD than in HD patients. 25,26
There are other limitations that need to be acknowledged. First, not all PD patients with COVID-19 in the participating hospitals may have been recorded in ERACODA. HD patients need to visit their dialysis unit often, and some centres introduced regular screening of asymptomatic HD patients, which is not done in PD patients. It may therefore be that in PD patients the more severe cases of COVID-19 have been captured in ERACODA when compared to HD patients. This is confirmed by the comparison of disease characteristics at presentation between HD and PD patients in ERACODA, with most symptoms being somewhat more severe in PD than in HD patients, possibly because PD patients present later after the onset of symptoms at the hospital. Second, due to the design of the study the assessment of functional and mental health status was performed by physicians, because we could not contact patients. This may have yielded less precise scores which may have led to misclassification, although we cannot predict in which direction. Furthermore, there was a relatively high proportion of missing data for the clinical frailty scale among PD patients (higher than among other patient groups within ERACODA). Even though we used multiple imputation to compensate for this missingness, the data regarding frailty score should be interpreted with caution.
Strengths of this study are that it is based on the large, international ERACODA database of patients with kidney replacement treatment admitted for COVID-19. The database contains specifically collected granular data at individual patient level over a relatively long follow-up period of 3 months, which enables detailed analysis of effects of COVID-19 and enabled us to study one of the largest populations of PD patients with COVID-19 worldwide.
The results of our study clearly demonstrate that PD patients are a vulnerable group in the COVID-19 pandemic and need special attention and specialised care. This pandemic has shown that there are more and more options for ‘healthcare from a distance’. Especially for PD patients the options for remote patient monitoring could be further explored to improve the care for these patients. 27
In conclusion, the mortality of PD patients in the first 3 months after presentation with COVID-19 is exceptionally high, especially among those who were hospitalised. PD patients with COVID-19 had a higher mortality risk than HD patients, even after adjustment for patient characteristics and disease severity, and they presented at the hospital significantly later in the course of the disease than HD patients. The majority of surviving PD patients recovered both functionally and mentally from COVID-19 within 3 months, although recovery was also somewhat poorer than HD patients. Based on the study results, we recommend greater vigilance for COVID-19 in PD patients who are older and frail, and reporting shortness of breath, adopting strategies that combine vaccination drive with early diagnosis and access to therapeutics to improve clinical outcomes.
Supplemental material
Supplemental Material, sj-docx-1-ptd-10.1177_08968608221144395 - Outcomes of COVID-19 in peritoneal dialysis patients: A report by the European Renal Association COVID-19 Database
Supplemental Material, sj-docx-1-ptd-10.1177_08968608221144395 for Outcomes of COVID-19 in peritoneal dialysis patients: A report by the European Renal Association COVID-19 Database by Alferso C Abrahams, Marlies Noordzij, Eric Goffin, J Emilio Sanchez, Casper FM Franssen, Priya Vart, Kitty J Jager, Madelon van Agteren, Adrian Covic, Sandip Mitra, Carlo Basile, Constantijn Konings, Marc H Hemmelder, Raphaël Duivenvoorden, Luuk B Hilbrands, Ron T Gansevoort and ERACODA Collaborators in Peritoneal Dialysis International
Footnotes
Acknowledgements
The ERACODA collaboration is an initiative to study prognosis and risk factors for mortality due to COVID-19 in patients with a kidney transplant or on dialysis that is endorsed by the ERA. The organisational structure contains a Working Group assisted by a Management Team and Advisory Board.
ERACODA Working Group: Franssen CFM, Gansevoort RT (coordinator), Hemmelder MH, Hilbrands LB and Jager KJ; ERACODA Management Team: Duivenvoorden R, Noordzij M, and Vart P; ERACODA Advisory Board: Abramowicz D, Basile C, Covic A, Crespo M, Massy ZA, Mitra S, Petridou E, Sanchez JE, and White C.
Data sharing statement: Collaborators that entered data in ERACODA remain owner of these data. The database can therefore not be disclosed to any third party without the prior written consent of all data providers, but the database will be made available to the editorial offices of medical journals when requested.
We thank all people that entered information in the ERACODA database for their participation, and especially all healthcare workers that have taken care of the included COVID-19 patients.
Albert Schweitzer Hospital, Dordrecht, The Netherlands
- Jeroen B van der Net, MD, PhD
Ambroise Pare Hospital, APHP Paris-Saclay University, Boulogne Billancourt, France
- Marie Essig, MD, PhD
Amphia Hospital, Breda, The Netherlands
- Peggy WG du Buf-Vereijken, MD, PhD
- Betty van Ginneken
- Nanda Maas
Amsterdam UMC, Amsterdam, The Netherlands
- Brigit C van Jaarsveld, MD, PhD
- Frederike J Bemelman, MD, PhD
- Farah Klingenberg-Salahova, MD
- Frederiek Heenan-Vos, MD, PhD
- Marc G Vervloet MD, PhD
- Azam Nurmohamed, MD, PhD
- Liffert Vogt, MD, PhD
Antwerp University Hospital, Antwerp, Belgium
- Daniel Abramowicz, MD, PhD
- Sabine Verhofstede
Avicennes Military Hospital, Faculty of Medicine, Cadi Ayyad University, Marrakech, Morocco
- Omar Maoujoud, MD, PhD
AZ Delta, Roeselare, Belgium
-Thomas Malfait, MD
B. Braun Avitum, Litomerice, Czech Republic
- Jana Fialova, MD
Bellvitge University Hospital, Hospitalet de Llobregat, Barcelona, Spain
- Edoardo Melilli, MD, PhD
- Alexandre Favà, MD
- Josep M Cruzado, MD, PhD
- Nuria Montero Perez, MD, PhD
Bernhoven Hospital, Uden, The Netherlands
- Joy Lips, MD
Bravis Hospital, Roosendaal/Bergen op Zoom, The Netherlands
- Harmen Krepel, MD, PhD
Cantonal Hospital Zenica, Bosnia and Herzegovina
- Harun Adilovic, MD
‘Carol Davila’ University of Medicine and Pharmacy, Bucharest, Romania/Emergency Clinical Hospital ‘Sf. Ioan’
- Daniela Radulescu, MD, PhD
Catharina Hospital, Eindhoven, The Netherlands
- Maaike Hengst, MSc
Central Clinical Hospital of the Ministry of Interior, Warsaw, Poland
- Andrzej Rydzewski, MD, PhD
Centre Hospitalier du Nord, Luxembourg
- Philippe Braconnier, MD
- Daniel Weis, MD
Centre of Postgraduate Medical Education, Poland
- Ryszard Gellert, MD, PhD
Centrodial, São João da Madeira, Portugal
- João Oliveira, MD, PhD
Centro Hospitalar Vila Nova de Gaia/Espinho, Vila Nova de Gaia, Portugal
- Daniela G Alferes, MD
City Hospital n.a. S.P. Botkin, Moscow, Russia
- Elena V Zakharova, MD, PhD
City Hospital Zürich, Switzerland
- Patrice Max Ambühl, MD
- Rebecca Guidotti, MSc
- Andrea Walker
Claude Galien Hospital Ramsay Santé, Quincy-sous-Sénart, France
- Fanny Lepeytre, MD
- Clémentine Rabaté, MD
- Guy Rostoker, MD, PhD
Clínica de Hemodiálise de Felgueiras, Felgueiras, Portugal
- Sofia Marques, MD
Clinical Centre of Vojvodina and Faculty of Medicine Novi Sad, University of Novi Sad, Novi Sad, Serbia
- Tijana Azasevac, MD
- Gordana Strazmester Majstorovic, MD, PhD
Croatian Society of Nephrology, Dialysis and Transplantation, Croatia
- Dajana Katicic, MD
CWZ Nijmegen, Nijmegen, The Netherlands
- Marc ten Dam, MD, PhD
DaVita Geilenkirchen, Geilenkirchen, Germany
- Thilo Krüger, MD, PhD
DaVita, Wrocław, Poland
- Szymon Brzosko, MD, PhD
1st Department of Internal Medicine, Medical School, Aristotle University of Thessaloniki, Thessaloniki, Greece
- Vassilios Liakopoulos, MD, PhD
Deventer Ziekenhuis, Deventer, The Netherlands
- Adriaan L Zanen, MD
Dianet Dialysis Center, Utrecht, The Netherlands
- Susan JJ Logtenberg, MD, PhD
Dialysis Center Bochum, Bochum, Germany
- Lutz Fricke, MD, PhD
Dnipro State Medical University, Dnipro, Ukraine
- Olexandr Kuryata, MD, PhD
Elyse Klinieken voor Nierzorg, Kerkrade, The Netherlands
- Jeroen JP Slebe, MD
Epidemiology Department – High Institute of Public Health – Alexandria University, Egypt
- Samar Abd ElHafeez, MD, DrPH
Erasmus MC Transplant Institute, Department of Internal Medicine, University Medical Center Rotterdam, Rotterdam, The Netherlands
- Jacqueline van de Wetering, MD, PhD
- Marlies EJ Reinders, MD, PhD
- Dennis A Hesselink, MD, PhD
- J Kal-van Gestel, MSc
Faculty of Medicine in Pilsen, Charles University, Pilsen, Czech Republic
- Jaromir Eiselt
- Lukas Kielberger
Faculty of Medicine-Alexandria University, Alexandria, Egypt
- Hala S El-Wakil, MD, PhD
Franciscus Gasthuis & Vlietland, Schiedam, The Netherlands
- Martine Verhoeven, MD
Freeman Hospital, Newcastle upon Tyne, UK
- Ian Logan, MD
Fundació Puigvert, Barcelona, Spain
- Cristina Canal, MD, PhD
- Carme Facundo, MD, PhD
Fundación Jiménez Díaz, Madrid, Spain
- Ana M Ramos, MD, PhD
Gdansk Medical University, Gdansk, Poland
- Alicja Debska-Slizien, MD, PhD
Gelre Hospital, Apeldoorn, The Netherlands
- Nicoline MH Veldhuizen, MD
General Hospital of Athens ‘G. Gennimatas’, Athens, Greece
- Eirini Tigka
General Hospital of Serres, Serres, Greece
- Maria Anna Polyzou Konsta, MD
General University Hospital of Alexandroupolis, Alexandroupolis, Greece
- Stylianos Panagoutsos
Grande Ospedale Metropolitano and CNR, Reggio Calabria, Italy
- Francesca Mallamaci, MD
- Adele Postorino, MD
- Francesco Cambareri, MD
Grigore T Popa University of Medicine and Pharmacy, Iasi, Romania/Dr Ci Parhon Hospital, Iasi, Romania
- Irina Matceac, MD
- Ionut Nistor, MD, PhD
Haaglanden Medisch Centrum, The Hague, The Netherlands
- JHM Groeneveld, MD
- Jolanda Jousma
Haga hospital, The Hague, The Netherlands
- Marjolijn van Buren, MD, PhD
Helsinki University Central Hospital and Helsinki University, Helsinki, Finland
- Patrik Finne, MD, PhD
Hospital Clínic de Barcelona, Barcelona, Spain
- Fritz Diekmann, MD, PhD
- Federico Oppenheimer, MD, PhD
- Miquel Blasco, MD, PhD
Hospital Curry Cabral – Central Lisbon University Hospital Center, Lisbon, Portugal
- Tiago Assis Pereira, MD
Hospital das Clinicas, Universidade Federal de Minas Gerais, Brazil
- Augusto Cesar S Santos Jr, MD, PhD
Hospital del Mar, Barcelona, Spain
- Carlos Arias-Cabrales, MD, PhD
- Marta Crespo, MD, PhD
- Laura Llinàs-Mallol, PhD
- Anna Buxeda, MD
- Carla Burballa Tàrrega, MD, PhD
- Dolores Redondo-Pachon, MD, PhD
- Maria Dolores Arenas Jimenez, MD
- Alberto Mendoza-Valderrey, PhD
Hospital de Santa Cruz, Centro Hospitalar de Lisboa Ocidental, Lisbon
- Ana Cristina Martins
- Catarina Mateus
- Goncalo Alvila
- Ivo Laranjinha, MD
Hospital Gelderse Vallei, Ede, The Netherlands
- Julia M Hofstra, MD, PhD
- Machiel A Siezenga, MD, PhD
Hospital General of Alicante, Alicante, Spain
- Antonio Franco
Hospital General Universitario Gregorio Marañón, Madrid, Spain
- David Arroyo, MD, PhD
- Sandra Castellano, MD
- Maria Luisa Rodríguez-Ferrero, MD
Hospital Obispo Polanco, Salud Aragón, Spain
- Sagrario Balda Manzanos, MD, PhD
Hospital Universitario Ramón y Cajal, Madrid, Spain
- R Haridian Sosa Barrios, MSc
Imelda Hospital, Bonheiden, Belgium
- Wim Lemahieu, MD, PhD
Isala, Zwolle, The Netherlands
- Karlijn Bartelet, MD
Istanbul Faculty of Medicine, Istanbul University, Istanbul, Turkey
- Ahmet Burak Dirim, MD
- Erol Demir, MD
- Mehmet Sukru Sever, MD
- Aydin Turkmen, MD
- Seda Şafak, MD
Jeroen Bosch Ziekenhuis, Den Bosch, The Netherlands
- Daan AMJ Hollander, MD, PhD
Klinikum Aschaffenburg-Alzenau, Aschaffenburg, Germany
- Stefan Büttner, MD
Leiden University Medical Center, Leiden, The Netherlands
- Aiko PJ de Vries, MD, PhD
- Soufian Meziyerh, MD
- Danny van der Helm, PhD
- Marko Mallat, PhD
- Hanneke Bouwsma, MD
Lister Hospital, Stevenage, UK
- Sivakumar Sridharan, PhD
Lithuanian University of Health Sciences, Lithuania
- Kristina Petruliene, MD, PhD
Luzerner Kantonsspital, Luzern, Switzerland
- Sharon-Rose Maloney, MD
Maasstad Ziekenhuis, Rotterdam, The Netherlands
- Iris Verberk, MD, PhD
Maastricht University Medical Center, Maastricht, The Netherlands
- Frank M van der Sande, MD, PhD
- Maarten HL Christiaans, MD, PhD
Manipal Hospital, Manipal, India
- Mohan Kumar N
Marche Nord Hospital, Pesaro, Italy
- Marina Di Luca, MD, PhD
Marmara University School of Medicine, Istanbul, Turkey
- Serhan Z Tuğlular, MD
Martini Ziekenhuis, Groningen, The Netherlands
- Andrea B Kramer, MD, PhD
Maxima Medisch Centrum, Veldhoven, The Netherlands
- Charles Beerenhout, MD, PhD
Meander Medisch Centrum, Amersfoort, The Netherlands
- Peter T Luik, MD, PhD
Medical University Innsbruck, Innsbruck, Austria
- Julia Kerschbaum, MD (Austrian Dialysis and Transplant Registry)
- Martin Tiefenthaler, MD
Medical University of Vienna, Vienna, Austria
- Bruno Watschinger, MD
Medisch Centrum Leeuwarden, Leeuwarden, The Netherlands
- Aaltje Y Adema, MD, PhD
Moscow Regional Research and Clinical Institute, Moscow, Russia
- Vadim A Stepanov, MD, PhD
- Alexey B Zulkarnaev, MD, PhD
Necmettin Erbakan University, Meram School of Medicine, Konya, Turkey
- Kultigin Turkmen, MD
Nephrology Unit, Department of Medicine and Surgery, University of Parma, Parma, Italy
- Ilaria Gandolfini, MD
- Umberto Maggiore, MD
Nierenzentrum Reutlingen-Tübingen, Reutlingen, Germany
- Anselm Fliedner, MD
Norwegian Renal Registry, Oslo University Hospital – Rikshospitalet, Olso, Norway
- Anders Åsberg, PhD
- Geir Mjoen, PhD
OLVG, Amsterdam, The Netherlands
- Carola WH de Fijter, MD, PhD
Ospedale S Maurizio Bolzano, Bolzano, Italy
- Nicola Mongera, MD
Padua University Hospital, Padua, Italy
- Stefano Pini, MD
Radboud University Medical Center, Nijmegen, The Netherlands
- Consuelo de Biase, MD
- Angele Kerckhoffs, MD, PhD
- Anne Els van de Logt, MD
- Rutger Maas, MD, PhD
Regional Clinical Hospital, Yaroslavl, Russia
- Olga Lebedeva, MD
Regional Hospital of Malaga, Malaga, Spain
- Veronica Lopez, MD, PhD
Rijnstate Hospital, Arnhem, The Netherlands
- Louis JM Reichert, MD, PhD
- Jacobien Verhave, MD, PhD
RUDN University, Russia
- Denis Titov
Saint-Petersburg State University Hospital, Saint-Petersburg, Russia
- Ekaterina V Parshina, MD
San Marco Hospital, University of Catania, Catania, Italy
- Luca Zanoli, MD, PhD
- Carmelita Marcantoni, MD
Satakunta Central Hospital, Pori, Finland
-Kaisa Laine, MD
Saxenburgh Medisch Centrum, Hardenberg, The Netherlands
-Gijs van Kempen, MD
Sint Antonius Ziekenhuis, Nieuwegein, The Netherlands
- Liesbeth EA van Gils-Verrij, MD
Southern Health and Social Care Trust, Newry, Northern Ireland
- John C Harty, MD
Spaarne Gasthuis, Haarlem, The Netherlands
- Marleen Meurs, MD
SPWSZ Hospital, Szczecinie, Poland
- Marek Myslak
St. Anna University Hospital, Ferrara, Italy
- Yuri Battaglia, MD, PhD
St. Bassiano Hospital, Bassano del Grappo, Italy
- Paolo Lentini, MD, PhD
Streekziekenhuis Koningin Beatrix, Winterswijk, The Netherlands
- Edwin den Deurwaarder, MD
Swedish Renal Registry, Jönköping, Sweden
- Maria Stendahl, MD, PhD
Tehran University of Medical Sciences, Tehran, Iran
- Hormat Rahimzadeh, MD
Tergooi MC, Hilversum, The Netherlands
- Marcel Schouten, MD, PhD
Third Faculty of Medicine, Charles University, and Faculty Hospital Kralovske Vinohrady, Prague, Czech Republic
- Ivan Rychlik, MD, Ph.D
Toledo University Hospital, Toledo, Spain
- Carlos J Cabezas-Reina, MD
- Ana Maria Roca, MD
Treant/Scheper Ziekenhuis, Emmen, The Netherlands
- Ferdau Nauta, MD, PhD
Turgut Ozal Medical Center, Malatya, Turkey
- İdris Sahin, MD
Université Catholique de Louvain, Cliniques Universitaires St Luc, Brussels, Belgium
- Nada Kanaan, MD
- Laura Labriola, MD
- Arnaud Devresse, MD, PhD
- Alexandre Candellier, MD
University Clinical Hospital of Santiago de Compostela, Santiago de Compostela, Spain
- Anabel Diaz-Mareque, MD
University Clinical Hospital of Valladolid, Valladolid, Spain
- Armando Coca, MD, PhD
Universitary Hospital of Guadalajara, Guadalajara, Spain
- Gabriel de Arriba, MD, PhD
University Hospital Leuven, Leuven, Belgium
- Björn KI Meijers, MD, PhD
- Maarten Naesens, MD, PhD
- Dirk Kuypers, MD, PhD
- Bruno Desschans
University Hospital Martin and Jessenius Faculty of Medicine Comenius University, Martin, Slovakia
- Ivana Dedinska, MD, PhD
University Hospital Medical Center Verona, Verona, Italy
- Giuseppina Pessolano, MD
University Hospitals of Coventry and Warwickshire NHS Trust, Coventry, UK
- Shafi Malik, MD
University Hospital of Ioannina, Ioannina, Greece
- Evangelia Dounousi, MD, PhD
University Hospital of Patras, Patras, Greece
- Evangelos Papachristou, MD, PhD
University Medical Center Groningen, Groningen, The Netherlands
- Stefan P Berger, MD, PhD
- Jan Stephan F Sanders, MD, PhD
- Akin Özyilmaz, MD, PhD (Dialysis Center Groningen)
University Medical Center Ljubljana, Ljubljana, Slovenia
- Jadranka Buturović Ponikvar, MD, PhD
- Andreja Marn Pernat, MD
- Damjan Kovac, MD, PhD
- Miha Arnol, MD, PhD
University Medical Centre Maribor, Maribor, Slovenia
-Robert Ekart, MD, PhD
University Medical Center Utrecht, Utrecht, The Netherlands
- Femke M Molenaar, MD
- Arjan D van Zuilen, MD, PhD
- Sabine CA Meijvis, MD, PhD
- Helma Dolmans
University of California Irvine School of Medicine, Orange, California, USA
-Ekamol Tantisattamo, MD, MPH
University of Genoa, Genoa, Italy
- Pasquale Esposito, MD, PhD
University of Liège, Liège, Belgium
- Jean-Marie Krzesinski, MD, PhD
- Jean Damacène Barahira, MPH
University of Milan, Milan, Italy
- Maurizio Gallieni, MD
University of Navarra Clinic, Pamplona, Spain
- Paloma Leticia Martin-Moreno
University of Piemonte Orientale, Novara, Italy
- Gabriele Guglielmetti, MD
Valais Hospital, Sion & Lausanne University Hospital, Lausanne, Switzerland
- Gabriella Guzzo, MD
Vall d’Hebron University Hospital, Barcelona, Spain
- Nestor Toapanta, MD
- Maria Jose Soler, MD, PhD
VieCuri Medical Centre, Venlo, The Netherlands
- Antinus J. Luik, MD, PhD
- Willi HM van Kuijk, MD, PhD
- Lonneke WH Stikkelbroeck, MD
- Marc MH Hermans, MD, PhD
Vilnius University, Vilnius, Lithuania
- Laurynas Rimsevicius, MD, PhD
Vimercate Hospital, Vimercate, Italy
- Marco Righetti, MD
Zonguldak Ataturk State Hospital, Zonguldak, Turkey
- Mahmud Islam, MD
Zuyderland Medical Center, Geleen and Heerlen, The Netherlands
- Nicole Heitink-ter Braak, MD
Author contributions
Research idea and study design: ACA, RTG, LBH, CFMF, KJJ, MHH; data analysis/interpretation: ACA, MN, RTG, EG, JES, CFMF, PV, RD; statistical analysis: MN, PV. Each author contributed to the data collection, contributed important intellectual content during the interpretation of the results and manuscript drafting and agrees to be personally accountable for the individual’s own contributions.
Declaration of conflicting interests
The author(s) declared potential conflicts of interest with respect to the research, authorship, and/or publication of this article. SM is supported by the National Institute for Health Research at Manchester, UK, and Devices for Dignity MedTech & In vitro Diagnostics Co-operative, Sheffield, UK.
Ethical approval
ERACODA was approved by the Institutional Review Board of the University Medical Center Groningen, who deemed the collection and analysis of data exempt from ethics review regarding the Medical Research Involving Human Subjects Act (WMO).
Funding
The author(s) received financial support for the research, authorship, and/or publication of this article: Unrestricted research grants were obtained from the European Renal Association (ERA), the Dutch Kidney Foundation, Baxter and Sandoz. Neither organisation had any role in the design of the study, interpretation of results nor in writing of the manuscript.
Informed consent to participate
Because of the international design of ERACODA, laws and regulations for informed consent were different in the different participating countries.
Informed consent to publish
All authors (full and collaborative) gave their consent for publication of this article.
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
