Abstract
Background:
Computerised tomographic (CT) peritoneography is performed on peritoneal dialysis (PD) patients to identify peritoneal boundary defects, dialysate maldistributions and loculated fluid collections. Iodinated contrast media are added to dialysate and infused through the dialysis catheter, and CT images are obtained. Chemical compatibility of contrast media with dialysis solutions has not been studied. In some institutions, pharmacists charged with oversight of compounded sterile preparations have placed a moratorium on the use of contrast media-dialysate mixtures until compatibility data become available. This study was undertaken to examine the compatibility of non-ionic iodinated contrast agents added to PD solution for the performance of CT peritoneography.
Methods:
100 mL of three non-ionic iodinated contrast agents, iopamidol 370 mgI/mL, iohexol 300 mgI/mL and iodixanol 320 mgI/mL, were mixed with 2 L 1.5% dextrose PD solution and stored at 2–8°C, 25°C and 40°C. Observations at predefined intervals were made over 5 days for visual appearance, turbidity, pH, drug concentration and chemical degradation.
Results:
Iopamidol, iohexol and iodixanol were stable for 5 days under study conditions. The contrast-dialysate mixture remained clear and colourless, no turbidity changes observed, pH and drug concentrations were stable and no increase in existing impurities or new impurities were detected.
Conclusions:
The addition of commonly used non-ionic iodinated contrast agents to 1.5% dextrose dialysis solution is chemically stable, meeting the criteria set forth in the standards and guidelines of the US Pharmacopeia and the Institute of Safe Medication Practices. A protocol for performing CT peritoneography is recommended herein to facilitate patient safety and diagnostic reliability of the imaging study.
Keywords
Introduction
Computerised tomographic (CT) peritoneography has been used to identify leaks from peritoneal boundary defects, dialysate maldistributions and loculated unopacified fluid collections in peritoneal dialysis (PD) patients. 1 –4 The diagnostic study consists of infusing iodinated contrast media mixed with PD solution through the PD catheter into the peritoneal cavity followed by CT imaging. No immediate adverse drug reactions were reported when either ionic or non-ionic iodinated contrast media were used for the study. 1 –6 No change in residual kidney function was observed in 25 patients 1 week following CT peritoneography using non-ionic contrast. 7 Uremic animal studies utilising a range of non-ionic contrast media-dialysis solution concentrations for CT peritoneography with prolonged post-imaging solution exposure had small effects on the peritoneal membrane and residual kidney function. 8
Addition of the contrast media to the dialysis solution is typically performed in the radiology department by the radiologist, a qualified radiology technician or by arranged attendance of a PD nurse. Under policies for patient safety, the preparation of a contrast media-dialysate mixture meets the criteria for the immediate-use provision of the US Pharmacopeia (USP) for compounding of sterile preparations (CSP). 9 USP standards are legally recognised in the United States and elsewhere and are used in more than 140 countries. 10 Additional guidelines for CSP are provided by the Institute of Safe Medication Practices (ISMP). 11 Both USP and ISMP state that the compounding supervisor, for example, the director of pharmacy, is responsible for oversight and ongoing monitoring of CSP occurring in any department within the institution. Regardless of point of service, some healthcare systems require that all immediate-use CSPs must be formulated within the pharmacy except under emergency conditions. 12 In compliance with USP and ISMP standards and guidelines, pharmacists have raised the issue of an absence of compatibility data for contrast media-dialysis solution mixtures with regard to chemical stability and occurrence of unintended impurities. As a result, some institutions have placed a moratorium on performing CT peritoneography until such compatibility data become available. This study was undertaken to examine the compatibility of three non-ionic iodinated contrast agents added to PD solution for the performance of CT peritoneography. In addition, the use of contrast agents and the steps in conducting CT peritoneography to promote safe procedure performance and to minimise risks of complications were reviewed.
Methods and materials
Dialysate-contrast compatibility includes the following elements: stability of the contrast agent and dialysate over a defined time, absorption into the dialysate container (contrast-container interactions) and the dialysate-contrast interactions. Stability is the extent to which the contrast and dialysate retains, within specific limits and throughout its period of storage and use, the same properties and characteristics possessed at the time of its manufacture. 13,14
Materials
The compatibility and stability of non-ionic iodinated contrast media were tested with a commercially available PD solution, Dianeal 1.5% dextrose, low calcium formulation (Baxter Healthcare Corporation, Deerfield, Illinois, USA). Dianeal solution is hermetically sealed inside a one-chamber bag manufactured from medical grade plasticised polyvinyl chloride (PVC). The solution is sterile and nonpyrogenic and contains no bacteriostatic or antimicrobial agents. The three non-ionic iodinated contrast agents studied were iopamidol 370 mgI/mL, iohexol 300 mgI/mL and iodixanol 320 mgI/mL, supplied in glass vials by Unijules Life Sciences, Ltd (Nagpur, Maharashtra, India). USP injections of iopamidol, iohexol and iodixanol may contain small amounts of buffers and edetate calcium disodium as stabilisers. 15 –17
Experimental design
The study was designed to simulate clinical end-user conditions as closely as possible. Three 2 L bags of Dianeal 1.5% dextrose, low calcium solution were used for each of the three contrast agents, therefore, three sets of three bags. A 100 mL volume of each contrast agent was aseptically introduced through the injection port of three dialysate bags using a sterile syringe. Each of the three dialysate-contrast mixtures were stored without light protection at three different temperatures. The three storage temperatures selected were based upon USP definitions. 14 Refrigerated storage was selected as a temperature maintained thermostatically between 2°C and 8°C. Storage at 25°C was selected as a customary room temperature experienced in pharmacies and hospitals. The third storage temperature was 40°C, exceeding normal body temperature and above which was defined as excessive heat.
Compatibility and stability investigations of the dialysate-contrast mixtures were performed over a 5-day (120-h) period. Dialysate-contrast solution samples were taken at time points 0 (immediately after addition of the contrast agent), 8, 24, 48, 72, 96 and 120 h for bags stored at 2–8°C; time points 0, 2, 4, 6, 8, 24, 28, 31, 48, 55, 72, 96 and 120 h for bags stored at 25°C; and time points 0, 1, 3, 5, 7, 24, 28, 31, 48, 55, 72, 96 and 120 h for bags at 40°C. Wider intervals of sampling were performed for the 2–8°C group since dialysate-contrast solution refrigeration was a less likely condition for immediate-use CSP. Because of the time required to immediately perform analyses, collection of samples was staggered at alternate hours during the first 8 h for the 25°C and 40°C groups. Before each sample collection, the bags were thoroughly shaken by hand for 1 min, after which aliquots for analysis were immediately withdrawn through the injection port at the above time intervals.
Physical compatibility assessment
All samples were visually inspected inside a booth with black- and whiteboard as background to detect change in colour, cloudiness and precipitation.
Turbidity and pH
Deionised water was used as the comparator solution for turbidity and pH. Turbidity of each solution was measured using Turbiquant Turbidimeter (Merck KGaA, Darmstadt, Germany). The pH level was determined with a Metrohm 780 pH metre (Metrohm, Herisau, Switzerland).
Concentration assays
Concentrations of iohexol and iodixanol in dialysis solution were measured at predefined time intervals and temperature conditions using the potentiometric titration procedure described in the USP monographs for both of these agents. 16,17 Potentiometric titrations were performed with the 905 Titrando (Metrohm, Herisau, Switzerland). Concentrations of iopamidol in dialysis solution were measured by high-performance liquid chromatography (HPLC) applying the procedure described in the USP monograph for this agent using the Waters HPLC e2695 Separation Module (Waters, Milford, Massachusetts, USA). 15
Organic impurities assays
Changes in existing impurities or appearance of new impurities were investigated for iohexol and iodixanol in dialysis solutions for all predefined time intervals and temperature conditions using HPLC according to the assay procedures described in the USP monographs for these agents. 16,17 The USP monograph for iopamidol does not describe a specific method for organic impurity testing; therefore, the HPLC assay method used for measurement of iopamidol concentration was utilised to look for organic impurities. 15
Data analysis
Analyses began immediately upon collection of samples from the dialysate-contrast mixtures. Results are presented as descriptive analyses. Target concentrations of contrast in dialysate were calculated from the dose of contrast solution added to a 2.06 L volume of dialysis solution. The manufacturer’s target overfill volume of dialysate for a 2 L bag is 2.06 L (range 2.04–2.08 L). Contrast concentration measured immediately after mixing with the dialysis solution was defined as baseline concentration. Changes in contrast concentrations over time were calculated as percentage differences between baseline and each scheduled time period. Contrast stability was defined as a change in concentration of less than 10%. 13,14 Signs of contrast degradation as expressed by an increase in existing impurities or new impurities were examined at each scheduled time interval by comparing the chromatograph of the dialysate-contrast sample to a USP-equivalent reference standard.
Results
Physical compatibility assessment
All three dialysate-contrast solution mixtures remained colourless and clear without visible precipitation at all time intervals and storage temperatures during the 120-h study period.
Turbidity and pH
No substantial change in turbidity was observed for any of the dialysate-contrast mixtures (Figures 1(a) to 3(a)). Turbidity of the dialysate-contrast solutions was similar to deionised water. The solution pH remained stable at all time intervals and temperature conditions throughout the 120-h study period (Figures 1(b) to 3(b)).
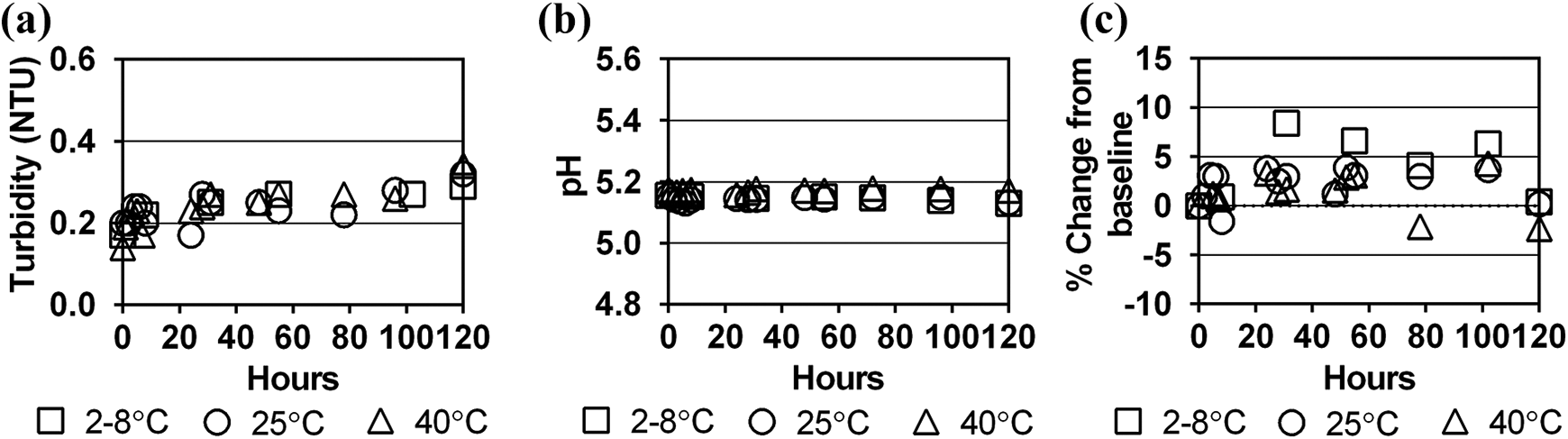
Stability of iopamidol in Dianeal 1.5% dextrose solution at three storage temperatures over 120 h as measured by (a) turbidity (NTU), (b) pH and (c) per cent change in contrast concentration from baseline concentration. NTU: Nephelometric Turbidity Units.
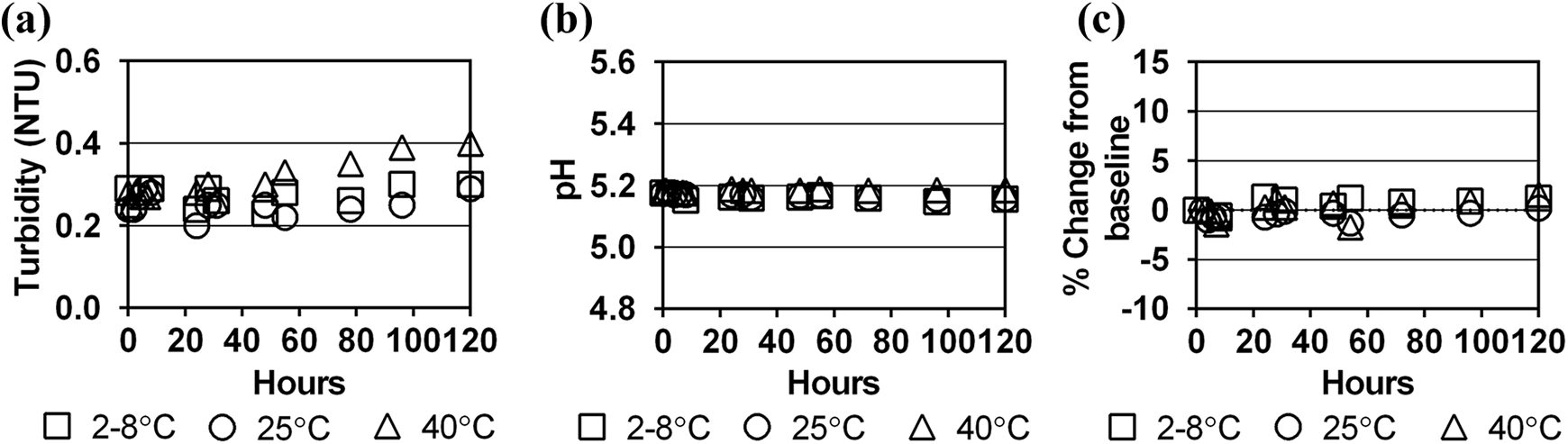
Stability of iohexol in Dianeal 1.5% dextrose solution at three storage temperatures over 120 h as measured by (a) turbidity (NTU), (b) pH and (c) per cent change in contrast concentration from baseline concentration. NTU: Nephelometric Turbidity Units.
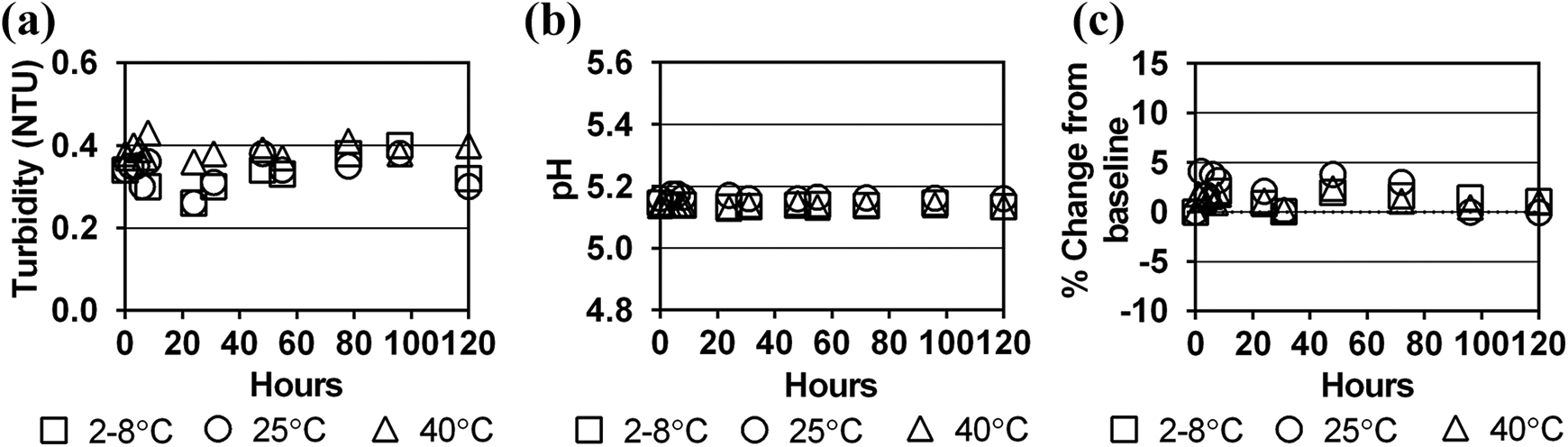
Stability of iodixanol in Dianeal 1.5% dextrose solution at three storage temperatures over 120 h as measured by (a) turbidity (NTU), (b) pH and (c) per cent change in contrast concentration from baseline concentration. NTU: Nephelometric Turbidity Units.
Concentration assays
Baseline concentrations of iopamidol, iohexol and iodixanol in a 2 L bag of dialysate differed by +1.2% to −6.5% from target levels calculated from the contrast dose and diluent admixtures (Table 1). Since the study simulated clinical end-user conditions, immediate absorption of the contrast into the PVC bag material could not be assessed due to the range of dialysate bag overfills, range of contrast concentrations in the manufactured product and imprecise syringe measurements during contrast transfers. However, compared to the baseline period, concentrations of the three contrast agents in dialysate remained stable over the 120-h study period for all storage temperatures (Figures 1(c) to 3(c)). All per cent differences in concentration from baseline concentration at each sampling period and storage temperature were <10%, meeting established criteria for drug stability. 13,14
Difference in target concentration of contrast agent and measured baseline concentration in 2.06 L dialysis solution.a
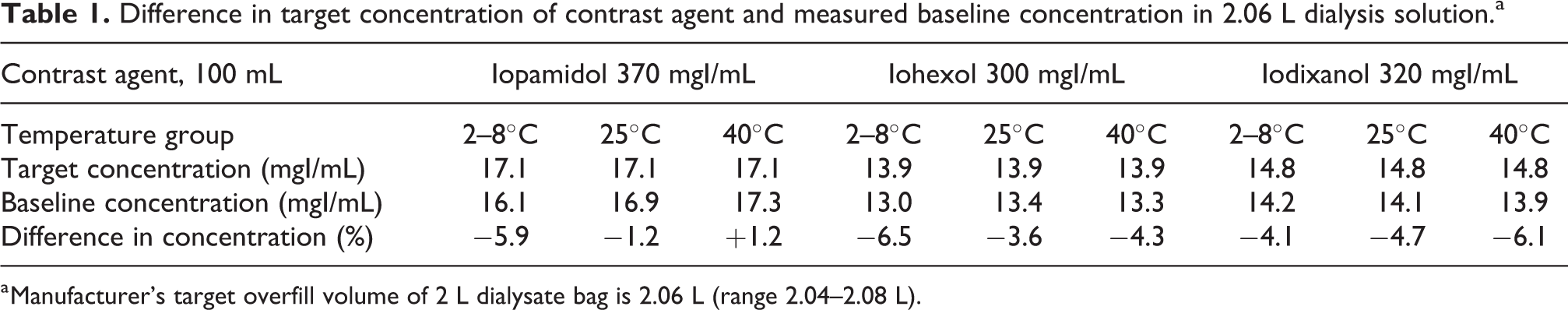
a Manufacturer’s target overfill volume of 2 L dialysate bag is 2.06 L (range 2.04–2.08 L).
Organic impurities assays
Chromatographs of the three dialysate-contrast solutions stored at the three temperature conditions showed no increase in existing impurities or the appearance of new impurities over the 120-h study period indicating stability of the dialysate-contrast mixture. Figure 4 is an example of the identical results obtained for the three temperature groups showing chromatographs of the three dialysate-contrast mixtures stored at 40°C.
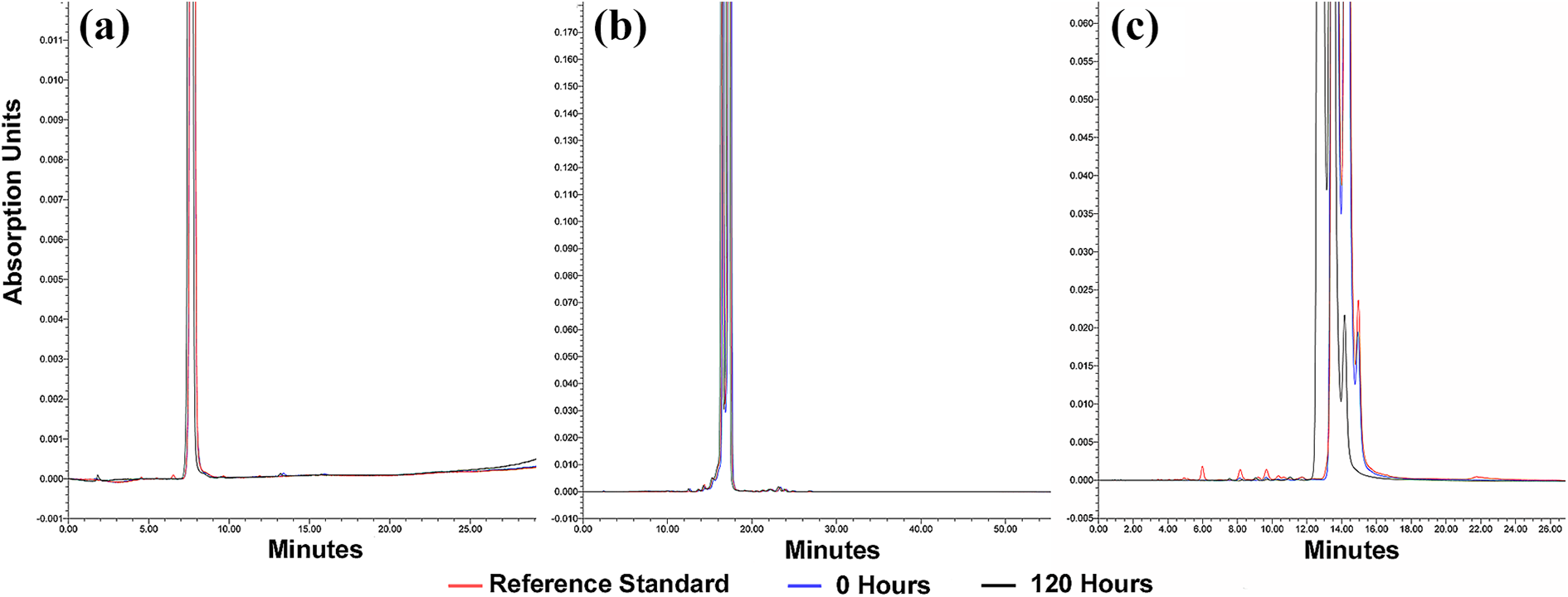
Chromatographs for the three dialysate-contrast solutions stored at 40°C to assess for presence of impurities. Chromatographic read-outs of tested samples are overlaid for comparison of the reference standard with each dialysate-contrast mixture at time 0 and the mixture at 120 h. Lower portions of the chromatographic printouts have been enlarged for illustrative purposes to permit inspection of the read-out for impurities; therefore, the peaks of the reference standard and contrast agents have been truncated. (a) Iopamidol elutes as identical single peaks for all three samples and shows no evidence of increase in existing impurities or new impurities. (b) Iohexol elutes as two major peaks and are identical for all three samples without evidence of impurities. (c) Iodixanol elutes as three major peaks and are identical for all three samples without impurities.
Discussion
Non-ionic iodinated contrast media have replaced ionic agents in clinical use because of a lower incidence of adverse reactions, especially with regard to the occurrence of contrast-induced nephropathy. 18 Frequently used for CT peritoneography, iopamidol and iohexol were studied for chemical compatibility with dialysis solution. These second-generation agents are classified as low-osmolar contrast media to distinguish them from the first-generation high-osmolar ionic agents but are still higher in osmolality than blood. Although no reports are currently found for use of iodixanol in CT peritoneography, its compatibility with dialysis solution was included in the study because it is iso-osmolar relative to blood and possibly associated with a lower incidence of contrast-induced nephropathy. 18,19 Each of these contrast agents were mixed in a 2 L bag of a commonly used 1.5% dextrose dialysis solution. The use of lower osmolarity 1.5% dextrose solution is important since higher dextrose concentrations produce significant ultrafiltration that can potentially increase contrast-related kidney toxicity due to extracellular fluid volume depletion. 20
Remarkably, CT peritoneography has been used to investigate PD complications since 1984 without formal studies to assess chemical compatibility of contrast media mixed with PD solutions. Incompatibilities go beyond obvious visible discolouration, cloudiness or presence of particulates in the solution. Comprehensively, the present compatibility study has further documented no substantial changes in turbidity or pH, absence of drug degradation or increase in existing organic impurities or appearance of new impurities, partially satisfying USP criteria for safe CSP. 9 Description of additional USP requirements for immediate-use CSP will follow.
As noted above, the addition of the three non-ionic iodinated contrast agents under study to dialysis solutions produced no chemical incompatibilities through the 5-day period of observation. This provides a wide margin of safety for chemical compatibility of the mixture since USP requirements for immediate-use CSP state that administration should begin no later than 1 h following the start of the preparation. 9 Moreover, to prevent contamination and medication errors, USP requirements for immediate-use CSP relative to CT peritoneography include aseptic technique during preparation, transfer of the contrast to the dialysis solution with no more than two needle entries into any one container, the compounding procedure is a continuous process not exceeding 1 h, and if administration is not witnessed by the preparer, the solution is appropriately labelled with patient identification, name and dose of contrast, name of the preparer and the 1-h beyond use time. 9
Investigations following CT peritoneography are scarce and, at most, suggest minimal or transient changes in kidney function. 7,8 Short-term animal studies have shown an increase in peritoneal membrane thickness and mesothelial cell number after exposure to non-ionic iodinated contrast agents. 8 Further research is warranted to determine if these histological changes are reversible or if they could contribute to epithelial-to-mesenchymal transition with long-term changes in peritoneal function, such as ultrafiltration loss and decreased solute clearance. Consequently, it is worthwhile to review the use of contrast agents and the steps in conducting CT peritoneography to promote safe procedure performance and to minimise risks of complications.
Iopamidol 21 –25 and iohexol 6,26,27 are the most frequently reported contrast agents used in performing CT peritoneography; although, iopromide 28,29 has been cited or the identities of the non-ionic agents were not stated. 30 –33 The most commonly used iodine concentration is 300 mgI/mL 22,23,25,27,28,30 –32 ; however, 350 mgI/mL 6,26 and 370 mgI/mL 21 have also been utilised. Volume of contrast added to 2 L of dialysate is usually 100 mL, 21,23,26,27,31 –33 but as little as 50 mL 22 to as much as 150 mL 25 are reported. While adding a fixed volume of contrast to 2 L of dialysate simplifies solution preparation, perhaps making compounding less error-prone, some clinical reports individualise the contrast-dialysate mixture to patient body weight, adding 1 mL/kg contrast solution to 30 mL/kg dialysate. 7,28,30 The issue with this customised formula is that it generates excessively large infusion volumes for patients weighing greater than 70 kg and potentially suboptimal volumes for petite or paediatric patients. A practical approach to be considered is to add 100 mL of a non-ionic contrast agent, 300 mgI/mL, to a 2 L bag of dialysate, a volume easily accommodated by adults, and limit the infusion in smaller patients to correspond to their usual PD exchange volume.
All non-ionic iodinated contrast media currently available possess a level of kidney toxicity. 34 The degree of contrast-induced nephropathy of non-ionic agents is primarily related to the contrast dose, its osmolality and viscosity and the hydration status of the patient. 34 –38 In hydrated patients, there does not appear to be any significant advantage of iso-osmolar iodixanol over low-osmolar contrast media except possibly in the case of iohexol. 37,39 In volume-depleted patients, the increase of iodixanol viscosity in the kidney tubules may actually increase the risk of contrast-induced nephropathy compared to low-osmolar contrast agents but further study is required. Given the relative lack of obvious advantages for use of iodixanol, its higher cost emerges as an important factor in the choice of contrast agents.
CT peritoneography is greatly facilitated by using a 2 L twin-bag continuous ambulatory peritoneal dialysis (CAPD) system which enables having to make only one connection and disconnection to the PD catheter for infusion and drainage of the contrast-dialysate solution mixture. Minimising the number of connections and disconnections reduces the opportunity for touch contamination and iatrogenic peritonitis. A twin-bag CAPD system is comprised of a dialysate-filled infusion bag and an empty drain bag joined together by a length of tubing to a Y-junction where a short third limb (patient connector line) attaches to the PD catheter transfer set. An illustration of twin-bag CAPD system components is provided in Online Supplemental Figure S1. Logistically, if a PD nurse is in attendance, they can supply the twin-bag system; otherwise, the patient can be provided the bags from the PD unit to bring with them to the radiology department. In addition, the patient should bring two tubing clamps and a replacement disinfection cap (MiniCap) containing povidone-iodine solution for the transfer set. While patients frequently initiate PD using twin bags, many have advanced to automated PD and may require a brief refresher course for a twin-bag CAPD manual exchange so they can assist the radiology staff with the connections. For time efficiency, the patient should drain the peritoneal cavity at home before coming to the radiology department.
After the contrast media have been added to the dialysate bag, the patient connector line at the Y-junction is aseptically attached to the catheter transfer set after discarding the cap. The catheter transfer set is opened to the drainage bag to allow egress of any remaining peritoneal fluid and then closed. The dialysate bag is opened to the drainage bag to allow flushing of air from the tubing. The tubing to the drainage bag is clamped and the transfer set is opened to allow infusion of the contrast-dialysate solution mixture into the peritoneal cavity. Once infused, the transfer set is closed and the dialysate bag infusion tubing is clamped. The empty twin bags remain attached to the PD catheter while the patient is ambulatory for at least 1 h to allow for thorough intraperitoneal dialysate distribution before undergoing CT imaging. If the patient is unable to ambulate, frequent turning from side to side should be performed. When CT peritoneography is being performed for a suspected abdominal wall leak, consider performing the CT scan with the patient in prone position. 31,32 The genitalia are included in the scan if genital oedema is present. 26,31 The lower thorax is included if a leak into the pleural space is suspected. 23 Following the imaging study, the patient should drain the dialysate to limit exposure to the contrast media. After draining is complete, the twin-bag CAPD system is disconnected from the closed transfer set and a new cap applied. A detailed checklist of procedure steps for performing CT peritoneography can be downloaded from Online Supplemental Table S1.
Compared to intra-arterial and intravenous injections of contrast, dilution of the contrast in dialysate minimises the rate of systemic absorption through the peritoneal capillaries and lymphatics. Removal of contrast following the imaging procedure by dialysate drainage further limits exposure. Reduced systemic exposure of the contrast media may account for the negligible or only transient changes observed in kidney function after CT peritoneography. 7,8 The magnitude of peritoneal absorption and kidney excretion of contrast media during CT peritoneography has not been studied; nevertheless, considering the importance of preserving residual kidney function in kidney failure patients, every precaution should be exercised to minimise any degree of kidney toxicity. This is best achieved by assuring that infused contrast-dialysate mixtures are chemically compatible without potentially injurious impurities, using doses of contrast media not exceeding that necessary for acceptable imaging, avoiding performance of the procedure under conditions of extracellular volume depletion, draining the contrast-dialysate mixture following the study, and using a procedure protocol that diminishes the risk of iatrogenic complications.
The limitation of this study is that chemical compatibility of low-osmolar non-ionic iodinated contrast media with dialysis solutions tested only two agents in predominant use, iopamidol and iohexol. Other low-osmolar non-ionic contrast agents include iopromide, ioversol, ioxaglate, iobitridol and iomeprol; however, frequency of their use for CT peritoneography is unknown. While iso-osmolar agents include iodixanol and iotrolan, testing was done only with iodixanol; though, there are no reports of either being used for CT peritoneography. The dialysis test solution in this study was Dianeal with 1.5% dextrose, low calcium. Due to differences in the composition of current PD solutions and drug-container interactions, contrast compatibility data generally cannot be extrapolated from one solution to another or from one container material to another. 13
Supplemental material
Supplemental Material, sj-docx-1-ptd-10.1177_08968608221096562 - Compatibility and stability of non-ionic iodinated contrast media in peritoneal dialysis solution and safe practice considerations for CT peritoneography
Supplemental Material, sj-docx-1-ptd-10.1177_08968608221096562 for Compatibility and stability of non-ionic iodinated contrast media in peritoneal dialysis solution and safe practice considerations for CT peritoneography by Jayan Rappai, John H Crabtree, Ann Mancini, Sudheer Kumar Badugu, Anuj Kaushal and Mary E Gellens in Peritoneal Dialysis International
Supplemental material
Supplemental Material, sj-docx-2-ptd-10.1177_08968608221096562 - Compatibility and stability of non-ionic iodinated contrast media in peritoneal dialysis solution and safe practice considerations for CT peritoneography
Supplemental Material, sj-docx-2-ptd-10.1177_08968608221096562 for Compatibility and stability of non-ionic iodinated contrast media in peritoneal dialysis solution and safe practice considerations for CT peritoneography by Jayan Rappai, John H Crabtree, Ann Mancini, Sudheer Kumar Badugu, Anuj Kaushal and Mary E Gellens in Peritoneal Dialysis International
Footnotes
Acknowledgements
The authors gratefully acknowledge the administrative support of Shincy P George, Swarna-gowri Addepalli, Sirisha Annam, Sharmila Muthukrishnan and Yogesh K Potdar, Baxter Innovations and Business Solutions Pvt. Ltd, Bengaluru, India. The authors thankfully acknowledge Lucy Todd and Lisa Ales for their input on PD exchange procedures.
Author contributions
JR, JHC, AM and MEG conceived the study and study design. JR, SKB and AK performed testing and data collection. JR, JHC and AM analysed the data and drafted the manuscript. All authors participated in critical review of the manuscript and approved the final version.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: JR, AM, SKB and AK are employees of Baxter Healthcare Corporation. JHC receives consultancy fees from Baxter Healthcare and DaVita Kidney Care and speakers’ honoraria from Baxter Healthcare, DaVita Kidney Care and Fresenius Medical Care. MEG is Senior Medical Director at Baxter Healthcare Corporation.
Ethical approval
Not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Contrast-dialysate compatibility investigation was funded by Baxter Healthcare Corporation.
Informed consent
Not applicable.
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
