Abstract
The wave of kidney and heart outcome trials, showing multiple potential benefits for sodium-glucose co-transport 2 (SGLT2) inhibitors, have excluded patients with an estimated glomerular filtration rate below 25 ml/min/1.73 m2. However, dialysis patients are at the highest risk of cardiovascular disease and would benefit most from effective cardioprotective therapies. There is emerging evidence from experimental studies and post hoc analyses of randomised clinical trials that SGLT2 inhibitors are well tolerated and may also be effective in preventing cardiovascular and mortality outcomes in patients with severe chronic kidney disease, including patients receiving dialysis. As such, extending the usage of SGLT2 inhibitors to dialysis patients could provide a major advancement in their care. Peritoneal dialysis (PD) patients have an additional unmet need for effective pharmacotherapy to preserve their residual kidney function (RKF), with its associated mortality benefits, and for treatment options that help reduce the risk of transfer to haemodialysis. Experimental data suggest that SGLT2 inhibitors, via various mechanisms, may preserve RKF and protect the peritoneal membrane. There is sound physiological rationale and an urgent clinical need to execute robust randomised control trials to study the use of SGLT2 inhibitors in PD patients to answer important questions of relevance to patients and healthcare systems.
Introduction
Peritoneal dialysis (PD) has gained popularity during the COVID-19 pandemic which has re-prioritised the need to explore novel and effective treatments to improve the outcomes of these patients. 1 Important therapeutic goals in PD include maintaining residual kidney function (RKF), protecting the peritoneal membrane integrity and reducing cardiovascular events in this high-risk group. 2 A relatively new agent, the sodium-glucose co-transport 2 (SGLT2) inhibitors, originally developed as oral antihyperglycaemic drugs for type 2 diabetics, have shown specific kidney cardioprotective effects in numerous studies over the last 5 years. In the chronic kidney disease (CKD) population, these agents delay disease progression in those with an estimated glomerular filtration rate (eGFR) >25 ml/min/1.73 m2 and proteinuria as well as improve survival. 3 –10 There is uncertainty as to whether these benefits can be extrapolated to patients with more advanced CKD, particularly those on PD who have been understudied with respect to cardiovascular risk reduction. Therefore, leveraging on evolving clinical evidence of potential benefits and expanding indications, there exists an unmet need to review SGLT2 inhibitors in this population.
SGLT2 inhibitors in CKD
SGLT2 inhibitors have emerged as an innovation to slow the progression of CKD. Secondary kidney benefits of SGLT2 inhibitors were found consistently in the landmark cardiovascular outcome trials of this drug class. 3 –5,11 Canagliflozin and Renal Outcomes in Type 2 Diabetes and Nephropathy (CREDENCE) was the first kidney-focused outcome trial with SGLT2 inhibitors in diabetic CKD, in this trial canagliflozin significantly slowed the progression of kidney function decline and reduced albuminuria. 7 Most recently, Dapagliflozin in Patients with Chronic Kidney Disease (DAPA-CKD) looked at the effect of dapagliflozin on morbidity and mortality in patients with both diabetic and non-diabetic proteinuric CKD (eGFR 25–75 ml/min/1.73 m2). 6 Kidney protective benefits were found across all demographic categories, geographical locations and causes of CKD. 6 Both trials also demonstrated a clear benefit of SGLT2 inhibitors on cardiovascular outcomes and all-cause death in patients with CKD. 6,7
The primary action of SGLT2 inhibitors is through the inhibition of the coupled reabsorption of sodium and glucose from the proximal tubules in the kidney, leading to natriuresis, glycosuria and diuresis. Since SGLT2 transporters are expressed nearly exclusively in the kidney, their action is attenuated at reduced eGFR levels due to a reduced number of functioning nephrons with proximal tubules containing SGLT2 receptors. 12 Consequently, they have not been thought to be effective in more advanced CKD. However, there is developing evidence from post hoc analyses, experimental and preclinical trials that support the premise that even in the setting of minimal diuresis (and little disposition of SGLT2 inhibitors to the transporter as expected in dialysis patients) SGLT2 inhibitors may exert favourable direct and indirect effects in preventing cardiovascular and mortality outcomes and others. For example, in DAPA-CKD, 14% of patients (with and without diabetes) had CKD 4 (eGFR < 30 ml/min/1.73 m2) and dapagliflozin safely and significantly reduced major kidney and cardiovascular events and attenuated progressive eGFR reduction. 13 Similarly in a post hoc analysis of CREDENCE data, where 4% of participants had an eGFR < 30 ml/min/1.73 m2, progression of CKD was slowed even in advanced diabetic nephropathy. 14 A study using serum of patients with end-stage kidney disease found that empagliflozin reduced the harmful effects of the uremic milieu on the heart. 15 This experiment was performed in an in vitro system and cardiomyocytes express SGLT1 (not SGLT2), implying that empagliflozin exerts direct effects on endothelial-cardiac tissues. SGLT2 inhibitors have been shown to bind to and inhibit the Na+/H+ exchange transporter in cardiac tissues which reduces cytosolic Na+ and Ca2+ levels improving cardiac function. 16 Further, empagliflozin and canagliflozin promote coronary vasodilation and improve cardiac energetics. 17,18
Current registered clinical trials looking at various utilities for SGLT2 inhibitors in dialysis will hopefully help bridge existing knowledge gaps. They include (1) SGLT2 Inhibition in Hemodialysis (EMPA-HD), assessing empagliflozin for cardiac mass reduction as measured by MRI at baseline and after 6 months of treatment with a planned sample size of 108 haemodialysis (HD) patients (NCT05179668), (2) a pilot study testing the safety of dapagliflozin for heart failure treatment in HD patients (NCT05141552) and (3) a PD study in 40 patients designed to assess the hypothesis that dapagliflozin can decrease glucose absorption from the PD fluid thereby improving ultrafiltration with a reduction in damaging intraperitoneal glucose (NCT04923295). Notably, no existing trial is looking at the use of SGLT2 inhibitors for the outcome of preservation of RKF in PD patients.
SGLT2 inhibitors in PD patients: RKF protection and other possible benefits
The predictive value of RKF was highlighted in the CANUSA study. Every 0.5 ml/min higher RKF was associated with a 9% lower risk of death, and total small solute clearance, both peritoneal and kidney, was a predictor of mortality. 19 However, subsequent evidence showed that the small solute clearance by the kidneys is associated with outcome, not by the peritoneum. 20,21 The apparent survival advantage conferred by RKF could be attributed to multiple factors, including urine volume excretion allowing better attainment of euvolemia and normokalaemia, some preserved kidney endocrine function (greater endogenous production of erythropoietin and calcitriol) and better clearance of middle and larger molecular weight uremic toxins. 22
Peritonitis is the most common cause for transitioning to HD from PD. 23 RKF loss, often portending inadequate dialysis, is another important reason to transition from PD and can be represented by both the slope of the decline of calculated creatinine clearance (the mean of the 24-h creatinine clearance and urea clearance) and the time to anuria. 22 Despite improvements in PD technique survival up to 70% of patients in a multicentre US study switched from PD to HD within the first 2 years. 24 In another large Canadian cohort, the median time to PD technique failure was less than 2 years. 25 Similarly, analyses carried out using data from the United States Renal Data System, on 66,381 chronic PD patients from 1996 to 2004, found that only 50% of patients were still on PD 2 years after initiation. 26
Current strategies to preserve RKF are limited such as restricting the duration of interval HD, prevention of peritonitis, avoidance of nephrotoxins, use of biocompatible PD fluids and the usage of renin–angiotensin–aldosterone system (RAAS) inhibitors. 27 –29 Aside from the expected physiological decline in kidney function over time, various modifiable factors are known to contribute to the loss of RKF in PD. In a cohort of new PD patients in Hong Kong proteinuria, glucose exposure and the number of peritonitis episodes were independent predictors of progression to anuria. 30 In a group of patients in the Netherlands, baseline factors that were negatively associated with RKF included higher urine protein loss and higher diastolic blood pressure. 31 The presence of diabetes, nephrotoxin use, poorly controlled hypertension, left ventricular hypertrophy, coronary artery disease and congestive heart failure have all been associated with a faster decline in RKF in dialysis patients. 27
For a clinical trial assessing the outcome of slowing the trajectory of decline of RKF in PD, estimating that eGFR declines on PD initiation (using available limited data) at 2.5 ml/min/1.73 m2/year 30,32 and a difference of 1 ml/min/1.73 m2/year supports clinical benefit, 33,34 a sample size of 309 participants in total would provide 90% power and a 5% two-sided α, to detect this annual relative difference in eGFR decline. This accounts for a 10% dropout rate, 62.5% technique failure rate, 33% mortality rate and 50% transplantation rate over the entire study period. 35 Based on CREDENCE and DAPA-CKD, we would expect a low rate of loss of follow-up (approximately 0.1–0.3%) as the outpatient population enrolled is stable and ‘captive’ in the real world. Similarly, assuming a loss of about 750 ml urine output per year on PD initiation, 36,37 and CANUSA showing that a 250 ml increment in 24-h urine volume conferred a 36% decrease in the relative risk of death, 21 168 patients in total would be required to detect a 250 ml difference in urine output loss. Although RKF preservation would be an important secondary outcome, all-cause mortality would be the primary outcome of interest in this population with an existing low 5-year survival rate. 2 With a yearly all-cause mortality rate of 13% in the control group and a relative hazard reduction of 25% (hazard ratio 0.75), a total sample size of 811 patients, with similar attrition rates over the study period and power, would be needed for this outcome. 6,7 However, the anticipated relative risk reduction for all-cause mortality with SGLT2 inhibitors would likely be lower in PD than what was found in CKD studies, a larger sample size would ultimately be required for adequate power.
On review of the risk factors for RKF loss, SGLT2 inhibitors, although not reducing hyperglycaemia in dialysis patients, should have beneficial effects in albuminuria reduction, which could also aid in reducing protein wasting in proteinuric PD patients with preserved urine output. In an animal model of diabetic kidney disease, dapagliflozin also reduced by 50% the biomarkers Kidney Injury Molecule-1 (KIM-1) and Neutrophil gelatinase-associated lipocalcin (NGAL), which are highly sensitive indicators of tubular damage also responsible for kidney inflammation and fibrosis. 38 Intradialytic hypotension, in patients transitioned temporarily to HD, may hasten the loss of RKF by producing intermittent abrupt volume depletion. SGLT2 inhibitors theoretically could protect RKF during periods of HD as evidenced in ischaemia–reperfusion injury models. 39,40 Further, SGLT2 inhibitors have been demonstrated to have antifibrotic and anti-inflammatory effects in various aetiologies of CKD, as well as improving podocyte dysfunction and loss. 9,41 –43 In addition to this, in a gentamicin-induced nephrotoxicity rat model, dapagliflozin showed early protection against drug-induced kidney injury. 9 Finally, knowing that sympathetic nervous system activation is associated with the progression of CKD, canagliflozin reduced kidney oxidative stress and inflammation in a mouse model which mimicked sympathetic nervous system activity. 9 Taken together, it is biologically plausible that SGLT2 inhibitors may well have the ability to slow the natural progression of CKD (and the loss of RKF) by multiple possible mechanisms, even in those on kidney replacement therapy.
The possible benefits of SGLT2 inhibitors on the peritoneal membrane in PD patients are under investigation. In animal models, dapagliflozin significantly reduced effluent transforming growth factor concentrations, peritoneal thickening and fibrosis, as well as microvessel density, resulting in improved ultrafiltration. 43 Although experimentally empagliflozin was not found to reduce glucose absorption, and therefore would not increase osmotic water transport during PD and improve ultrafiltration, this trial was performed in a rat model therefore it is unclear as to whether this can be generalised to humans. 44 Further, in an experimental analysis of human peritoneal biopsies, expression of SGLT2, glucose transporter 1 and glucose transporter 3 was found. Protein expression of SGLT2 increases with PD duration and is significantly enhanced in encapsulating peritoneal sclerosis patients suggesting that this upregulation may be associated with pathological changes in the peritoneal membrane in this syndrome. 45 Since preclinical studies in mice show that downregulation of SGLT2 ameliorated pathological changes in the peritoneum, 40 SGLT2 inhibitors may delay the functional deterioration of the peritoneal membrane in the long term.
Given their pleiotropic effects, SGLT2 inhibitors (at least conceptually) could be a viable treatment option for improving health-related quality of life (by cardiovascular effects and lowering the risk of transfer to HD), slowing the decline of RKF in PD patients and preserving the longevity of PD treatment (Figure 1).
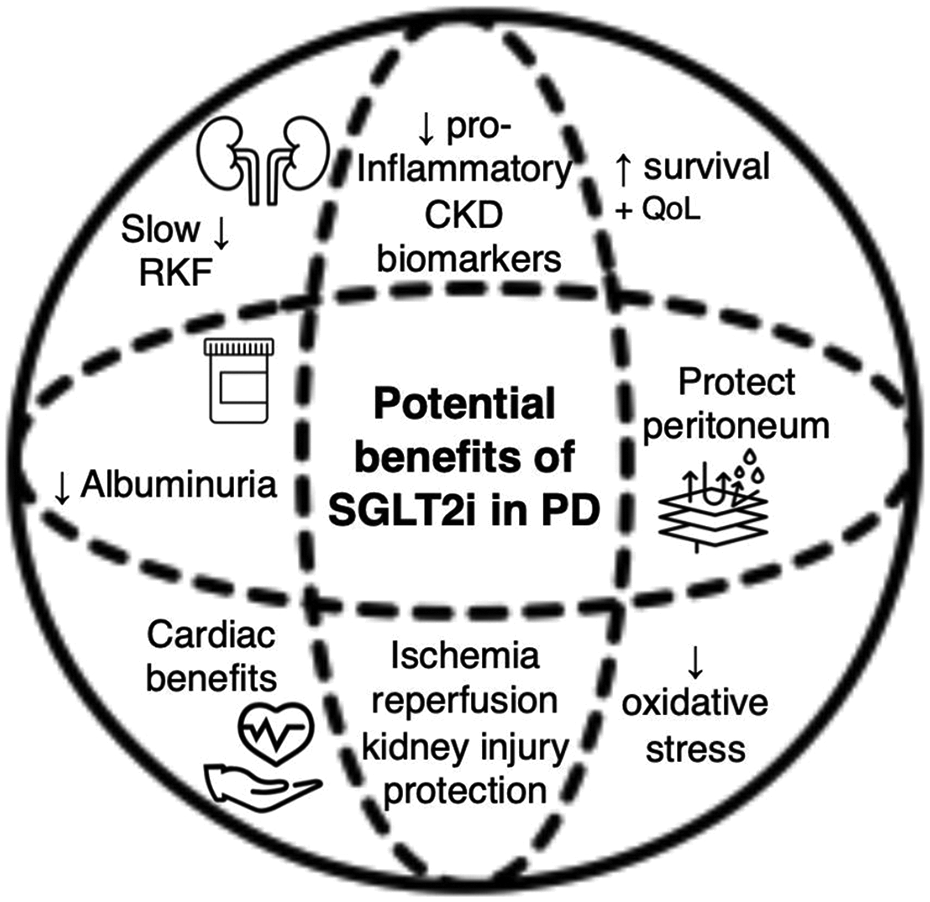
Potential benefits of SGLT2 inhibitor use in PD patients.
What are the safety concerns for SGLT2 inhibitor use in PD patients?
Overall, SGLT2 inhibitors have consistently shown a good safety profile, in both diabetic and non-diabetic patients with CKD. 6,46 A major concern with the use of these drugs is the risk of euglycaemic diabetic ketoacidosis (DKA), glycosuria is central to the proposed mechanism behind this risk where kidney glucose clearance exceeds endogenous production. 47 Another adverse effect that has consistently been reported involves genital mycotic infections related to glycosuria. 48 SGLT2 inhibitors have also been associated with rare but life-threatening Fournier’s gangrene of the perineum. 49 However, since the glycosuric effect of this drug dissipates beyond stage 3 CKD, the risk of euglycaemic DKA, genitourinary infections and even hypoglycaemia should not be of concern in the PD population. 12 Due to no prior clinical trials of SGLT2 inhibitors in PD patients, it is unclear if their use would increase the risk of fungal peritonitis (both primarily and in patients undergoing treatment for bacterial peritonitis). Interestingly, higher serum hepcidin-25 level is associated with an increased risk of all-cause and infection-related mortality in PD patients, SGLT2 inhibitors have been associated with a reduction in the level of hepcidin therefore may have a protective role in cases of PD peritonitis. 50
The second safety concern is that SGLT2 inhibitors have not yet been studied in patients with eGFR < 25 ml/min/1.73 m2. However, both in DAPA-CKD and CREDENCE, patients continued on active drugs until the commencement of dialysis, and in DAPA some continued despite starting dialysis. In the subgroup analyses of DAPA-CKD and CREDENCE, of participants with an eGFR < 30 ml/min/1.73 m2 at baseline randomisation, rates of serious adverse events (including acute kidney injury and hyperkalaemia) were similar among participants, even those with low eGFR, treated with dapagliflozin or placebo. 13,14 Moreover, in a meta-analysis, including four studies and a total of 38,723 participants, SGLT2 inhibitor usage was also significantly associated with a 25% reduction in the risk of acute kidney injury, including for participants with a baseline eGFR 30–45 ml/min/1.73 m2. 51
A third safety concern related to SGLT2 inhibitors is the initial reversible acute drop in eGFR, believed to be a haemodynamic effect, both with the concurrent use of RAAS blockers (prescribed to many PD patients for peritoneal membrane protective benefits) and in isolation. RAAS blockade and SGLT2 inhibitors reduce intraglomerular pressure (SGLT2 inhibitors by afferent arteriolar vasoconstriction, RAAS blockers by efferent arteriolar vasodilation) and subsequently reduce albuminuria. In EMPA-REG, it was observed that concomitant RAAS use enhanced the effect of empagliflozin to reduce albuminuria and progressive kidney disease, although empagliflozin had these protective effects even in the absence of intercurrent RAAS blocker use. Importantly, there were no additional safety findings for the empagliflozin–RAAS inhibitor combination compared to empagliflozin alone. 51,52 Similar results were seen in CREDENCE. 51 The initial dip in eGFR with SGLT2 inhibitor usage occurs regardless of diabetes status and CKD stage and starts at 2 weeks with a subsequent gradual return to baseline from 4 weeks and stabilisation of eGFR. 8 In the context of incident PD patients, this may only limit initial assessment of the effect of dialysis itself. The magnitude of the acute drop is more dramatic in patients with a higher eGFR as demonstrated in a secondary analysis of CREDENCE data. 53 The extent of the initial drop in eGFR has no effect on the subsequent trend in eGFR except in patients with more advanced CKD in whom a larger initial drop in eGFR is actually beneficial as it is associated with a slower decline in the eGFR slope overtime. 53
A final, less common concern is related to lower limb amputations. The Food and Drug Administration had previously issued a black box warning for canagliflozin regarding atraumatic leg and foot amputation risk based on results from two clinical trial results. 7,11 This was removed in 2020 but remained described in the ‘Warnings and precautions’ section of the prescribing information. 54
Conclusion
In light of emerging evidence and ongoing limitations in current therapeutic strategies to optimise outcomes in PD patients, there is accumulating rationale to support SGLT2 inhibitor use in this population. The ability to preserve RKF in PD patients and the potential to protect the peritoneal membrane and improve cardiovascular outcomes may all be interrelated in this unique population. A rigorous, adequately powered clinical trial should be conducted to ascertain the safety and efficacy of SGLT2 inhibitors in PD patients.
Footnotes
Acknowledgements
None
Author contributions
MB and AL researched literature and conceived the review. MB and AL wrote the first draft of the manuscript. All authors reviewed, gave input and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
None
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
None
