Abstract
Background:
Technique survival is a core outcome for peritoneal dialysis (PD), according to Standardized Outcomes in Nephrology-Peritoneal Dialysis. This study aimed to identify modifiable causes and risk factors of technique failure in a large Dutch cohort using standardised definitions.
Methods:
Patients who participated in the retrospective Dutch nOcturnal and hoME dialysis Study To Improve Clinical Outcomes cohort study and started PD between 2012 and 2016 were included and followed until 1 January 2017. The primary outcome was technique failure, defined as transfer to in-centre haemodialysis for ≥ 30 days or death. Death-censored technique failure was analysed as secondary outcome. Cox regression models and competing risk models were used to assess the association between potential risk factors and technique failure.
Results:
A total of 695 patients were included, of whom 318 experienced technique failure during follow-up. Technique failure rate in the first year was 29%, while the death-censored technique failure rate was 23%. Infections were the most common modifiable cause for technique failure, accounting for 20% of all causes during the entire follow-up. Leakage and catheter problems were important causes within the first 6 months of PD treatment (both accounting for 15%). APD use was associated with a lower risk of technique failure (hazard ratio 0.66, 95% confidence interval 0.53–0.83).
Conclusion:
Infections, leakage and catheter problems were important modifiable causes for technique failure. As the first-year death-censored technique failure rate remains high, future studies should focus on infection prevention and catheter access to improve technique survival.
Introduction
Peritoneal dialysis (PD) is an established treatment for kidney failure, offering patients more flexibility and independence compared to in-centre haemodialysis. 1,2 Improving the technique survival of PD, that is, preventing technique failure, remains a challenge despite advances in technique survival over the past decades. 3 –5 In fact, technique survival was chosen as one of the five core outcomes for PD according to the Standardized Outcomes in Nephrology-Peritoneal Dialysis study. 6
Identifying modifiable causes and risk factors of technique failure could contribute to develop strategies to improve PD technique survival. Previous research has identified causes and risk factors of technique failure during the first months of PD treatment. 7 –9 Although technique failure after the first months of PD treatment is also relevant for the loss of prevalent PD patients, few studies have explored the various causes over an extended period of PD treatment. 10 –12
Moreover, comparing previous research on technique failure is hampered by the lack of standard definitions. 8 Technique failure is defined differently in almost every other study, especially in handling death as a cause of technique failure. Lan et al. therefore advocated the use of a standardised definition of technique failure, including both transfer to in-centre haemodialysis (CHD) and death. 13 Few studies to date have used this standardised definition. 3,7
In addition, the characteristics of PD patients have changed over time and studies on technique failure in the current PD population are scarce. Therefore, this study aims to investigate the causes, risk factors and centre variation of PD technique failure in a recent Dutch cohort, all according to the standardised definitions.
Methods
Study design and research population
Patients were enrolled from the retrospective Dutch nOcturnal and hoME dialysis Study To Improve Clinical Outcomes, a multi-centre cohort study in the Netherlands. In this study, 33 centres included PD patients, representing nearly two thirds of all dialysis centres in the Netherlands. Eligible patients were adults who started PD between 1 January 2012 and 1 January 2017 and had a minimum PD treatment duration of 14 days. Patients who were previously treated with dialysis or kidney transplantation were also included. Patients who stopped dialysis or died within 30 days after dialysis initiation were excluded. Patients were followed until kidney transplantation, wish to stop dialysis, death or end of study period on 1 January 2017. Local medical ethics committees of all participating dialysis centres approved the study. Reporting of the study conforms to broad STROBE guidelines. 14
Definition of PD technique failure
The primary outcome of this study was PD technique failure, defined as a transfer to CHD for ≥ 30 days, death on PD or death within 30 days after transfer to CHD, in accordance with the previously proposed standardised definition. 13 In patients with multiple episodes of technique failure, only the first episode of technique failure was analysed. The following causes for technique failure were collected from the electronic patient charts: PD-related infections consisting of PD peritonitis and exit-site infections, catheter-related problems, clearance or ultrafiltration (UF) problems, peritoneal leakage, psychosocial problems, risk for or diagnosis of encapsulating peritoneal sclerosis (EPS), another reason, stop dialysis and death. 15
In addition, patients were stratified into an early and a late technique failure group. Early technique failure was defined as technique failure during the first 6 months after start of PD, and late technique failure was defined as technique failure that occurred more than 6 months after start of PD. 8,9,16
Secondary outcomes were death-censored technique failure, death and permanent technique failure, the latter was defined as a transfer to CHD for ≥ 180 days, death on PD or death within 180 days after transfer to CHD. 13
Covariates
Demographic, clinical and dialysis-related data at dialysis initiation were collected from electronic patient charts. These included age, sex, ethnic background, employment status, smoking, body mass index (BMI), primary kidney disease, comorbid conditions, dialysis vintage and kidney transplant history. PD modality, that is, continuous ambulatory PD (CAPD) or automated PD (APD), was defined as the modality the patient used most of the time during follow-up. BMI was divided into three groups according to the World Health Organization classification: BMI <25 kg/m2, BMI 25–30 kg/m2 (overweight) and BMI ≥30 (obese). Comorbid conditions were scored into three groups according to the Charlson Comorbidity Index (CCI): low (2 points, since patients with kidney failure by definition already have 2 points), intermediate (3–4 points) and severe comorbidity (≥5 points). 17 Causes of death, coded according to the ERA-EDTA coding system, were retrieved from the Dutch renal registry (RENINE). 18 For each participating centre, PD volume was calculated from data provided by RENINE, as mean annual number of prevalent patients, and divided into tertiles. 19 Variation in practice patterns was collected with an additional questionnaire that was send to the local investigators of the participating centres.
Statistical analysis
Baseline characteristics were expressed as number with percentages for categorical variables and as mean with standard deviation or median with interquartile range (IQR) for continuous variables. Incidence of all-cause technique failure was presented as a Kaplan–Meier curve. Cumulative incidence curves of cause-specific technique failure were calculated using a competing risk model. 20 Causes of early and late technique failure were shown as percentages.
To investigate the association between possible risk factors and technique failure, a cox regression model was conducted. This model was censored for kidney transplantation. BMI and PD modality were selected as potentially modifiable patient-specific risk factors according to literature. 3,7,9,12,16 Each potentially modifiable risk factor was adjusted for plausible predetermined confounders (age, sex, employment status, BMI, CCI and centre PD volume). The proportional hazard assumption was verified in the unadjusted models on the basis of Schoenfeld residuals and Kaplan–Meier graphs. Several sensitivity analyses were conducted. First, a competing risk model was used to investigate the association between possible risk factors and technique failure in the presence of a competing event. 20 In such a model, a participant with the competing event (i.e. kidney transplantation) remains in the analysis. This model was also used to investigate the association between possible risk factors and death-censored technique failure, in which both kidney transplantation and death were competing events. Second, hypothesising that PD modality at PD cessation might be different from PD modality used most of the time and be related to technique failure, in patients with technique failure the PD modality at PD cessation was used.
Finally, a funnel plot was constructed to evaluate the early technique failure rate of the participating centres, adjusted for age and sex. This is a graphical method to evaluate centre performance with a reference standard, that is, the overall early technique failure rate, and an indication of precision through control limits based on sample sizes. 21,22 The early technique failure rate was chosen, because especially early failure is associated with catheter-related problems and thus possible modifiable causes. 8
Missing confounders (maximum of 25% missing for BMI and CCI) were imputed using standard multiple imputation techniques in SPSS (10 repetitions and predictive mean matching). All analyses were performed using SPSS Statistics version 26 (IBM) or STATA 14 (StataCorp LP, College Station, Texas, USA). A
Results
A total of 708 adult patients started PD treatment between 2012 and 2016 in the participating centres, of whom 13 patients were excluded since they had a total PD duration of less than 14 days. The study population thus consisted of 695 patients (see flow diagram, Figure 1). Baseline characteristics are presented in Table 1. Mean age at dialysis initiation was 62.9 ± 15.1 years and 27% of patients had a high CCI score indicating severe comorbidity. A history of previous dialysis was present in 15% of patients. APD was the predominantly used PD modality in 29% of patients with early technique failure and 53% of patients with late technique failure, reflecting common practice in the Netherlands to start PD therapy with CAPD. The median PD follow-up time for all patients was 13 months [IQR 6–22.2 months], with a minimum of 0 and a maximum of 59 months.
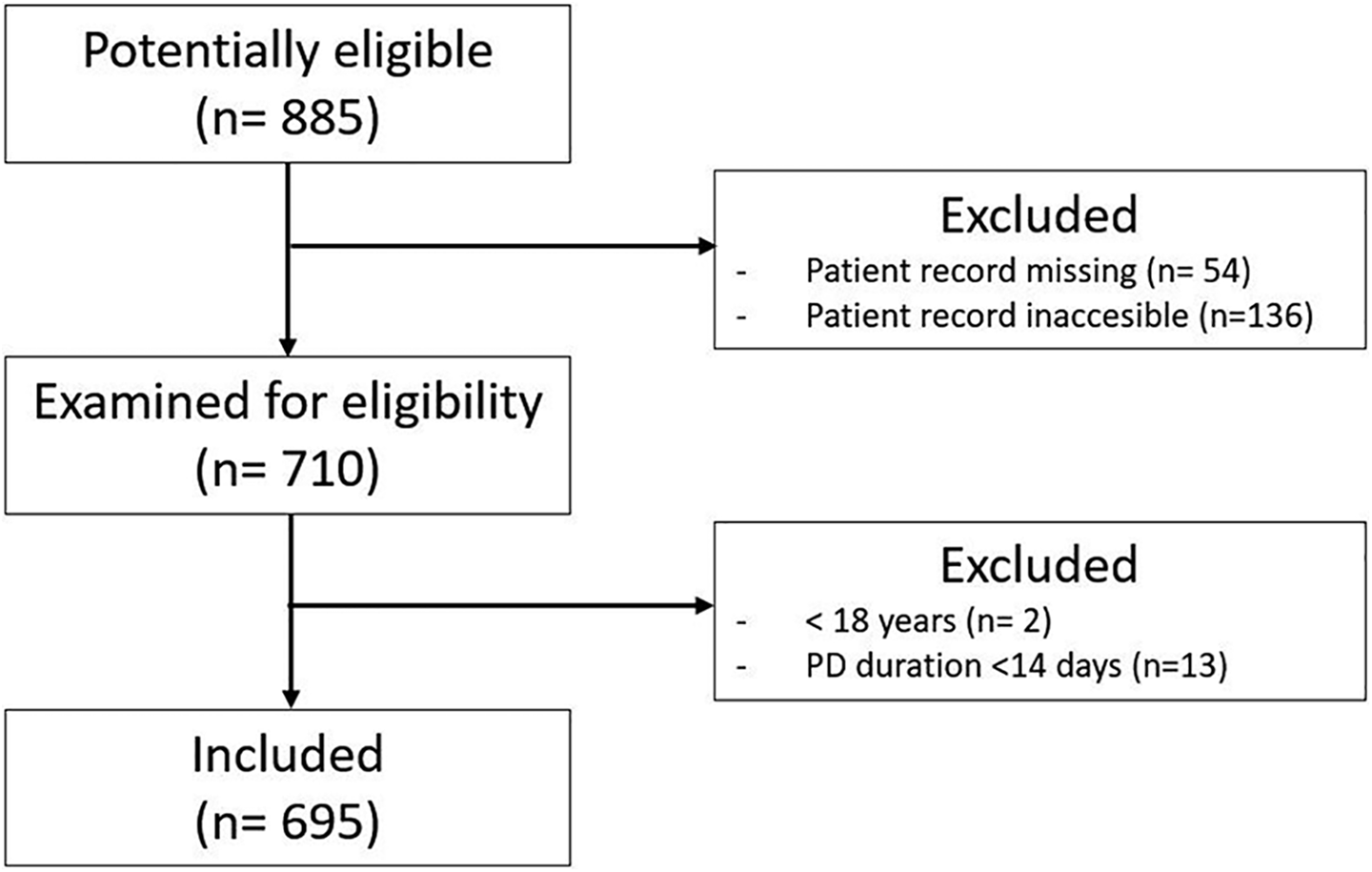
Flow chart of the patients included in the study.
Baseline characteristics of 695 patients treated with peritoneal dialysis.a
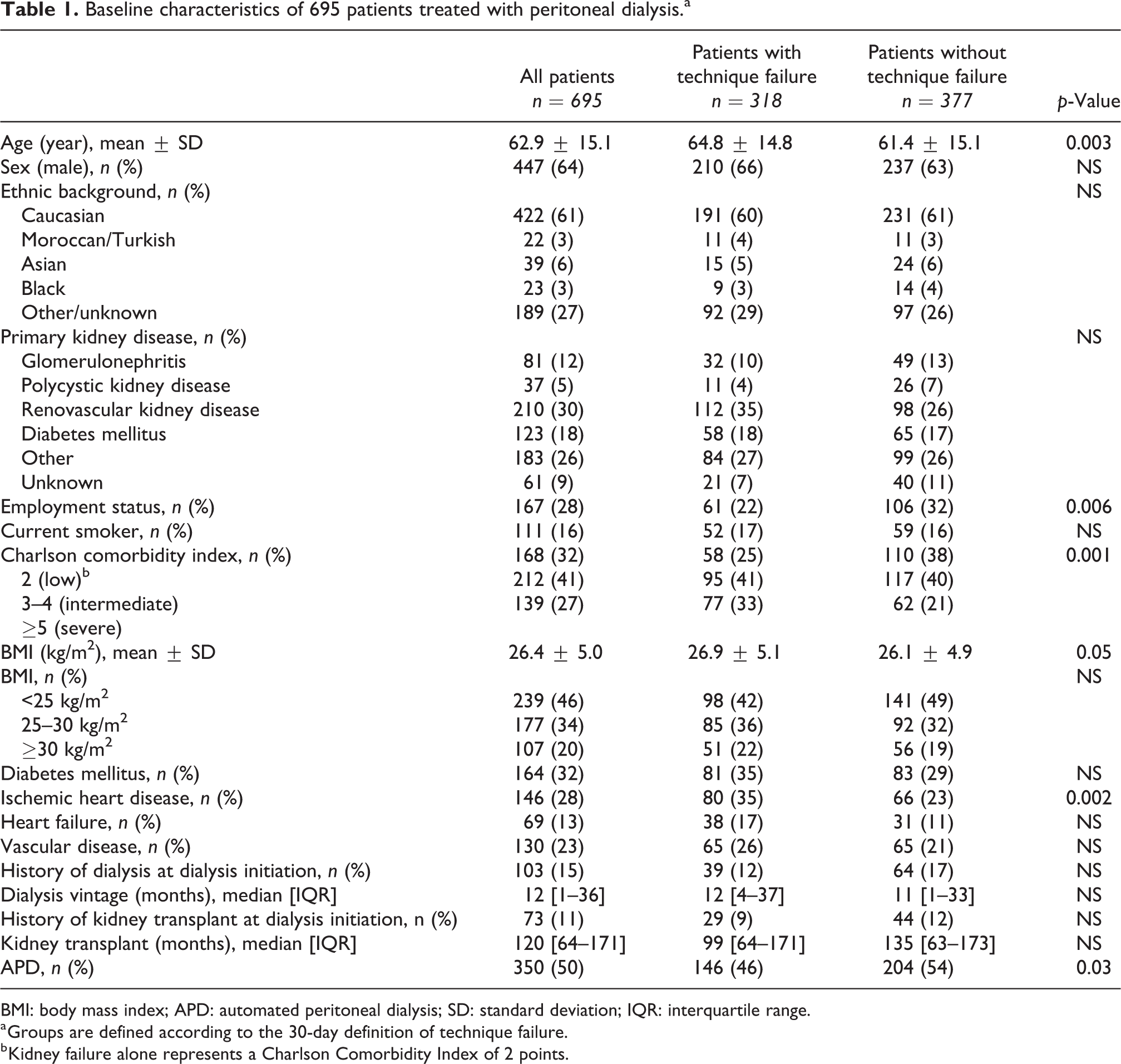
BMI: body mass index; APD: automated peritoneal dialysis; SD: standard deviation; IQR: interquartile range.
a Groups are defined according to the 30-day definition of technique failure.
b Kidney failure alone represents a Charlson Comorbidity Index of 2 points.
Incidence of technique failure
A total of 318 patients developed technique failure during the study, of whom 22 patients experienced a recurrent episode of technique failure. The PD patients experienced a mean of 0.36 episodes of technique failure per person-year of follow-up. The 1- and 2-year technique failure rates were 29% and 52%, respectively (Figure 2(a)). The median time to technique failure was 1.85 years. Patients with technique failure were older, had higher comorbidity scores, were more likely to have ischemic heart disease and were more frequently treated with CAPD (Table 1). A total of 202 patients developed death-censored technique failure during the study (0.24 episodes of death-censored technique failure per person-year). The 1- and 2-year death-censored technique failure rates were 23% and 35%, respectively (Figure 2(b)). The median time to death-censored technique failure was 3.58 years.
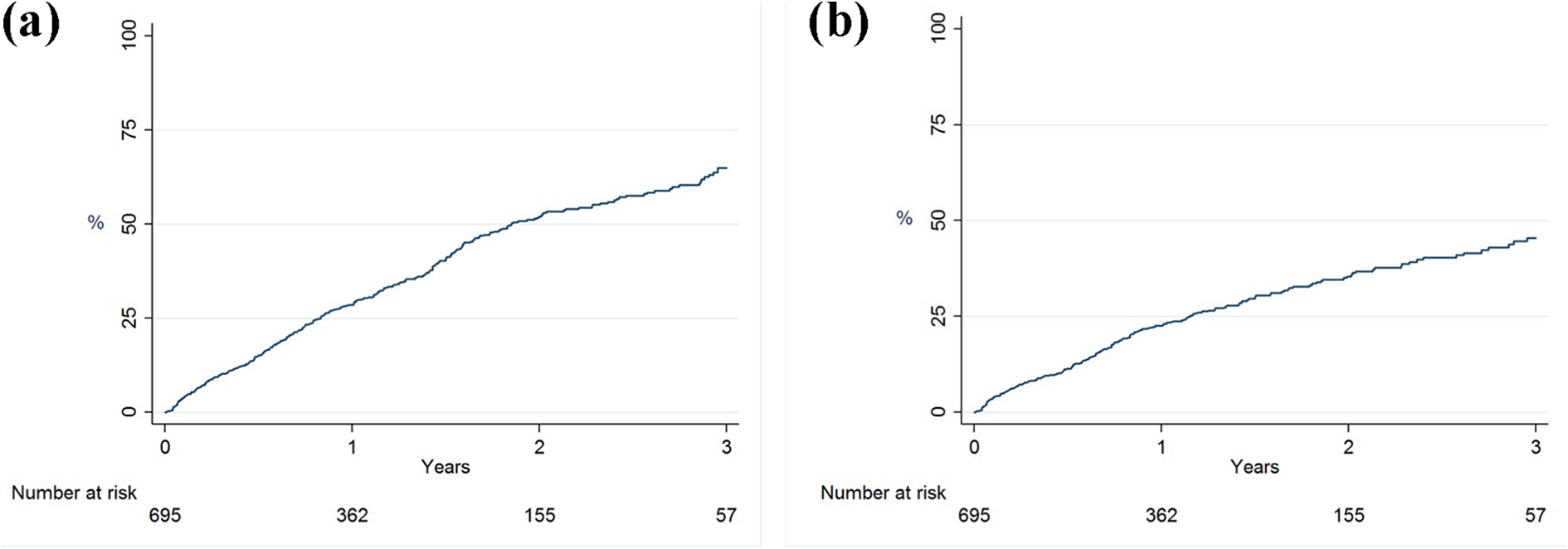
Technique failure, as a composite outcome (with transfer to CHD or death) (a) and as death-censored technique failure (b). Technique failure was defined as a transfer to CHD for ≥30 days, death on PD or death within 30 days after transfer to CHD. First day of receiving CHD was the date assigned as technique failure. PD: peritoneal dialysis; CHD: in-centre haemodialysis.
Causes of technique failure
Figure 3 shows that death was the most common cause of technique failure, followed by PD-related infections (20%). The other causes of technique failure occurred in about 10% or less than 10% of the patients who experienced technique failure. The predominant causes for death were cardiovascular disease (28%), infections other than PD peritonitis (15%) and malignancies (13%). None of the deaths were attributable to a PD peritonitis.
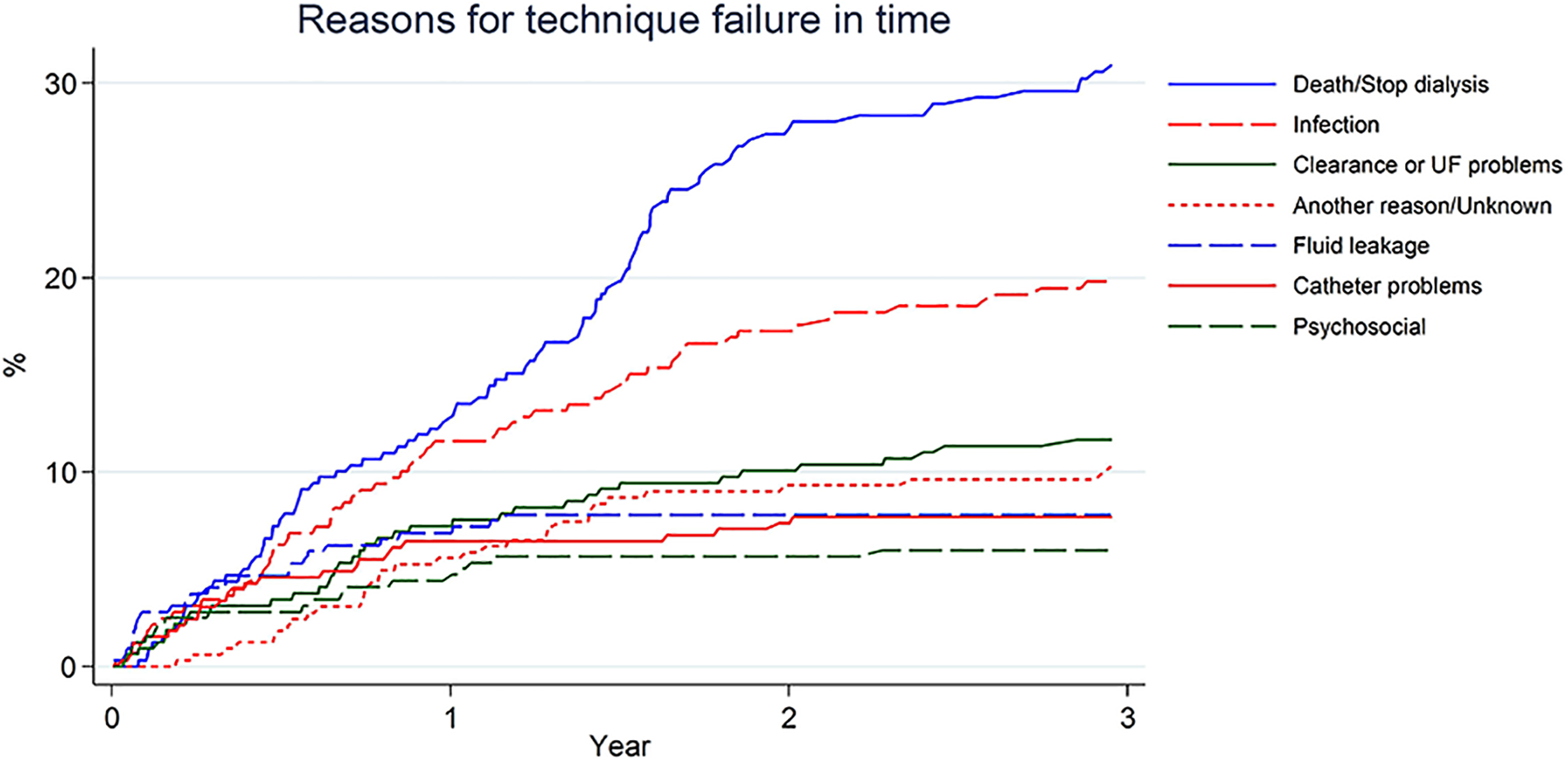
Cumulative incidence of different causes for technique failure shows the occurrence of different causes for technique failure over time in a population of patients with technique failure (
Figure 4 shows the different causes of early (i.e. during the first 6 months after start of PD) and late (i.e. more than 6 months after start of PD) technique failure. A total of 99 patients developed early technique failure, and 219 patients developed late technique failure. Catheter-related problems were the cause of early technique failure in 15% of patients, whereas this was the cause of late technique failure in only 5% of patients. Similarly, PD fluid leakage was the cause in 15% and 5%, respectively. Infections and clearance problems were a major cause of both early and late technique failure; infections were in 20% of patients the cause of technique failure and clearance problems in 11–12% of patients. EPS was a cause of technique failure in less than 1% of patients. The group of ‘other reasons’ included (temporary) discontinuations of PD due to major (abdominal) surgery with hospitalisation.
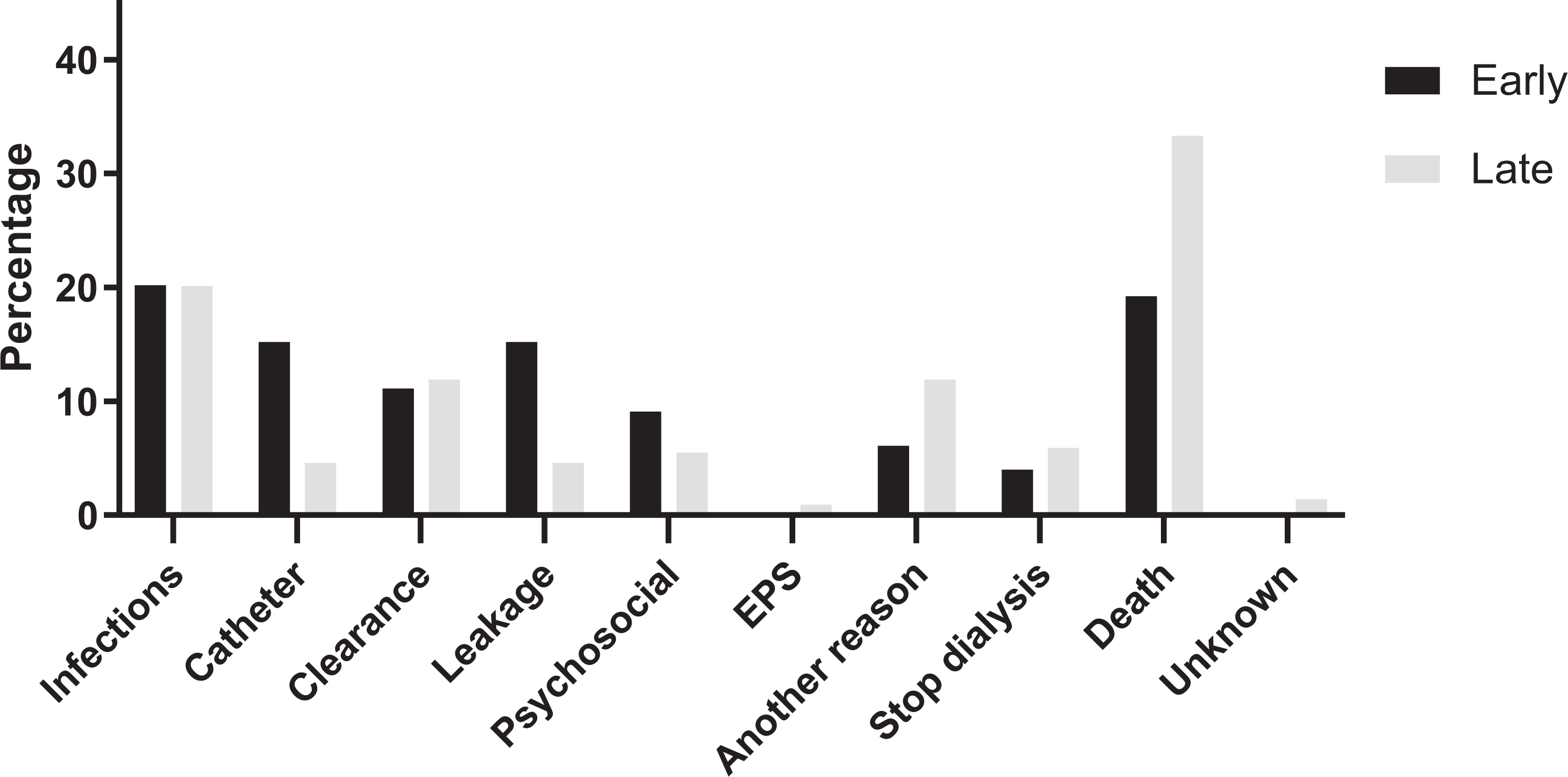
Comparison of causes of early and late technique failure. Early PD technique failure is defined as occurrence of technique failure in the first 6 months after start of PD (
Risk factors
The patient-specific risk factors sex, age, employment status and BMI were not associated with technique failure (Table 2). APD compared to CAPD was associated with a reduced risk of technique failure (adjusted hazard ratio (HR) 0.66 (95% confidence interval (CI) 0.53–0.83). The patient-specific risk factors for death-censored technique failure were similar to those for technique failure including death in the definition (Supplementary Table S1); only APD was associated with a reduced risk of death-censored technique failure (adjusted HR 0.60, 95% CI 0.46–0.80). In addition, APD use was not associated with death as a separate outcome while age was associated with death (Supplementary Table S2).
Risk factors associated with technique failure in a Cox regression model.a
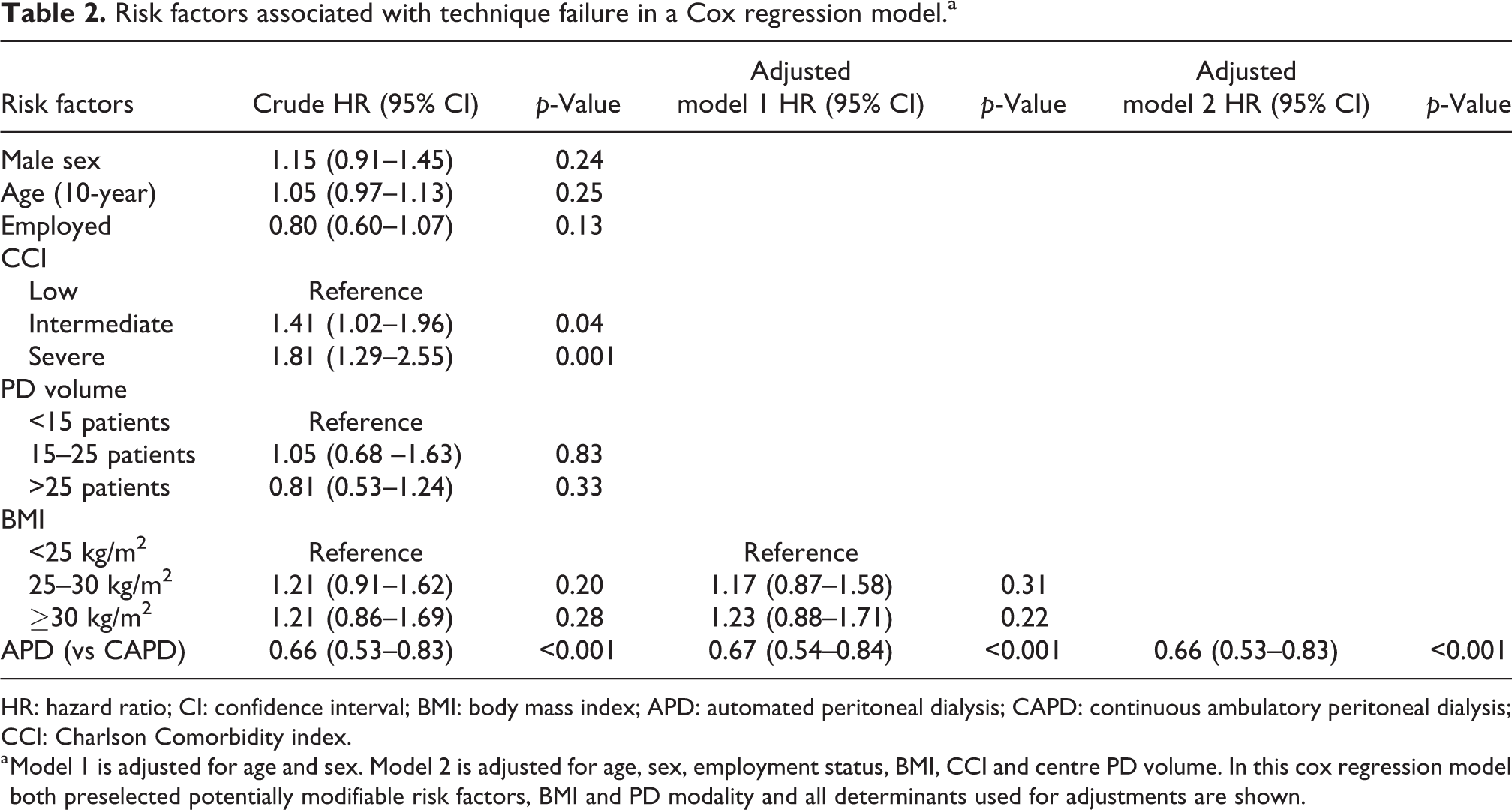
HR: hazard ratio; CI: confidence interval; BMI: body mass index; APD: automated peritoneal dialysis; CAPD: continuous ambulatory peritoneal dialysis; CCI: Charlson Comorbidity index.
a Model 1 is adjusted for age and sex. Model 2 is adjusted for age, sex, employment status, BMI, CCI and centre PD volume. In this cox regression model both preselected potentially modifiable risk factors, BMI and PD modality and all determinants used for adjustments are shown.
The sensitivity analysis in which the association between patient-specific risk factors and technique failure was investigated with a competing risk model, showed similar results for these associations as the original analyses (Supplementary Table S3). In a sensitivity analysis using PD modality at PD cessation, similar results were found (for APD compared to CAPD, adjusted HR 0.60 (95% CI 0.47–0.75)).
Centre variation in technique failure
All centres used icodextrin and antibiotic prophylaxis during PD catheter insertion (Supplementary Table S4). Most centres used neutral pH low glucose degradation products solutions (91%) and exit-site antibiotic prophylaxis (79%). The initial antibiotic regimen for peritonitis varied across centres and antifungal prophylaxis during antibiotic therapy was provided only in 6% of centres.
The centre variation in technique failure rate is shown in Supplementary Figure S1. The overall early technique failure rate, shown as the reference standard, was 16%, which is the total number of patients with early technique failure divided by the total number of PD patients from all centres that were not lost to follow-up at 6 months (due to transplantation or study end,
Permanent technique failure
A total of 254 patients developed permanent technique failure during the study: that is, at 180 days after transfer to CHD, they had not returned to PD (0.26 episodes of permanent technique failure per person-year). The 1- and 2-year permanent technique failure rate was 22% and 43%, respectively (Supplementary Figure S2). The median time to permanent technique failure was 2.7 years. The most common cause of permanent technique failure was death, followed by infections. A total of 72 patients developed early permanent technique failure and 182 patients developed late permanent technique failure. Again, early technique failure was associated with catheter-related problems and leakage, while infection and clearance problems were important causes for both early and late technique failure (Supplementary Figure S3 and Supplementary Table S5).
Discussion
In this cohort of 695 Dutch patients who were treated with PD between 2012 and 2017, the technique failure rate within the first year of PD treatment was 29%. Death was the most common cause of technique failure. Death-censored technique failure rate at 1 year was 23%. In 20% of patients with technique failure, infections were a possible modifiable cause. In addition, early technique failure was frequently caused by catheter-related problems and leakage (both accounting for 15%). We found that APD use had a protective effect on technique failure.
Only few studies to date have used the standardised technique failure definition as proposed by Lan et al. 3,7,13 See et al. 7 reporting on Australian patients who started PD between 2000 and 2014, also used the standardised 30-day definition and found a first year technique failure rate of 26%. In an older study by Descoeudres et al., 23 not using the standard definition but a similar definition of technique failure including death by any cause, the technique failure rate at 1 year was 25%. The technique failure rate in our study is thus comparable to other studies that included death as a cause for technique failure. Death was the most common cause for technique failure during the entire follow-up, as would be expected in a study on dialysis patients since mortality rates of both PD and CHD patients are high. 24 Yet the death-censored technique failure rate was still high. This, in addition to the decline of the number of PD patients in the Netherlands, underscores the need to find modifiable causes for technique failure.
In recent decades, significant advances in PD treatment have declined the overall rate of technique failure. 3 –5 Boyer et al. stated that this is, in addition to improved patient survival, attributable to less infection-related technique failure. 5 Nevertheless, infections were still an important cause of technique failure – both in early and late technique failure – indicating that prevention of infections is pivotal in technique survival. Recommendations for the prevention of peritonitis from the ISPD, including exit-site prophylaxis and antibiotic prophylaxis during PD catheter insertion, were generally well followed by participating centres especially if compared to international data from PDOPPS. 25 –27 In a recent study by PDOPPS, antibiotic prophylaxis during PD catheter insertion was indeed associated with a lower peritonitis risk. 28 On the other hand, most centres in the Netherlands did not use antifungal prophylaxis during antibiotic therapy, although prophylaxis was associated with a significant risk reduction of fungal peritonitis in a systematic review. 29 According to the results of PDOPPS antifungal prophylaxis was also variably used across countries, the lowest in Japan (8% of facilities) and the highest in Australia (89%). 27 So a greater reduction in infections may be possible if all centres would adhere to current guidelines.
The ISPD guidelines refrain from recommending a specific antibiotic regimen for peritonitis based on a Cochrane systematic review due to lack of superiority.
25,30
As a result, the initial antibiotic regimen varied across centres. Of note, one third of all centres used a combination with glycopeptides, possibly based on a systematic review in which glycopeptides were proven most effective in combination with ceftazidim.
31
Also in PDOPPS, a variable use of vancomycin across countries has been reported.
27
However, because evidence for antibiotic regimens including glycopeptides remain weak,
30
future clinical trials may evaluate good practices from single centres. Examples are temporary discontinuation of PD without removing the catheter (peritoneal rest) combined with intravenous meropenem and meropenem intracatheter as lock (Mero-PerRest protocol) in case of enteric peritonitis and the treatment with amphotericin B catheter lock for salvage of the PD catheter in case of
Catheter-related problems have been identified as an important cause of early technique failure in previous studies. 10,23 In this study, we identified leakage as another important cause of early technique failure. This underscores the need for a multidisciplinary team with sufficient experience in catheter care and insertion. 34 In a study from Australia and New Zealand, small centre volume – possibly indicative of low centre experience – was associated with technique failure due to mechanical complications. 3 A striking variation in PD catheter survival among different centres in the United Kingdom suggests differences in access protocols. 15 Still, previous studies have not yielded results that could lead to recommendations for the preferred use of a catheter delivery technique or specific PD catheter type. 34,35 The workgroup PD catheter access of PDOPPS hypothesises that standardised protocols for catheter insertion will be associated with a reduction of technique failure, the results of this working group are thus eagerly awaited. 15
A possible other reduction in technique failure might be the increased interest in assisted PD due to the ageing dialysis population. 36 Within this demographic shift, assistance during PD treatment is a mean to provide home dialysis to elderly patients who may be unable to perform PD themselves due to frailty or physical impairments. In a recent study, family-assisted PD was associated with lower risk on catheter-related technique failure. 37 The authors hypothesised that involving family members in dialysis treatment may lead to better adherence to diet restrictions resulting in less constipation. Of note, in this study also a lower risk on technique failure due to clearance problems was found in both family-assisted and nurse-assisted PD. The nurse or family member supervising the treatment likely ameliorates the patient’s adherence to dialysis prescriptions. 37 Clearance problems, in our report the main cause of death-censored technique failure following infections, may thus also be perceived as a modifiable cause for technique failure. These aforementioned modifiable causes – infections, leakage, catheter-related problems and clearance problems – accounted for 48% of technique failure within our cohort; hence, quality improvements aimed at these causes can have a major impact on technique survival.
APD use had a protective effect on death-censored technique failure in our analysis, even after adjustments for age and comorbidity. In recent literature, conflicting results have been presented: APD use was associated with an adjusted lower technique failure rate and higher patient survival in one study, 38 while in other studies APD use was associated with a higher risk of technique failure. 3,7 There may be a link with infections, since CAPD use was associated with a higher rate of peritonitis in recent studies. 28,39 Also in the only two randomised controlled trials to date – although originating from <2000 – higher peritonitis rates with CAPD use were found. 40,41 This association with peritonitis might be due to better adaptation of therapy to patient needs, as the authors of a recent study suggest, 39 or to fewer connections between catheter and dialysis bags when using APD instead of CAPD and thus less risk of breaching hygiene measures. Although the suggestion of fewer connections resulting in less infections is disputed, 25 new devices that assist the patient are hypothesised to reduce infection risk. 42 APD might also be used more often by patients themselves than for assisted PD. 37 which could explain the protective effect since self-care may be associated with a lower peritonitis rate. 43,44 However, the association between APD use and technique failure may also reflect long-term PD treatment, as patients with early technique failure may not be able to transfer to APD (in other words: confounding by indication). In the Netherlands, most patients start PD treatment with CAPD to familiarise themselves with performing exchanges by hand prior to a transfer to APD. The reason for the protective effect of APD is thus uncertain, therefore the choice for APD or CAPD should ideally be based on patient preference 25
In a previous study from the Netherlands by Huisman et al., smaller centres with on average less than 20 PD patients had a significantly higher risk of technique failure than larger centers. 45 The association between centre volume and technique failure however likely reflects centre experience. 16 Indeed, others confirmed that in larger centres technique failure due to modifiable causes, that is, infections, catheter – and UF problems, were less common. 46 Guillouët et al. found that centre volume and patients characteristics alone could not fully explain the centre effect on technique failure. They suggested that factors of centre experience such as patient education and nephrologist’s views on home dialysis play an important role in technique failure. 16 Contributing to this, we showed that the early technique failure rate – often caused by infections, leakage and catheter-related problems – was similar across all centres and was not related to the number of incident study patients. This probably indicates that it is not the centre volume itself that matters, but the experience within a centre and having a dedicated team.
In this study, technique failure consisted of a composite outcome of death and transfer to CHD, in accordance with the standardised definition. 13 Death is an objective measure but transfer to CHD is subjective; often a choice is made by the nephrologist to discontinue treatment and this decision will be weighed differently by each nephrologist. A considerable proportion of the causes of technique failure may have been modifiable, that is, infections, leakage and catheter problems, since practice variation exists in peritonitis rate and in the treatment of infections and access. 27,39 Because the definition of technique failure partly consists of the decision to discontinue PD, studies on infection prevention and catheter access such as the PDOPPS will help to increase technique survival. 15
Strengths of this study include the use of the standardised definitions of technique failure, including the death-censored and permanent definition, the analysis of causes of both early and late technique failure, the use of a patient cohort reflecting current practice patterns and extensive adjustments for confounders. In addition, most studies were conducted on registry data whereas our cohort study enabled to identify the causes of technique failure in more detail. Yet, the study sample of this analysis was relatively small and the study was conducted in a single country. The study duration of this study was a respectable 5 years, yet the median follow-up duration was 13 months. As a result, the proportion of technique failure after 1 year should be interpreted with caution.
In conclusion, in this multi-centre Dutch study of PD patients PD-related infections, leakage and catheter problems were important modifiable causes for technique failure. As almost a quarter of patients experience death-censored technique failure within the first year, future studies should emphasise on prevention of infections and PD catheter access problems to improve technique survival.
Supplemental material
Supplemental Material, sj-pdf-1-ptd-10.1177_08968608221077461 - Technique failure in peritoneal dialysis: Modifiable causes and patient-specific risk factors
Supplemental Material, sj-pdf-1-ptd-10.1177_08968608221077461 for Technique failure in peritoneal dialysis: Modifiable causes and patient-specific risk factors by Anna A Bonenkamp, Anita van Eck van der Sluijs, Friedo W Dekker, Dirk G Struijk, Carola WH de Fijter, Yolande M Vermeeren, Frans J van Ittersum, Marianne C Verhaar, Brigit C van Jaarsveld, Alferso C Abrahams and on behalf of the DOMESTICO study group. in Peritoneal Dialysis International
Footnotes
Authors’ note
The DOMESTICO study group:
Acknowledgements
We would like to thank the patients and local investigators of all participating centres of the DOMESTICO study. We also acknowledge all local investigators of the centres in this study for providing answers to an additional questionnaire and Prof. Dr JWR Twisk for his guidance on mixed model analysis.
Author contributions
AAB, AES, BCJ and ACA designed the research question. AES and AAB collected data. AAB performed the statistical analyses. AAB, AES, FWD, FJI, BCJ and ACA interpreted the data. FWD, DGS, CWF, YMV and MCV provided intellectual content of critical importance to the work described. AAB drafted the manuscript. All authors critically edited the manuscript and approved the final version.
Data availability statement
The data underlying this article will be shared upon reasonable request to the corresponding author and approval of the DOMESTICO steering committee.
Declaration of conflicting interests
The author(s) declared potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AES and ACA have received speaker honoraria from Baxter Healthcare. All other authors declare no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The retrospective part of the ‘Dutch nOcturnal and hoME dialysis Study To Improve Clinical Outcomes’ is supported by a grant of the Dutch Kidney Foundation (grant no: A2D4P02). The sponsor had no role in the design and conduct of the study and no role in writing or in the decision to publish this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
