Abstract
Transport of serum proteins from the circulation to peritoneal dialysate in peritoneal dialysis patients mainly focused on total protein. Individual proteins have hardly been studied. We determined serum and effluent concentrations of four individual proteins with a wide molecular weight range routinely in the standardised peritoneal permeability analysis performed yearly in all participating patients. These include β2-microglobulin, albumin, immunoglobulin G and α2-macroglobulin. The dependency of transport of these proteins on their molecular weight and diffusion coefficient led to the development of the peritoneal protein restriction coefficient (PPRC), which is the slope of the relation between the peritoneal clearances of these proteins and their free diffusion coefficients in water, when plotted on a double logarithmic scale. The higher the PPRC, the more size restriction to transport. In this review, we discuss the results obtained on the PPRC under various conditions, such as effects of various osmotic agents, vasoactive drugs, peritonitis and the hydrostatic pressure gradient. Long-term follow-up of patients shows an increase of the PPRC, the possible causes of which are discussed. Venous vasculopathy of the peritoneal microcirculation is the most likely explanation.
Keywords
Introduction
Most studies on peritoneal transport in patients treated with peritoneal dialysis (PD) focused merely on small solutes and to a lesser extent fluid removal. However, peritoneal effluent also contains proteins. These are partly the result of local production or release from peritoneal tissues, but the quantitatively largest portion is due to transport of serum proteins from the circulation. Studies using electron microscopy and tracers suggested transmembrane transport through vesicles, 1 but a more recent study in mice lacking endothelial caveolae showed even increased macromolecular transport compared to control animals and provided evidence of transport through pores in the vascular wall. 2 In PD, the pathway for plasma proteins is through inter-endothelial pores in the microcirculation, via interstitial tissue and the mesothelial layer to the dialysate-filled peritoneal cavity. The mesothelium is not considered to be an important transport barrier. 3 In the initial period of chronic PD treatment, the interstitium consists of extracellular matrix components with only a few fat cells and fibres. The cellular content of the interstitium changes and increases with PD duration 4 as well as the quantity of collagen. 5 Consequently, the contribution of the interstitium to transport may be irrelevant in the beginning of PD but increases with its duration.
Plasma proteins can pass the microvascular wall to be taken up in interstitial tissue. Assessment of this phenomenon is hampered by the difficulty of obtaining interstitial fluid. This is also the case for glomerular filtration of proteins, because these are almost completely reabsorbed in the tubular system. However, in patients with nephrotic syndrome, the quantity is so large that tubular reabsorption is overloaded and the filtered proteins can be detected and studied in urine. It appeared that a size dependency was present for glomerular proteinuria and this led to the development of the so-called selectivity index in which urinary protein and plasma concentrations were related to their molecular weights (MWs) in patients with nephrotic syndrome. 6,7 Tubular reabsorption is not present in peritoneal transport and the presence of dialysis fluid in the peritoneal cavity provides immediate access to the interstitial tissue. The general aim of the studies discussed in the present review was to deepen our insight in the peritoneal restriction barrier to protein permeability by assessment of relationships between clearances of plasma proteins and their free diffusion coefficients, and the peritoneal protein restriction coefficient (PPRC). The use and value of this parameter in various conditions will be examined.
Physiology of peritoneal protein transport
According to the generally accepted pore theory, solute transport across the vascular wall occurs mainly through small inter-endothelial pores with radii of about 40 Å that constitute 90% of all pores 8 and, additionally, through large pores with radii exceeding 150 Å. Serum proteins can pass through these pores depending on their molecular size as reflected by their MW. Peptides and small proteins like β2-microglobulin (β2-Mi) with MW of 11.8 kD and radius of 16 Å will pass through the small pores, while proteins with radii exceeding 40 Å can only be transported across the large pore system. These include immunoglobulin G (IgG), MW 150 kD, radius 54 Å and α2-macroglobulin (α2-Ma), MW 720 kD, radius 89 Å. Albumin with a MW of 67 kD and radius of 36 Å has an intermediate position. The anatomic equivalent of the large pores has not been established with certainty but likely consists of endothelial gaps between endothelial cells in venules that can be provoked by vasoactive substances, such as histamine. 9 If this is the case, the number and radius of large pores may be variable.
Peritoneal transport of small solutes occurs mainly by diffusion and its velocity is dependent on its MW or free diffusion coefficient. The relationship between transport and size of various solutes can be described as a power function, meaning linearity when plotted on a double logarithmic scale. 10 The slope of the obtained line represents the size selectivity of peritoneal transport. A slope <0.5 indicates free diffusion, so the absence of a size-selective barrier. 11,12 However in contrast to small solutes, the slope is different for serum proteins, meaning the presence of size-selectively restricted peritoneal transport. 11 This is illustrated in Figure 1. Assuming diffusion as an important mechanism, also for peritoneal transport of macromolecules, the PPRC was defined in 1993 as the exponent of the power relationship between the peritoneal clearances of β2-Mi, albumin, IgG and α2-Ma versus their free diffusion coefficients in water; in other words: the slope of the straight line between these parameters when plotted on a double logarithmic scale. 13 This is illustrated in Figure 2. The higher the PPRC, the more size-selective hindrance to peritoneal protein transport. In this approach, effects of convective transport are assumed to be stable. With 1.36/1.5% glucose dialysate during a 4-h dwell, the median value was 2.3 and ranged between 1.9 and 3.2, 14 indicating restricted diffusion. In contrast, a similar calculation using mass transfer area coefficients of urea, creatinine, urate and β2-Mi yielded no evidence for a size-selective hindrance to peritoneal transport of solutes that diffuse through the small inter-endothelial pores from the microcirculation to the peritoneal cavity. A clinically useful interpretation is that the transport rate of small solutes is dependent on the number of perfused peritoneal microvessels, so the number of small pores which can be designated as the effective peritoneal surface area, while the PPRC represents the large pore radius, also indicated as the intrinsic permeability to macromolecular transport. 15 No relationship is present between peritoneal clearances of individual proteins and the PPRC, the coefficient of intra-individual variability of which averaged 5%. 16
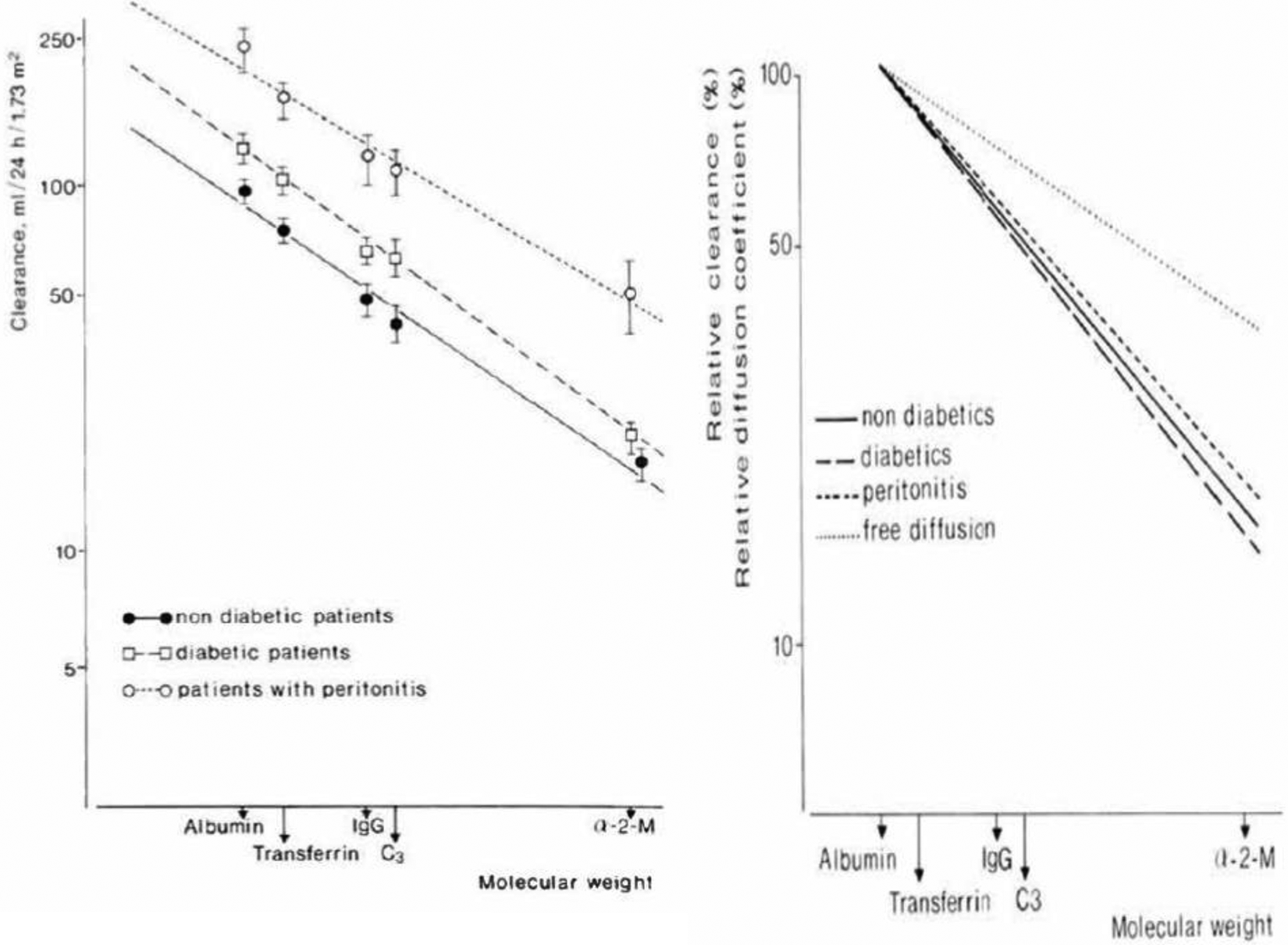
Left panel. The relationship between peritoneal protein clearance and molecular weight plotted on a double logarithmic scale in three patients groups. Mean values with SEM are given, and the regression lines are drawn using albumin, transferrin, immunoglobulin G, complement C3 and α2-macroglobulin. The mean slopes of the three lines ranged from −0.62 (during peritonitis) to −0.75 (diabetic patients). Right panel. The three regression lines of the right panel, but now expressed as relative clearance, when compared to albumin clearance. The upper line represents the relationship between the free diffusion coefficients in water for the investigated proteins and their molecular weight. The slope of the upper line was −0.36, which is markedly blunted compared to the slopes of the clearances, so indicating a size-selective restriction of the transport of serum proteins. Taken from Krediet et al. 11 with permission of Karger AG.
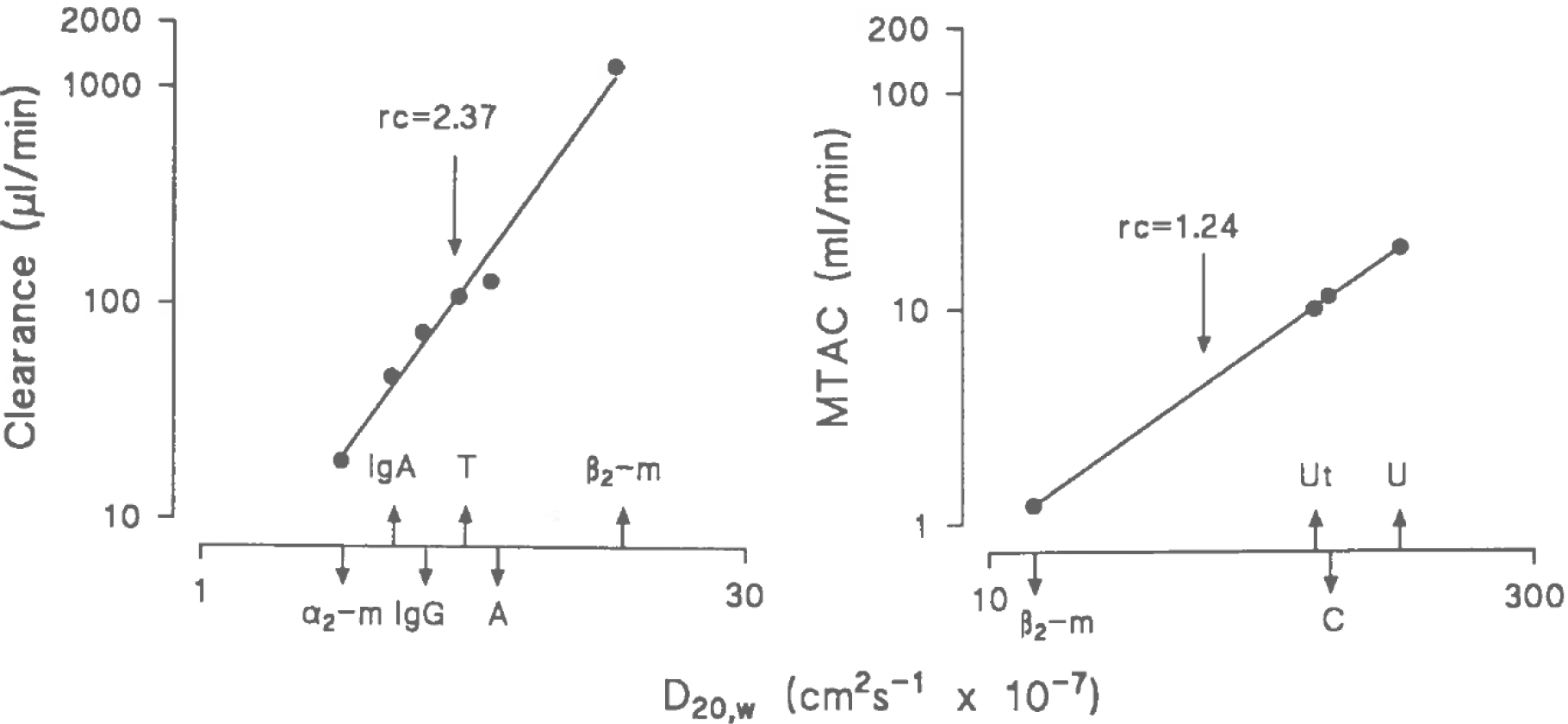
Left panel. The relationship between the peritoneal clearances of β2-microglobulin (β2-m), albumin (A), transferrin (T), IgG, IgA and α2-macroglobulin (α2-m) and their free diffusion coefficient in water (D20,w), plotted on a double logarithmic scale. The slope of the line represents the restriction coefficient to proteins (PPRC). A mean value of 2.37 ± 0.04 was found. Right panel. A similar plot for the mass transfer area coefficients (MTAC) of β2-m, urate (Ut), creatinine (C) and urea (U). In this analysis of small solute transport, a mean slope of 1.24 ± 0.03 was found. Taken from Imholz et al. 13 with permission of Elsevier.
Up to now our group has been the only one that reported on the PPRC in PD patients. This review summarises the results of various clinical studies in them and reports on how analysis of this PPRC contributed to understanding the peritoneal alterations that can occur in PD patients and also how the PPRC changed the interpretation of the physiology of peritoneal protein transport. A comparison between the traditional view on peritoneal protein loss and the insights obtained with the studies discussed in the following sections, as presented in Table 1.
Transport of plasma proteins from the circulation tot the dialysate-filled peritoneal cavity.
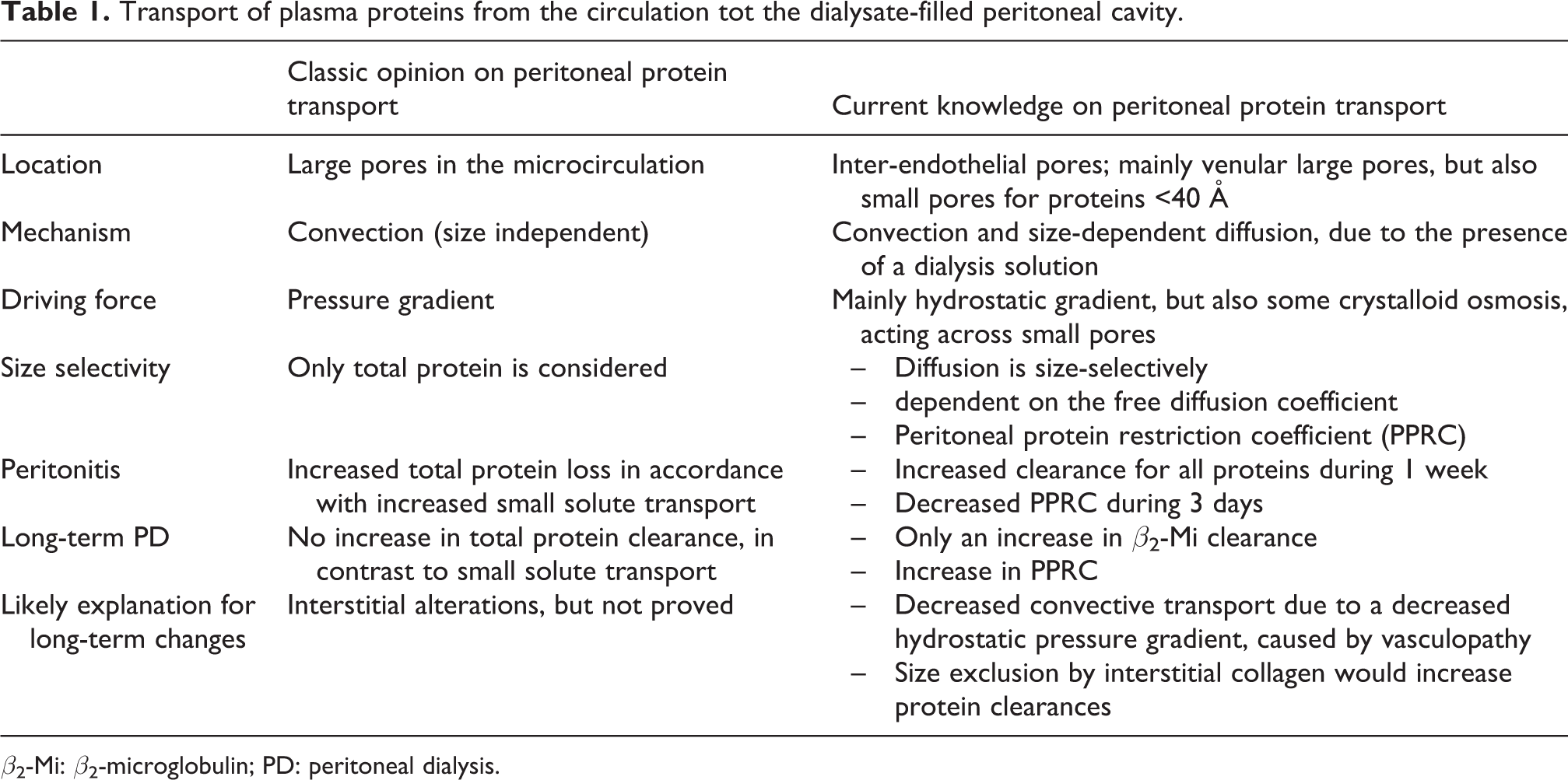
β2-Mi: β2-microglobulin; PD: peritoneal dialysis.
Effect of osmotic agents
The crystalloid osmotic pressure gradient influences the clearance of β2-Mi, but not of proteins that are transported through the large pore system. 13 This effect of convection leads to higher normal values when a 3.86/4.25% glucose solution is used for peritoneal function assessment. This increased the median value of the PPRC from 2.3 to 2.6 with a range from 1.4 to 3.2, 17 due to the increase in β2-Mi transport. A comparison of 1.36% glucose with 1.4% glycerol only had a marginal effect on the PPRC. 18 Also a comparison between a 1.1% amino acids and a 1.36% glucose solution showed similar values. 19 As expected, the use of 7.5% icodextrin during 4-h dwells yielded values of the PPRC in-between those obtained with 1.36% and 3.86% glucose. 20 It can therefore be concluded that the tonicity of dialysis solutions influences osmotic convective transport through small pores but has no effect on that across the large pores.
Effect of vasoactive drugs
A number of intraperitoneally administered vasodilators has been studied in the past with regard to their effects on peritoneal transport during intermittent PD. 21 Of these, the venodilator nitroprusside showed the largest and most consistent increase in small solute transport and also increased peritoneal protein loss. The intraperitoneal administration of 4.5 mg/L nitroprusside during a standard peritoneal permeability analysis using 1.36% glucose had no effect on systemic blood pressure but increased the mass transfer area coefficient of creatinine with 40%, while the clearance of albumin increased 75% that of IgG 98% and α2-Ma clearance 137%. 22 The PPRC decreased from 2.29 to 1.86, suggesting an increase is the average large pore radius. This was confirmed using kinetic modelling, irrespective of assuming convection or diffusion as the main transport mechanism. 22 Repeating this study with icodextrin instead of glucose affected the PPRC similarly. 23 It can be concluded that the nitroprusside studies confirm the contention of Fox et al. 9 that the large pore system is a dynamic structure in the venular part of the microcirculation.
Effects of peritonitis
Acute peritonitis is associated with an increase in peritoneal transport of small solutes and proteins, the latter being larger. 12,24 The PPRC had a low median value of 2.04. Daily follow-up during treatment showed that β2-Mi clearance representing small pore surface area took about 1 week to decrease to control values, while the increase to control was only 3 days for the PPRC. 25 Furthermore, the time course of β2-Mi clearance was associated with that of effluent interleukin-6 (Il-6) and tumour necrosis factor-α, while that of the restriction coefficient showed associations with effluent Il-6 and prostaglandin E2 (PGE2). These findings suggest that Il-6 is involved in the regulation of the number of perfused peritoneal microvessels during an acute inflammatory process, while vasodilating prostaglandins affect the large pore radius. Intraperitoneal administration of indomethacin, 25 mg/2 L for 3 consecutive days directly following the diagnosis of peritonitis, inhibited effluent PGE2 and was associated with a reduced decrease of the PPRC. 26 In contrast, indomethacin administration had no effect on β2-Mi clearance or effluent Il-6. The relationships between PGE2, the PPRC and the effect of indomethacin were only found in the acute phase of peritonitis. In stable, non-infected patients, the drug had no effect on effluent PGE2 or parameters of peritoneal fluid, solute and protein transport, including the PPRC. 27 No data are available on the PPRC and micro-inflammation, but a relationship has been reported between total protein clearance and effluent Il-6 in otherwise stable patients. 28 It should however be appreciated that such relationships in cross-sectional studies must be interpreted cautiously, because they may be influenced by mathematical coupling between protein transport and the Il-6 protein, which enters the peritoneal cavity by transport from the circulation and by local intraperitoneal production. 29
Effects of the hydrostatic pressure gradient
Peritoneal protein transport in PD is probably the result of two transport mechanisms, that is, hydrostatic convection as shown in experimental studies 30 and diffusion. The last mechanism, that is, diffusion is relevant in PD due to the presence of a dialysis solution in the interstitial tissue surrounding the microvessels. In vitro studies on endothelial monolayers also are also supportive of a combination of hydrostatic convection and diffusion. 31 We assumed that important changes in the hydrostatic pressure gradient would be absent in PD patients, favouring the use of the PPRC as a marker of the intrinsic peritoneal permeability to macromolecules. In retrospect this assumption was incorrect, as illustrated by two recent studies on the effect of venous congestion due to fluid overload on the peritoneal clearance of total protein in starting PD patients. 32,33 The presence of peritoneal alterations is unlikely in this population. Using multivariable analysis, it was shown in both studies that parameters of fluid overload pointing to an increased venous pressure and thereby augmenting convection were the only significant predictors of peritoneal total protein clearance. 32,33 The relationship between left ventricular dysfunction and protein loss reported in the Stokes study is in accordance because left ventricular dysfunction often causes venous congestion. 28 These findings on the importance of venous pressure are in accordance with previous ones in the opposite situation, that is, lowering the hydrostatic pressure gradient on peritoneal transport of individual proteins by decreasing the pressure gradient due to increasing the dialysate volume. An increase from 2 to 3 litres in PD patients raised mean intraperitoneal pressure from 13 cmH2O (9.6 mmHg) to 16 cmH2O (11.8 mmHg), 34 but the effects on lowering the mass transfer area coefficients of albumin and IgG were marginal. 35 However, when intraperitoneal pressure was raised from 8 to 18 mmHg by the application of external pressure, the peritoneal clearances of albumin, IgG and α2-Ma decreased in a size-dependent way, that is, 17% for albumin, 27% for IgG and 32% for α2-Ma with a consequent increase of the PPRC. 36 As diffusion is dependent on molecular size while convection is not, the contribution of convection to the transport of an individual protein compared to diffusion will be larger the higher its MW. So, α2-Ma will be more affected by convective changes than albumin clearance, which has obvious effects on the PPRC. A lower hydrostatic pressure gradient may also explain the lower normal value of the PPRC after 4-h dwells with 1.5 compared to 2 litres 3.86% glucose. 17 It underlines the importance of standardisation for longitudinal follow-up of peritoneal transport in patients, not only with regard to glucose concentration and dwell time but also the instilled dialysate volume.
Effects of PD duration
The PPRC increases with the duration of PD. 37 The PPRC increase was also present in patients who developed encapsulating peritoneal sclerosis (EPS) but of a similar magnitude as in time-matched control patients with or without ultrafiltration failure. 38 Also the presence of previous peritonitis episodes had no effect on its time course. 39 This increase of the PPRC does not imply that the clearances of all constituents of total protein are higher but only that changes occur in some of these, as discussed in the section on the importance of vasculopathy. Irrespective of the mechanism, the alteration in the size selectivity of protein transport likely reflects changes in peritoneal morphology. Besides loss of mesothelial cells, long-term PD is associated with vascular and interstitial remodelling. 40 These consist of vasculopathy and fibrotic alterations in the interstitial tissue composed of myofibroblasts and collagen fibres. Both interstitial fibrosis and vasculopathy will be discussed in the following sections.
Effects of PD duration on peritoneal protein clearances
For interpretation of the time course of the PPRC, that of peritoneal protein clearances will be discussed first. Peritoneal small solute transport increases with PD duration. 41,42 This is probably due to an increase of the vascular surface area, caused by the formation of many immature microvessels. 43 As serum proteins and small solutes are transported by the same microcirculatory system, relationships between the two can be expected and have indeed been found in the initial phase of PD. 44 However, a progressive uncoupling between small solute transport rate and peritoneal total protein clearance occurred with PD duration, resulting in absence of an increase of the latter with time. 44 This absence was confirmed for the clearances of individual serum proteins, except for β2-Mi, which is transported across the small pore system, just like creatinine. 39
The relative decrease of peritoneal protein transport with PD duration can either be due to a decreased hydrostatic filtration pressure resulting from vasculopathy or to interstitial changes. Also a survival bias due to the relationship between peritoneal protein clearance and overhydration cannot be excluded with certainty. 32,33 However, this is not very likely, because of the well-documented increase is small solute transport with PD duration. Both interstitial changes and vasculopathy as possible explanations for the absence of a rise in protein clearance in long-term PD are discussed in the following sections.
Importance of interstitial changes
The peritoneal interstitium in PD consists of fluid, extracellular matrix components, adipocytes and collagen producing myofibroblasts. The collagen and the hyaluronan content of the extracellular matrix bind water, which causes a reduction of the distribution volume of proteins in the fluid space of the interstitium 45 The effects of this size exclusion by collagen-1 have been shown in an elegant in vitro study using a chromatography column composed of collagen-1, that was obtained from bovine Achilles tendon tissue. 46 Perfusion of this column with of a mixture of glucose and fetuin (a protein with MW 48 kD) dissolved in a buffer, showed the presence of fetuin in the elution volume of 51 mL, but that of glucose in the 80 mL eluent. This difference is in line with the presence of a smaller distribution volume of macromolecules compared to that of small solutes, because only small molecules can enter the water bound by collagen fibres, which influences their volume of distribution. The presence of a smaller distribution volume of a protein in the interstitium in combination with an unaltered microcirculatory transport will increase the concentrations in the dialysate and thereby also the peritoneal clearance to the dialysis fluid. This hypothesis is supported by analysis of relationships between effluent markers of peritoneal fibrosis like metalloproteinase-2 (MMP-2) and plasminogen activator inhibitor-1 (PAI-1), and the peritoneal clearances of β2-Mi, albumin, IgG and α2-Ma in the 86 prevalent PD patients in whom we reported the results of these fibrosis markers. 47 Moderate positive relationships were present between locally produced MMP-2 and PAI-1, and the clearances of the above mentioned serum proteins, as shown for albumin in Figure 3. These clinical observations and the discussed in vitro data make it likely that interstitial fibrosis would increase protein transport in long-term PD patients, instead of the observed relative decrease, which argues against peritoneal fibrosis as the explanation of the absence a time-related rise in peritoneal protein transport.
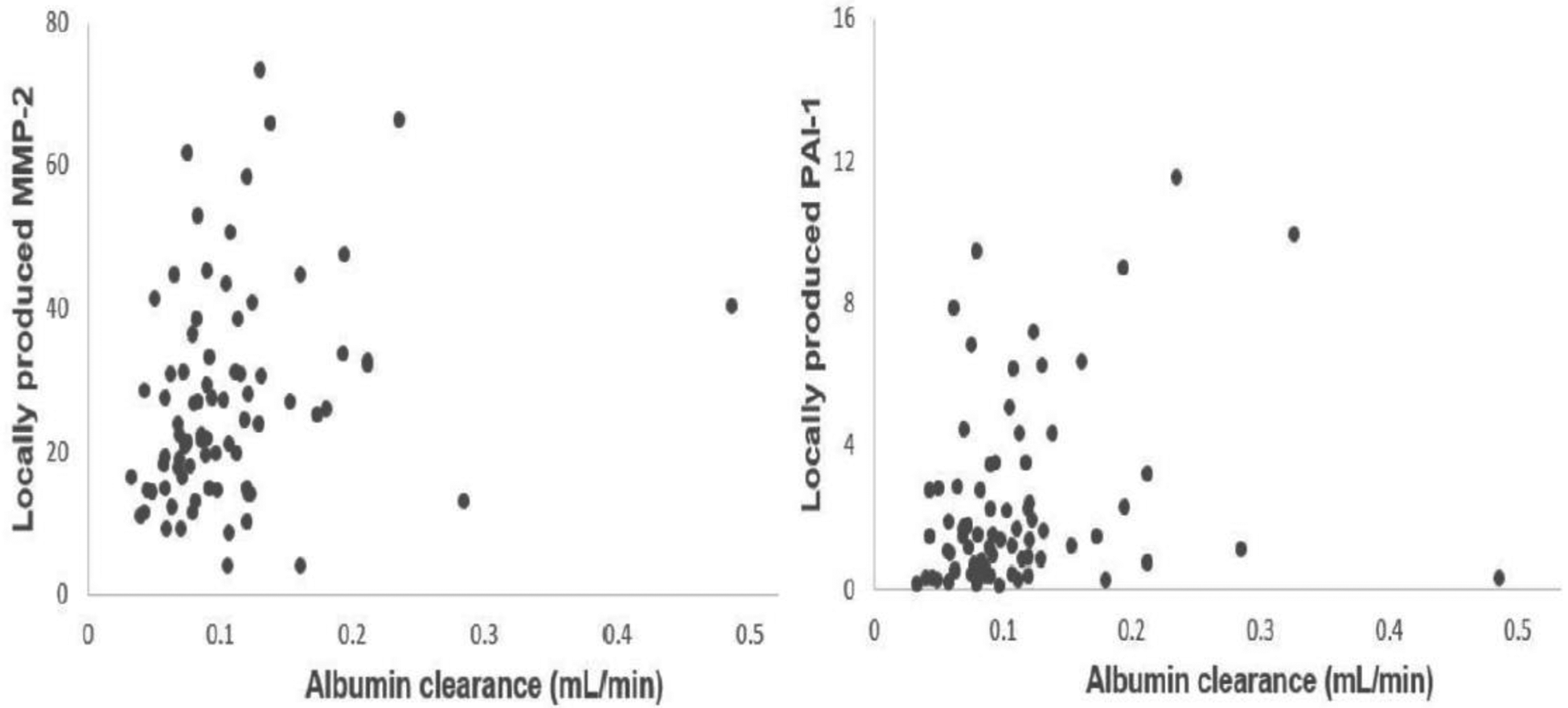
Relationship between peritoneal albumin clearance and locally produced effluent MMP-2 (left panel) and locally produced effluent PAI-1 (right panel). The explained variance was 9% for both biomarkers of fibrosis. MMP-2: metalloproteinase-2; PAI-1: plasminogen activator inhibitor-1.
Importance of vasculopathy
Vasculopathy of the peritoneal microcirculation is probably due to deposition of advanced glycosylation end products (AGE’s). 48 This process induces subendothelial hyalinosis leading to various localised luminal narrowings that are the most likely cause of a reduction in the hydrostatic pressure gradient, similar to the post stenotic pressure drop in renal artery stenosis. This reduced hydrostatic pressure gradient explains the decrease in small pore fluid transport with PD duration. 49 As stated above, a low hydrostatic pressure gradient will affect convective solute transport. Convection is not only important in the transport of solutes with a low diffusive capacity but also in the absence of a marked concentration difference between two compartments as is the case for sodium in PD. 50 It is remarkable that in a multivariable analysis, peritoneal fluid and sodium removal were significant predictors of total protein clearance in the Stoke study. 44 In the absence of information on the univariable analyses and on the explained variance in that study, the results are difficult to interpret, because sodium and fluid transport occur mainly through the small pores, while proteins have been shown to leave the intravascular compartment mainly by large pores in the venular part of the microcirculation. 9 Furthermore, the hydrostatic pressure in the arteriolar part of the peritoneal microcirculation is by autoregulation modified systemic pressure, while it has been made likely that the pressure in the venular part is regulated by the central venous pressure, at least in the absence of vasculopathy. 32,33 To investigate the role of small and large pores in more detail, we analysed relationships between hydrostatic fluid transport, which mainly but not exclusively, occurs through the small inter-endothelial pores and the clearances of individual proteins in the patients described in our longitudinal study on the time course of peritoneal transport using conventional dialysis solutions. 51 No relations were present for any of the investigated proteins during the first 2 years of treatment. Only after 3 years, some relationships were found between small pore fluid transport and peritoneal clearances of β2-Mi and albumin but with very low explained variances of 16% and 8%, respectively, as illustrated in Figure 4. No relationships were present between small pore fluid transport and peritoneal IgG and α2-Ma clearance, confirming that these large proteins are only transported through the large pores.
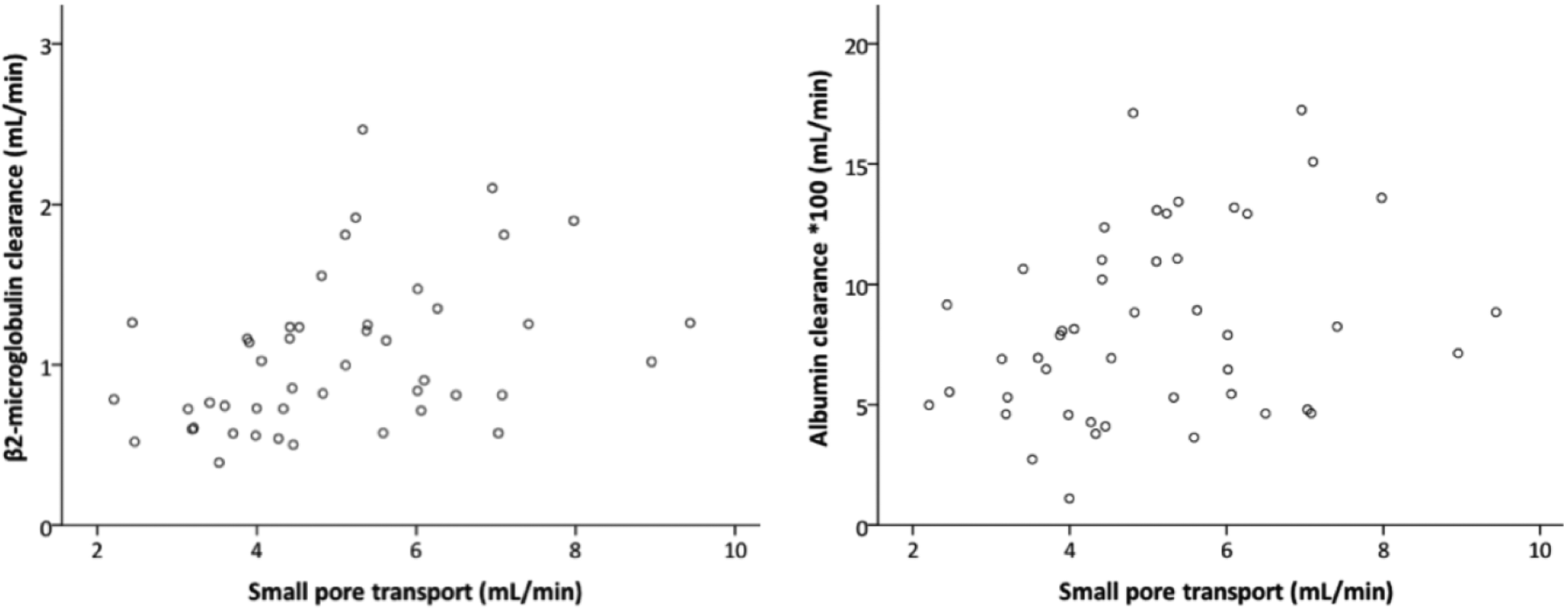
Relationship between peritoneal small pore fluid transport and β2-microglobulin clearance (left panel) and albumin clearance (right panel) after 3 years PD duration. The explained variances were 16% (left panel) and 8% (right panel).
The reduction of convective transport will affect overall peritoneal transport of solutes with low diffusion rates more than that of solutes with higher diffusion rates. Consequently, peritoneal clearance of α2-Ma will decrease more than that of albumin. This likely explains the increase in the PPRC found during longitudinal follow-up of PD patients.
Summary and conclusions
The PPRC has been developed to characterise peritoneal permeability to macromolecules by diffusion. The aim was to have a clinically useful parameter for assessment of changes in the large pore radius. Our studies have shown that this is probably only the case in the initial phase of acute peritonitis and intraperitoneal administration of nitroprusside. More importantly, the PPRC is strongly affected by the effect of the hydrostatic pressure gradient on protein convection, as is evident from our data on intraperitoneal pressure and the findings in long-term PD. The PPRC decrease in long-term patients points to the presence of venous vasculopathy, while lower small pore fluid transport rates in time indicate arteriolar vasculopathy. More longitudinal studies with individual proteins in combination with transport parameters of small solutes, free water transport and small pore fluid transport are required for final interpretation of these functional parameters of the peritoneum used as a dialysis membrane.
Footnotes
Acknowledgements
None.
Author contributions
Raymond T Krediet is the first author and has co-authored all reviewed papers from the Amsterdam group, often as senior author.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
No approval by a committee on Medical ethics is required for this review of clinical studies, that have all been published in the past, after approval by a committee on Medical ethics.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Not applicable.
