Abstract
Background:
Peritoneal dialysis (PD) is the preferred dialysis modality for paediatric patients with end-stage kidney disease. Frequently, malnutrition is encountered. Percutaneous endoscopic gastrostomy (PEG) is the preferred mode of feeding because of its minimal invasive mode of placement and easy handling in daily life. However, reports of a high risk for early post-interventional peritonitis hampered this procedure during PD and controlled studies on the benefit of peri-interventional management to prevent peritonitis are lacking. Here, we report the safety profile of PEG insertion among a cohort of children on PD by using a prophylactic antibiotic and antifungal regimen as well as modification of the PD programme.
Methods:
We performed a single-centre analysis of paediatric PD patients receiving PEG placement between 2015 and 2020. Demographic data, peri-interventional prophylactic antibiotic and antifungal treatment as well as modification of the PD programme were gathered and the incidence of peritonitis within a period of 28 days after PEG was calculated.
Results:
Eight PD patients (median weight 6.7 kg) received PEG insertion. Antibiotic and antifungal prophylaxis were prescribed for median time of 4.0 and 5.0 days, respectively. After individual reduction of PD intensity, all patients continued their regular PD programme after a median of 6 days. One patient developed peritonitis within 24 h after PEG insertion and simultaneous surgery for hydrocele.
Conclusions:
Applying an antibiotic and antifungal prophylactic regime as well as an adapted PD programme may reduce the risk for peritonitis in paediatric PD patients who receive PEG procedure.
Keywords
Introduction
Failure to thrive is common in children with chronic kidney disease and end-stage kidney disease (ESKD). 1,2 The insufficient intake of nutrients is caused by lack of appetite, nausea and vomiting as well as dietetic restrictions, 3 aggravated by dialysis-associated loss of water soluble vitamins and proteins. 4,5 To overcome these problems, gastrostomy has been implemented especially in infants and toddlers 6 and is recommended as standard of care by current guidelines. 7 –9 Thus, complications of a nasogastric tube (e.g. trauma of frequent replacement, inhibition of development of speech and swallowing, increased risk for aspiration and obvious sickness) are avoided.
Insertion of gastrostomy can be performed using open, percutaneous laparoscopic, percutaneous radiological or percutaneous endoscopic gastrostomy (PEG) techniques. In contrast to the surgical options of open and laparoscopic gastrostomy, PEG is placed under endoscopic view by the pull-through or by push technique without sutures to the abdominal wall, hence being less invasive. Percutaneous laparoscopic-assisted PEG (PLAG) and laparoscopic gastrostomy need additional surgical intervention but offer the ability to visualise the stomach during placement of the gastric tube, 10,11 which might be beneficial in special anatomic circumstances (e.g. adhesions after previous surgery or peritonitis).
Placement of PEG in children on peritoneal dialysis (PD) is considered as an additional risk factor for PD-associated peritonitis. 12,13 Therefore, PEG placement is recommended before PD initiation. 13 Only limited data are available about peri-interventional strategies to minimise the risk of peritonitis when PEG insertion is performed after initiation of PD. In general, the use of antibiotic and antimycotic drugs and open or laparoscopic gastrostomy or PLAG have been reported. 12,13 Furthermore, a temporary discontinuation of PD after gastrostomy placement and reduced fill volume after resumption are suggested even if there is a lack of data to prove the benefit of these measures. 12,14 Here, we report on children who received PEG placement during PD under a peri-interventional prophylactic antibacterial and antifungal therapy accompanied by a modified PD programme.
Materials and methods
Study design
We conducted a single-centre analysis of paediatric patients who received PEG placement during chronic PD. Based on dialysis records, we identified all children who started with PD between January 2015 and February 2020 (Supplemental Figure 1,
Technique of PEG placement
All patients underwent gastrostomy by an experienced paediatric gastroenterologist. A flexible gastroscope Olympus Evis Exera III was used. The stomach was inflated and the gastric puncture site was identified by diaphanoscopy. The skin was incised a few millimetres. One of eight patients had received local anaesthesia before. A cannula was inserted into the stomach under endoscopic visual control and the PEG tube (Freka PEG Set Gastric FR 15, ENFit) was placed by pull-through technique without any kind of retention suture. Then, it was fixed to the abdominal wall with a fixation device on the tube.
Peri-interventional management
Before starting endoscopic intervention, the last session of PD ended with the complete drainage of the dialysate. After PEG placement, PD was paused at least until the first post-operative day. The interruption of PD was extended if tolerable or restarted according to the paediatric nephrologist’s decision based on clinical and laboratory issues. Whenever possible, PD was restarted with a modified programme: manual PD changing system or automated PD cycles with reduced fill volume.
According to International Society for Peritoneal Dialysis (ISPD) 2012 recommendations, 12 all patients received antibacterial and antifungal therapy to prevent infectious complications. Cefotaxime was used as standard antibiotic prophylaxis taking into account centre-specific susceptibility patterns. Colonisation with multidrug-resistant pathogens, former peritonitis episodes with proven causative organisms, and additional surgical interventions at the abdomen or genitourinary tract at the time of PEG placement were considered when choosing the antibacterial treatment. For antifungal prophylaxis, all patients received echinocandins (micafungin or caspofungin). Six hours after PEG placement, administration of water or tea was started with 2–3 ml/kg body weight as bolus and, in case of good tolerance, tube feeding was started.
Results
Patient characteristics
Eight PD patients with a median age of 6.1 months (IQR 0.4–20.5) at the initiation of PD received PEG placement. Patient baseline characteristics are shown in Table 1. Congenital anomalies of the kidney and urinary tract (CAKUT) with posterior urethral valves were the most common cause of ESKD (three patients). Five of eight (62.5%) patients were male. All patients were receiving continuous cycling peritoneal dialysis (CCPD). PEG insertion was performed at a median of 162 days after the initiation of PD to treat poor growth and feeding disorders. At this time, the median weight was 6.7 kg (IQR 6.0–10.2). PEG intervention was performed in a planned inpatient admission in six patients. In two cases, patients were hospitalised for other reasons.
Patient characteristics and outcome.
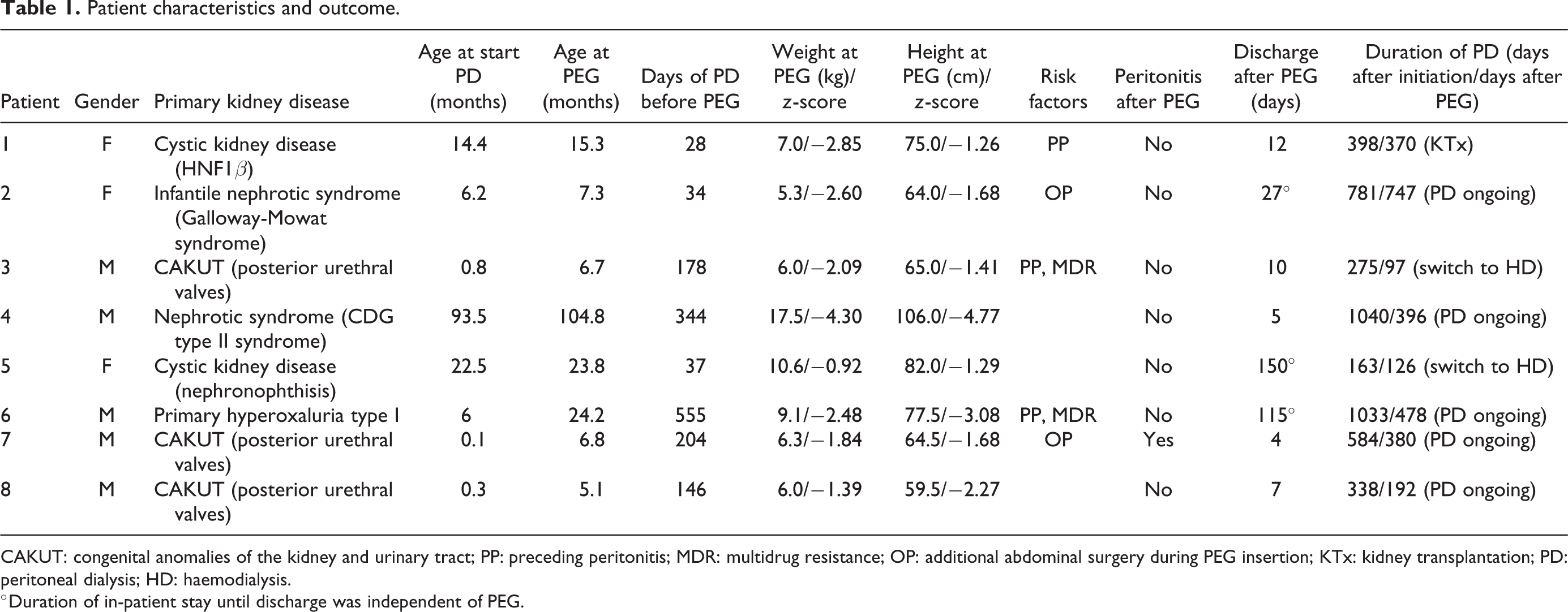
CAKUT: congenital anomalies of the kidney and urinary tract; PP: preceding peritonitis; MDR: multidrug resistance; OP: additional abdominal surgery during PEG insertion; KTx: kidney transplantation; PD: peritoneal dialysis; HD: haemodialysis.
°Duration of in-patient stay until discharge was independent of PEG.
Peri-interventional management
Preceding peritonitis episodes, colonisation with multidrug-resistant pathogens and additional abdominal or genitourinary surgery at PEG placement were considered as additional risk factors for peritonitis after PEG insertion (Table 1). Before PEG insertion, three patients had episodes of PD-related peritonitis (patients # 1, 3 and 6). In patient #1, a causative pathogen was not revealed. Patient #3 experienced two episodes caused by different 2MRGN strains. Patient #6 was treated in his country of origin before and two episodes of peritonitis caused by Gram positive pathogens were recorded. None of our patients was colonised with 3MRGN/4MRGN or MRSA, but two of them were found to have 2MRGN. When the PEG procedure was performed, two patients received concurrent inguinal interventions, hernia and hydrocele surgery, respectively.
The peri-interventional procedure including antibiotic and antifungal therapy and modification of PD schedule in all eight patients is summarised in Figure 1.
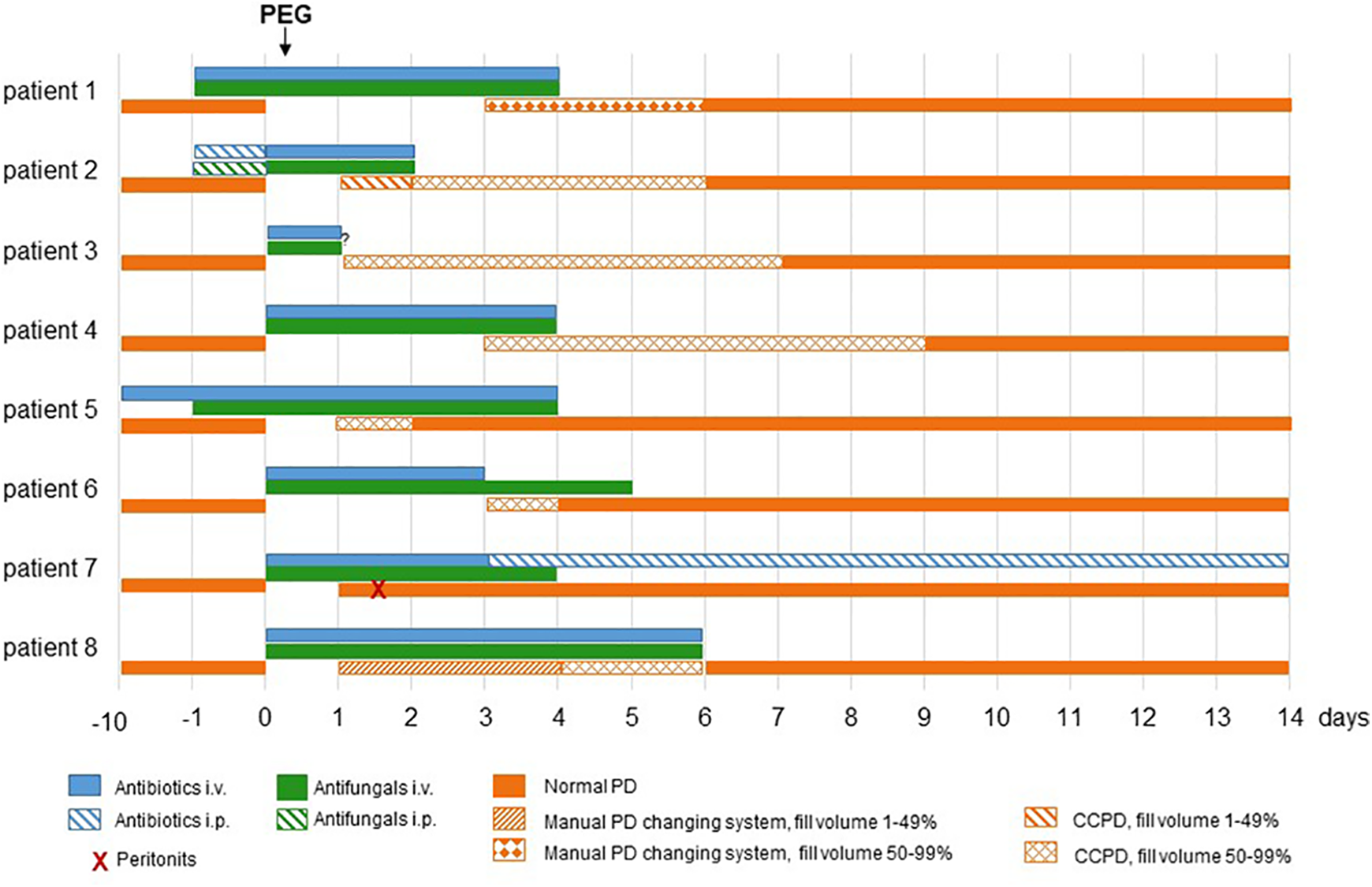
Observed peri-interventional procedure for prevention of peritonitis.
Dependent on individual infection risks, the standard antibiotic therapy with cefotaxime was adapted (Supplemental Table 1): Ampicillin was administered in patients with prior findings of
Except for patient #5 who was receiving antibiotic treatment of pyelonephritis since day -10, antibiotic prophylaxis was initiated within 24 h before PEG insertion (at induction of anaesthesia at the latest) and continued for 1–5 days according to the individual course. For patient #3, only information about antibiotic and antifungal substance starting at day 0 before PEG insertion was available, but not dosing and duration. The median duration of antibiotic prophylaxis (
Interruption of PD and reduced programmes differed between patients according to their individual ability to cope with limited dialysis conditions. Figure 1 shows interruption and course of resumption of PD in the eight patients. In general, automated PD was preferred for resumption of PD (
Peritonitis after PEG placement and outcome
One episode of peritonitis was observed during the first 28 days after PEG placement (Table 1; Figure 1). Patient #7 developed signs of infection with fever and rising inflammation parameters at day 1 after PEG insertion and concurrent hydrocele surgery. Besides, he had already started with automated PD at this day with full fill volume. The effluent was cloudy and elevated leucocyte count (1026/µl) was observed, matching the criteria for peritonitis. Antibiotic therapy was adapted by teicoplanin i.v. and ceftazidime i.v. and was continued intraperitoneally from day 3 until day 14 (Figure 1). Microbiological analysis of the dialysate did not reveal a causative pathogen, but cell count and differentiation of leucocytes with 80.9% polymorph nuclear granulocytes in dialysate were suspicious for bacterial peritonitis. Automated PD was re-established without problems by applying the patient’s regular fill volume from day 2. The patient showed rapid clinical improvement from day 2 onwards with dialysate looking clinically unremarkable in the control and was discharged 4 days after PEG insertion. All patients were discharged after a median time of 11 days (IQR 5.5–93) after PEG placement (Table 1).
Discussion
We report a single-centre study about PEG insertion during PD in eight patients with ESKD. By applying an antibiotic and antifungal prophylaxis as well as a reduction of PD programme within the first days after PEG insertion, our results indicate a low rate of PD-associated peritonitis. Within 28 days after PEG placement, we observed peritonitis in only one of eight patients even though the age at onset of PD was lower (≤2 years) than in other reports. 14,16
Former studies reported high rates of complications after PEG insertion in PD patients. In 4 of 5 PD patients, peritonitis occurred early after PEG insertion, 14 while others found early onset peritonitis (<7 days after PEG) in 36% of patients and early fungal peritonitis in 4 of 27 patients. 16 In four cases, a change to haemodialysis (HD) became necessary and the death of two patients was described related to later PEG complications. Zaritsky et al. described an up to three-fold increased risk of peritonitis associated with PEG insertion after PD initiation compared to PEG before or at PD catheter placement. 17 In contrast, a study by Rahim et al. found no difference in the peritonitis rate depending on the time point of gastrostomy. 18 As a consequence and, in the absence of larger prospective studies, current clinical practice recommendations by Pediatric Renal Nutrition Taskforce suggest open gastrostomy or PLAG in patients already treated with PD, as these techniques are supposed to have a lower risk of peri-interventional peritonitis. 13
Theoretically, the surgical procedure limits the risk of infection, as the stomach is secured to the abdominal wall by sutures. 3 For laparoscopic gastrostomy a similar safety profile is reported compared to open gastrostomy, 19 while the incidence of peritonitis after laparoscopic gastrostomy was lower compared to open gastrostomy. 14 In contrast, in PEG via pull-through technique, there are no sutures securing the gastrostomy with the potential risk of minimal gastric content leakage within the first days after PEG insertion until the stomach fully attaches to the abdominal wall. 3 Therefore, PEG needs to be tightly closed after insertion procedure keeping the gastrostomy tight which should prevent relevant amounts of gastric content leak.
In our cohort, we used PEG which offers the advantage of a more rapid and less invasive method of gastrostomy with less incisional pain, shorter inpatient stay and, thus, a reduction of health care burden. 20 All patients received pre-operative antibiotics and antifungal prophylaxis in line with current guidelines. 13 In our cohort, only one of eight patients suffered from early peritonitis without any further complications. Fungal infections were not encountered. The variability of prophylactic treatment in our cohort reflects the individual conditions of the patients but also the need of strict implementation of standardised procedure. Considering the expected bacterial spectrum and the local resistance situation, we decided to use cefotaxime as the standard antibiotic prophylaxis in derogation of ISPD recommendations, 12 which is according to the current standard procedure of PEG insertion at our paediatric gastroenterological centre. For antifungal prophylaxis, echinocandins were used in all patients, as their dose is independent of estimated glomerular filtration rate and of dialysis. Because of these advantages compared to fluconazole echinocandins were used deviating from ISPD recommendations. These agents were reported to have effectivity against a wide spectrum of invasive infections especially by yeasts like candida which are leading cause of PD-associated fungal peritonitis. Among echinocandins, micafungin and caspofungin are approved for treatment in children. 21 Patients #1–6 received micafungin, but caspofungin was administered for prophylaxis after a recommendation was released for a more restrictive use of micafungin due to severe hepatotoxic effects in animal models. Both substances were well tolerated by our patients. Our observation underlines the protective effect of peri-interventional antimicrobial prophylaxis at PEG insertion in PD. We interrupted PD after PEG insertion as long as tolerable for each patient and reduced fill volume as long as possible.
Based on our presented retrospective data, we propose a standardised procedure as summarised in Figure 2. Our scheme provides antibiotic and antifungal treatment over 3 days in order to lengthen protective effects during the first days when PEG is still a fresh wound, and abdominal manipulation occurs by water/nutrition application and PD resumption. Choice of antibiotic substance needs regular re-evaluation in the future due to possible changes of local resistance situation. An optimal anti-infectious regimen has not yet been determined by any clinical study and should be determined by local situations. Based on our data, we recommend interruption of PD after PEG insertion as long as tolerable for each patient (1–2 days) and reduced fill volume afterwards for another 2 days in our peri-interventional protocol. However, if minimal 24 h interruption and subsequently reduced PD programme are not feasible, temporary HD might be discussed as another approach. Whether the start of PD resumption with a manual PD changing system offers any benefit, is unknown. Most of our patients were treated with CCPD right from the restart of PD.
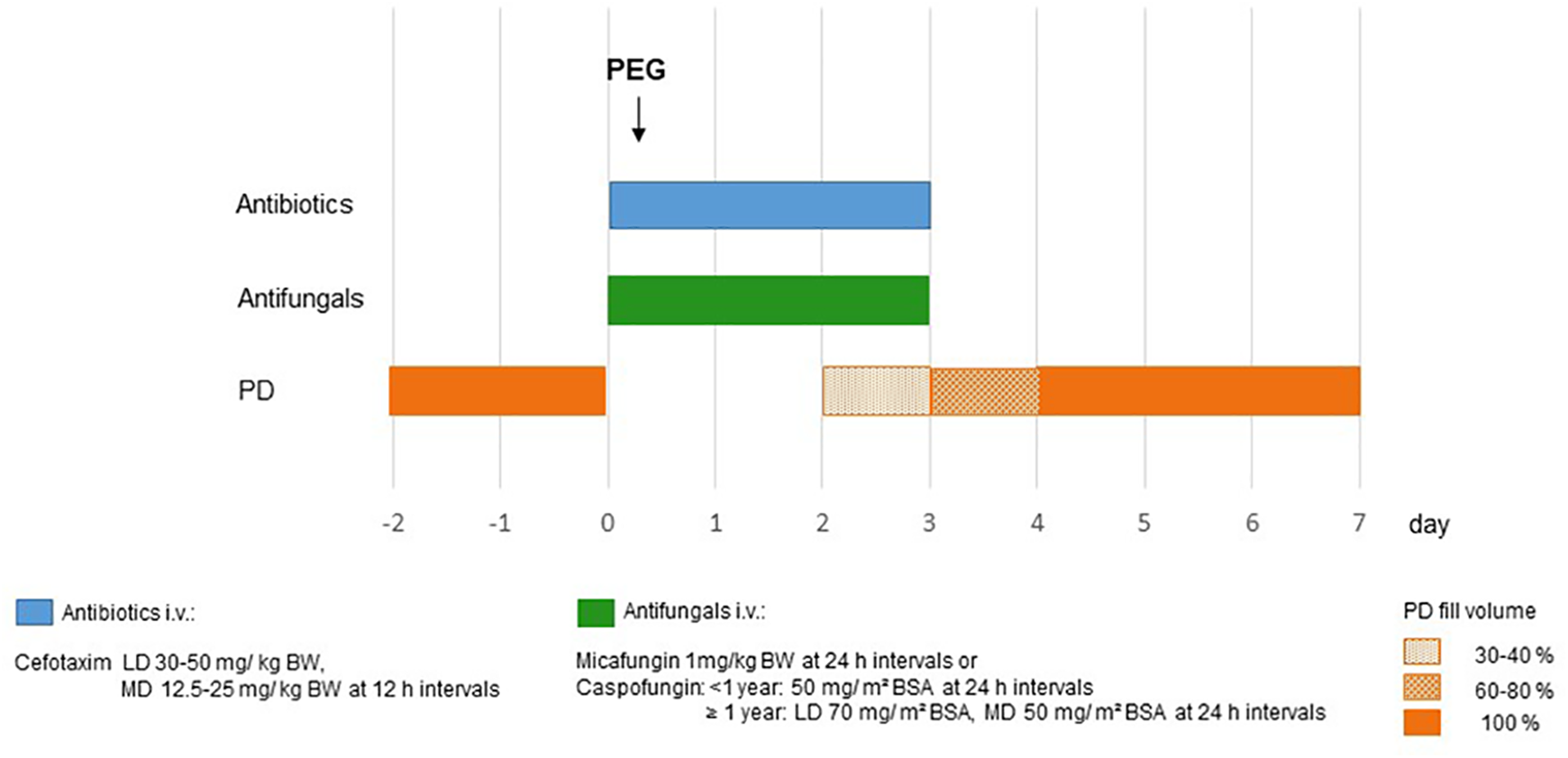
Scheme of the intended peri-interventional procedure for prevention of peritonitis. Antibiotic prophylaxis for 3 days, starting before PEG insertion with loading dose. Maintenance dose adapted to GFR. Antifungal prophylaxis for 3 days, starting before PEG insertion. Interruption of PD: day of PEG insertion and day 1. Resumption of PD: day 2 with 30–40%, day 3 with 60–80% of filling volume, from day 4 after PEG: 100%. BW: body weight; BSA: body surface area; LD: loading dose; MD: maintenance dose; PD: peritoneal dialysis.
Limitations of this study are its small cohort and retrospective character. This causes a high variability of treatment data limiting conclusions. We focused on early complications after PEG insertion in PD patients as we aimed to implement a safe peri-interventional procedure for this situation. Potential advantages of the recent direct puncture technique for PEG insertion combined with percutaneous gastropexy need evaluation in ESKD patients treated with PD.
In conclusion, we report a cohort of children with ESKD on PD undergoing PEG placement. By using antibiotic and antifungal prophylaxis and withholding PD after PEG insertion for as long as tolerable, we observed low incidence of peritonitis. We assume PEG insertion in patients during PD is a safe option at an experienced centre when adequate preventive therapeutic measures are taken.
Supplemental material
Supplemental Material, sj-docx-1-ptd-10.1177_08968608211057651 - Feasibility of percutaneous endoscopic gastrostomy insertion in children receiving peritoneal dialysis
Supplemental Material, sj-docx-1-ptd-10.1177_08968608211057651 for Feasibility of percutaneous endoscopic gastrostomy insertion in children receiving peritoneal dialysis by Caroline Kempf, Johannes Holle, Susanne Berns, Stephan Henning, Philip Bufler and Dominik Müller in Peritoneal Dialysis International
Supplemental material
Supplemental Material, sj-docx-2-ptd-10.1177_08968608211057651 - Feasibility of percutaneous endoscopic gastrostomy insertion in children receiving peritoneal dialysis
Supplemental Material, sj-docx-2-ptd-10.1177_08968608211057651 for Feasibility of percutaneous endoscopic gastrostomy insertion in children receiving peritoneal dialysis by Caroline Kempf, Johannes Holle, Susanne Berns, Stephan Henning, Philip Bufler and Dominik Müller in Peritoneal Dialysis International
Footnotes
Authors’ note
Caroline Kempf and Johannes Holle contributed equally to this study.
Acknowledgements
None declared.
Author contributions
CK and JH: conception and design of the study. CK, JH and SB: collection of data, data analysis and interpretation. CK and JH: drafting of the article. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments or comparable standards.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Not applicable.
Trial registration
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
