Abstract
Life participation requiring physical activity and physical function is a key patient-reported outcome for people receiving peritoneal dialysis (PD). Clinician guidance is required from multidisciplinary sources regarding exercise and activity advice to address the specific needs of this group. From August 2020 through to June 2021, the Global Renal Exercise Network and the International Society for Peritoneal Dialysis reviewed the published literature and international clinical experience to develop a set of clinical practice points. A set of questions relevant to physical activity and exercise were developed from the perspective of a person receiving PD and were the basis for the practice point development. The GRADE framework was used to evaluate the quality of evidence and to guide clinical practice points. The review of the literature found sparse quality evidence, and thus the clinical practice points are generally based on the expert consensus of people receiving PD, PD exercise expert clinicians and experienced PD exercise researchers. Clinical practice points address timing of exercise and activity (post-catheter insertion, peritoneal space empty or full), the uptake of specific activities (work, sex, swimming, core exercise), potential adverse outcomes related to activity and exercise (exit site care, perspiration, cardiovascular compromise, fatigue, intra-abdominal pressure), the effect of exercise and activity on conditions of interest (mental health, obesity, frailty, low fitness) and exercise nutrition.
This is a visual representation of the abstract.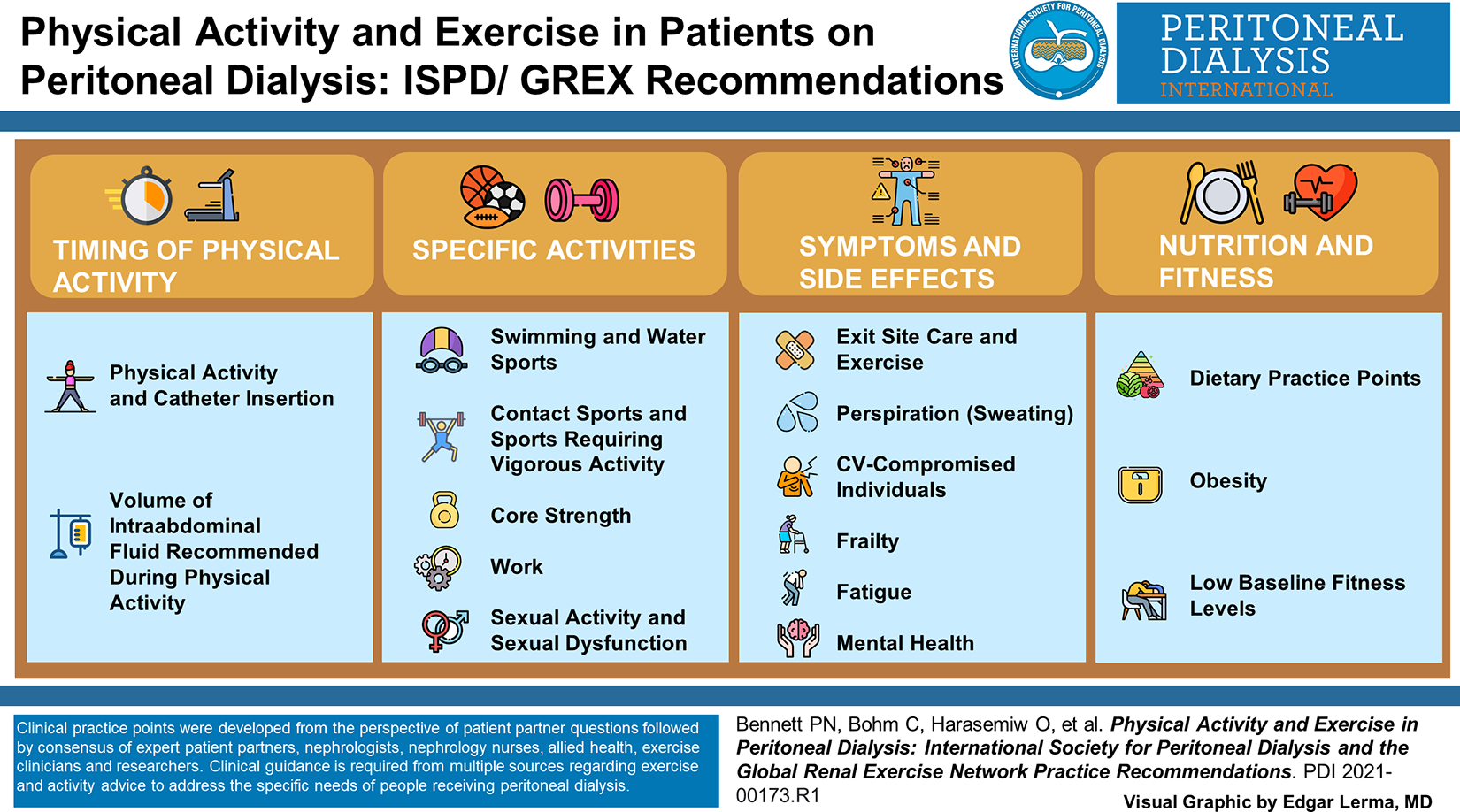
Background
Functional impairment is highly prevalent in people receiving peritoneal dialysis (PD). 1 Life participation requiring physical activity and physical function is a key patient-reported outcome in PD. 2 Given that most PD clinicians are not exercise experts, guidance is required from multidisciplinary sources regarding physical activity (defined as any bodily movement resulting in energy expenditure) and exercise (a type of planned and structured physical activity with the objective of improving or maintaining physical fitness) advice for the PD population.
Methods
From August 2020 through to June 2021, the Global Renal Exercise Network (GREX) in association with the International Society for Peritoneal Dialysis (ISPD) consulted 22 experts in exercise and PD to develop clinical practice points. A review of the literature was performed using the Pub Med database with the search terms ‘exercise’ OR ‘physical activity’ AND ‘peritoneal dialysis’ (January 2010 to July 2020). To identify the expert group, invitations were sent to the lead authors from the identified peer-reviewed PD exercise and physical activity intervention publications and expressions of interest were invited from GREX, an international group formed in 2019 to foster research and innovation across multiple disciplines to develop strategies to increase physical activity and improve health outcomes for people with chronic kidney disease (https://grexercise.kch.illinois.edu/). Only professionals who had researched or were involved in clinical PD exercise programs were included in the practice point development group. This group consisted of nephrologists experienced with the care of people on PD (CB, DJ, DWJ, JM, AYMW, ST, DZ, PKTL), 1 x physical therapist (NSR), kinesiologists (KP, KS, IG), exercise physiologists (CJL, MiW, BT), 1 x researcher (OH), dietitians (KL, AM), 1 x PD nurse (PNB) and 1 x PD nurse practitioner (LB).
Additionally, a group of patient partners with lived experience of PD (NSR, NV, MaW) was formed to advise the PD professionals throughout the process. The patients were from Canada, Australia and the United Kingdom. Although a patient with PD experience was requested from a low-income country, the group was unsuccessful in sourcing a person with this profile.
Common questions asked from the perspective of a person receiving PD and uncertainties relevant to physical activity and exercise in the PD population were identified through consensus by the author group and the patient partner group. Through literature review and clinical experience, a set of clinical best practices for use by clinicians was developed that addressed these questions and uncertainties. These practice points consist of activity timing issues (post-catheter insertion, peritoneal space empty or full); guidance for specific activities (work, sex, swimming, core exercise); potential adverse outcomes related to activity and exercise (exit site care, perspiration, cardiovascular compromise, fatigue, intra-abdominal pressure (IAP); effect of exercise and activity on select conditions of interest (mental health, obesity, frailty, low fitness) and exercise nutrition. Finally, the practice points presented in this report were reviewed by the GREX patient partner steering committee to ensure the appropriateness and relevance to people living with kidney disease.
The global perspective of this report emphasises the relevance of physical activity and exercise to all people receiving PD, particularly in low-income countries (LICs) and low to middle-income countries (LMICs). Thus, the practice points, where possible, have been written to be applicable to LICs and LMICs. The GRADE (Grading of Recommendations, Assessment, Development and Evaluations) framework was used to evaluate the quality of evidence and to guide recommendations. 3 The strength of each recommendation is indicated as either Level 1 (we recommend) or Level 2 (we suggest), and the certainty of the supporting evidence is shown as A (high certainty), B (moderate certainty), C (low certainty) or D (very low certainty). The summary of the consensus process can be seen in Figure 1.
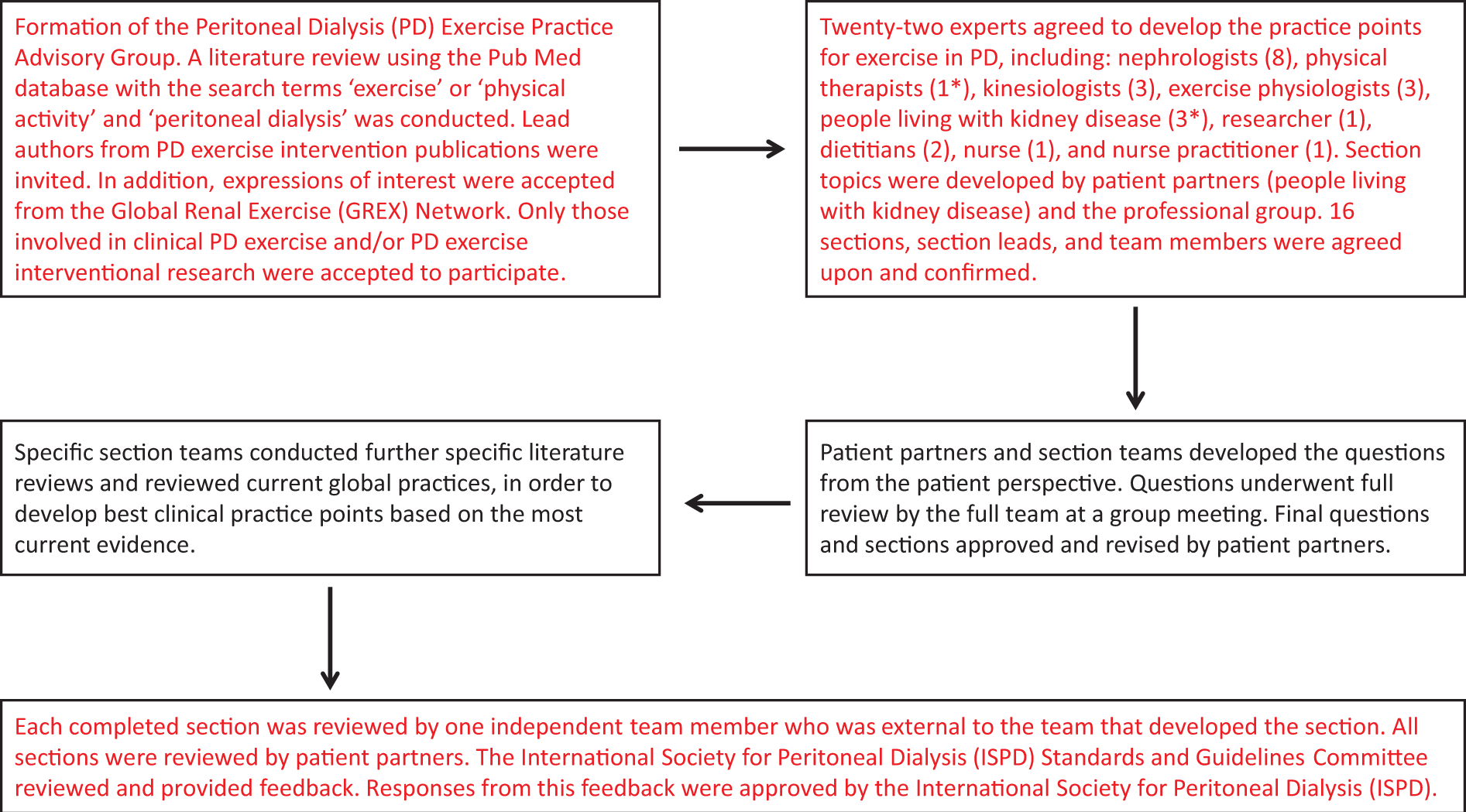
Development process for the peritoneal dialysis physical activity and exercise best clinical practices. *One member was a physiotherapist and a peritoneal dialysis recipient.
Practice point implementation
Maintaining physical function through physical activity and exercise can be challenging for people on PD. There is limited high-quality evidence, and thus the clinical practice points are predominantly based on low-quality evidence and clinical experience. 4 Unfortunately, given this lack of evidence and clinician exercise experience, people on PD are often discouraged from participating in exercise programs because of perceived barriers and uncertainty about the best exercise regimen. 5
It is important to address barriers and provide recommendations to assist in implementing clinical practice points. Although local and global barriers to exercise and physical activity exist for people on PD, clinical and policy recommendations have been previously proposed to address these barriers. 6 Nephrologists and physicians can include physical function as part of the medical history, proactively discuss physical activity to modify chronic disease risk and promote participation in exercise studies. Dialysis care providers can include physical assessment in clinical pathways, develop physical activity key performance indicators, advocate for exercise professionals, seek opportunities and partnerships with universities and design activity centres in future home dialysis clinics. Dialysis funders can include exercise professionals in future funding models, physical activity credits to offset medication or insurance costs, promote fitness membership in insurance packages and provide incentives for clinics that promote physical activity. 6
For LICs and LMICs, exercise and physical activity do not have to be expensive. For example, elastic fitness bands for resistance exercises are inexpensive and easy to store and can be used for frail, intermediate and high functioning PD patients for use at home. Although high level, personal training professionals are out of the financial reach of many people, cheap alternatives such as group activities can be encouraged by PD providers.
Although PD practices vary worldwide, 7,8 one constant is the lack of physical activity and physical capacity in this group. 9,10 Few providers offer exercise programs, with notable exceptions in some regions of Mexico, Portugal, Germany, the United Kingdom and Canada. 11,12 If we do not address the low physical function of people on PD, many will be lost to this modality, decreasing their independence and quality of life. The following practice points have been generated to provide clinical guidance and accelerate the conversation around keeping this group physically functional and independent to maintain quality and quantity of life.
Summary of clinical practice points
1. Timing of physical activity (practice points 1.1–1.2)
1.1. Physical activity and catheter insertion
Regardless of surgical technique, walking is safe and should be encouraged as soon as possible following catheter insertion.
Activities that are associated with an increase in intra-abdominal pressure (IAP) (e.g. lifting anything over 5–10 kg, chopping wood, shovelling snow, vacuuming, sit-ups) should be delayed for at least 2 to 3 weeks after buried/laparoscopic catheter insertion and for 4 to 6 weeks after open surgery.
For PD catheters that are to be used before complete healing, delay activities that increase IAP for 4 to 6 weeks.
1.2. Volume of intra-abdominal fluid recommended during physical activity
For activities that are not associated with a significantly higher IAP such as walking, hiking and jogging, PD fluid does not need to be drained prior to exercise unless the ‘fullness’ contributes to patient discomfort.
For activities such as weightlifting, snow shovelling and jumping, which are associated with much higher IAP, PD fluid should be drained prior to exercise.
2. Specific activities (practice points 2.1–2.5)
2.1. Swimming and water sports
We suggest that swimming or other water sports should preferably take place in either sea water or swimming pools that are known to be well maintained (private or municipal) to limit exposure to waterborne pathogens.
We suggest avoiding swimming or other water sports in open water directly after storms to limit exposure to waterborne pathogens.
A clear waterproof dressing or colostomy bag to secure and protect the catheter and exit site from getting wet during bathing or swimming, although it remains uncertain as to whether this strategy mitigates the risk of infection.
We recommend that routine exit-site care should be performed after swimming and water sports.
2.2. Contact sports and sports requiring vigorous activity
Participation or gradual return to sport is recommended for people on PD.
Resumption of physical activity and exercise are discussed with the patient in the post-operative period following appropriate local interdisciplinary collaboration and consultation.
Contact sports where there is risk of physical trauma or repetitive rubbing occuring to the catheter site are not recommended.
Sports that require frequent bending, squatting or lifting may be best done without PD fluid in situ. When performing such movements, the following factors which may place participants at increased risk of adverse outcomes should be considered: age, physical capacity, comorbidities, PD vintage, previous injuries.
Patients may prefer their peritoneum to be empty for greater comfort when participating in sports.
The use of a PD belt during sports may provide enhanced protection and comfort to the catheter and catheter site.
Patients may need to temporarily modify or cease their sporting activities if their PD fluid becomes pink or red-tinged, suggesting intra-peritoneal bleeding. Following resolution and, when approved by a relevant professional within the PD care team, activity may resume.
2.3. Core strength
Core strengthening exercises are recommended for PD patients.
Stronger core muscles can support the low back and prevent/manage low back pain.
Stronger transverse abdominal muscles can support increased IAP secondary to PD fluid, potentially reducing hernia risk.
2.4. Work
People receiving PD should be encouraged to continue to work.
Clinicians should support patients to liaise with their employer to discuss ways of continuing employment.
Clinicians should discuss if the person’s employing organisation has a corporate wellness program that the person receiving PD could safely participate in.
For those who are primarily sitting at work, intermittent movement should be performed throughout the work shift. For example, perform sit-to-stands, calf raises, arm circles, marching in place every hour.
Functional exercises targeted to work tasks should be performed. For example, those who are performing lifting tasks much of the day, exercises such as targeted stretching, core strengthening, shoulder press, seated/standing row and deadlifts could be recommended.
2.5. Sexual activity and sexual dysfunction
The risk of PD fluid leak or dislodgement during sexual activity (after 4 weeks post insertion of PD catheter) is low.
Factors such as age, blood pressure, medications, iron treatment, glucose levels and lipid levels can contribute to sexual dysfunction and should be assessed.
Pharmaceutical treatment of erectile dysfunction should be discussed on an individual basis.
Fatigue can have a significant impact on sexual activity.
Clinicians should provide education and support regarding how the type of PD (i.e. automated PD versus continuous ambulatory PD) may impact sexual activity
Patients’ concerns regarding body image, impact of scars, weight changes and safety of sexual activity need to be addressed by clinicians, especially the fear of catheter dislodgement.
Symptoms/side effects (practice points 3.1–3.6)
3.1. Exit site care and exercise
We suggest that applying a non-occlusive dressing, such as gauze, may reduce microtrauma to the skin that may result from skin friction during moderate to vigorous activity.
We recommend wearing breathable and freshly laundered clothing during physical activity to decrease sweating near the exit site.
We suggest that, when engaging in moderate to vigorous activity, the catheter should be immobilised with tape to prevent trauma to the catheter or the exit site.
We recommend the exit site should be cleaned immediately after it becomes soiled or wet during exercise and that the exit site be cleaned routinely at least twice per week.
3.2. Perspiration (sweating)
Shower and clean the PD exit site shortly after any vigorous exercise that leads to sweating to limit any potential risk of infection risk associated with perspiration.
Individuals who are on PD and are obese may be at higher risk of exit site complications associated with perspiration.
3.3. Cardiovascular-compromised individuals
A personalized approach is required for exercise prescription, taking into consideration the severity and nature of heart disease, medical comorbidities and usual functional status.
Personalized short-term and long-term training goals should be clearly defined.
A multidisciplinary approach (nephrologist, nurse, dietitian, local or remote exercise professional) should be adopted in the prescription, monitoring and supervision of exercise.
A graduated approach in exercise prescription with gradual progression in exercise intensity and duration as tolerated should be adopted.
3.4. Frailty
Physical activity and/or exercise training programs can prevent and reduce physical frailty and reduce the risk of disability.
Use frailty screening tools to identify those who would benefit most from physical activity (e.g. FRAIL scale, Fried phenotype, Clinical Frailty Scale).
Use physical function tests to assess strength and balance (e.g. Short Physical Performance Battery (SPPB), Sit-To-Stand (STS), Timed-Up-and-Go (TUG), gait speed) to identify those with decreased function status/frailty and those who would benefit from exercise. Cut-off points for predicting development of disability as follows: SPPB score ≤9, STS-5 ≥10 s, TUG ≥9 s.
Use screening tools and physical function test results to guide exercise prescription.
Design exercise programs that fit into each individual’s lifestyle and provide education on the benefit of physical activity in maintaining function, for example, climbing stairs. The PD care team (nephrologist, dialysis nurse, social worker, dietitian) will determine the appropriate assessment lead given the varying resources from centre to centre and country to country.
Consider exercises, such as walking or stationary cycling, body weighted resistance exercises and balance exercises, particularly for those at high risk of falls.
3.5. Fatigue
Physical activity and exercise may reduce fatigue symptoms and should be encouraged.
Exercise programs should aim to elicit an intensity of Rating of Perceived Exertion (RPE) 11-16 (using the Borg RPE Scale 6-20).
A walking program (outdoor or treadmill) employing a graded approach (i.e. incremental over time) is well tolerated and may decrease fatigue experience and increase an individual’s ability to perform daily activities
3.6. Mental health
Regular, moderate intensity exercise 20–30 minutes, 3-5 times per week is likely to improve or maintain mental health (including symptoms of anxiety and depression).
Screening for depression, recognition of causes and a discussion with the patient should ensue to develop an individualized approach for the treatment of depression.
Nutrition and fitness (practice points 4.1–4.3)
4.1. Dietary practice points
Eat a small meal or snack containing carbohydrate before exercise.
If exercise duration is longer than 60 minutes, additional carbohydrate intake may be required during exercise.
Fluid intake during exercise should match but not exceed sweat losses and be individualized in context with usual urine output and fluid allowance.
It is reasonable to consume 20 grams of high-quality protein immediately after resistance exercise to support protein synthesis.
Sports nutrition supplements, such as protein supplements and sports drinks, should be used under clinical supervision.
Where resources allow, dietitians are a valuable member of the PD team to advise on nutrition and exercise.
4.2. Obesity
Physical activity and exercise are safe in obese individuals and should be recommended.
Consider performing vigorous exercise with an empty (PD fluid) peritoneum, as this may: • Improve one’s capacity to exercise. • Mitigate the risk of abdominal and musculoskeletal strain if performing vigorous activities that may increase IAP.
Consider performing low-impact exercises such as swimming and cycling, as these may: • Improve one’s capacity to exercise. • Mitigate the risk of musculoskeletal injury. • Exercise is an effective adjunctive therapy for weight management and weight loss, particularly with the supervision of a specialized dietitian.
4.3. Low baseline fitness levels
Any increase in daily physical activity and decrease in sedentary time are likely to have health benefits for people receiving PD.
Individuals should start exercising slowly and at a low intensity to understand their baseline level of fitness and to minimize the risk of injuries.
Incorporate both aerobic (e.g. walking, dancing, seated marching, seated cycling) and resistance (e.g. sit-to-stands, seated row, seated leg curl with resistance bands) activities in a stepwise manner.
Track exercise performance, with a view to steadily increase the frequency, intensity and time spent exercising.
In line with World Health Organization guidelines, individuals with very low activity levels should gradually work towards achieving over time: • 150–300 minutes of moderate-intensity aerobic physical activity (e.g. walking, swimming, dancing) per week OR • 75–150 minutes of vigorous-intensity aerobic physical activity (e.g. jogging, competitive sports) per week OR • An equivalent combination of moderate intensity and vigorous intensity aerobic activity AND • 2 or more days per week of muscle-strengthening activities (e.g. push-ups, squats)
Older adults should include activity that focuses on balance and strength training to improve functional capacity and reduces falls risk.
Patient question, rationale, evidence, practice points and further research
1. Timing of physical activity (practice points 1.1–1.2)
1.1. Physical activity and catheter insertion
Question from the perspective of the person living with PD:
‘Following PD catheter insertion, how long must I wait to exercise and what exercises are safe to do?’
Question Rationale:
Recommendations for resumption of physical activity and exercise post PD catheter insertion vary and are inconsistent. Uncertainties include when to resume activity, what type of activity is safe to resume and at what intensity. Concerns regarding resumption of activity include delayed wound healing/dehiscence, peritonitis, peritoneal leak and hernia development. Moreover, there may be differences depending on surgical technique used to insert the PD catheter such as buried, laparoscopic or open.
Evidence and/or clinical experience
IAP is greatest when sitting followed by standing and supine. 13,14
Tensile wound strength increases dramatically from post-operative days 5 to 15. 15
Several programs recommend walking immediately following PD catheter insertion but delay any type of strenuous activity for 2 to 6 weeks. 16 –18
Several programs recommend avoid swimming after PD catheter insertion until either advised by their healthcare team (14) or for 4 to 6 weeks. 17
Practice points
Regardless of surgical technique, walking is safe and should be encouraged as soon as possible following catheter insertion.
Activities that are associated with an increase in IAP (e.g. lifting anything over 5–10 kg, chopping wood, shovelling snow, vacuuming, sit-ups, etc.) should be delayed for at least 2 to 3 weeks after buried/laparoscopic catheter insertion and for 4 to 6 weeks after open surgery.
For PD catheters that are to be used before complete healing, delay activities that increase IAP for 4 to 6 weeks.
Further research
Does baseline fitness, including abdominal strength, affect time to wound healing?
Is there any additional benefit in waiting to exercise for time periods beyond 2 weeks?
Does cycling (due to increase IAP with sitting) require delayed return to sport compared to standing activities?
1.2. Volume of intra-abdominal fluid recommended during physical activity
Question from the perspective of the person living with PD:
‘Should I leave the PD fluid in my abdomen when I exercise?’
Question Rationale:
IAP increases with certain types of exercise and it is not clear to what extent the presence of PD fluid contributes to increased IAP.
Evidence and/or clinical experience
Studies suggest that increasing the volume of PD fluid in the abdomen results in higher IAP, although not in a linear fashion. 19,20
Different body position also impacts the IAP and is lowest with the supine position, higher with standing and highest with sitting. 21,22
The type of exercise affects the IAP differently and is lowest with walking and jogging and highest with weightlifting and jumping. 22
There is no consistent correlation between PD fluid volume and subsequent risk of hernia 23 or PD fluid leaks. 21
The presence of PD fluid in the abdomen does not appear to have any consequences on exercise capacity, as shown by an unchanged V02 max and aerobic threshold with and without a 2-litre fill. 22
The presence of PD fluid in the abdomen is considered akin to performing exercise while holding additional weight and exercise time may need to be shortened in this setting. 22
Practice points
For activities that are not associated with a significantly higher IAP such as walking, hiking and jogging, PD fluid does not need to be drained prior to exercise unless the ‘fullness’ contributes to patient discomfort.
For activities such as weightlifting, snow shovelling and jumping, which are associated with much higher IAP, PD fluid should be drained prior to exercise.
Further research
Are the effects of specific exercises on IAP that were measured in very selective populations generalisable to the broader groups of patients treated with PD?
Is there a relationship between the sustained IAP associated with exercise and subsequent PD complications, such as leaks and hernia?
Are there specific patient characteristics, such as polycystic kidney disease, high body mass index and core fitness that modify IAP?
2. Specific activities (practice points 2.1–2.5)
2.1. Swimming and water sports
Question from the perspective of the person living with PD:
‘What do I need to consider if I want to go swimming or engage in activities involving water? How do I use a colostomy bag to protect my exit site and catheter? Do clear waterproof dressings or colostomy bags reduce the risk of infection?’
Question rationale:
The presence of a peritoneal catheter presents the question of whether engaging in water sports, in particular swimming, places people receiving PD at increased risk of infection. PD clinicians should provide information on what people receiving PD need to consider prior to water sport activity (such as how to protect the exit site and connection).
Evidence and/or clinical experience
Exit site infections or peritonitis are rarely reported in those who swim. 24
It is a common practice to have appropriate protection for the catheter and exit site during bathing or swimming. 25
European PD centres allow bathing, swimming and saunas. 26
Swimming or saunas can contribute significantly to an improved quality of life in many dialysis patients. 27
85% of Australian PD nurses recommend swimming is safe in private pools with 90% recommending safety in sea water as long as protocols are adhered to. 24
Swimming can be safe in lakes and public pools if strict pre- and post-exit site care are adhered to. 28
Practice points
We suggest that swimming or other water sports should preferably take place in either sea water or swimming pools that are known to be well maintained (private or municipal) to limit exposure to waterborne pathogens.
We suggest avoiding swimming or other water sports in open water directly after storms to limit exposure to water-borne pathogens.
A clear waterproof dressing or colostomy bag to secure and protect the catheter and exit site from getting wet during bathing or swimming, although it remains uncertain as to whether this strategy mitigates the risk of infection.
We recommend that routine exit-site care should be performed after swimming and water sports.
Further research
Is there a significant association between water activities, exit site infection and peritonitis?
2.2. Contact sports and sports requiring vigorous activity
Question from the perspective of the person living with PD:
‘What considerations must be made when participating in active sports?’
Question rationale:
People receiving PD can suffer from social isolation contributing to decreased quality of life. 29 Sports participation can provide people receiving PD with the opportunity to partake in physical activity whilst promoting increased socialisation, enhanced self-efficacy and mental well-being. 30 Given the method of treatment for people receiving PD, considerations must be taken when determining the appropriateness and physical demands of sports.
Evidence and/or clinical experience
Bending, squatting and lifting may increase IAP. Patients should be educated on correct breathing technique when physically exerting. 13
A graded return to participation following illness or surgery is encouraged, with guidance from a relevant exercise professional.
Sports with minimal to no contact (such as lawn bowling, tennis, cycling, golf) are well tolerated and should be encouraged.
Participation in contact sports needs to be evaluated case-by-case based on the potential for trauma to the catheter site, and the physical demands of the sport. 30
Strengthening of abdominal musculature may assist in managing IAP and reducing adverse events. 31
Practice points
Participation or gradual return to sport is recommended for people on PD.
Resumption of physical activity and exercise are discussed with the patient in the post-operative period following appropriate local interdisciplinary collaboration and consultation.
Contact sports where there is risk of physical trauma or repetitive rubbing occur to the catheter site are not recommended.
Sports that require frequent bending, squatting or lifting may be best done without PD fluid in situ. When performing such movements, the following factors which may place participants at increased risk of adverse outcomes should be considered: age, physical capacity, co-morbidities, PD vintage, previous injuries.
Patients may prefer peritoneum to be empty for greater comfort when participating in sports.
The use of a PD belt during sports may provide enhanced protection and comfort to the catheter and catheter site.
Patients may need to temporarily modify or cease their sporting activities if their PD fluid becomes pink or red-tinged, suggesting intra-peritoneal bleeding. Following resolution and, when approved by a relevant professional within the PD care team, activity may resume.
Further research
What are the effects of physical movements (e.g. squatting, bending, lifting, etc.) and breathing technique on IAP?
What is the best way to quantify abdominal strength?
What is the incidence of adverse events related to specific exercise and sports?
What are the experiences of people receiving PD who continue to participate in both contact and non-contact sports?
2.3. Core strength
Question from the perspective of the person living with PD:
‘How do I safely strengthen and maintain my abdominal core muscles when I am on PD?’
Question rationale:
Weak core abdominal muscles may increase hernia risk and place additional strain on the lumbar spine, particularly when accompanied by 1–2 liters of PD fluid in the peritoneal space. Strengthening core muscles may further support the PD dwell and potentially reduce the risk of catheter leaks and hernias.
Evidence and/or clinical experience
A randomized controlled trial examining resistance training in PD patients found the intervention was well tolerated and no adverse effects were reported. 32
Increasing abdominal core strength may reduce the risk of inguinal and umbilical hernia occurrence. 33
The lowest IAP occurs in the supine position. 14
Strengthening abdominal muscles may be safest in the supine position.
Strengthening abdominal muscles can protect and prevent back pain in PD patients. 34
Practice points
Core strengthening exercises are recommended for PD patients.
Stronger core muscles can support the low back and prevent/manage low back pain.
Stronger transverse abdominal muscles can support IAP secondary to PD fluid, potentially reducing hernia risk.
Further research
Are interventions to improve core strength safe and what are their effects?
What is the association of core strength with incidence of hernia, back pain and catheter leaks?
2.4. Work
Question from the perspective of the person living with PD:
‘What activities do I need to consider in my daily job if I am on PD?’
Question rationale:
Continuing to work is vital for individual well-being in addition to financial and social reasons. 26 With the presence of a peritoneal catheter, the question of safety while engaging in manual labour arises. Fitness, strength and ergonomic factors are important considerations while performing manual labour and/or office work with a PD catheter.
Evidence and/or clinical experience
Maintaining normal activities, including the ability to work is critical to the quality of life of patients and their families. 35
The ability to maintain correct posture/position and an activated core while on continuous ambulatory PD is feasible and safe.
Fatigue may affect biomechanics during manual work.
Modified work duties, such as office-based tasks, may be required during times of fatigue.
Practice points
People receiving PD should be encouraged to continue to work.
Clinicians should support patients to liaise with their employer to discuss ways of continuing employment.
Clinicians should discuss if the person’s employing organisation has a corporate wellness program that the person receiving PD could safely participate in.
For those who are primarily sitting at work, intermittent movement should be performed throughout the work shift. For example, perform sit-to-stands, calf raises, arm circles, marching in place every hour.
Functional exercises targeted to work tasks should be performed. For example, those who are performing lifting tasks much of the day, exercises such as targeted stretching, core strengthening, shoulder press, seated/standing row and deadlifts could be recommended.
Further research
Is there a safe weightlifting limit for people on PD (both when empty and full of PD fluid)?
What is the total volume of weight lifted throughout a workday that would be safe to prescribe for a person on PD?
What effect does core strengthening have on the ability to lift safely?
2.5. Sexual activity and sexual dysfunction
Question from the perspective of the person living with PD
‘What do I need to consider regarding sexual activity and intimacy when undertaking peritoneal dialysis?’
Question rationale
The presence of the peritoneal catheter can create a question about safety, timing and type of sexual activity. There may be concerns regarding whether sexual activity can place a person at risk of infection, catheter dislodgement or damage to the peritoneum. There may also be concerns about the impact of PD on sexuality. These questions and concerns are very rarely addressed when education is provided about PD or while the person is undertaking PD.
Evidence and/or clinical experience
Fear of catheter dislodgement and kidney health can impact sexual function. 36
Sexual dysfunction is highly prevalent and strongly impacts quality of life in PD patients.
Each gender has differing variables impacting sexual dysfunction. 37
Fatigue and having no energy can affect sexual function.
Altered menstrual patterns related to kidney failure and PD treatment can affect sexual function.
Altered self-image (catheter, skin conditions) can affect sexual function, and symptoms of uremia (e.g. metallic taste) can affect intimacy.
Practice points
The risk of PD fluid leak or dislodgement during sexual activity (after 4 weeks post insertion of PD catheter) is low.
Factors such as age, blood pressure, medications, iron treatment, glucose levels and lipid levels can contribute to sexual dysfunction and should be assessed.
Pharmaceutical treatment of erectile dysfunction should be discussed on an individual basis.
Fatigue can have a significant impact on sexual activity.
Clinicians should provide education and support regarding how the type of PD (i.e. automated PD versus continuous ambulatory PD) may impact sexual activity.
Patients’ concerns regarding body image, impact of scars, weight changes and safety of sexual activity need to be addressed by clinicians, especially the fear of catheter dislodgement.
Further research
How can clinicians promote the discussion of sexuality and related issues experienced by people receiving PD?
What is the impact of feelings of sexual unattractiveness due to body image changes?
Are there temporal factors that impact sexual activity (e.g. empty or full peritoneum, how long to wait post insertion of PD catheter)?
What is the effect of educational strategies on sexual activity and sexual intimacy?
3. Symptoms/side effects (practice points 3.1–3.6)
3.1. Exit site care
Question from the perspective of the person living with PD
‘How can I minimize the risk of exit site damage related to exercise?’
Question rationale
Exit site complications such as subcutaneous tunnelling, and catheter site infection are common complications of PD. It is not known whether the skin surface defences may be impaired by sweat and microtrauma associated with vigorous activity, thereby increasing the risk of PD exit site complications.
Evidence and/or clinical experience
Before and after exercise, it is essential to inspect the exit site for signs and symptoms of infection or trauma, and to notify the PD unit of any changes in the site.
During exercise, if the exit site becomes soiled or wet, it should be cleaned immediately and covered with a fresh clean dressing. 21
During exercise, the catheter exit site dressings should be changed any time they become soiled or wet. 38
When engaging in moderate to vigorous activity, it is common practice to immobilise the catheter with tape to prevent trauma to the catheter or the exit site. 21
Practice points
We suggest that applying a non-occlusive dressing, such as gauze, may reduce microtrauma to the skin that may result from skin friction during moderate to vigorous activity.
We recommend wearing breathable and freshly laundered clothing during physical activity to decrease sweating near the exit site.
We suggest that, when engaging in moderate to vigorous activity, the catheter should be immobilised with tape to prevent trauma to the catheter or the exit site.
We recommend the exit site should be cleaned immediately after it becomes soiled or wet during exercise.
Further research
What is the effect of exercise on the immune system and on the risk of exit site infection in people requiring PD?
What are the effects of new approaches to catheter stabilisation and catheter placement to minimise friction during activity?
3.2. Perspiration (sweating)
Question from the perspective of the person living with PD
‘Does sweating increase my risk of PD-related infections (peritonitis, PD exit site and tunnel infections)?’
Question rationale
Perspiration has been suggested as a risk factor for peritonitis, and therefore clinicians may advise individuals who are on PD to avoid exercise due to a risk of infection associated with perspiration (especially early after PD catheter placement).
Evidence and/or clinical experience
Evidence in this area is scarce limiting the strength of our practice points.
There is no evidence for higher risk of infection with perspiration.
Exit site care should be prioritized if exercise is performed before the catheter wound is healed. 39
Higher risk of infection with perspiration occurs in obese patients due to skin folds. 40,41
Following physical exertion with sweating, the exit site around the catheter site should be cleansed with antibacterial soap or antiseptic and antibiotic cream to the catheter site. 25
Practice points
Shower and clean the PD exit site shortly after any vigorous exercise that leads to sweating to limit any potential risk of infection risk associated with perspiration.
Individuals who are on PD and are obese may be at higher risk of exit site complications associated with perspiration.
Further research
Are there significantly different peritonitis rates in individuals on PD who exercise compared to those who do not exercise?
3.3. Cardiovascular-compromised individuals
Question from the perspective of the person living with PD
‘I have a bad heart and poor blood vessels, so how can I safely improve my cardiovascular and physical fitness?’
Question rationale
Cardiovascular disease is the leading cause of death in patients receiving PD and causes significant morbidity. However, clinical trials in PD patients and specifically in PD patients with cardiovascular compromise are extremely limited.
Evidence
Systematic reviews showed that regular exercise training, regardless of the modality of exercise, incurred significant beneficial effects on physical fitness, walking and aerobic capacity, cardiovascular dimensions including blood pressure and heart rate, health-related quality of life and some nutritional parameters in adults with CKD. 42 –45
No rigorous studies have reported exercise in cardiovascular compromised participants in the dialysis context.
Practice points
A personalized approach is required for exercise prescription, taking into consideration the severity and nature of heart disease, medical comorbidities and usual functional status.
Personalised short-term and long-term training goals should be clearly defined.
A multidisciplinary approach (nephrologist, nurse, dietitian, local or remote exercise professional) should be adopted in the prescription, monitoring and supervision of exercise.
A graduated approach to exercise prescription with gradual progression in exercise intensity and duration as tolerated should be adopted.
Future research
What is the feasibility, safety and effectiveness of graded exercise programs in people on PD with cardiovascular disease?
3.4. Frailty
Question from the perspective of the person living with PD
‘What exercises can I do to maintain my physical condition, and to better prepare me for a transplant?’
Question rationale
Frailty is characterized by the loss of physiologic reserve across multiple systems, which leads to a vulnerable state. Frailty in people receiving dialysis is associated with higher mortality, falls, hospitalizations, cognitive impairment, loss of independence in activities of daily living and poorer quality of life. 46,47 In kidney transplant recipients, frailty is associated with increased risk of delayed graft function and early hospital readmission. 48,49
Evidence and/or clinical experience
In a 12-week home-based exercise program (aerobic exercise thrice weekly and resistance training twice weekly), improvements in aerobic capacity of PD patients were observed without any adverse effects. 32
Physical function tests, such as Sit-To-Stand (STS), Timed Up and Go (TUG) and pinch-strength tests, can be used to assess independence and quality of life as these relate to important PD activities including being able to stand from a chair, walk 8 feet and connect and disconnect PD catheter exchanges. 50
A personalized, home-based, low-intensity exercise program may improve physical performance and quality of life in patients on dialysis. 51
Regular physical exercise is associated with favourable outcomes for reduction in falls and higher health-related quality of life scale scores including frailty, disability and exhaustion compared with inactive patients. 52
Pre-dialysis education and exercise program can increase uptake of PD and improvements in physical capacity and decrease hospital admissions. 53
Practice points
Physical activity and/or exercise training programs can prevent and reduce physical frailty and reduce the risk of disability.
Use frailty screening tools to identify those who would benefit most from physical activity (e.g. FRAIL scale, Fried phenotype, Clinical Frailty Scale).
Use physical function tests to assess strength and balance (e.g. Short Physical Performance Battery (SPPB), Sit-To-Stand (STS), Timed-Up-and-Go (TUG), gait speed) to identify those with decreased function status/ frailty and those who would benefit from exercise. Cut-off points for predicting development of disability as follows: SPPB score ≤9, STS-5 ≥10 s, TUG ≥9 s.
Use screening tools and physical function tests results to guide exercise prescription
Design exercise programs that fit into each individual’s lifestyle and provide education on the benefit of physical activity in maintaining function, for example, climbing stairs. The PD care team (nephrologist, dialysis nurse, social worker, dietitian) will determine the appropriate assessment lead given the varying resources from centre to centre and country to country
Consider exercises, such as walking or stationary cycling, body weighted resistance exercises and balance exercises, particularly for those at high risk of falls.
Further research
What are the best measures to assess the effect of exercise on frailty?
What is the optimal exercise prescription and the additive benefit of nutrition and/or pharmacological agents in improving frailty?
3.5. Fatigue
Question from the perspective of the person living with PD
‘I feel tired most of the time so how can I exercise?’
Question rationale
Fatigue is a major barrier to exercise participation and, of high importance to people receiving PD in-part due to the implications on quality of life and clinical outcomes. 2,54,55 Exercise has been shown to decrease fatigue experience leading to enhanced overall quality of life in individuals on haemodialysis. Clinicians require strategies to help people continue activity even when fatigued.
Evidence and/or clinical experience
Exercise interventions do not increase the fatigue experience. 55
Physical activity may decrease the severity of the fatigue experience. 56
Both aerobic and resistance exercise are appropriate modalities. 57
A patient program commencing with two 7-minute walks per day at a Borg RPE of 10–12, increasing the variables (time, distance or intensity) of the walks gradually (e.g. 5 minute per walk/week, RPE increase of 1–2/week) found no exacerbation of fatigue experience (clinical experience)
Practice points
Physical activity and exercise may reduce fatigue symptoms and should be encouraged.
Exercise programs should aim to elicit an intensity of Rating of Perceived Exertion (RPE) 11–16 (using the Borg RPE Scale 6–20).
A walking program (outdoor or treadmill) employing a graded approach (i.e. incremental over time) is well tolerated and may decrease fatigue experience and increase an individuals’ ability to perform daily activities.
Further research
What is the effect of exercise and/or physical activity on fatigue experience?
How do different exercise modalities (e.g. cycling, strength training etc.), durations (i.e. dose–response) and intensities (e.g., low versus moderate–vigorous) effect fatigue experience?
3.6. Mental health
Question from the perspective of the person living with PD
‘Would exercise benefit my mental health?’
Question rationale
Mental health, which encompasses a person’s psychological, emotional and social well-being, is an important component of quality of life. Individuals receiving dialysis experience increased mental and emotional distress due to the burden of dialysis. 58 In the general population, exercise has shown to benefit mental health, and there is some evidence for improvement in symptoms of anxiety and depression with exercise in people on haemodialysis.
Evidence and/or clinical experience
There is evidence in the haemodialysis population that both depression and anxiety (including stress and worry), as well as general mental health, are improved with aerobic exercise. 59,60
In the PD population, there is evidence that physical activity in people receiving PD improves general mental health. 55,61,62
Practice point
Regular, moderate-intensity exercise 20–30 minutes, 3–5 times per week is likely to improve or maintain mental health (including symptoms of anxiety and depression).
Screening for depression, recognition of causes and a discussion with the patient should ensue to develop an individualized approach for the treatment of depression.
Further research
What type, duration and intensity of exercise provides the greatest benefit on mental health in people receiving PD?
4. Nutrition and fitness (practice points 4.1–4.3)
4.1. Dietary practice points
Question from the perspective of the person living with PD
‘Is there a role for specific foods or eating strategies to enhance the benefits of exercise? Should I take protein powders to optimize my exercise?’
Question rationale
Health professionals are frequently asked questions about diet from people with kidney disease. Many people undertaking PD may be younger in age and have been familiar with or undertaking regular exercise prior to commencing PD. There is a large evidence base for dietary interventions to support safe exercise and optimise exercise performance in the general population. 63 This creates the question if these recommendations can be applied to people receiving PD for similar benefits.
Evidence and/or clinical experience
There is a paucity of evidence regarding nutrition and dietary interventions to support exercise in people receiving PD.
It is reasonable to apply evidence from high-quality studies and guidelines for nutrition interventions in exercise in the general population with respect to exercise preparation and recovery. (Opinion)
People with diabetes who use insulin should monitor blood glucose levels before and after exercise and treat symptomatic hypoglycaemia. (Opinion)
Practice points
Eat a small meal or snack containing carbohydrate before exercise.
If exercise duration is longer than 60 minutes, additional carbohydrate intake may be required during exercise.
Fluid intake during exercise should match but not exceed sweat losses and be individualized in context with usual urine output and fluid allowance.
It is reasonable to consume 20 grams of high-quality protein immediately after resistance exercise to support protein synthesis.
Sports nutrition supplements, such as protein supplements and sports drinks, should be used under clinical supervision.
Where resources allow, dietitians are a valuable member of the PD team to advise on nutrition and exercise.
Further research
What is the effect of nutrition assessment, counselling, appropriate pre-exercise preparation and nutrition recovery for people receiving PD?
What are the best nutritional strategies to optimize exercise outcomes?
4.2. Obesity
Question from the perspective of the person living with PD
‘What exercises can I do if I am overweight?’
Question rationale
The prevalence of obesity continues to increase in people receiving PD. 64,65 Up to 60% of people receiving PD gain more than 3% of body weight within the first year of PD commencement. 64 Exercise in obese individuals receiving PD can be challenging in the context of additional abdominal and musculoskeletal strain imparted by PD fluid as well as the propensity for ongoing weight gain.
Evidence and/or clinical experience
There is no specific evidence that one type of exercise is superior for people who are overweight/obese and undertaking PD. As such, all types of exercise are to be encouraged.
Adverse effects of exercise have not been rigorously evaluated in randomised trials involving overweight or obese dialysis patients. 42
A graded, individualized approach to exercise is likely to be safe for most individuals. 66
Prescription of exercise to counteract the caloric load of PD may be a worthwhile strategy. 67
Practice points
Physical activity and exercise are safe in obese individuals and should be recommended.
Consider performing vigorous exercise with an empty (PD fluid) peritoneum, as this may: • Improve one’s capacity to exercise. • Mitigate the risk of abdominal and musculoskeletal strain if performing vigorous activities that may increase IAP.
Consider performing low impact exercises such as swimming and cycling, as these may: • Improve one’s capacity to exercise. • Mitigate the risk of musculoskeletal injury.
Exercise is an effective adjunctive therapy for weight management and weight loss, particularly with the supervision of renal dietitian.
Further research
Does exercise with and without intra-abdominal PD fluid have a different effect on cardiovascular, biochemical, fitness and patient-reported outcomes?
Can exercise prescription (based on calorie expenditure) counteract the anticipated weight gain (caloric load from PD exposure) in people receiving PD?
Can progressive resistance training facilitate positive changes in body composition for people undertaking PD compared to aerobic exercise?
What core training can obese PD patients undertake to strengthen core muscles?
4.3. Low baseline fitness levels
Question from the perspective of the person living with PD
‘I want to start exercising but I am not sure at what level or intensity to start’
Question rationale
Beginning an exercise regimen can be challenging for anyone, let alone for individuals receiving PD. The metabolic and physical impacts of the PD fluid can pose unique and significant barriers to exercise performance. Although graded, individualized programs are likely to be most effective, general exercise recommendations according to baseline levels of fitness may improve wider uptake and generalizability.
Evidence and/or clinical experience
Practice points
Any increase in daily physical activity and decrease in sedentary time are likely to have health benefits for people receiving PD.
Individuals should start exercising slowly and at a low intensity to understand their baseline level of fitness and to minimise the risk of injuries.
Incorporate both aerobic (e.g. walking, dancing, seated marching, seated cycling) and resistance (e.g. sit-to-stands, seated row, seated leg curl with resistance bands) activities in a graduated stepwise manner.
Track exercise performance, with a view to steadily increase the frequency, intensity and time spent exercising.
In line with World Health Organization guidelines, individuals with very low activity levels should gradually work towards achieving: • 150–300 minutes of moderate intensity aerobic physical activity (e.g. walking, swimming, dancing) per week OR • 75–150 minutes of vigorous intensity aerobic physical activity (e.g. jogging, competitive sports) per week OR • An equivalent combination of moderate intensity and vigorous intensity aerobic activity AND • 2 or more days per week of muscle-strengthening activities (e.g. push-ups, squats)
Older adults should include activity that focuses on balance and strength training to improve functional capacity and reduces falls risk.
Future research
What are the effects of exercise programs that are tailored according to baseline level of fitness and goals (e.g. functional independence, minimize falls, lifting PD bags) for people receiving PD?
Supplemental Material
Supplemental Material, sj-pdf-1-ptd-10.1177_08968608211055290 - Physical activity and exercise in peritoneal dialysis: International Society for Peritoneal Dialysis and the Global Renal Exercise Network practice recommendations
Supplemental Material, sj-pdf-1-ptd-10.1177_08968608211055290 for Physical activity and exercise in peritoneal dialysis: International Society for Peritoneal Dialysis and the Global Renal Exercise Network practice recommendations by Paul N Bennett, Clara Bohm, Oksana Harasemiw, Leanne Brown, Iwona Gabrys, Dev Jegatheesan, David W Johnson, Kelly Lambert, Courtney J Lightfoot, Jennifer MacRae, Anthony Meade, Kristen Parker, Nicole Scholes-Robertson, Krista Stewart, Brett Tarca, Nancy Verdin, Angela Yee-Moon Wang, Madeleine Warren, Mike West, Deborah Zimmerman, Philip Kam-Tao Li and Stephanie Thompson in Peritoneal Dialysis International
Footnotes
Acknowledgements
This study is endorsed by the Global Renal Exercise (GREX) network and the International Society for Peritoneal Dialysis (ISPD). The interpretation and conclusions contained herein are those of the researchers and do not represent the views of GREX.
Author contributions
All authors were involved in developing the patient questions, conducting the literature reviews, developing the practice points and drafting the manuscript. All authors reviewed and approved the final document.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
(include full name of committee approving the research and if available mention reference number of that approval)
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank Satellite Healthcare, Inc. San Jose, California for their unrestricted grant to support the Global Renal Exercise Network (GREX).
Informed consent to participate
N/A
Informed consent to publish
N/A
N/A
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
