Abstract
Background:
The benefits of automated peritoneal dialysis (APD) have been established, but patient adherence to treatment remains a concern. Remote patient monitoring (RPM) programs are a potential solution; however, the cost implications are not well established. This study modeled, from the payer perspective, expected net costs and clinical consequences of a novel RPM program in Colombia.
Methods:
Amarkov model was used to project costs and clinical outcomes for APD patients with and without RPM. Clinical inputs were directly estimated from Renal Care Services data or taken from the literature. Dialysis costs were estimated from national fees. Inpatient costs were obtained from a recent Colombian study. The model projected overall direct costs and several clinical outcomes. Deterministic and probabilistic sensitivity analyses (DSA and PSA) were also conducted to characterize uncertainty in the results.
Results:
The model projected that the implementation of an RPM program costing US$35 per month in a cohort of 100 APD patients over 1 year would save US$121,233. The model also projected 31 additional months free of complications, 27 fewer hospitalizations, 518 fewer hospitalization days, and 6 fewer peritonitis episodes. In the DSA, results were most sensitive to hospitalization rates and days of hospitalization, but cost savings were robust. The PSA found there was a 91% chance for the RPM program to be cost saving.
Conclusion:
The results of the model suggest that RPM is cost-effective in APD patients which should be verified by a rigorous prospective cost analysis.
Introduction
The benefits of automated peritoneal dialysis (APD) have been established in the medical literature. 1 –3 A concern in APD, however, has been the challenge of maintaining patient adherence to treatments. Past evidence is variable, but studies suggest a third or more of APD patients may end up with unsuitable adherence which increases the risks of complications. 4,5 A potential solution exists in remote patient monitoring (RPM) programs which are designed to give health-care staff more awareness regarding patient adherence and, in fact, there has been some recent evidence of improved adherence with RPM programs. 6,7 Further, recent studies suggest a potential for improved economic and clinical outcomes associated with an RPM program for APD patients. For example, a recent simulation study designed by international clinical experts to reflect likely impacts on utilization in several health systems concluded that remote therapy management programs would lead to lower resource utilization. 8 In addition, a recent observational study in Colombia found an association between the implementation of an RPM program and a reduction in the risk of hospitalization for APD patients. 9 However, currently, there are no published evidence-based economic evaluations of implementing an RPM program.
The purpose of this study was to quantitatively assess the net monetary costs of a recently implemented RPM program to assist with APD therapy in adult patients with chronic kidney disease, incident renal replacement therapy, and who were considered candidates for APD for their initial dialysis modality. This study provides an evidence-based projection of net costs and clinical outcomes to help inform the decision-making process regarding the reimbursement of RPM programs by Colombian payers and the Ministry of Health in Colombia. This study also provides a framework to evaluate similar programs in other settings.
Methods
The intervention of interest was an RPM program designed by the renal care center of Baxter Renal Care Services (RCS) in Colombia referred to here as the APD with the RPM program. The program was implemented in Colombia using a specialized remote monitoring device embedded within APD cycler machines (Home Choice Claria with the platform
A Markov type model was built for the analysis using Excel (Excel 2016; Microsoft, Redmond, Washington, USA) based on five health states: controlled, hospitalization without technique failure, peritonitis without technique failure, technique failure for any reason, and death. The model used monthly cycles to estimate costs and outcomes of a hypothetical cohort of incident APD patients, with and without an RPM program over a 1-year time horizon. The selection and definitions of the relevant health states and the nature of the transitions between health states to best summarize the relevant outcomes were based on the results of a focus group conducted with five RCS experts made up of nephrologists and nurses specialized in nephrology. Following the focus group, two additional nephrologists validated these health states and the model structure.
Figure 1 illustrates the basic structure of the model. The model begins with 100% of patients in the controlled health state after which portions of patients may transition during the monthly cycles to any of the other four health states which are mutually exclusive. Patients who transition to hospitalization without technique failure and to peritonitis without technique failure are assumed to all return to being controlled after one cycle. Death and APD technique failure were modeled as absorbent health states, meaning that after patient’s transition to APD technique failure or to death they remain there. Transition probabilities from the controlled health state to any of the other health states were estimated from the annualized rate of occurrence converted to monthly transition probabilities. 9,10 A time horizon of 1 year was used, given that there was no evidence available showing benefits of the program after the first year and consequently the model did not incorporate discounting.
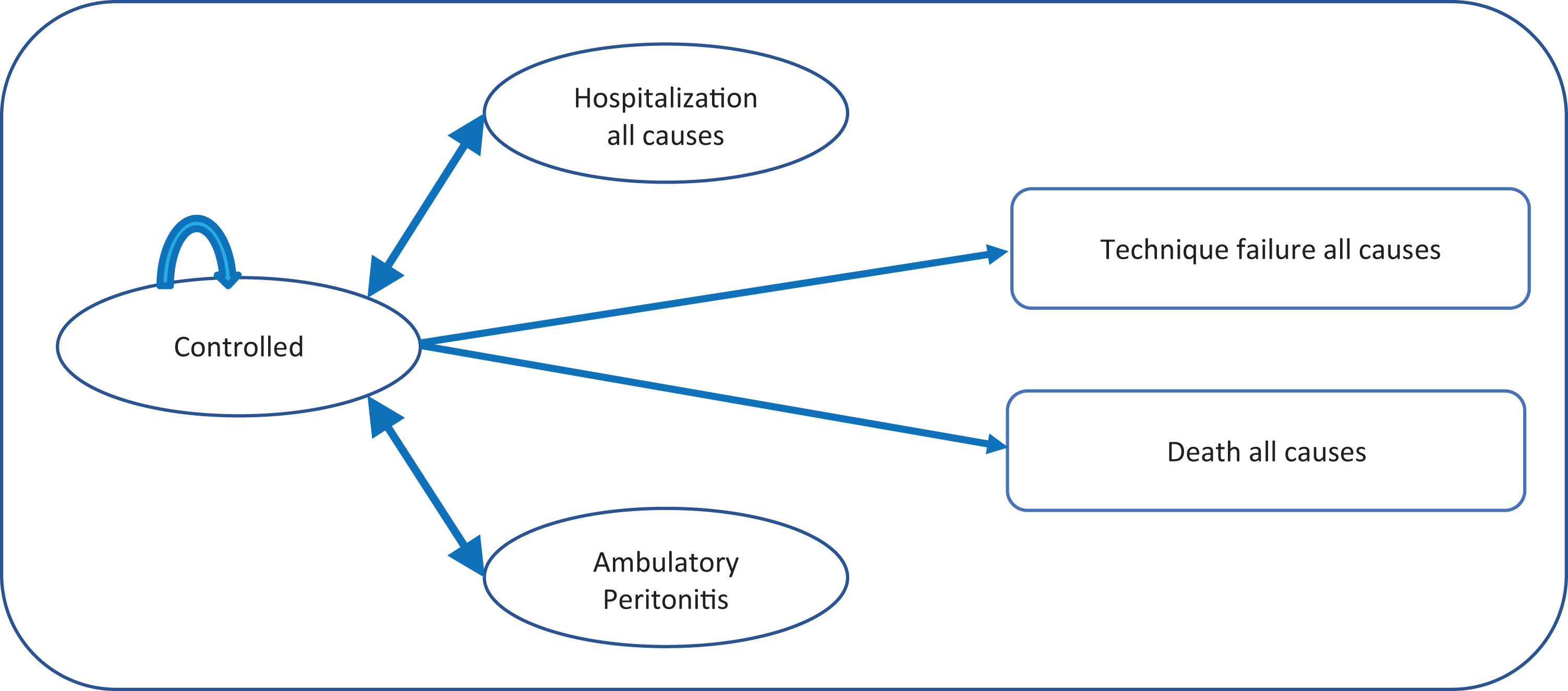
Markov chain model structure.
The model was used to estimate the net annual direct costs of the RPM program from a payer perspective as well as changes in key clinical consequences including time free of complications, hospitalization episodes, days of hospitalization, and episodes of peritonitis. Hospitalization episodes and related days of hospitalization in the model only occur in the health state “hospitalization without technique failure.” Time free of complications is counted as the number of patient months in the health state “controlled.” For ease of interpretation, the model is scaled to represent the outcomes of 100 patients.
Model inputs
The effectiveness of the interventions in reducing the risk of hospitalization and hospitalization days was based on a recent retrospective cohort study conducted in 28 dialysis centers belonging to the RCS network located in six major cities of Colombia. 9 In that study, 63 patients with RPM propensity matched to 63 patients without RPM were found to have significantly fewer hospitalizations per year (incidence rate 0.61) and even more substantially fewer hospital days per year (incidence rate of 0.46). The rate of hospitalization per controlled patient with and without RPM and the average length of stay in patients with and without RPM are used as key inputs in the model. An internal logistic regression type analysis of the same matched sample was also used to inform the incidence of peritonitis with and without RPM in the model. Given the limited evidence, the risk of APD technique failure and mortality were assumed to be equal with and without RPM. Here the rates of technique failure and death were derived based on average rates seen in the aforementioned cohort study and assumed to be equal with and without RPM. 9
Outpatient resources and fees associated with the comprehensive management of patients on APD were estimated from the national pricing guide for medical and surgical procedures in Colombia. 11,12 The cost of the RPM program was based on the monthly fee set by RCS for 2018 in Colombia. Inpatient-day costs of a patient on dialysis were taken from a published abstract of hospital costs based on claims data collected from patients on dialysis in Colombia. 13 Specifically, 752 dialysis hospitalizations were identified from the RCS Disease Management Program and matched to billing data. Hospital costs per episode related to dialysis were Colombian Pesos (COP) 7,088,009 and in that data, the average length of stay was 8.002 days. The cost of outpatient treatment of peritonitis was based on a recent estimate in the literature. 14 Costs associated with switching of dialysis modality due to APD technique failure were estimated internally based on published costs of associated medical and surgical procedures in Colombia. 11,12 Specifically, APD technique failure was assumed to include the cost of switching the patient to hemodialysis, which involves the insertion of a temporary central venous catheter, an arteriovenous fistula surgical procedure, and the need for professional services to support patients switching therapy. The fees, taken from the national fee schedule for medical and surgical procedures in Colombia, were updated to the year 2018 based on adjustments of the current legal minimum wage. Also, adjustment using relative value units was used as a lower bound. For mortality costs, the average length of stay that patients in RCS spend (8.4 days) before dying was used along with the claims-based estimated cost of a hospital day described above for this population. 13 It is however noted that these presumed hospital days were only used to estimate a cost of mortality and were not added to the reported hospital day estimate.
All costs in the model were first estimated in COP and then converted into US$ at an exchange rate of US$1 = COP 2956 based on the average exchange rate for January–December 2018 reported by the Colombian Central Bank. 15
A one-way deterministic sensitivity analysis (DSA), meaning that single inputs in the model were varied one at a time across a set range, was performed to assess the individual effect of parameter uncertainty on each of the model inputs. Uncertainty was also considered using a probabilistic sensitivity analysis (PSA) where distributions were assigned to the inputs and Monte Carlo simulations drawing from those distributions and then running the model were performed. Specifically, 1000 Monte Carlo simulations were run based on beta and log-normal distributions for variables measured as rates, and gamma distributions for those variables measured in terms of costs or amounts of resource utilization. Variables used as inputs for our model, as well as their values, ranges, and distribution parameters applied in the DSA and PSA, are given in Table 1.
Variables, values, and parameters.
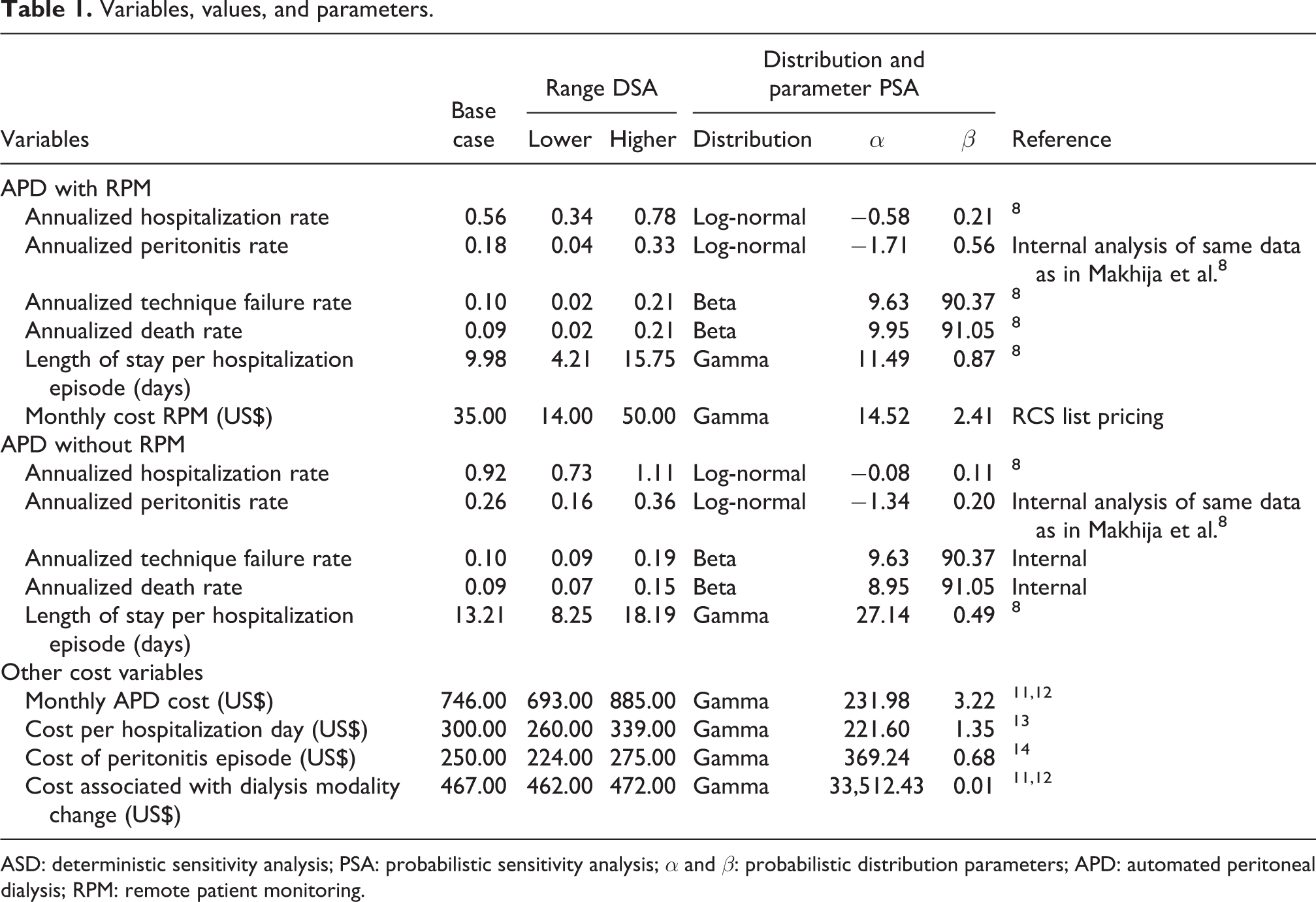
ASD: deterministic sensitivity analysis; PSA: probabilistic sensitivity analysis; α and β: probabilistic distribution parameters; APD: automated peritoneal dialysis; RPM: remote patient monitoring.
Results
Base case results
The base case results reflect the costs and outcomes of a hypothetical cohort of APD patients treated with and without an RPM program analyzed from a payer perspective over a time horizon of 1 year. The results are given in Table 2. Although the transition rates with and without RPM in the model from controlled to technique failure and from controlled to death were the same, the percent of patients that end up after a year in those states is slightly different across the arms. The reason is that patients in the hospitalization state and in the peritonitis state cannot transition to death or treatment failure, and patients are less likely to experience hospitalization and peritonitis in the RPM arm. Also, the total costs of the controlled health state are higher in the RPM arm for two reasons. First, more patients end up in the controlled state in the RPM arm where they accrue dialysis costs and second because the costs of the RPM program itself accrue in that health state. On net, primarily because of reduced hospitalization in the RPM arm, when the RPM program is implemented, the expected total cost of APD was projected to be lower by US$121,233 for every 100 patients, which corresponds to a saving of 9.5% compared to the cost of APD without an RPM program. The model also projected that the implementation of an RPM program would result in fewer episodes of intra- and extra-hospital complications and reduced length of stay.
Base case results per 100 patients per year: cost consequence analysis.
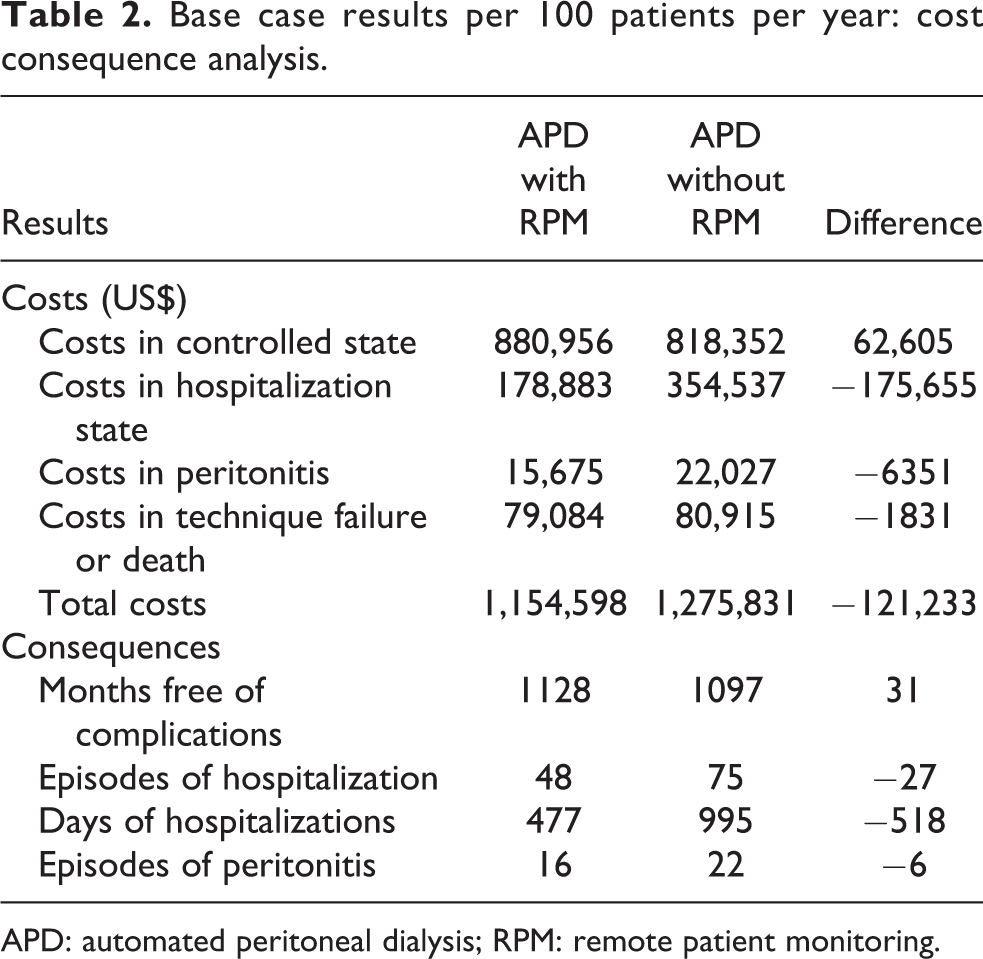
APD: automated peritoneal dialysis; RPM: remote patient monitoring.
Sensitivity analysis
The DSA shows that the cost-saving results of the RPM program in this model were robust (see Table 3). Model results were most sensitive to the change in the hospitalization rate with RPM as well as the length of stay related to hospitalizations, but cost savings remain anticipated over the likely ranges of those variables.
Deterministic sensitivity analysis.
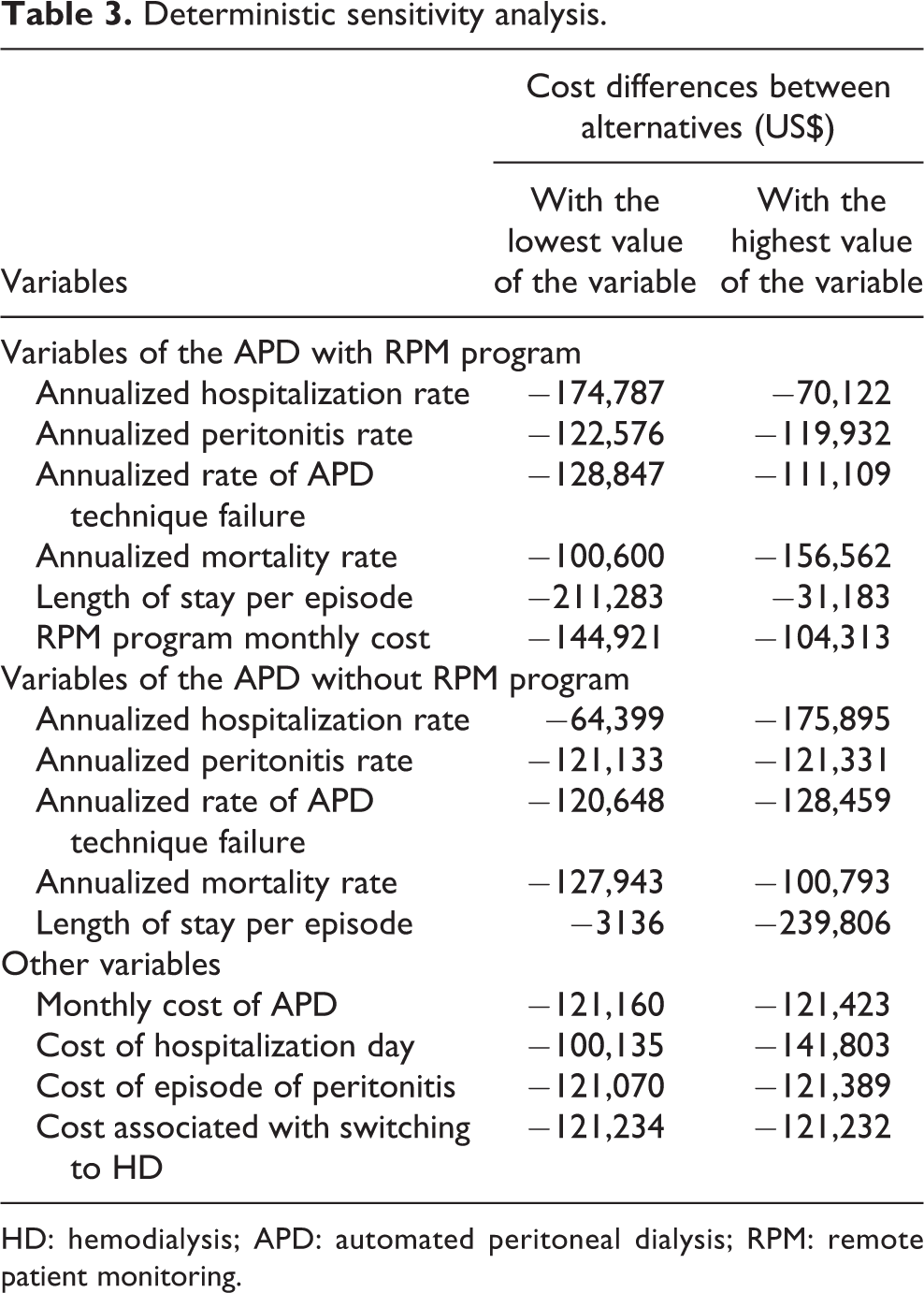
HD: hemodialysis; APD: automated peritoneal dialysis; RPM: remote patient monitoring.
The PSA demonstrated that APD with RPM program at the base case cost of US$35 per month was projected to have cost savings in 91% of the Monte Carlo simulations (see Table 4) and net annual costs of less than US$500 per patient year in 97% of the simulations. Similar results are seen for other potential costs of the program.
Pricing scenarios of RPM and net cost impact.
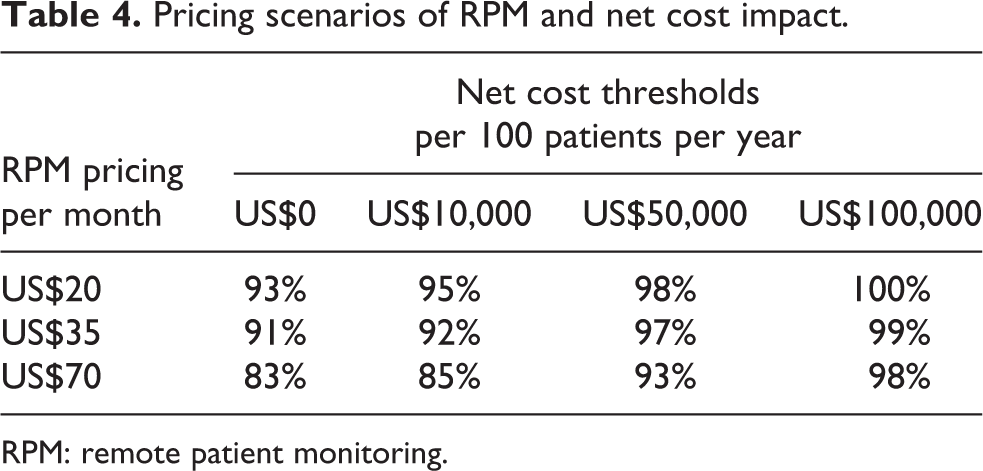
RPM: remote patient monitoring.
Discussion
This study was among the first to provide an evidence-based economic evaluation of an RPM program in APD. The main findings were that implementation of an RPM program in APD incident patients was likely to be a cost-saving intervention compared to traditional APD and that it would be expected to improve patient outcomes. In comparing the model results to the underlying clinical study, there was a greater reduction in hospital days per patient in the clinical study than what is projected by the model even though the model uses the same rate of hospitalization for controlled patients as seen in that study. The reason there is a slightly lower reduction in hospital days projected by the model is that the model includes consideration of treatment failure and death and hence there were less than 100% of patients in the controlled state where the hospitalization rates apply.
Decision makers should consider the results of this model as reassuring evidence in making additional investments for RPM in the Colombian health system. However, there are several limitations to keep in mind. Since this was an RPM program based on a very recently developed remote monitoring device, clinical evidence with high internal validity (e.g. randomized clinical trials) was not available. The association between the RPM program and the key clinical effect of reduced hospitalizations used in this model was instead based on observational evidence. While the study that provided the hospitalization results did include adjustment by propensity score which was designed to control for selection bias, there is still a possibility of confounding by unobserved variables as well as other inherent limitations of analyses based on observational data. Further, the sensitivity analysis showed that this model is particularly sensitive to the risk of hospitalization and length of hospital stay associated with the RPM. Clearly, it would be beneficial to continue developing scientific evidence with improved statistical power for hospitalization-related outcomes. Nonetheless, since the economic benefit of the RPM is directly related to the positive effect that an RPM program may have on the risk of hospitalization, the potential economic impact of the implementation of an RPM program is likely to be more positive in renal centers experiencing less control over their APD patients. Similarly, for those health-care systems with a higher cost per hospital day, the RPM program would have a better chance of being a low-cost and/or cost-saving alternative.
Uncertainty in other outcomes considered in this model, such as risk of peritonitis, risk of technique failure, and mortality, did not affect the conclusions as could be seen in the sensitivity analysis. Hence, potential uncertainty in those inputs is less of a concern and it is also unlikely that the potential for savings in the use of RPM would be related to those characteristics.
The model makes several conservative assumptions including equal rates of mortality and technique failure and limiting the time horizon of the analysis to 1 year. Hence, though the extent is not known, it is possible that the long-term effects of the RPM program were underestimated by the model. Here again, future research on long-run outcomes associated with remote monitoring would be valuable.
This model also makes other standard assumptions of Markov models including that the transition probabilities do not have any memory and they remain constant across the cycles. The model makes a further simplifying assumption that patients experiencing hospitalization or peritonitis without treatment failure return to being controlled for at least one cycle. However, relaxing that assumption would add conditional transitions where there are no data and would be unlikely to change the conclusions. Furthermore, the available evidence does not allow the potential for individual patient characteristics to modify outcomes. Future evidence may show important conditional transition probabilities and/or predictive patient characteristics for consideration.
The structure of this model was built based on the available evidence and conceptualized by clinical experts trying to characterize, in a simplified manner, the natural history of the disease as well as clinically relevant outcomes where the RPM program could have a significant effect. In this respect, it could be expected that the modeling structure could be adopted as a framework for economic assessments in other contexts. On the other hand, the RPM program and the clinical evidence considered here are confined to the context of the very specialized renal model, costs structure, and clinical practice of Colombia. Therefore, any generalizing of these findings to other contexts should be done with caution. Finally, since this is a remote monitoring system, clinical outcomes will depend largely on specifics of its use, learning curves of the users, and ultimately on the quality of the decisions taken by the medical staff using the system. Overall, the results of the model should be viewed as motivation for a rigorous prospective cost analysis to confirm the main findings.
Conclusion
From a payer perspective and with a time horizon of 1 year, the model suggests that the RPM program implemented in Colombia has a strong potential to be a cost-saving intervention, a finding that proved to be robust in the sensitivity analyses. Rigorous prospective cost analyses of RPM versus standard of care in real-world settings that utilize APD should be conducted to confirm the findings of our model.
Supplemental material
Supplemental Material, PTD896880_supp_mat - Evaluating a remote patient monitoring program for automated peritoneal dialysis
Supplemental Material, PTD896880_supp_mat for Evaluating a remote patient monitoring program for automated peritoneal dialysis by Juan G Ariza, Surrey M Walton, Mauricio Sanabria, Alfonso Bunch, Jasmin Vesga and Angela Rivera in Peritoneal Dialysis International
Footnotes
Acknowledgments
We acknowledge Baxter Renal Care Services Colombia and Baxter Healthcare Corporation for the financial support provided for the project.
Author contributions
JA, SW, MS, and AB made substantial contributions to the design of the study. JA, MS, and AB led the data analysis and primary construction of the model. All of the authors contributed to the interpretation of the data as well as drafting and revising the manuscript for clarity and scientific accuracy. All of the authors have met the journal’s requirements for authorship, and all authors agree to be accountable for all aspects of the work. All of the authors approved the final submitted draft of the manuscript.
Declaration of conflicting interests
The author(s) declared following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Baxter Healthcare Corporation, Deerfield, IL, USA, provided funding to SW to assist with the model design and support the preparation of the manuscript. JA is an employee of Baxter Latin America, Bogotá, DC, Colombia. MS and AB are employees of BRCS Latin America, Bogotá, DC, Colombia. JV is an employee of BRCS Colombia, Bogotá, DC, Colombia. AR is an employee of Baxter Healthcare Corporation, Deerfield, IL, USA. None of the authors except SW received funding related to the development of the manuscript.
Ethical approval
Ethical approval was not sought for the present study because it did not involve human subjects. This study was completed in accordance with the Helsinki Declaration as revised in 2013.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received the financial support of Baxter Renal Care Services (BRCS) Colombia, an independent entity owned by Baxter International Inc.
Informed consent to participate
Informed consent was not sought for the present study because it did not involve human subjects.
Informed consent to publish
Informed consent was not sought for the present study because it did not involve human subjects.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
