Abstract
There are a number of misconceptions around the identified early survival benefit of peritoneal dialysis (PD) relative to hemodialysis (HD), including that such benefits “even out in the end” since the relative risk of death over time eventually encompasses 1.0 (or even an estimate that is unfavorable to PD); that the early benefit is, in fact, most likely due to unmeasured confounding; and such benefits are only due to the influence of central venous catheters and “crash starters” in the HD group. In fact, the early survival benefit results in a substantial gain of patient life years in PD cohorts relative to HD ones, even if it the benefit appears to “even out in the end,” is relatively insensitive to unmeasured confounding, and persists even when the effects of central venous catheters are accounted for. In this review, the calculations and arguments are made to support these tenets. Survival on dialysis is still one of the most important considerations for all stakeholders in the end-stage kidney disease community, including patients who rank it among their top priorities. Shared decision-making is a fundamental patient right and requires both balanced information and an iterative mechanism for a consensual decision based on shared understanding and purpose. A cornerstone of this process should be an explicit discussion of the early survival benefit of PD relative to HD.
Introduction
The comparatively better survival of peritoneal dialysis (PD) patients relative to hemodialysis (HD) patients in the first year of dialysis was first described 40 years ago. 1 This early survival benefit has also been evident in the majority of studies in the more modern era, as will be shown below. The nature of this benefit, however, is widely misunderstood by stakeholders in the dialysis community. The main misconceptions include that such benefits “even out in the end” with no net gain in survival since the relative risk of death over time eventually encompasses 1.0 (or even an estimate that is unfavorable to PD); that the early benefit is, in fact, most likely due to unmeasured confounding; and such benefits are only due to the influence of central venous catheters and “crash starters” in the HD group. These tenets are, in the main, incorrect. In this narrative, I will address these issues and suggest clinical implications for care, based on the general position that the relationship between modality and early survival is both real and likely to be causal.
Why early, beneficial hazard ratios do not “even out in the end” if the hazard ratio subsequently encompasses 1.0
In medical research, comparing the relative risk of death between two treatments involves the calculation of a ratio: a rate ratio, an odds ratio, or a hazard ratio (HR) depending on the type of statistical test used for the computation. A Poisson model compares the frequency (rate ratio), a logistic model the odds (odds ratio), and a Cox model the hazard (HR) of the event of interest, here being death. All of these ratios are roughly comparable when they are used for large samples and when the outcome is common (e.g. death in dialysis populations). By far, the most common method used for comparing the risk of death in epidemiological research is the Cox model. For all practical purposes, the “hazards” calculated by the Cox model can be thought of as incidence rates, and thus, the HR can be roughly interpreted as the incidence rate ratio. 2
So, as we all should know, an HR for of 1.2 for death between treatment A and treatment B at time T indicates the following: there are 20% more deaths on treatment A and treatment B in those followed up to time T. However, this interpretation assumes that the HR is constant over time (“proportional hazards”). In reality, the HR at time T ignores the distribution of events before (and obviously after) time T, and should be regarded as an “instant” ratio through to and calculated at the time of follow-up. 2 If the HR has not been constant at each point up until time T, the HR at that time is certainly not interpretable as an “average” effect over the period of observation, and at best can be regarded as a smoothed estimate.
Using an example quoted in a seminal article by Hernán et al., 2 consider the Women’s Health Initiative, a randomized clinical trial that compared the risk of coronary heart disease of women allocated to combine hormone therapy versus placebo. 3 This mean follow-up time in the study was 5.2 years. The primary result from the trial was expressed as the HR for coronary artery disease—“Combined hormone therapy was associated with a hazard ratio of 1.24.” However, the HRs varied markedly depending on duration of follow-up: 1.81, 1.34, 1.27, 1.25, 1.45, and 0.70 for follow-up to years 1, 2, 3, 4, 5, and 6 or more, respectively. The 24% increase in the rate of coronary heart disease that many consider as the definitive effect of combined hormone therapy arises from the decision of investigators to define follow-up as being 5.2 years. As pointed out by Hernan in his paper, a trial with a shorter follow-up would have reported an 80% increase, whereas a longer trial would have reported little or no increase at all. 4
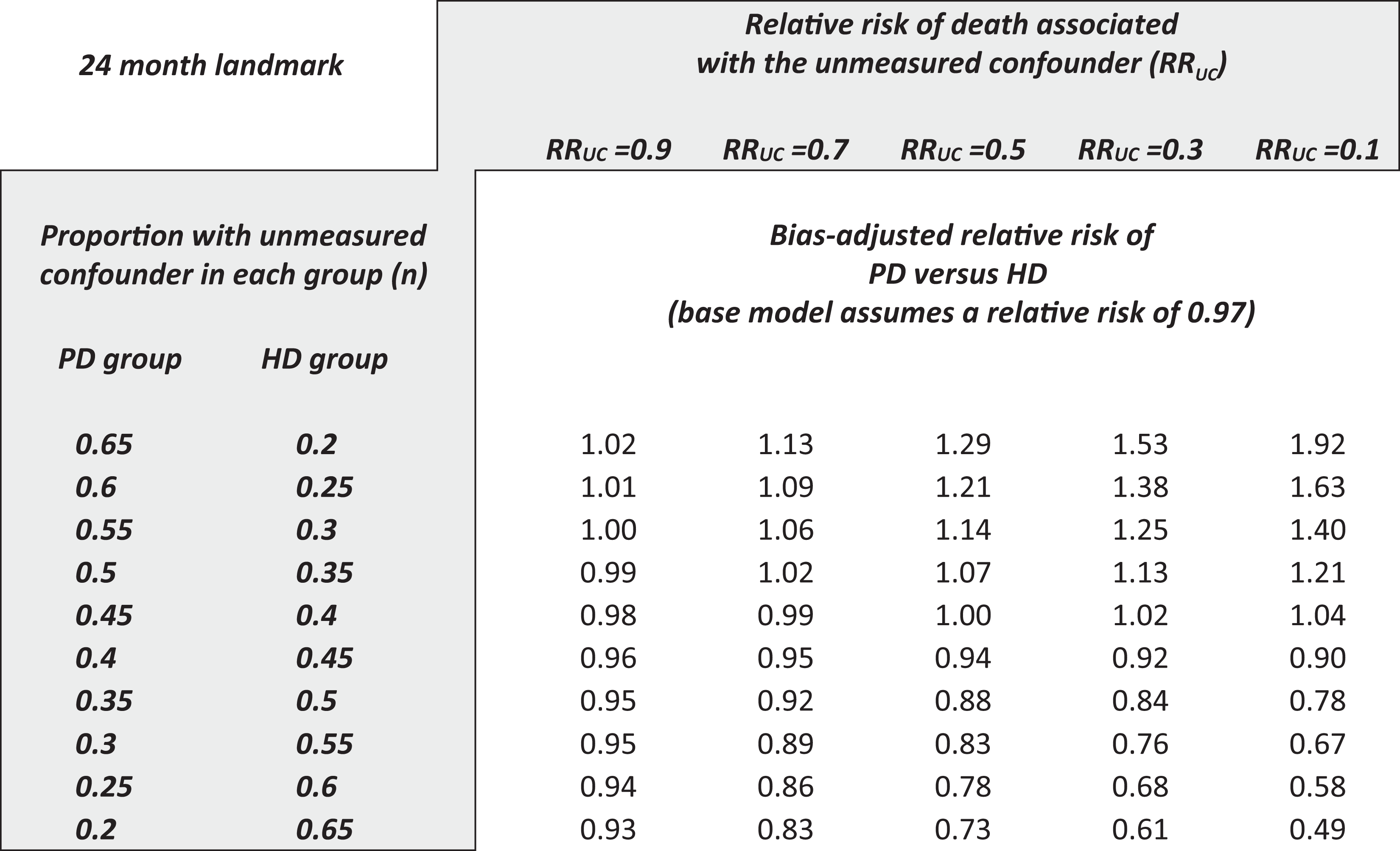
How then we should then interpret time-varying HRs, if they appear to “even out” in the end? Consider another example, namely the survival of general populations in Liberia and the United States. The survival of these populations is well defined in life tables from the World Health Organization (https://www.who.int/gho/mortality_burden_disease/life_tables/life_tables/en/). As shown in Figure 1, the rate of death in Liberia is 11.1-fold higher than that in the United States during childhood but is only marginally higher at complete follow-up. There is, therefore, an early survival benefit in the United States, although mortality risk appears to mostly “even out” by the end of follow-up. The reason for this, of course, is that most people are now dead in both countries. At any one point in time, however, many more patients are alive in the United States compared to Liberia. The early survival benefit in the United States means that more patients live longer in that country, despite the apparent “evening out” at long-term follow-up.
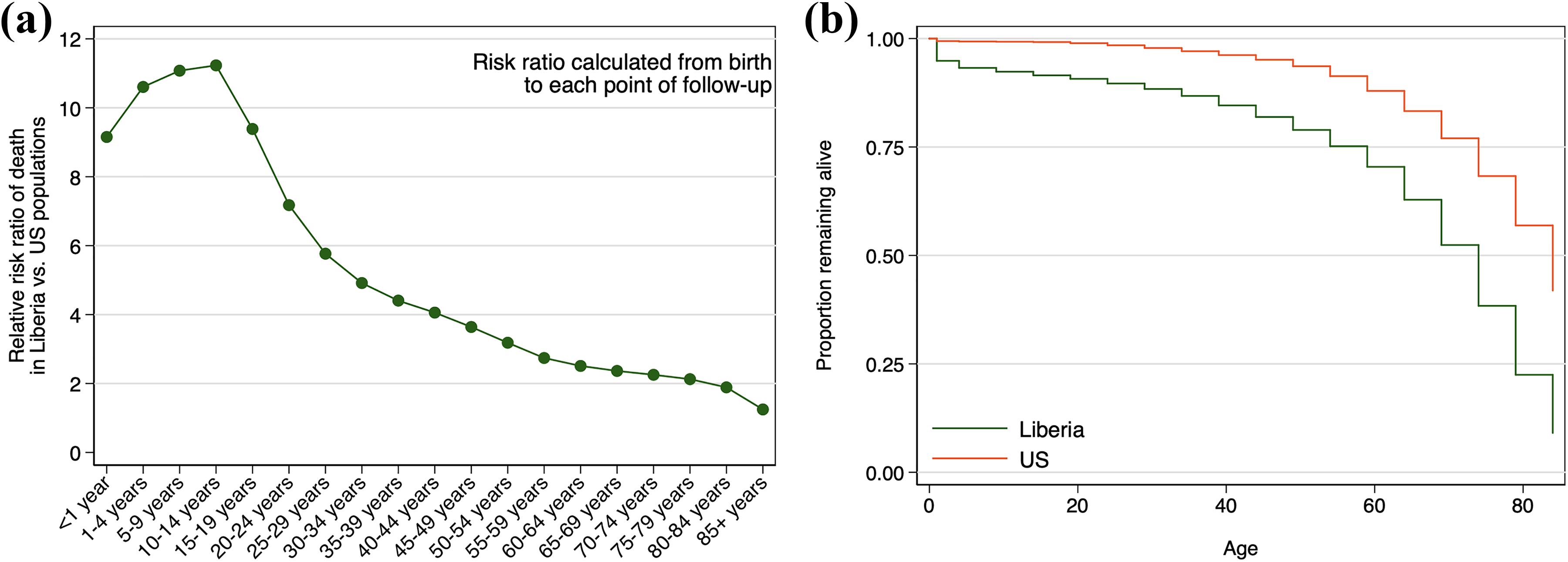
(a) The ratio of death rates between Liberia to the United States appear to “even out” in the end, (b) but there is still much better survival of people in the United States (right).
This interpretation also applies to the comparison of PD versus HD. As an example, consider a hypothetical scenario of 1000 patients starting PD and 1000 identical ones starting HD, as modeled in Figure 2 (see Stata code in Online Supplementary Material). In this example, the annual mortality is set at an unrealistically high rate for the purposes of illustration, and there is no dropout or loss to follow-up. The mortality risk with PD has been simulated such that it is lower than that with HD initially, but higher later on. Note, however, that HR for death on PD versus HD over the entire period is equal to 1.00 (95% confidence intervals (CIs) 0.91, 1.10). However, despite the equal hazards at 5 years, the typical longevity of PD patients is 2.35 years, as opposed to 1.83 years for those on HD. A greater number of patients in the cohort benefit from the early survival advantage of PD, and a lower number are harmed by the late survival disadvantage. The net effect is patient life years saved with PD compared to HD, despite the relative risk of death that appears to “even out” in the end.
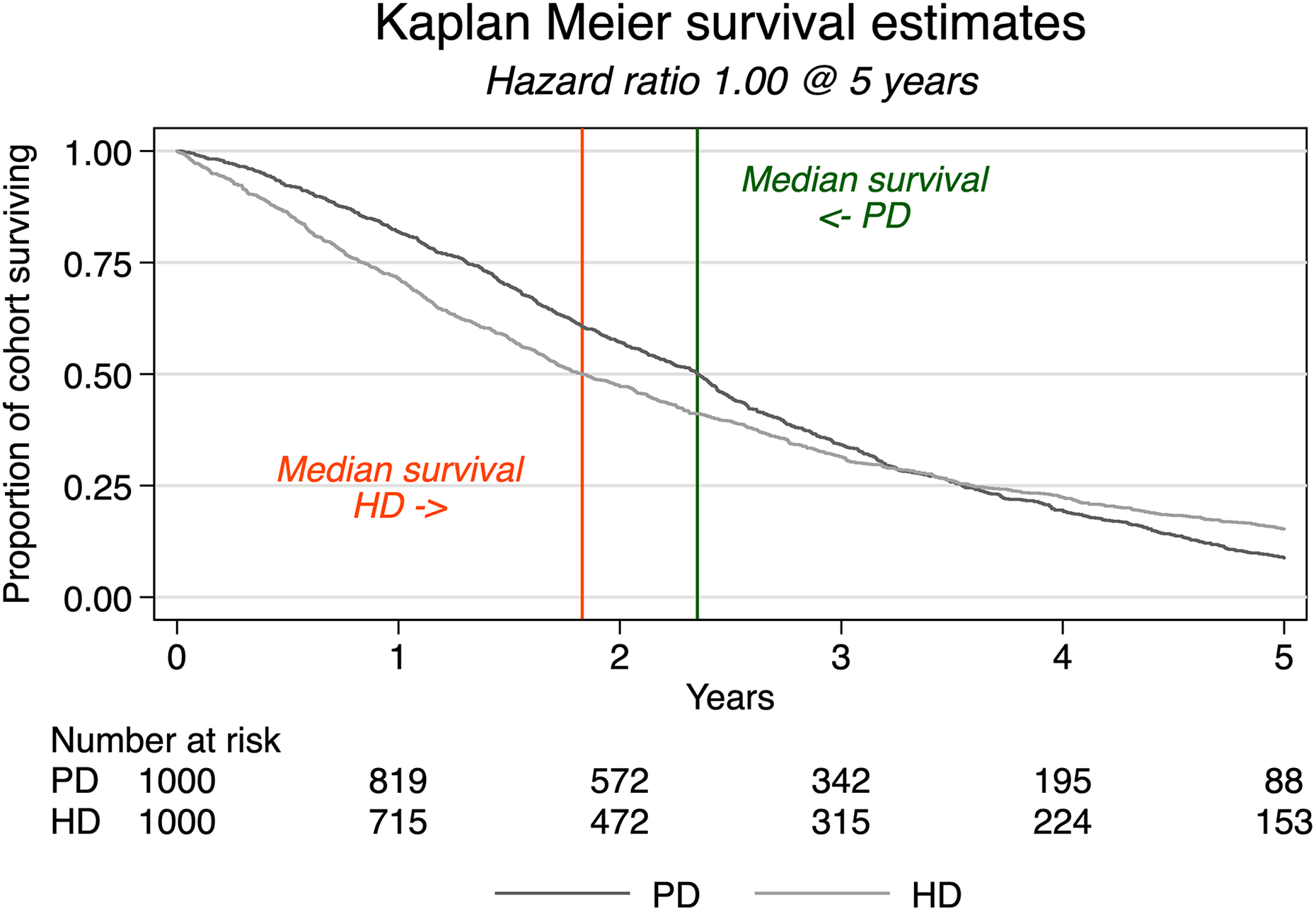
In this simulation, the hazard rate for PD is an increasing function over time with a lower hazard rate early in follow-up compared to HD and eventually crossing over to be higher than the HD hazard rate, thereby resulting in a time-dependent hazard ratio. When analyzed as a proportional hazards model, the overall hazard ratio is estimated to be 1.00 contrary to the true underlying time-dependent hazard ratios. Restricted to a follow-up of 5 years, longevity is greater in the PD group. PD: peritoneal dialysis; HD: hemodialysis
Finally, let us examine a real-world example, using the study by Kumar et al. 5 This study is notable for several reasons. First, it can be regarded as one of the most methodologically rigorous analyses comparing mortality risk with PD versus HD and used a methodology that computes a cumulative HR, not (as is usual) the “instant” HR. Second, it makes adjustments for not only typical demographic and clinical confounders but also neighborhood family income and neighborhood education level. Third, the study demonstrates convergent validity using both “intention to treat (ITT)” and “as treated (AT)” frameworks. 6 Finally, the sampling frame excluded any HD who initiated dialysis urgently with a catheter in an effort to reduce case-mix bias. In this study, the cumulative survival favors PD over HD for the first 2–3 years, with little in the way of significant difference in adjusted survival thereafter (Figure 3). In Table 1, the marginal gain in life years for the PD cohort relative to the HD cohort is presented when follow-up of this cohort is restricted to 1, 2, 3, 4, and finally, 5 years. As can be seen, in the AT analysis, the average life expectancy of a PD patient is approximately 0.14 years longer at the 5-year landmark than that of a corresponding HD patient. Once again, despite the survival probabilities appearing to “even out” in the end, there is a net gain in patient life years saved from the early survival benefit on PD in this study.
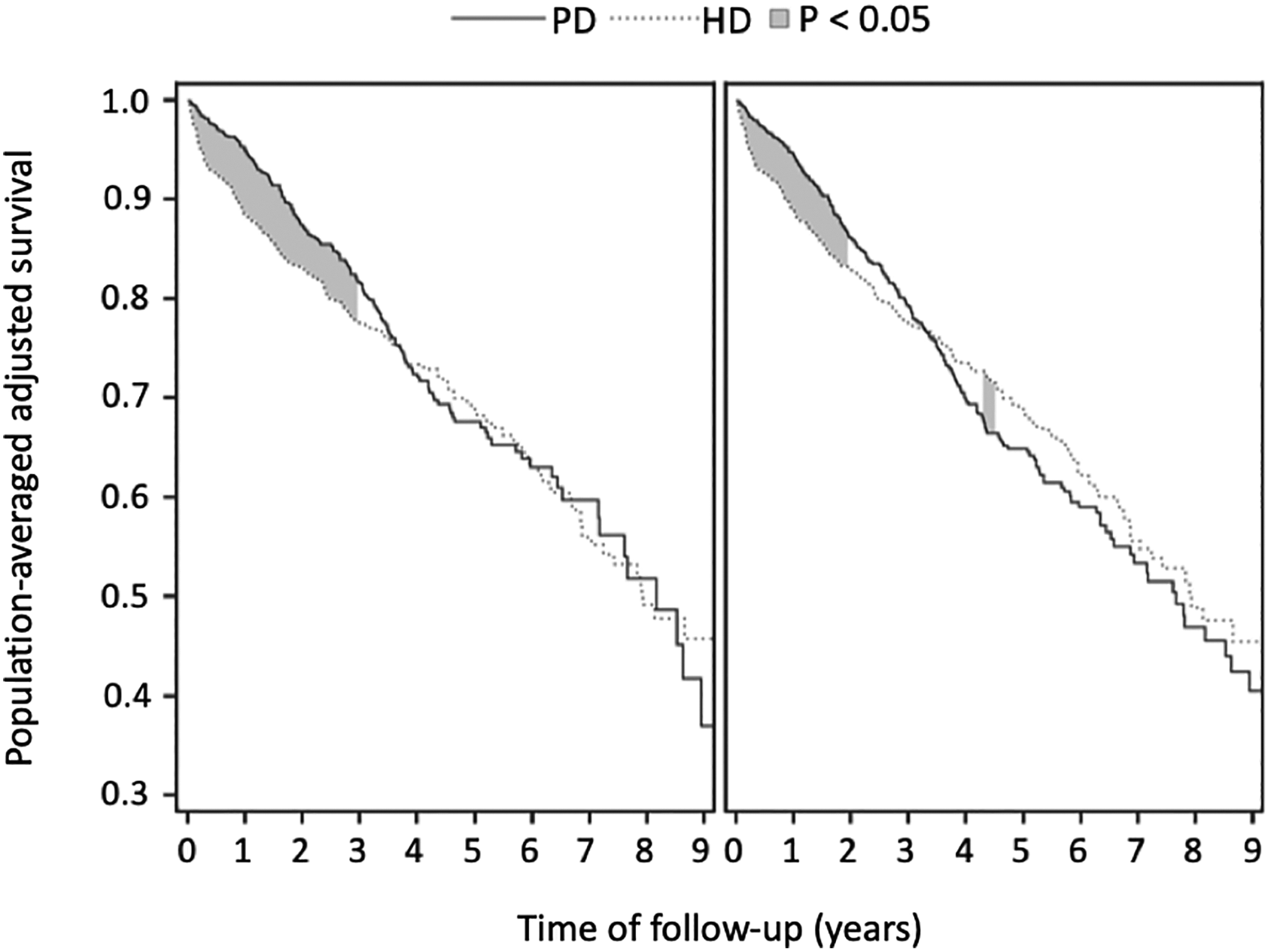
Population-averaged adjusted patient survival by modality, reproduced with permission from Kumar et al. 5 The shaded areas reflect where the pointwise differences in the two survival curves differ. On the left is the as-treated analysis, and on the right is the intention-to-treat analysis.
Marginal, covariate adjusted, survival estimates from Kumar et al. 5 in “as-treated” (AT) and “intention-to-treat” (ITT) analyses, respectively, and their difference (absolute risk reduction) and RSMT through 5 years and the corresponding life years gained (differences in RMST) through the first 5 years, between PD and HD cohorts (courtesy of Edward F Vonesh and Victoria A Kumar, personal communication, 27 June 2019).
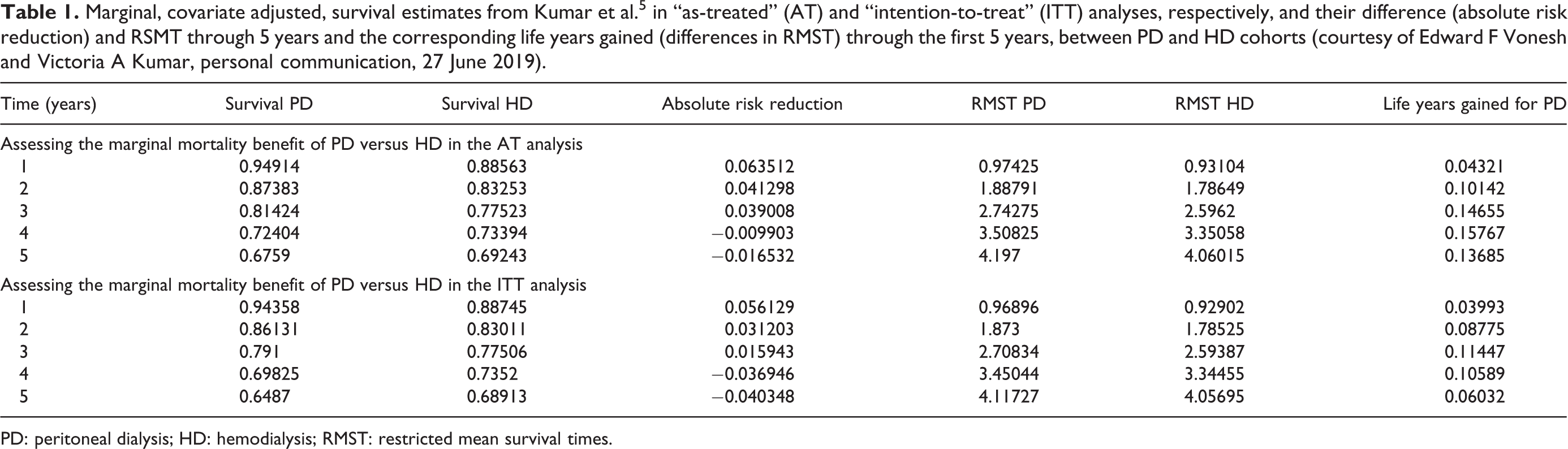
PD: peritoneal dialysis; HD: hemodialysis; RMST: restricted mean survival times.
In short, there is a disconnect between typical survival time and the HR from conventional Cox models when comparing PD versus HD because of the time-varying HR. The single number that is generated from such models over the entire period of observation does not provide an accurate reflection of an “average” effect. Should such a number be required, a valid estimate can be generated using more complicated (and seldom performed) computations, for instance, discrete-time hazard models 7,8 or cumulative HRs, as developed by Vonesh, and used in a number of papers (including Kumar et al. above) that generate adjusted population-averaged survival curves. 5,9,10 In the literature at large, however, the majority of computations report standard HRs using conventional Cox models. These estimates must be interpreted correctly.
Why early benefit from PD versus HD is unlikely to be explained in entirety by unmeasured confounding
When discussing the early survival benefit of PD versus HD, the question of causality always arises. Is this effect of modality on mortality an actual one or merely an association that is apparent because we cannot adjust for all important confounders? This is a key question. Under what must be currently considered a strong assumption of causality, there are potentially hundreds of thousands of life-years to be gained from greater exposure to a lower death rate early after dialysis inception, given the millions of dialysis patients on the planet today. It behooves us to understand if this is a real opportunity or not.
In assessing whether this early survival benefit may be an artifact of residual confounding, there are two approaches to be taken. The first is to triangulate finding with those in randomized controlled trials (RCTs). Of course, it should be remembered that RCTs are not always a better reflection of reality that observational studies, despite their well-accepted higher ranking in the evidence hierarchy. 11 Randomized clinical trials are known to systematically underrepresent older, frailer, and less well-educated patients, who are often not considered for participation. 12 –18 The results of RCTs are therefore often difficult to generalize to unselected populations. Such is the case with the largest trial ever performed in dialysis patients, yielding an unreasonably young and healthy patient sample, with results that are of questionable relevance to the “real world.” 19 Notwithstanding, clinical trials are a useful tool with which to assess causality (can the intervention work), before such steps are taken to show clinical effectiveness (does it work).
The second approach is to estimate the extent to which bias due to unmeasured confounding may have over- or underestimated the true effect. In this technique, the prevalence of a hypothetical unmeasured confounder is varied among patients in the reference and comparator groups as well as the strength of association between the hypothetical unmeasured confounder and the outcome of interest (here being mortality risk). The models are created to reflect plausible scenarios in which unmeasured confounding might be at play. The resulting impact on the baseline relative risk ratio (RR) can be computed in a number of ways but is most easily modeled using bias formulas. 20 –24 Such modeling shows what sort of unmeasured confounder is necessary to abrogate the association of interest, in terms of both the distribution of the confounder between groups and its strength of association with the outcome (e.g. see literature 25 –27 ). If such an unmeasured confounder may plausibly exist, then the association of interest is deemed sensitive to unmeasured confounding. If not, then the measured association can be considered as being closer to a causal one.
So, using these two approaches, how robust is the tenet of an early survival benefit of PD versus HD? As a first step, it is appropriate to quantify the extent of this benefit from cumulative clinical experience. There is no adequate synthesis of evidence in the literature, although an attempt is planned.
28
It is difficult to meta-analyze studies comparing PD and HD due to the heterogeneity of data, differences and deficiencies concerning various computations within articles, and often variable and patchy reporting. To provide some substance for further discussion, however, an informal meta-analysis of the literature is presented below. In this analysis, the following studies were included: Only studies published from 1997, to exclude studies based on obsolete practices. Only studies published up to 18 October 2018, when the literature search was completed. Excluding combination dialysis, where PD is augmented by one to two sessions of HD per week
29
Where possible, including publications that excluded deaths up to 90 days (eliminating the interim or short-term HD patients who have very high mortality due to factors independent of dialysis modality). Where possible, including publications and results where there has been some statistical adjustment for confounding factors. Publications not using any sort of statistical adjustment were not included in the synthesis unless they were also considered in previous in-depth narrative reviews or published health technology assessments.
30
–33
Where relative RR estimates were reported for different eras, including only those pertaining to the most recent data (e.g. literature
10,34,35
) Where relative RR estimates were reported only visually, digitizing figures using Web Digitizer 2.6.6
36
to include numerical data. Where relative risks were reported as a ratio using PD as the reference group, using the inverse of the ratio in meta-analyses to ensure correct modeling of the effect of PD relative to HD. Where possible, seperating relative RR estimatesderived from the ITT framework (“did exposure that the patient initially received affect mortality, irrespective of subsequent changes that occurred along the way?”) from those derived from the AT framework (“did the exposure that the patient actually received affect mortality?”).
6,37
The initial pooled data include patients from 38 countries, and >150,000 PD and >1 million HD patients, and were analyzed in such a way to determine “instant” relative RR up to the landmarks of 6 months, 1 year, and 2 years after dialysis inception. 5,10,34,35,38 –75
Importantly, not all of the literature reported on whether an early survival benefit was present, or had available data for extraction. 41,43 –45,50,52,54,57,62,63,65,70,72,76 Such studies were excluded from the analysis. Also, an early survival benefit was sought but not found in a smaller number of studies and patients. 10,56,61,65 –67,71,73 Where estimates were available from these negative studies, these were included in the meta-analysis. 10,65,71 Of note, most of these “negative” studies had some degree of nonproportionality on visual inspection of PD versus HD survival curves, indicating different HRs at different follow-up landmarks. However, this nonproportionality was either not formally tested for statistical significance, 56,65 or was statistically nonsignificant on formal testing, 10,67 or was demonstrative of survival that was equal between PD and HD early on but worse with PD at a later vintage. 71 Of the three studies that demonstrated no nonproportionality at all, one was underpowered, 66 and one had an extraordinary sampling frame creating an artificially well-cared-for cohort that excluded 80% of patients. 61 The remaining study has no particular explanation, although it is interesting to note the higher use of diuretics in HD patients than PD patients, allowing speculation that residual renal volume (but not necessarily function) might be unusually high in the HD group, contrary to customary findings in the literature. 73
The final pooled data for the meta-analysis included 811,319 patients from 18 countries. Extraction of data measurements was performed in adherence of standard operating procedures in the Cochrane Handbook for Interventions in Systematic Reviews (https://training.cochrane.org/handbook), and results were expressed as relative RR with 95% confidence intervals (CIs). 77 Data were meta-analyzed with RevMan version 5.3 using random-effects models. For RCTs, the Mantel–Haenszel relative RR was computed from reported numbers of deaths. For observational studies, the DerSimonian and Laird inverse variance method was used to calculated relative RRs from natural logarithms of adjusted odd ratios or HRs (and their standard errors). 78 The details of the meta-analysis are shown in Figure 4 and summarized in Figure 5. Overall, there is a notable survival benefit in the first year of dialysis in these studies, which often extends up to 2 or more years.
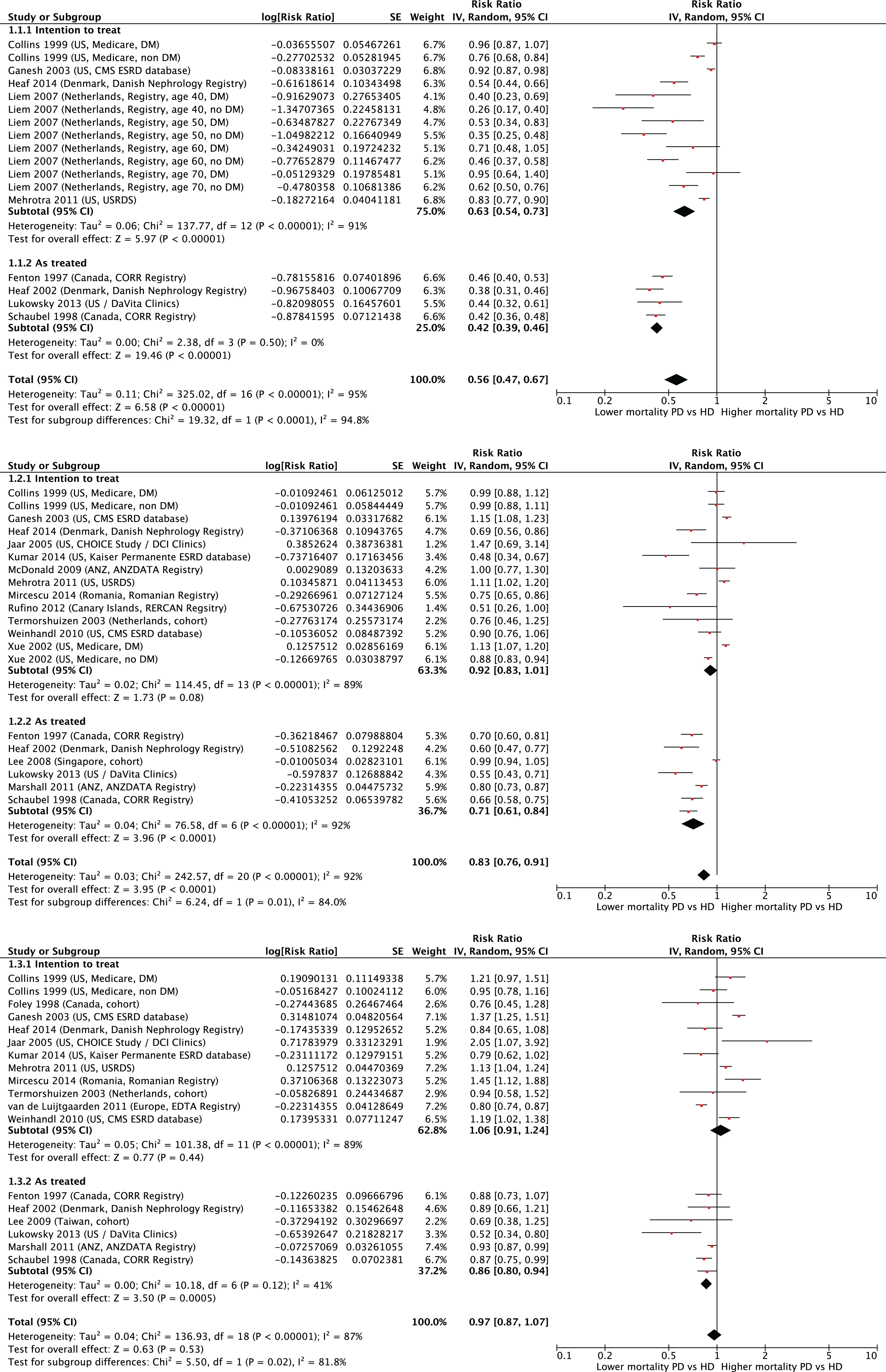
Forest plots from the meta-analysis of cohort studies comparing risk of death in those treated with HD versus PD, summarized for landmarks of 6 months (top panel), 1 year (middle panel), and 2 years follow-up (lower panel), subgrouped according to the intention-to-treat and as-treated analytical frameworks.
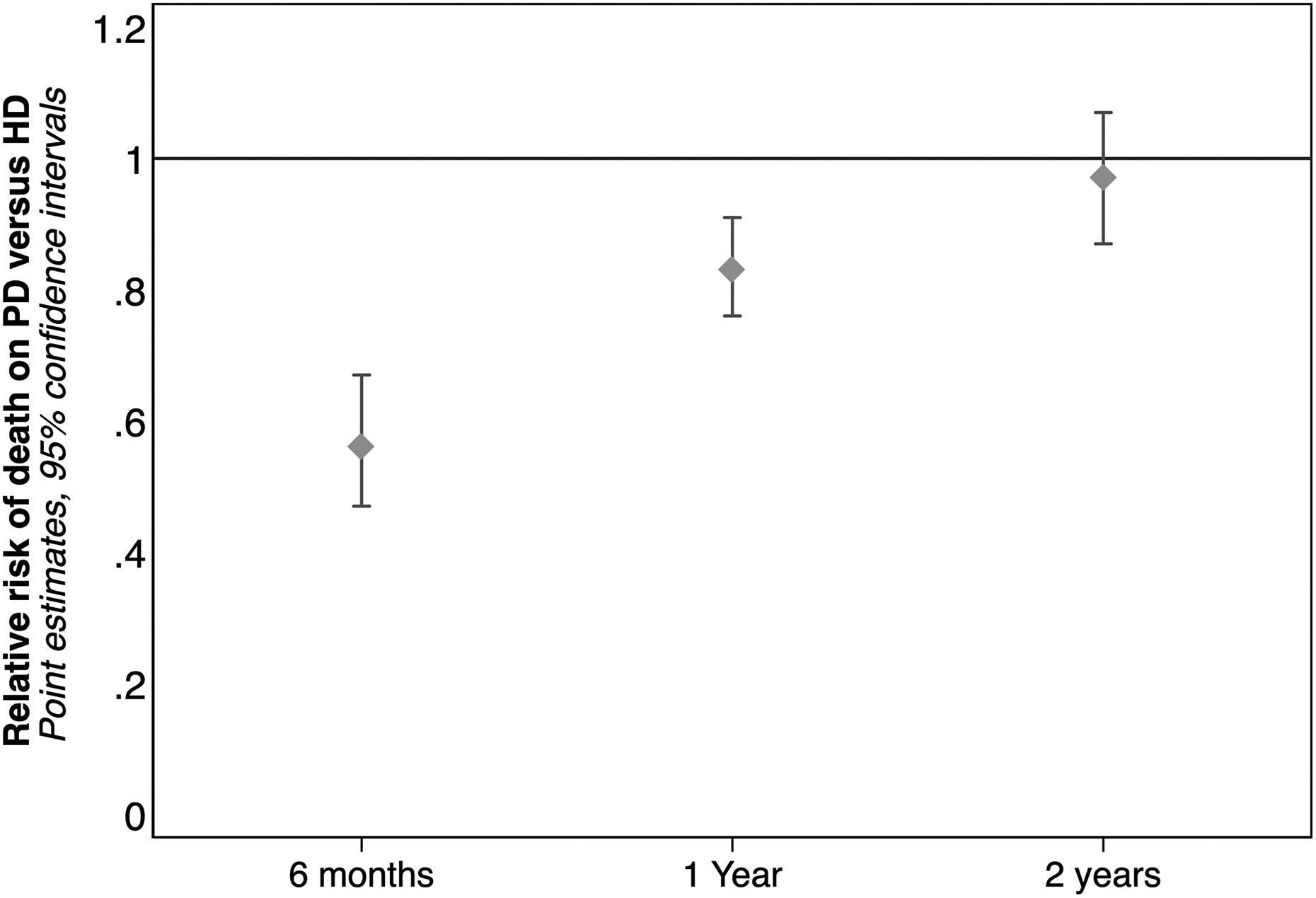
Summary of relative risks of death from the meta-analysis at landmarks of 6 months, 1 year, and 2 years.
As can be seen, there is a high degree of heterogeneity in these analyses. This is not only related to the different sampling frames and eras of studies, but also from variations in how they handle data. First, some studies use the ITT framework for analysis and others the AT framework. These frameworks provide different insights and often disparate statistical estimates. 6,37 Both have their drawbacks. In general, ITT models tend to underestimate differences in risk between groups due to progressive contamination that occurs over time that is ignored. On the other hand, AT models can overestimate differences in risk between groups, due to switching of patients that is accounted for, but often without including changes in patient health status that also occur concomitantly along the way. The impact of these different frameworks can be readily appreciated from the forest plots. The second methodological source of heterogeneity is variation around how HRs are calculated in the studies. All give relative RRs at landmarks of 6 months, 1 year, and 2 years postdialysis inception. However, some articles calculate these as instantaneous HRs, 34,39,47,51,58 –60,64,79 while some calculate a genuinely cumulative estimates, 5,80 and yet others have interval-specific estimates computed over anywhere between 3 42,53 and 12 month 48,75 intervals. These two sources of variation result in an inevitable degree of heterogeneity within the meta-analysis, that cannot be ameliorated in any way.
Notwithstanding these difficulties, a robust observation can be made of a significant early survival benefit with PD versus HD. The question then arises: Is this effect due to unmeasured confounding? At a minimum, these studies all adjusted for differences in patient’s age, gender, diabetic status, and race between groups. At a maximum, they also accounted for detailed comorbidity, center effect, era, and socioeconomic and marital status. However, none of the studies could possibly have included all factors that are important to patient outcomes. For instance, unmeasured differences in adherence and functionality might be present between groups and contributing to the observed differences in outcomes. How can we assess the causal veracity of the early survival benefit?
As described above, the first approach is to use RCTs. There have been two RCTs of PD and HD in the modern era, and one in an older era which is invalidated by obsolete practices. 81 The most recent of the modern ones is still to be published in full, but has been presented at the Annual Scientific Meeting of the American Society of Nephrology in the High Impact Clinical Trials session, and is published as a peer-reviewed abstract (ChinaQ: trial registration NCT01413074 at clinicaltrials.gov). 82 The single fully reported RCT in the modern era is the Netherlands Cooperative Study on the Adequacy of Dialysis (NECOSAD) study. 83 No other trials have been identified in systematic reviews. 84 In the ChinaQ study, 668 patients from 21 centers in China were randomized to either PD or HD with a weighted mean patient follow-up of 39.1 weeks. In the NECOSAD study, 38 patients were randomized, with an overall weighted mean patient follow-up of 2.23 years. Combining the results of both to these trials, the weighted mean relative RR for death on PD versus HD is 0.61 (95% CI 0.36, 1.06) at a weight mean follow-up time of 43.2 weeks (see forest plot in Figure 6). While this is statistically nonsignificant as a result of imprecision, it is nonetheless directionally consistent with those of the meta-analysis reported above.
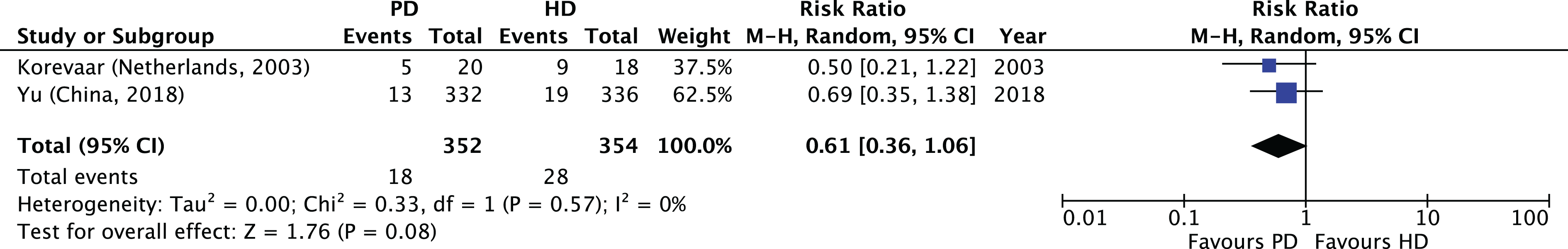
Forest plot of mortality risk from the two randomized controlled trials in the modern era.
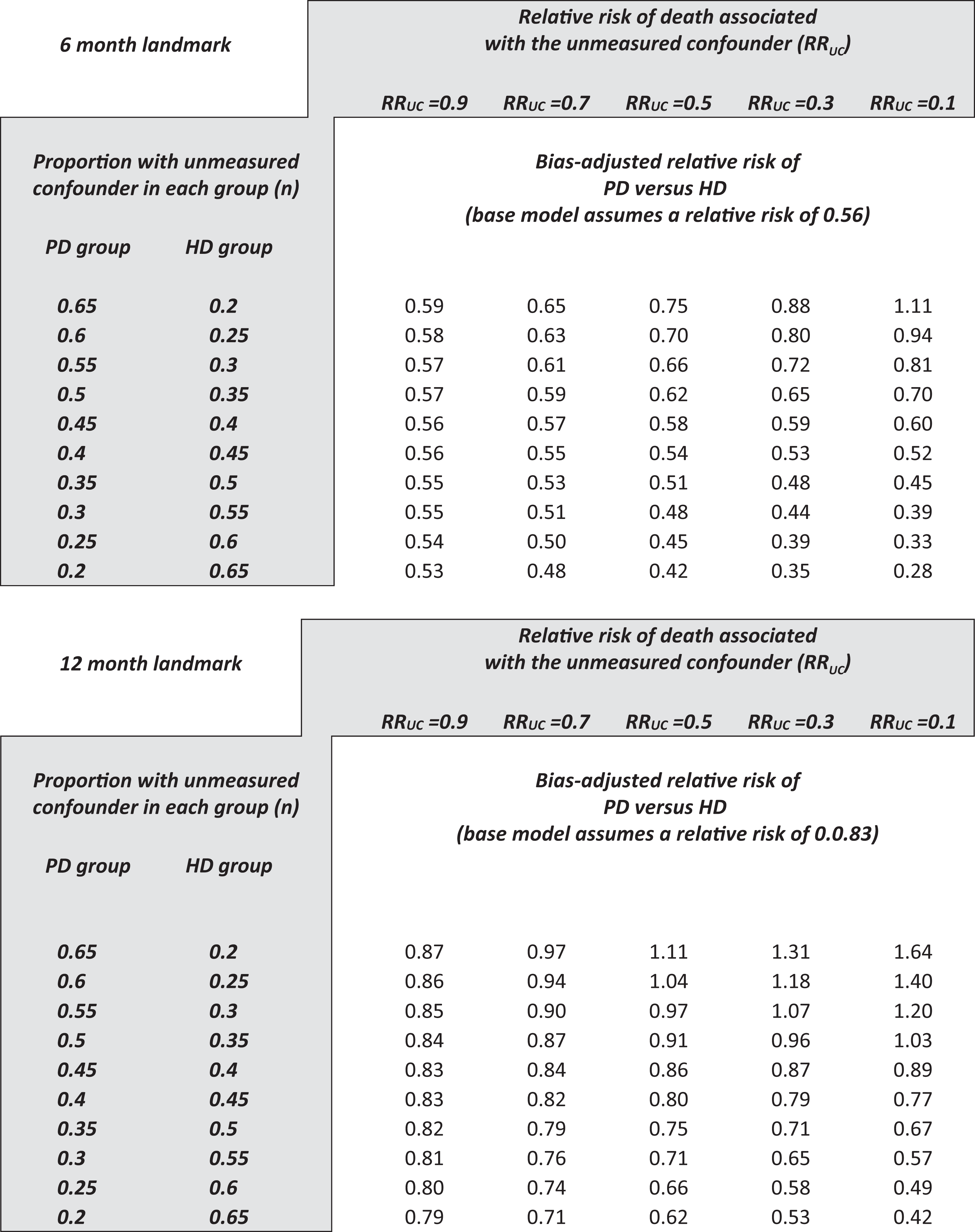
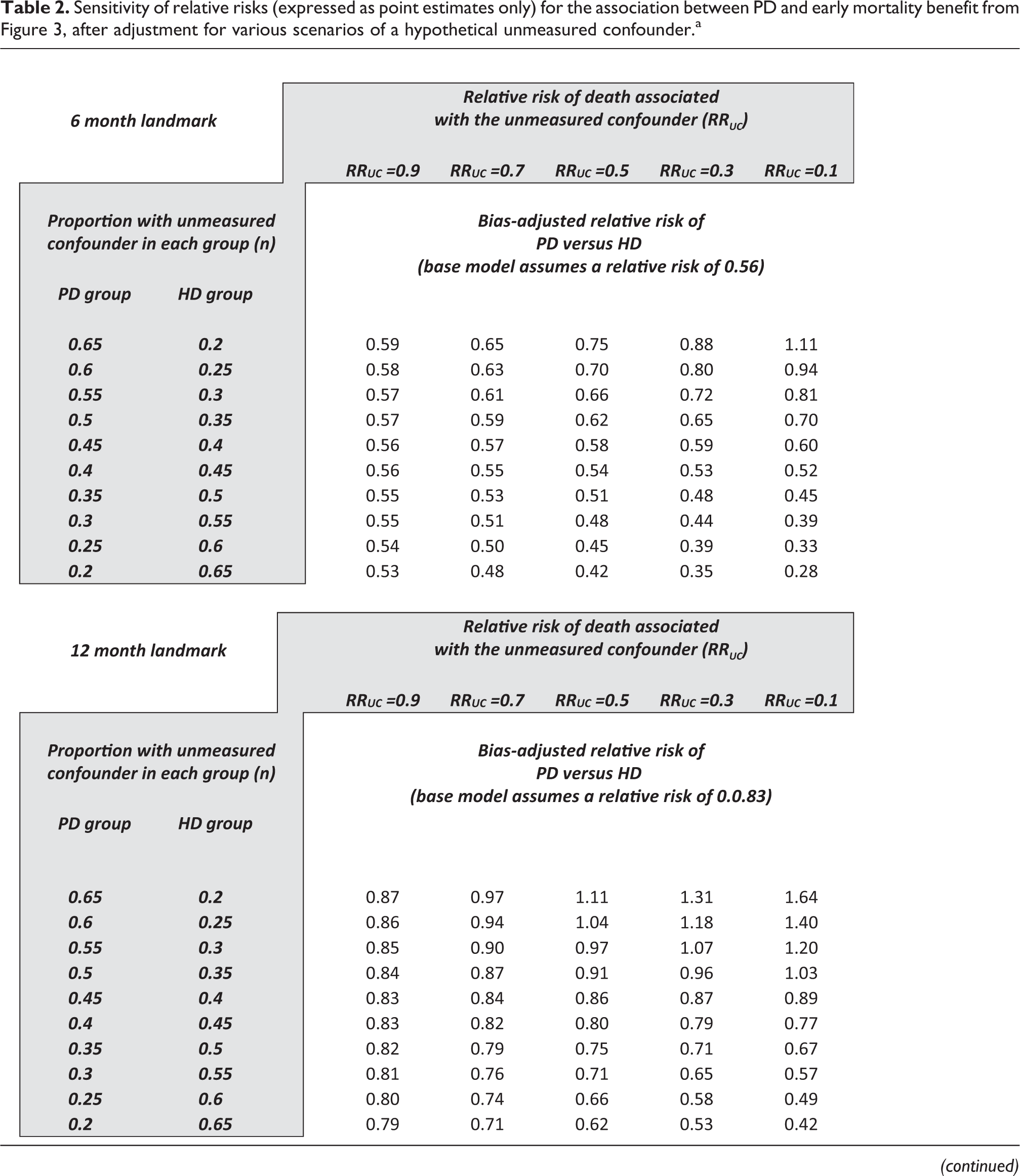
The second approach uses a sensitivity analysis and can be performed using bias formulas. 20 –24 Table 2 presents various scenarios around a hypothetical unmeasured confounder and the resulting impact on the baseline relative RR. These are shown for the landmarks of 3 months, 1 year, and 2 years. The rows show a different prevalence of the unmeasured confounder in each group, and the columns show a different strength of association between the unmeasured confounder and death. When the unmeasured confounder is introduced into the models, the relative risks identified in the meta-analysis are only sensitive to an implausibly strong and/or implausibly unbalanced confounder. For example, to account for the association at 6 months, the unmeasured confounder would have been needed to be three to four times more prevalent in patients in one group than the other and would have been needed to have a relative risk for mortality of around 10. The effect identified in the meta-analysis is otherwise insensitive and persistent after adjustment for other degrees of confounding. A similar situation exists at the 12-month landmark but not the 24-month landmark. For completeness, it should be acknowledged that these conclusions are subject to several strong assumptions—these bias formulas work best with rare events, and when the effect-size of the unmeasured confounder is small. 24,85 In general, however, the technique shows acceptable approximations even if the events are not rare, and bias is only slight as the effect-size of the unmeasured confounder increases 24
Sensitivity of relative risks (expressed as point estimates only) for the association between PD and early mortality benefit from Figure 3, after adjustment for various scenarios of a hypothetical unmeasured confounder.a
Given the consistency of the observational data, the directional corroboration of experimental data, the modest sensitivity of estimates to unmeasured confounding, one can make a strong case for the veracity of the association between modality and early survival benefit, and a reasonable case for a substantial element of causality in the relationship.
Why this early survival benefit is not likely to be from central venous catheter use alone (and why this is not a “deal-breaker” anyway)
If the relationship between modality and early survival benefit has a substantial element of causality, what is the said cause? There are three main hypotheses: Better preservation of residual kidney function (RKF) with PD compared to HD. RKF decreases the relative risk of death in both PD
86
and HD.
87
Moist et al. showed that PD had a 65% lower risk of loss of RKF compared to HD during first year of dialysis in incident end-stage kidney disease (ESKD) patients.
88
Wang et al. reported a 50% reduction in all-cause mortality and cardiovascular death in PD patients for every 1 ml/min increase in residual glomerular filtration rate.
89
Morbidity and mortality associated with central venous catheter (CVC) use. Around the world, patients most often initiate HD using a CVC.
90
–92
At the most recent reporting, this figure was 80% in the United States.
93,94
Studies have clearly documented that the use of CVCs is associated with increased mortality compared to the use of arteriovenous fistulas (AVFs),
95
–97
mainly mediated by increased infectious complications.
98
Patients are yet to face increasing peritoneal membrane exposure to dialysis solutions, which eventually lead to changes in the peritoneal membrane with more fibrosis,
99
decreased ultrafiltration capacity, poorer volume control,
100
and possibly loss of nutrients due to rapid transport, thus increasing the mortality risk over time.
101,102
The point about CVCs is an important consideration. Perl et al. compared approximately 7400 PD patients, 6600 HD patients using AVFs or arteriovenous prosthetic bridge grafts (AVGs), and 24,000 HD patients using CVCs, all of whom started dialysis in 2001–2008. They found that the HD patients using AVFs had a similar or better survival compared to PD patients, whereas the CVC patients had 80% higher mortality than the PD patients. They explicitly conclude that “the use of CVCs in incident HD patients largely accounts for the early survival benefit seen with PD.” 103 Another study from Portugal has supported this tenet, 104 and a study from the United States Renal Data System 2010–2014. 105 On the other hand, Kumar et al. compared 1003 matched patients, who started PD and HD in “optimal” fashion; in particular, the HD patients all started with permanent vascular access. The early survival benefit for PD was still evident in these “optimal” patients (see Figure 1), indicating that factors other than vascular access are still at play. 5
From these two studies, it is reasonable to conclude that vascular access is a very important determinant of the early survival benefit observed with PD, but not the only one. Perhaps more importantly, it is not conceivable that there will be a time that HD will be initiated only with arteriovenous access, just as it is not conceivable that PD will be undertaken without the occasional case of PD-related peritonitis. In the real world, with real-world patients and real-world practice settings, the observed survival benefit of PD relative to HD is likely to persist for the foreseeable future.
Conclusions
Recently, a provocative editorial highlighted the focus of medical researchers with mortality risk on dialysis and the fact that they deprioritize of other important aspects of the patient experience. 106 Notwithstanding, the answer to the question that the authors pose (“Survival by dialysis modality—who cares?”) is in fact “everyone.” 107,108 Survival is among the top-ranked patient-centered outcomes, in addition to being a core clinical outcome. The authors of the provocative editorial are absoutely right of course—the spirit of their editorial is that patient-centered outcomes deserve more consideration by health-care professionals. However, the fact remains that survival on dialysis is still one of the most important considerations for all stakeholders in the ESKD community.
With this in mind, it behooves health-care professionals to tell patients they are likely to live a bit longer if they start their course on dialysis with PD, especially if they are to start HD with a CVC. Shared decision-making is a fundamental patient right and requires both balanced information and an iterative mechanism for a consensual decision based on shared understanding and values. 109 –111 It is known that well-implemented predialysis education and/or shared decision-making results in patients prospectively choosing PD ∼50% of the time, 112 –118 which is similar to the reported preference of health-care professionals when they are presented with the hypothetical case of their own ESKD. 119,120 There is an opportunity to improve patient satisfaction with the dialysis treatment through better shared decisions, 121 –130 with choices that better meet patient needs. 131 A cornerstone of this process should be an explicit discussion of the early survival benefit of PD relative to HD.
Supplemental material
Supplemental Material, 895177_supp_mat - The benefit of early survival on PD versus HD—Why this is (still) very important
Supplemental Material, 895177_supp_mat for The benefit of early survival on PD versus HD—Why this is (still) very important by Mark R Marshall in Peritoneal Dialysis International
Footnotes
Acknowledgment
The author would like to acknowledge the generous data sharing by Victoria A Kumar and Rajnish Mehrotra, the helpful comments from reviewers, and the insights and guidance of Edward F Vonesh over the years.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The author is a paid employee of Counties Manukau District Health Board, University of Auckland, and Baxter Healthcare (Asia) Pte Ltd.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.