Abstract
Background:
Peritonitis is more common in peritoneal dialysis (PD) patients nonadherent to the PD exchange protocol procedures than in compliant patients. We therefore investigated whether regular testing of PD knowledge with focus on infection prophylaxis could increase the time to first peritonitis (primary outcome) and reduce the peritonitis rate in new PD patients.
Methods:
This physician-initiated, open-label, parallel group trial took place at 57 centers in Sweden, Denmark, Norway, Finland, Estonia, Latvia, the Netherlands, and the United Kingdom from 2010 to 2015. New peritonitis-free PD patients were randomized using computer-generated numbers 1 month after the start of PD either to a control group (n = 331) treated according to center routines or to a retraining group (n = 340), which underwent testing of PD knowledge and skills at 1, 3, 6, 12, 18, 24, 30, and 36 months after PD start, followed by retraining if the goals were not achieved.
Results:
In all, 74% of the controls and 80% of the retraining patients discontinued the study. The groups did not differ significantly regarding cumulative incidence of first peritonitis adjusted for competing risks (kidney transplantation, transfer to hemodialysis and death; hazard ratio 0.84; 95% confidence interval (CI) 0.65–1.09) nor regarding peritonitis rate per patient year (relative risk 0.93; 95% CI 0.75–1.16).
Conclusions:
In this randomized controlled trial, we were unable to demonstrate that regular, targeted testing and retraining of new PD patients increased the time to first peritonitis or reduced the rate of peritonitis, as the study comprised patients with a low risk of peritonitis, was underpowered, open to type 1 statistical error, and contamination between groups.
Keywords
Introduction
Peritonitis is a major and potentially serious complication of peritoneal dialysis (PD), the most important risk factor for PD technique failure. 1,2 and may result in a mortality rate of 2–6%. 3 –5 According to a recent international survey, the absence of a PD-related infection is deemed more important than all other clinical outcomes by both patients on PD and their caregivers. 6 Peritonitis rates have declined over the last 30 years because of several improvements in PD treatment including the design of connection systems and antibacterial prophylaxis. 7 Recently reported peritonitis rates in adult PD patients vary greatly between countries from around 0.2 episodes per patient year in a few outstanding centers 8 –10 to between 0.6 and 0.9 episodes per patient year in others. 3,11,12 There is also a clear variation in peritonitis rates between centers within the same country. 3,13,14 The reasons for such variations are unclear but may be related to differences in patient training, infection-prevention protocols, and follow-up routines. 15,16 Patients who are noncompliant with the PD exchange protocol procedures experience higher peritonitis rates than compliant patients. 17 Moreover, according to an observational study from a center in Beijing, those with a poor PD bag exchange technique are reported to have an over fivefold increased risk of peritonitis. 18 Retraining may reduce the risk of peritonitis, 16,19 but this has only been investigated in a limited number of uncontrolled studies. 18,20 –24 Early testing and retraining of new PD patients with focus on hand hygiene and a correct connection technique may thus be a possible way of reducing the incidence of peritonitis.
The primary aim of this trial was to study whether a protocol that involves regular follow-up of new PD patients with testing of their theoretical and practical knowledge in addition to retraining with focus on peritonitis prevention (hereafter called “a new follow-up model”) could extend the time to the first peritonitis episode, while the secondary aim was to investigate whether this model could reduce the incidence of peritonitis. Further aims were to study the risk factors for peritonitis, PD technique failure rate, and peritonitis-related hospitalization time, which will be reported later.
Materials and methods
Trial design
This noncommercial, physician-initiated, randomized, controlled, open-label, parallel-group, multicenter trial started in January 2010 and was initially planned to include 750 incident PD patients over 2 years in Sweden, Norway, Denmark, Finland, Estonia, and Latvia. Further countries were recruited later (see Results section). Patients were eligible for inclusion in the study from the first day of PD at home and up to 6 weeks thereafter. After a baseline visit, patients attended follow-up visits at 1 month (within the 2 weeks before or after the due date) after the start of PD at home, followed by visits at 3 and 6 months, and every sixth month thereafter up to 36 months (Figure 1), which were expected to take place within the 3 weeks before or after the due date. The duration of the study was thus 36 months from the start of PD at home, that is, 35 months from the time of randomization. The trial was planned to continue until the last patient to be included had taken part for 1 year.
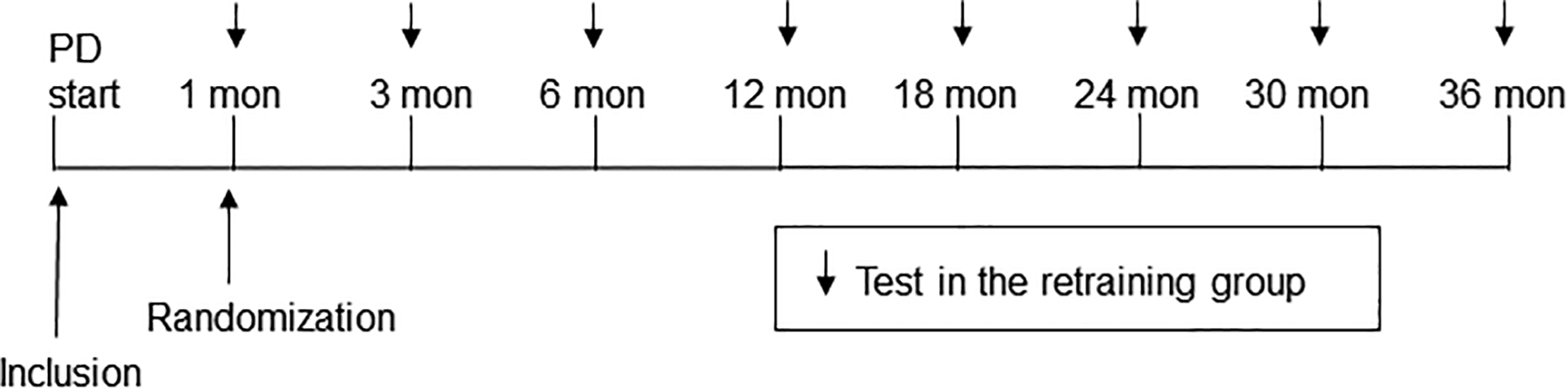
Design of the study.
The study was designed and supervised by a steering committee, coordinated at the Department of Nephrology, Sahlgrenska University Hospital, Gothenburg, Sweden, where the data were collected. The trial was carried out in accordance with the principles of the International Conference on Harmonization Good Clinical Practice guideline. The study protocol and amendments, which fully complied with the Helsinki Declaration, were approved by the ethics committees in the participating countries. The diagnosis of peritonitis was adjudicated by an end point committee based on predefined criteria. The study was monitored by an independent Data Safety and Monitoring Board, consisting of statisticians and nephrologists, who were not involved in the care of the study participants. Only this board had the authority to undertake interim analyses. There were no predefined criteria for ending the study.
The funders of the study had no role in its design, data collection, data analysis, data interpretation, or writing of the report. The corresponding author had full access to all the data as well as the final responsibility for the decision to submit the report for publication. The study protocol was not published prior to the start of the study. The trial is registered with CinicalTrials.gov number NCT01293799.
Participants
Patients aged at least 18 years who had recently started PD and able to perform the dialysis without assistance were eligible. Assistance with the handling of PD bags and exit-site care was allowed. Exclusion criteria included previous PD treatment during the past 2 years, peritonitis related to PD before inclusion, active malignancy, and participation in other studies that might affect the outcome of the present study. All participants provided written informed consent before participation.
Randomization
Participating patients who remained free of peritonitis were randomly assigned to one of two groups at 1 month after the start of PD at home (within the 2 weeks before or after the due date) and received either standard care in accordance with the routines of the center (control group) or “a new follow-up model” (retraining group) in addition to standard care. A permuted block randomization sequence was generated by computer using a block size of eight (1:1 ratio) by an independent manufacturing organization (APL Pharma Specials, Stockholm, Sweden). Patients were allocated to the trial groups at the participating hospitals using sealed, opaque, consecutively numbered envelopes, which were opened in numerical order by nephrologists taking part in the study.
Procedures
The baseline PD training program at all participating centers followed the recommendations of the International Society for Peritoneal Dialysis (ISPD) regarding the topics to be taught.
19
In addition, the alcohol-based hand-rub technique of all patients was checked using fluorescent alcohol and ultraviolet (UV) light at least once during the basic PD training at the study centers. After the basic training, this method was only used in patients recruited to the study and allocated to the retraining group. This group underwent testing at all follow-up visits, after every peritonitis episode, and when restarting PD treatment after a temporary break lasting 6 weeks or more. The tests were designed prior to the study by some of the authors with the help of PD nurses. The questionnaire was tested on a small number of PD patients who found the questions easy to understand. No formal validation of the efficiency of the tests was conducted. The testing, which required 2 to 2½ h and took place either at the PD center or in the patient’s home, was conducted by PD nurses and included the following procedures: Practical test (Supplemental material): The patient was asked to perform a PD exchange and exit-site care. Those who needed assistance with exit-site care could skip the latter step. Patients using automated PD were observed as they set up a cycler. A PD nurse was present during the test but did not interrupt the patient. Using a check list, the nurse recorded whether preparations for the PD exchange, hand hygiene using an alcohol-based hand-rub technique, PD exchange, exit-site care, and the securing of the catheter were correctly and safely performed. In addition, the alcohol-based hand-rub technique was checked using fluorescent alcohol and a UV lamp. The goal was for all steps in the test to be performed correctly. Questionnaire (Supplemental material): Participants were asked to complete a questionnaire with 24 multiple-choice questions on hygiene, infection prophylaxis, PD bag exchange technique, recognition of and appropriate action to deal with contamination, exit-site infection, and peritonitis. Thereafter, a nurse discussed the result with the patient. The goal was to achieve at least 80% of the maximum score. If the patient did not meet the goals of both tests, further training was provided, either on the same day or scheduled later, until the goals were achieved.
Participating centers were instructed to treat the control group in accordance with the ordinary routines without being influenced by the protocol used in the retraining group and not to change the routines for instruction and follow-up of patients pertaining to prophylaxis of PD-related infections during the course of the study.
Prior to the start of the study, each center reported the number of PD patients and PD staff including their roles, the incidence of peritonitis the year before taking part in the study, whether the center screened for and treated nasal carriers of Staphylococcus aureus, used topical antibiotic cream at the exit site, antibiotic prophylaxis prior to catheter insertion, and regularly checked the knowledge and skills of new patients.
Baseline laboratory tests were performed just before the start of PD. Serum albumin values analyzed using the bromcresol purple method were converted to the corresponding values of the bromcresol green method. 25 Physical status was estimated using the Karnofsky performance scale, 26 and the number of comorbid diseases was measured by the Stoke score. 27 At the follow-up visits we recorded the need for assistance with exit-site care as well as the occurrence of peritonitis episodes and exit-site infections 28 since the previous follow-up visit. The diagnosis of peritonitis was based on at least two of the following criteria in accordance with the recommendations of the ISPD 28,29 : (1) cloudy PD effluent fluid with leukocytosis corresponding to more than 100 white blood cells/µL (compulsory in the trial, and we regarded this criterion fulfilled by a positive dip-test for leukocytosis) and more than 50% polymorphonuclear cells (optional in this study); (2) symptoms of peritoneal inflammation; and (3) positive dialysis effluent culture.
Primary and secondary outcomes
The predefined primary outcome was time from randomization to the first peritonitis episode. The incidence of peritonitis during the study, which was a secondary outcome, is also reported. Further secondary outcomes, not reported here, include risk factors for peritonitis, PD technique failure rate, and peritonitis-related hospitalization time.
Statistical analysis
The sample size calculation was based on an analysis of all patients who started PD at the Sahlgrenska University Hospital, Gothenburg, Sweden, between 1 January 2004 and 31 December 2007 (n = 120). Up to 31 November 2008, 42% (50 of 120) of the patients had experienced a first peritonitis episode. The overall peritonitis rate was 0.48 episodes per patient year. The sample size was calculated for log-rank follow-up between the retraining group and the control group for the time up to the first peritonitis episode. The significance level was set to 5% and power to 80%. The calculations were based on a 2-year inclusion period with a prompt effect of the intervention declining over time. For an initial hazard ratio of 0.70, which corresponds to a 30% reduction of the peritonitis risk, the minimum sample size calculated via simulations was found to be 480 patients. With adjustment for potential withdrawals, which were expected to be around 30% during the first year, the sample size was estimated to be 750 patients.
Time to the first peritonitis episode was analyzed as the cumulative time without peritonitis using the Cox proportional hazards regression model from which unadjusted hazards ratios (HRs) were calculated. Actuarial survival curves showing proportions of peritonitis-free patients over time in the two groups were estimated by means of the Kaplan–Meier method. Log-rank follow-up was used to compare the survival curves. Patients who stopped PD treatment due to causes other than peritonitis were managed according to the intention-to-treat principle. Patients were censored when the PD treatment was stopped, at withdrawal of consent, and at the closing date of the study. Per-protocol analyses were also performed. The Fine and Gray model 30 was used to examine the cumulative incidence of first peritonitis considering kidney transplantation, transfer to hemodialysis, and death as competing events. The number of recurrent events between the two groups was tested by assuming a Poisson distribution of the number of events per follow-up time, log (follow-up time) as offset time, by using generalized linear models. The results were expressed as frequencies, percentages for categorical variables, mean and standard deviation (SD) for continuous normally distributed variables, and median and interquartile rate (IQR) for continuous non-normally distributed variables. All tests were two-tailed and conducted at a significance level of 0.05. The analyses were performed using SAS software version 9.4 (SAS Institute Inc., Cary, North Carolina, USA).
Results
Study population
Patient recruitment started on 18 January 2010 in Sweden, followed by Finland, Estonia, and Latvia, while in Norway and Denmark it began in 2011. As only 254 participants had been randomized after 2 years, the recruitment period was extended from the initially planned 2 years in two steps to 31 December 2014 (first to 4 years and then to 5 years). It was not feasible to prolong the inclusion further. In addition, participants from the Netherlands and the United Kingdom were recruited from 2012 and 2014, respectively. In October 2013, when 472 patients had been randomized, the Data Safety and Monitoring Board performed an interim analysis, showing that the conditional power of the study was low and therefore recommended termination of the enrollment. Due to the delay in the reporting of results from the study centers, the actual study data were incomplete when the interim analysis was performed, which possibly influenced the results. The Steering Committee therefore decided to continue the trial, which ended on 31 December 2015.
We included 713 patients from 57 centers located as follows: Sweden (25), Norway (11), Finland (7), Denmark (5), Estonia (1), Latvia (1), the Netherlands (5), and the United Kingdom (2). Of the 708 patients who met the inclusion criteria, 37 were not eligible for randomization, mainly due to peritonitis occurring after inclusion (n = 25; Figure 2). Of the 671 randomized patients, 331 were assigned to the control group and 340 to the retraining group.
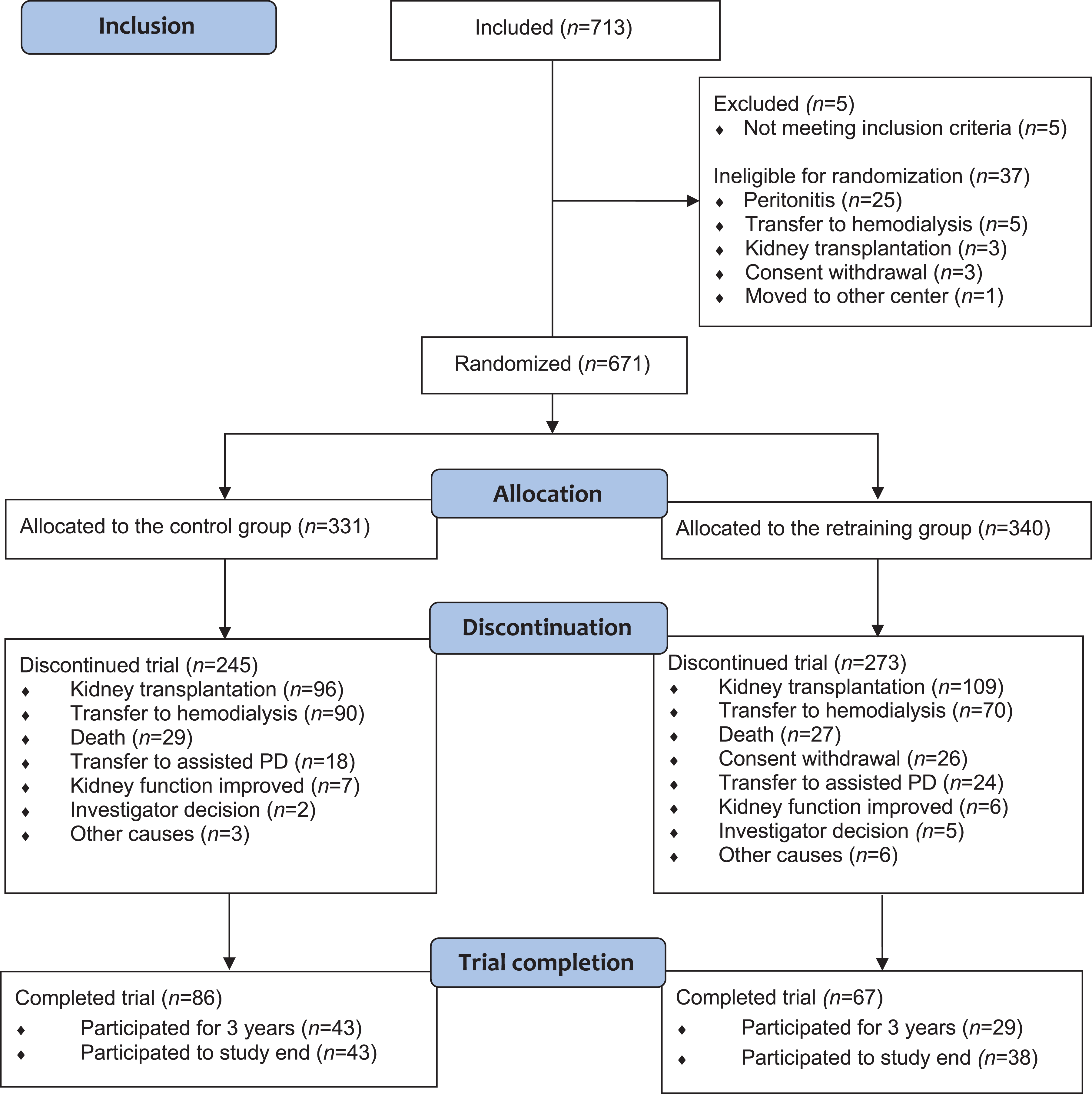
Patient flow diagram.
Baseline characteristics were similar with no significant differences between the groups, except for diabetic nephropathy as the primary cause of renal failure (p = 0.02; Table 1). The participants used multi-chamber bag dialysis fluids with low glucose degradation products, except for 2% (17 of 671) who used conventional dialysis fluid. In addition, icodextrin and amino acid solution were utilized.
Baseline characteristics of the study population.a

PD: peritoneal dialysis; CAPD: continuous ambulatory PD; APD: automated peritoneal dialysis; SD: standard deviation.
a Data are number (%), mean (SD), or median (interquartile rate).
b Data missing for one to three participants per group.
c Data missing for two to six participants per group.
d Connection system and PD fluid available throughout the study, even after change of brand name.
Seventy-four percent of the participants in the control group and 80% in the retraining group discontinued the study (Figure 2). The main reasons were kidney transplantation (29% vs. 32%), transfer to hemodialysis (27% vs. 21%), death (9% vs. 8%), transfer to assisted PD (5% vs. 7%), and withdrawal of consent (0 vs. 8%) in the control group and the retraining group, respectively (Figure 2). During the first year after randomization, 40% (n = 270) of the participants discontinued the trial, 35% (n = 117) in the control group and 45% (n = 153) in the retraining group. In the latter group, consent was withdrawn by 3% (n = 11) during the first 6 months, 7% (n = 24) during the first year, and later by 1% (n = 2). The number of participants who remained in the study at 24 and 35 months after randomization was 102 (31%) and 26 (8%) in the control group and 73 (22%) and 20 (6%) in the retraining group. The follow-up time after randomization was significantly longer in the control group (490.9 patient years; median days 509; interquartile range (IQR) 267–815) than in the retraining group (435.5 patient years; median days 410; IQR 186–684) (p = 0.003).
Time to the first peritonitis episode
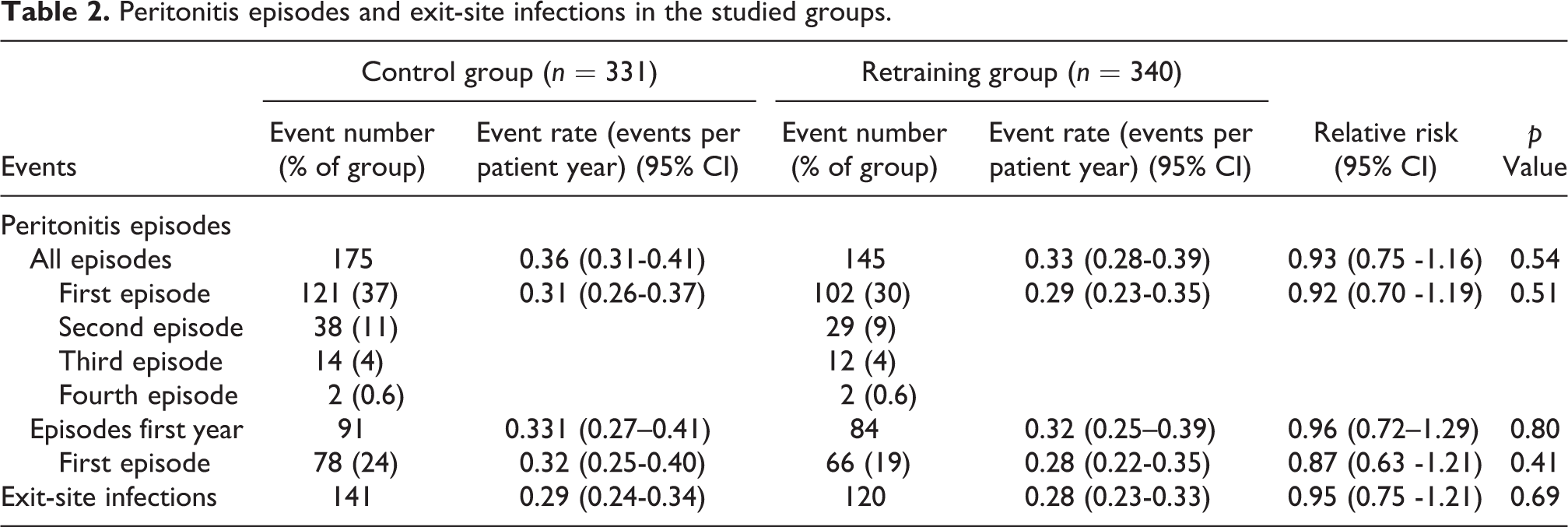
Of the 671 participants, 223 (33%) experienced a first peritonitis episode: 121 (37%) in the control group and 102 (30%) in the retraining group (Table 2). The mean incidence of first peritonitis per patient year did not differ significantly between the groups during the first year after randomization nor during the whole study (Table 2). According to the Kaplan–Meier analysis, the retraining group did not have a significantly longer time to first peritonitis than the controls during the whole study (HR 0.92, 95% CI 0.71–1.19; p = 0.52; Figure 3) or during the first year (HR 0.87, 95% CI 0.63–1.21, p = 0.41).
Peritonitis episodes and exit-site infections in the studied groups.
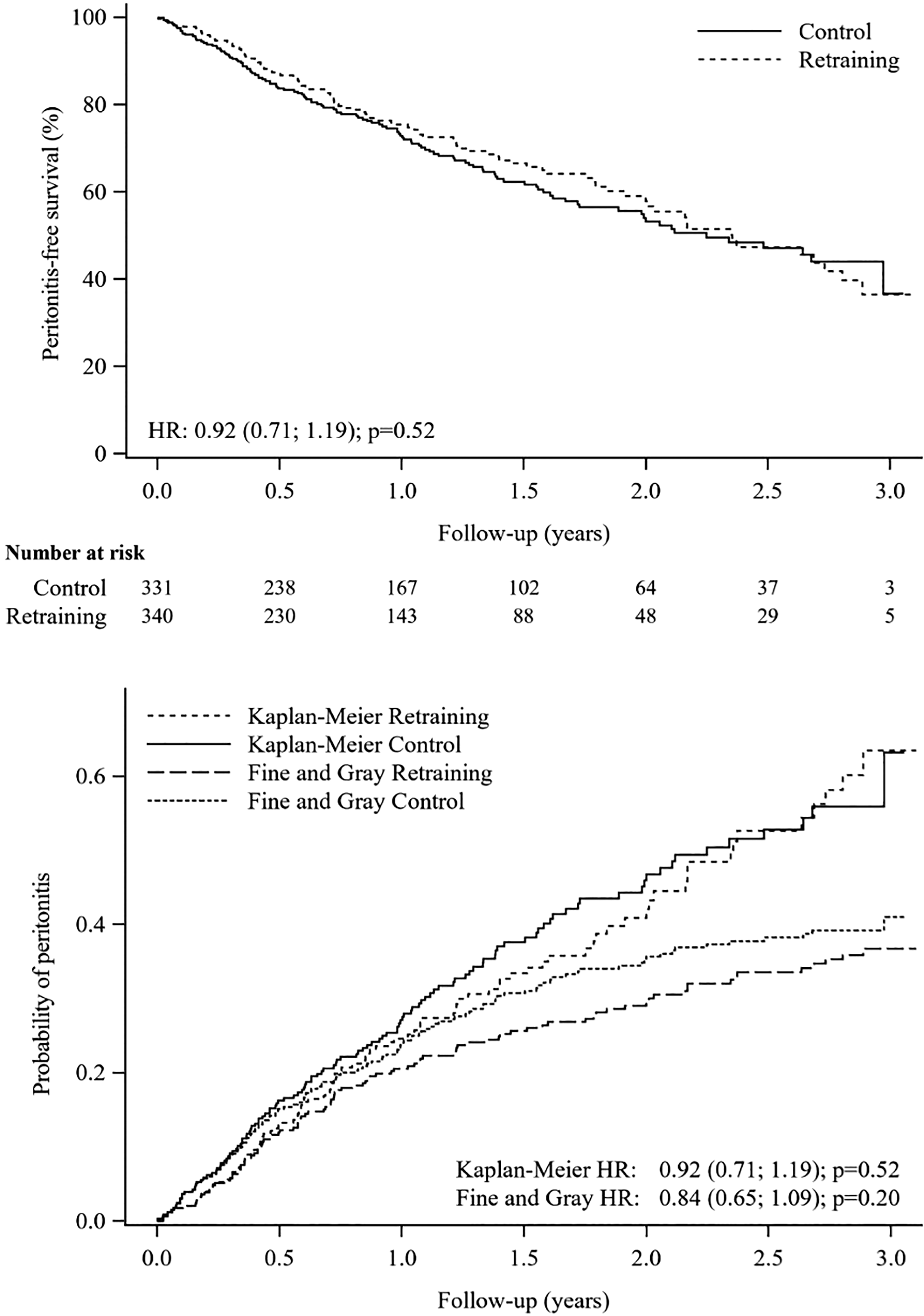
Kaplan–Meier and competing risk analysis curves for follow-up time from randomization to the first peritonitis episode for the control and retraining groups. The time of follow-up was 35 months from randomization, that is, to the time of the visit at 36 months after the start of PD at home. The numbers at risk refer to the number of peritonitis-free patients. Upper panel: Kaplan–Meier survival curves. Lower panel: Cumulative incidence curves adjusted for the competing risks of kidney transplantation, transfer to hemodialysis, and death in accordance with the Fine and Gray method.
Per-protocol analysis of 329 controls (two patients tested by mistake were excluded) and 319 patients in the retraining group (21 patients attending less than 75% of the planned tests were excluded) showed that 120 (36%) of the participants in the control group and 95 (30%) in the retraining group had experienced a first peritonitis episode, the rate per patient year being 0.31 (95% CI 0.26–0.37) and 0.28 (95% CI 0.23–0.34), respectively. In this population, the risk of a first peritonitis episode was not significantly lower in the retraining group compared to the controls (HR 0.89, 95% CI 0.68–1.17; p = 0.40).
The analysis of the cumulative incidence of the first occurrence of peritonitis after randomization taking the competing risks of kidney transplantation, transfer to hemodialysis, and death into account demonstrated slightly lower cumulative incidences than the Kaplan–Meier survival analysis but showed no significant difference between the controls and the retraining group (HR 0.84, 95% CI 0.65–1.09; p = 0.20); Figure 3).
Incidence of peritonitis and exit-site infection
The total incidence of peritonitis and exit-site infection per patient year did not differ significantly between the study groups (Table 2). The rate of gram-negative microorganisms per patient year was significantly lower in the retraining group compared to the control group. The rates of gram-positive and other microorganisms were similar in the two groups (Table 3). The outcome of peritonitis was similar in the study groups (Table 4). Cure was defined as resolution of the signs of peritonitis with antibiotic therapy and without the need for catheter removal. A peritonitis relapse was defined as a further episode of infection with either the same organism or negative culture within 4 weeks of antibiotic therapy cessation. 28
Peritonitis episodes and causative microorganisms.a
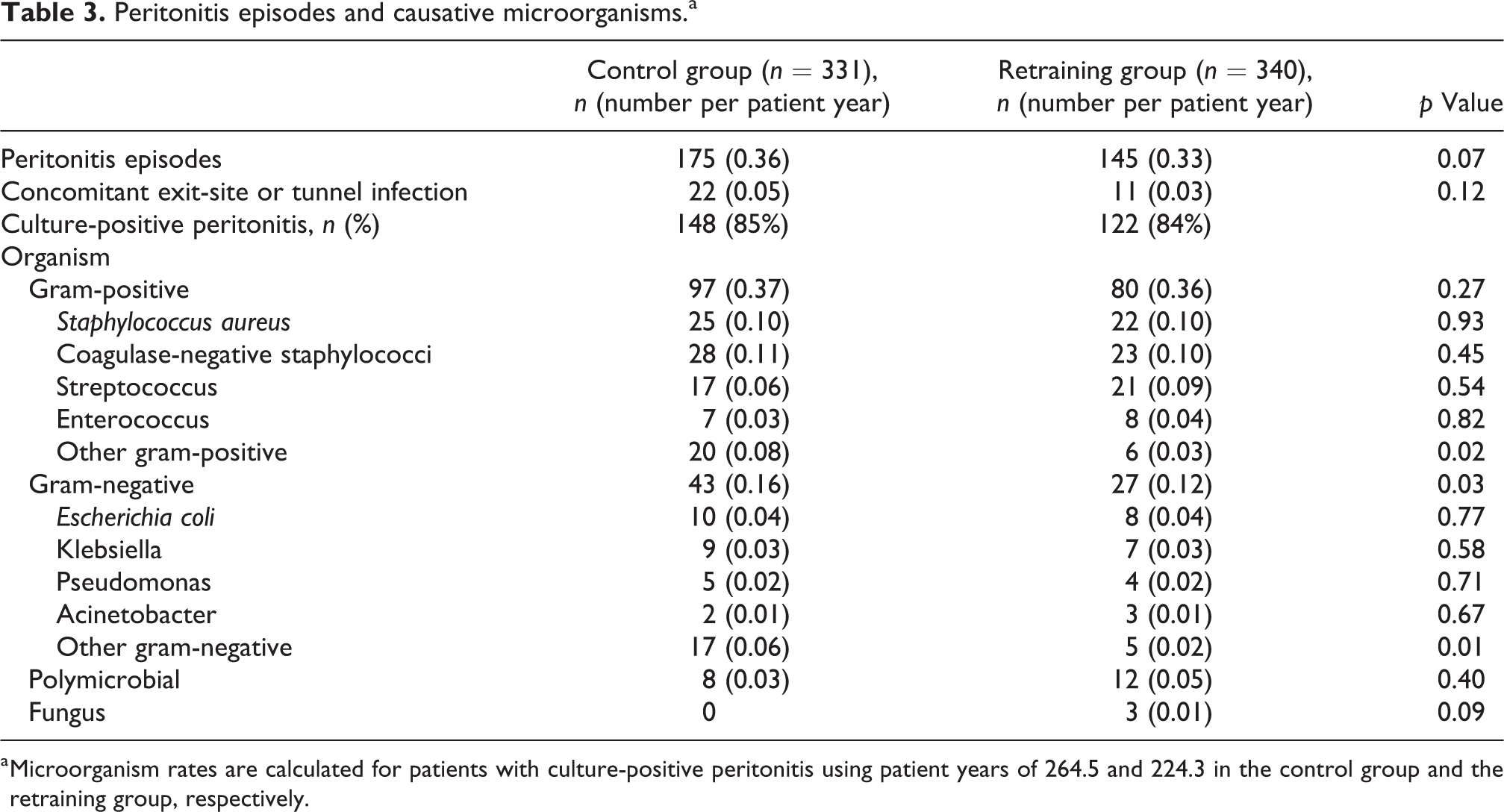
a Microorganism rates are calculated for patients with culture-positive peritonitis using patient years of 264.5 and 224.3 in the control group and the retraining group, respectively.
Outcome of peritonitis episodes in the studied groups.
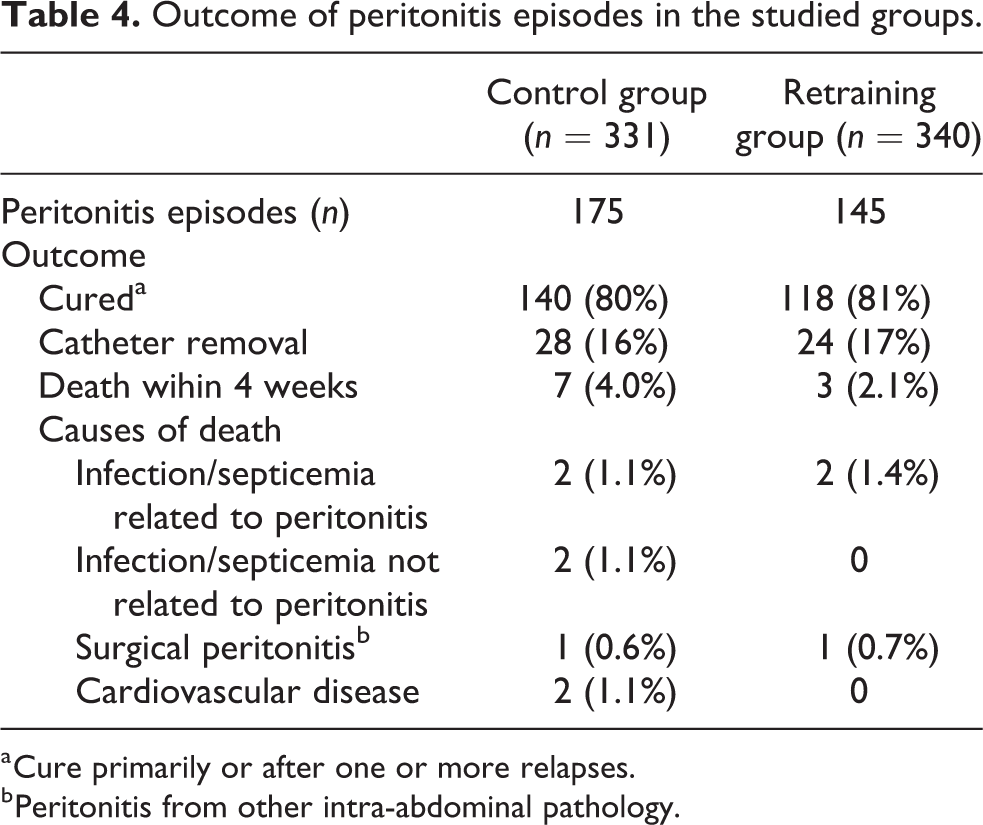
a Cure primarily or after one or more relapses.
b Peritonitis from other intra-abdominal pathology.
Characteristics of the study centers
At the time of inclusion of the first patient, the total number of PD patients at the 57 participating clinics was 1605 with a median of 21 (IQR 16–34) and the median ratio of PD patients to full-time PD nurses was 10.7 (IQR 8.5–13.3). A protocol for follow-up of new PD patients within the first 6 months after the start of PD formed part of the routine at 18% (10 of 57) of the centers. After the first 6 months, only 9% of the centers (5 of 57) performed follow-up every sixth month and 4% (2 of 57) only once a year. Most of these protocols included inspection of a PD exchange, exit-site care, and hand hygiene technique. Nasal screening and treatment of nasal carriers of S. aureus was routine at 16 (28%) of the centers, prophylactic use of topical antibiotic exit-site cream or ointment was regular at 3 (5%), and occasional at 10 (18%). The median peritonitis rate the year before taking part in the study was 0.46 (range 0.23–0.96; IQR 0.34–0.65). A Cox regression analysis of time to the first peritonitis episode per center, including the largest 10 centers with at least 20 participants, showed no center effect interaction on the treatment outcome (p = 0.26).
Discussion
The results of this randomized controlled trial of new PD patients showed that regular, targeted testing of PD knowledge and practical PD skills focusing on infection prophylaxis, as well as retraining if the goals were not achieved, was not associated with a significantly longer time to the first peritonitis episode or a significantly lower total peritonitis rate than standard care provided in accordance with the routines of the centers.
Our hypothesis that regular testing and retraining of PD patients may prolong the time to the first episode of peritonitis and reduce the peritonitis rate was based on the studies by Russo et al. 17 and Kazancioglu et al. 31 demonstrating that PD patients who were noncompliant with the PD exchange protocol procedures more often experienced a peritonitis episode compared to compliant patients. This has also been found in later studies. 18,22,24,32 In their investigation of prevalent Italian PD patients, Russo et al. 17 also found that only 74% of the 191 PD patients performed the PD procedure correctly in terms of infection prophylaxis. Based on this result and an assessment of the knowledge of 353 patients who completed a questionnaire, the authors estimated that 29% of the patients were in need of retraining of the PD exchange technique. In addition, a large Italian observational survey has shown that hospitals in which retraining of PD patients is provided had lower peritonitis rates than those that did not provide retraining. 33 The effect of a theory-based method for training and retraining of adult PD patients was investigated in a large, multicenter, longitudinal, prospective, non-randomized trial by Hall et al., 21 which showed an insignificantly lower peritonitis rate in the pilot clinics than in the control centers after 2 years.
Our results differ from those of a small number of observational studies, which have demonstrated significant improvement of the peritonitis rate at a clinic after introduction of a new PD education program. 20 –23 The disparity between the results of these studies and our trial most likely relates to differences in the study design and the characteristics of the PD populations investigated. Most observational studies have included prevalent PD patients who may have already experienced a peritonitis episode. As the present study only included incident peritonitis-free PD patients who were able to perform PD without assistance, the results cannot be generalized to regular PD populations. However, it is likely that the majority of incident PD patients at most PD units would meet the inclusion criteria of this trial. Differences in the use of prophylactic antibiotics may also influence the generalizability of the results. Thus, the ISPD guidelines for the prevention of peritonitis 29 recommending antibiotic administration prior to catheter insertion (level 1A) were followed by 91% of the study participants. The fact that exit-site antimicrobial prophylaxis (level 1B) was only used by 5% of the patients is likely due to the fact that the health authorities in the Scandinavian and Baltic countries do not encourage long-term prophylactic use of antibiotics in any patient category due to the risk of the development of resistant bacterial strains. It has also been demonstrated that in many countries it is very common not to follow these ISPD recommendations. 15
The fact that many patients discontinued the study, mainly due to kidney transplantation, transfer to hemodialysis, and death, could have altered the probability of a first peritonitis episode over the course of the study. We therefore used the Fine and Gray analysis to take these competing risks into account, as has been recommended for survival analysis in PD research. 34
The present study is, to our knowledge, the first randomized, controlled trial of the effect of retraining on the peritonitis rate in PD patients. In addition to its design, the strength of the study is its large sample size, enrollment of incident, peritonitis-free PD patients from a range of centers and countries with similar approaches to PD, and the fact that data could be collected for all participants.
The limitations of the study mainly concern its low power. The power calculation was based on a discontinuation rate of 30% of the participants after 1 year. However, 40% of the participants, 45% in the retraining group and 35% in the control group, had left the study by that time. As a consequence, the total number of patient years after randomization was significantly lower in the retraining group than in the control group. The main reason for this difference was that 8% of the participants in the retraining group withdrew consent and that half of these withdrawals occurred already within the first 6 months.
The power of the study may also have been weakened by other factors. Thus, the recruitment to the study was slower than anticipated, and the final number of randomized participants was only 89% (671 of 750) of the target, despite the fact that the inclusion time was extended from 2 years to 5 years and additional centers were included. The burden of the study on the PD centers could also have influenced the recruitment of patients, as regular PD staff performed the testing at most centers.
Randomization as late as 1 month after the start of PD also limited the recruitment to the study because 5% (37 of 708) of the included participants had become ineligible for randomization, mainly due to peritonitis. The reason for not randomizing earlier was that we wanted to be sure that this dialysis modality functioned in an acceptable manner and that the included participants were able to perform PD without assistance at home. The late randomization most likely resulted in the selection of less peritonitis prone patients, which might reduce the generalizability of the results of the trial. These factors may also explain why the incidence of peritonitis in the control group (0.36 episodes per patient year) was lower than expected, both compared with the pre-study peritonitis rate at the PD center on which the sample size calculation was based (0.48 episodes per patient year) and the pre-study median value of the participating centers (0.46 episodes per patient year). In addition, the participating PD centers had a slightly lower median pre-study peritonitis rate than those reported by many other PD units. 3,11,35 This finding might be explained by the Hawthorne effect, that is, the treatment of controls by the study center staff may have been modified due to their awareness of being observed. Another explanation could be selection bias, but this is less likely, since allocation concealment was used.
A further factor that could possibly have contributed to a lower-than-expected peritonitis rate in the control group was that all study centers routinely used fluorescent alcohol and an UV lamp during the basic hand hygiene training. This method was introduced at all study centers where it was not previously used because we wanted all potential participants to have comparable basic PD training.
The effectiveness of the intervention can be questioned, due to the fact that the practical test and the questionnaire were not validated prior to the study. In addition, 6% of the patients attended less than 75% of the planned tests.
Another factor that could have influenced the results was that 18% of the participating centers already provided some form of retraining before the study. However, only 9% performed regular follow-ups after the first 6 months of PD, and the retraining at these centers was not as rigorous as that in the study. Although we did not find any significant center interaction effect on outcome, the study may not have been adequately powered to assess this.
In addition, it cannot be excluded that the PD staff were unintentionally influenced by the study protocol used for the retraining group when taking care of the patients in the control group. Moreover, it was not possible to employ blinding in this study. To reduce the risk of possible contamination between groups, cluster randomization of comparable centers should ideally have been carried out. However, this would have required a larger number of participants in order to obtain equivalent statistical power compared to individually randomized trials. 36 Furthermore, in contrast to individual randomization, clusters are usually randomized at the same time. It may be easier to achieve parity of key factors in the intervention and the control arm in non-clustered clinical trials, especially if the participants have multiple chronic conditions, 37 which is common in PD patients. It was not possible to conduct cluster randomization in the present trial because most centers had to be recruited during the study and greater financial resources would have been required than were initially available.
Based on our experience of the present study, we suggest that future studies of the effect of retraining on the prevention of PD-related peritonitis should have sufficient power to compensate for the high turnover in this patient group, should employ cluster randomization, and should initiate testing and retraining as soon as possible after the start of PD.
Conclusions
This randomized controlled trial was unable to demonstrate that “the new follow-up model” with regular, targeted testing and retraining of new PD patients with focus on infection prophylaxis increased the time to the first episode of peritonitis (primary outcome) or reduced the risk of peritonitis compared with controls treated in accordance with the standard routines of the center. The main limitation of the study was its low power with a risk of type 1 statistical error. Moreover, the trial included patients at low risk of peritonitis, in addition to which there was a possibility of contamination between the groups. Recommendations for further studies on the effect of retraining for the prevention of PD-related peritonitis are presented.
Supplemental material
Supplemental_materials_887626 - Retraining for prevention of peritonitis in peritoneal dialysis patients: A randomized controlled trial
Supplemental_materials_887626 for Retraining for prevention of peritonitis in peritoneal dialysis patients: A randomized controlled trial by Susanne Ljungman, Jørgen E Jensen, Dag Paulsen, Aivars Petersons, Mai Ots-Rosenberg, Heikki Saha, Dirk G Struijk, Martin Wilkie, Olof Heimbürger, Bernd Stegmayr, Thomas Elung-Jensen, Ann-Cathrine Johansson, Margareta Rydström, Helga Gudmundsdottir, Max Petzold and on behalf of the Peritonitis Prevention Study (PEPS) Trial Investigators in Peritoneal Dialysis International
Footnotes
Authors’ note
PEPS TRIAL INVESTIGATORS: (The number of patients enrolled and randomized at each center in parenthesis.) Sweden: Ulf Persson, Susanne Ljungman, Sahlgrenska University Hospital, Gothenburg (67/65); Stefan B Jacobsson, Elena Fedchenko, Danderyd Hospital, Stockholm (36/35); Finn D Nielsen, Southern Älvsborg Hospital, Borås (30/28); Henrik Hadimeri, Margareta Rydström, Skaraborg Hospital, Skövde (26/26); Per Dahlberg Northern Älvsborg County Hospital, Trollhättan (25/25); Martin Lindgren, Helsingborg Hospital (25/24); Ann-Cathrine Johansson, Skåne University Hospital, Malmö (24/23); Knut-Christian Gröntoft, Eskilstuna Hospital, Eskilstuna (23/22); Malin Isaksson, Sunderby Hospital, Luleå (19/17); Lars Weiss, Karlstad Hospital, Karlstad (12/12); Antonija Jagunic, Varberg Hospital, Varberg (12/11); Ole Simonsen, Naomi Clyne, Skåne University Hospital, Lund (11/11); Hans Furuland, Uppsala University Hospital, Uppsala (11/11); Per Halvarsson, Gävle County Hospital, Gävle (11/11); Gudrun Malmsten, Örebro University Hospital, Örebro (10/10); Björn Rogland, Hässleholm Hospital, Hässleholm (10/10); Bernd Stegmayr, Umeå University Hospital, Umeå (8/8); Britta Hylander, Karolinska University Hospital, Solna (6/6); Mattias Tejde, Falun Central Hospital, Falun (6/5); Isabel Hernadez Bascaran, Eksjö Hospital, Eksjö (6/4); Olof Heimbürger, Karolinska University Hospital, Stockholm, Huddinge (5/5); Mattias Alvunger, Peter Åkesson, Kalmar County Hospital, Kalmar (4/4); Stefan Melander, Linköping University Hospital, Linköping (4/4); Erland Olausson, Sundsvall Hospital, Sundsvall (3/3); Nils-Johan Mauritz, County Hospital Ryhov, Jönköping (3/3); Latvia: Aivars Petersons, Ilze Puide, P. Stradins Clinical University Hospital, Riga (59/50); Estonia: Mai Ots-Rosenberg, Tartu University Hospital, Tartu (23/20); Denmark: Thomas Elung-Jensen, Rigshospitalet University Hospital, Copenhagen (19/18); Jørgen E Jensen, Odense University Hospital, Odense (17/13); Else Randers, Viborg Hospital, Viborg (14/13); Kjeld Erik Otte, Fredericia Hospital, Fredericia (10/10); Johan V. Povlsen, Aarhus University Hospital, Skejby, Aarhus (5/5); Norway: Dag Paulsen, Kolbjørn Høgåsen, Innlandet Hospital, Lillehammer (9/9); Øyvind Storset, Geir Mordal, Akershus University Hospital, Lørenskog (8/8); Ingrid Os, Helga Gudmundsdottir, Oslo University Hospital Ullevål, Oslo (8/8); Izeta Muijic, Haukeland Hospital, Bergen (7/7); Anne Kristine Fagerheim, Nordland Hospital, Bodø (6/6); Stig Arne Kjellevold, Tønsberg Hospital, Tønsberg (5/4); Julie Sunseth, Telemark Hospital, Skien (5/5); Kristian Selvig, Drammen Hospital, Drammen (5/4); Ingrid Toft, University Hospital of North Norway, Tromsø (2/2); Gunnar Fjermeros, Sørlandet Hospital, Kristiansand (2/2); Ingrid Lagreid, Trondheim University Hospital, Trondheim (1/1); The Netherlands: Dirk Struijk, Academisch Medish Centrum, Amsterdam (19/17); Jeroen Vincent, St Antonius Hospital, Nieuwegein (19/16); Tom Cornelis, Maastricht University Medical Center, Maastricht (8/7); Marc Hermans, VieCuri Medical Center, Venlo (7/7); Anita M. Schrander van der Meer, Rijnland Hospital, Leiderdorp (5/4); Finland: Seppo Ojanen, Lahti Central Hospital, Lahti (16/16); Heikki Saha, Tampere University Hospital, Tampere (7/7); Mika Huuskonen, Central Hospital of Kymenlaakso, Kotka (6/6); Kristiina Kananen, Kainuu Central Hospital, Kajaani (6/6); Marja Miettinen, Central Finland Central Hospital, Jyväskylä (4/4); Martti Metso, Jani Ahvonen, Hämeenlinna Central Hospital, Hämeenlinna (3/3); Maarit Vainiotalo, Satakunta Central Hospital, Pori (2/2); United Kingdom: Martin Wilkie, Sheffield Kidney Institute, Northern General Hospital, Sheffield (7/6); Richard Fluck, Janson Leung, Royal Derby Hospital, Derby (2/2).
Acknowledgments
The authors gratefully acknowledge the PD nursing staff and the investigators for their contribution and the participating hospitals for their generous support. We also wish to thank the NIHR clinical research nurses in the United Kingdom as well as research nurses at other study centers. Special thanks to research nurse Lena Heijdenberg, Sahlgrenska University Hospital, Gothenburg, Sweden, for her excellent assistance in coordinating the trial. We also acknowledge and thank the following individuals for their contributions: Data Safety and Monitoring Board members: Hans Wedel, chair, professor emeritus of epidemiology and biostatistics; Nils-Gunnar Pehrson, consulting statistician; Ola Samuelsson, Department of Nephrology, Sahlgrenska University Hospital, Gothenburg, Sweden, and James Heaf, Department of Medicine, Zealand University Hospital, Roskilde, Denmark. Clinical Endpoint Adjudication Committee member Börje Haraldsson, University of Gothenburg, Gothenburg, Sweden. This study was performed within the framework of the Nordic PD Council, an ad hoc group formed to promote investigator-driven research collaboration in peritoneal dialysis in the Nordic countries. The results in this article have not been presented elsewhere in whole or part, except in abstract form.
Author contributions
SL, DP, and JEJ conceived the study. SL obtained funding, was the principal investigator, and managed the trial. She participated in the design, data analysis and interpretation, and drafting of the first manuscript. DP, JEJ, OH, AP, MO-R, A-CJ, TEJ, BS, and MR participated in the design of the study. DP, HS, JEJ, MO-R, AP, HS, DS, and MW, who were chief investigators in their respective countries, translated the study documents into the language of their respective country and applied for ethical permission there. The biostatistician MP advised about the statistical analysis. As members of the steering committee SL, DP, JEJ, OH, A-CJ, TEJ, HG, HS, AP, MO-R, MP, DS, and MW approved the protocol, supervised the study, participated in the data analysis, and interpretation of the results. All authors commented on the draft and approved the final version.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SL received a research grant from Baxter Healthcare Corporation that partly funded this trial. DS received fees from a research foundation and personal fees from Shire/Takeda, that have no bearing on the submitted work. OH received personal fees from Astra Zeneca, Vifor, Gilead, Opterion, Baxter, Gambro, Fresenius, and Adcock Ingram, outside the submitted work. MW is the editor in Chief of Peritoneal Dialysis International. However, the handling of and decision-making regarding this manuscript was taken care of by other members of the editorial board.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article: This trial was supported by The Health & Medical Care Committee of The Regional Executive Board, Region West of Sweden, Sweden (VGFOUREG-78061, 226521, and 383641), Baxter Healthcare Corporation (McGaw Park, IL, USA; Clinical Evidence Council grant number 10CECEU1004), Swedish Society of Nephrology, The Swedish Kidney Association, The Society of Kidney Patients in Region West of Sweden, The John and Brit Wennerström Foundation, The Foundation for Kidney Failure (Sweden), The Swedish Kidney Foundation, and the Norwegian Society of Kidney Patients.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
