Abstract
Purpose:
Techniques that minimize dissection of neurovascular structures and the pelvic floor during radical prostatectomy improve perioperative outcomes and functional recovery. Previous groups have tried to reduce dissection by performing a transurethral prostatectomy. However, the vesicourethral anastomosis could not be reliably performed because of the limited instrumentation. We sought to address this with a concentric tube robot (CTR) system designed specifically for performing a transurethral vesicourethral anastomosis after transurethral prostatectomy, in a series of validated phantoms.
Materials and Methods:
We have constructed a CTR system specifically for transurethral surgery. The robot features needle-sized robotic arms that pass through a rigid transurethral endoscope and are composed of telescoping, curved, elastic tubes. By axially rotating these tubes and telescopically extending them, our robot provides surgeons with two small arms that can bend and elongate at the tip of a standard-sized endoscope. This enables suturing within the small lumen of the urethra. We evaluated the CTR in performing the vesicourethral anastomosis in a series of validated phantoms, after transurethral radical prostatectomy was performed manually with a fiber laser. Anastomosis success was evaluated using a leak test. Additionally, we evaluated the surgical time of prostate resection and the suturing time of vesicourethral anastomosis.
Results:
We performed transurethral radical prostatectomy and subsequent vesicourethral anastomosis in 11 phantoms. A successful anastomosis was performed in 10 out of 11 (91%) experiments. The median time of resection was 19 minutes (IQR: 18–21 minutes). The median suturing time was 103 minutes (IQR: 91–115 minutes).
Conclusions:
We demonstrated the use of a CTR system to perform a transurethral vesicourethral anastomosis in a series of experiments using validated phantoms. Our CTR overcomes the main barrier for providing a natural-orifice approach to radical prostatectomy by enabling intraluminal completion of the vesicourethral anastomosis.
Introduction
Prostate cancer is the most prevalent cancer in men in the United States, affecting 1 in 7 men in their lifetime.1,2 Radical prostatectomy is a common treatment (with ∼90,000 radical prostatectomies performed annually in the United States) and most often performed transabdominally. Transabdominal radical prostatectomy requires the dissection of the structures that surround the prostate including endopelvic fascia and prostatic ligaments, as well as manipulation of the neurovascular bundles. 3 This can impact functional outcomes such as sexual dysfunction and urinary incontinence.4–8 Additionally, dissection-related perioperative complications occur in up to 26% of cases and include rectal injury, deep vein thrombosis, urine leak, urine retention, and lymphocele. 8 Improved therapeutic approaches are needed to mitigate the risks of complications.
Historically, early efforts at transurethral radical prostatectomy between 2009 and 2011 demonstrated that the prostate could be successfully removed through a natural-orifice approach using holmium laser enucleation techniques.9–11 Humphreys et al. performed proof-of-concept transurethral prostatectomy using a rigid transurethral endoscope, with specimen removal through a small cystotomy and an attempted transurethral urethral anastomosis using a modified cannula scope and Ti-Knot device (LSI Solutions, Victor, NY). 10 However, these studies reported substantial difficulty maintaining visualization, directing instruments, and achieving stable tissue approximation during the anastomosis. As a result, progress stalled because the vesicourethral anastomosis could not be reliably completed with the rigid, limited-degree-of-freedom tools available at the time, and investigators concluded that new instrumentation would be required before transurethral prostatectomy could advance clinically. As the underlying clinical motivations remain highly relevant, our work directly addresses this unmet need by introducing a relatively new technology, the concentric tube robot (CTR), that we specifically design to enable intraluminal suturing in the constrained transurethral workspace.
We have previously demonstrated the use of CTRs for natural orifice surgery.12–15 CTRs are a class of continuum robots that allow dexterous movements in confined spaces. The robotic arms comprise nested, pre-curved tubes of nitinol that both rotate axially with respect to each other and telescope in and out of one another. As they rotate, the tubes elastically interact, allowing for controlled bending16–19 The arms are so small, less than 2 mm in diameter, that two of them can pass through a standard transurethral nephroscope, as shown in Figure 1 (bottom right). Our robotic system is tele-operated from user interfaces mounted directly to the robot, enabling a surgeon to manipulate the arms as well as maneuver the robot as a whole (Fig. 1), which enables tilting and positioning of the endoscope as desired. Prior preliminary studies for trans-urethral anastomosis focused solely on the suturing process in simpler models, where prostate resection was not possible. Through this system, we sought to enable CTR-based suturing, to perform the vesicourethral anastomosis after transurethral prostatectomy in a series of experiments in validated phantoms.

Robotic system used for suturing in this paper. An actuation unit controls two two-tube concentric tube arms passed through the nephroscope. (bottom right) The concentric tube arms exit below the endoscope camera. For inserting into the urethra, the outer sheathe of the endoscope is extended out to cover the arms and grasper, protecting both the urethra and the mechanisms. The needle arm is equipped with a 3-prong device for grasping the suture, and the manipulator arm with BIGopsy biopsy forceps (Cook Medical Inc).
Materials and Methods
Robot design
Our robot was specifically designed to deliver two, endoscopically deployed, concentric tube arms to perform an anastomosis. Each arm was designed to accomplish a specific task. One of the arms, the ‘needle arm’, comprises a pointed tip for tissue penetration with a retractable 3-prong grasper (Tricep 3.0 F Hooked Prong Grasping Forceps, Boston Scientific Corporation, Marlborough, MA, USA) that enables secure handling of the suture without compromising with tissue penetration (i.e., the prongs retract completely inside the tube). The other arm, the ‘gripper’ arm, uses forceps (BIGopsy Backloading Biopsy Forceps, Cook Medical, Bloomington, IN, USA) to manipulate tissue and suture.
Both arms are deployed through the working channel of a clinical nephroscope (Hopkins Telescope 6°, 27292AMA, Karl Storz, Tuttlingen, Germany) with a 26F operating sheath (27293BD, Karl Storz, Tuttlingen, Germany), which provides stable and controlled access to the surgical site. A key feature of our system is that the arms can swap positions without removing or repositioning the endoscope. This swapping mechanism takes advantage of the endoscope’s working channel, a small steel sheath through which the arms pass. At the distal end, each arm exits the sheath through its own eyelet. Because the entire working channel can rotate within the endoscope, this rotation also repositions the arms. A 180° rotation of the channel effectively swaps the arms’ exit points, enabling the needle arm to be brought to bear on all desired locations.
The robot is mounted inside a vertical bearing (to enable easy axial rotation about the endoscope axis) and is suspended from the ceiling via a counterbalance arm, enabling full orientation and position control of the robot by the surgeon. The robot’s user interface consists of two 3D Spacemouse Wireless Controllers (3Dconnexion, Munich, Germany). Both Spacemouse controllers are mounted directly onto the robot in such a way as to enable a surgeon to move the robot physically and operate the controllers simultaneously. This setup enables the surgeon to maneuver the endoscope easily — for tasks such as adjusting suture tension or changing the angle of approach—and then transition seamlessly to controlling the CTR manipulators. 20 Additionally, a simulated view of the robot arms is displayed alongside the endoscope view, shown in Figure 1, so that the surgeon can see the simulated arms from a variety of viewpoints. 21
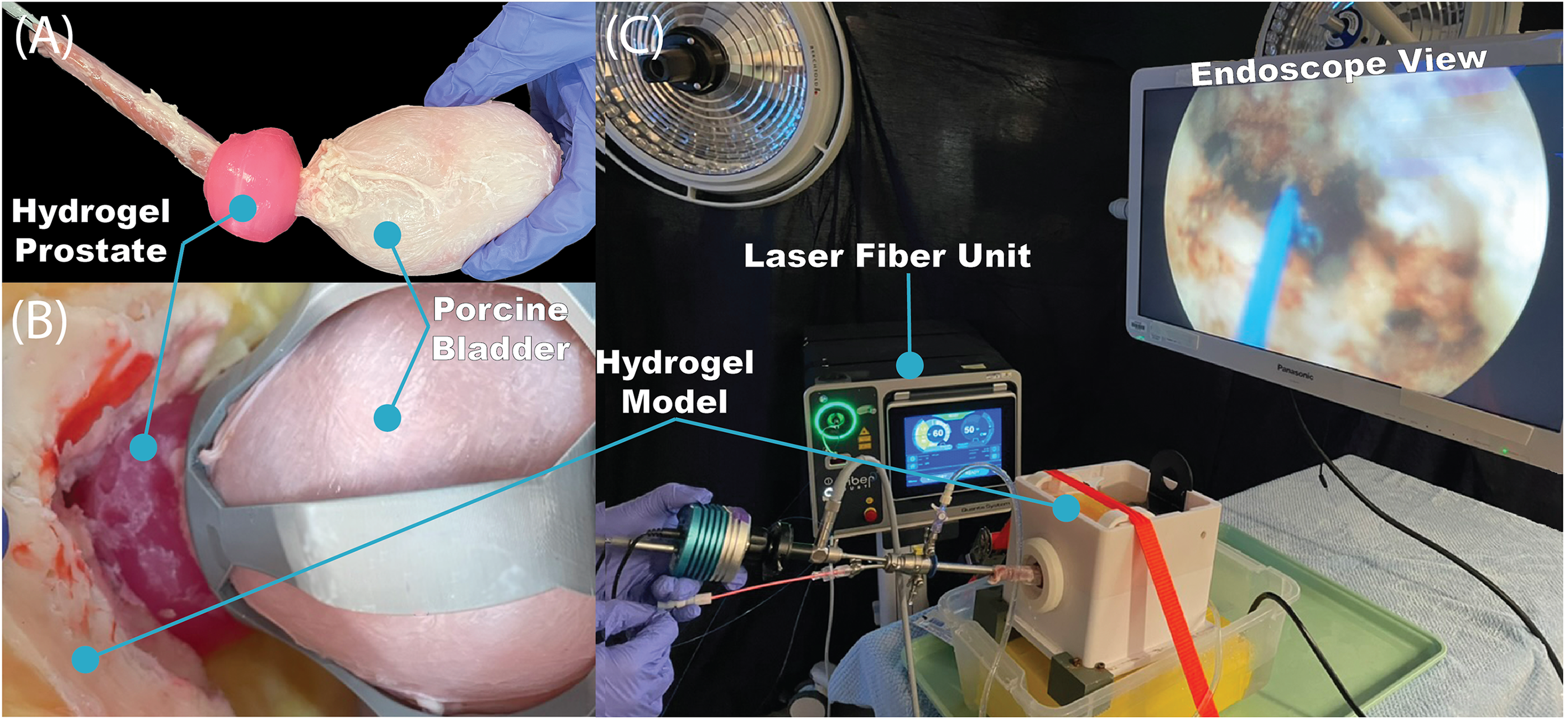
Prostate phantoms
We used a high-fidelity validated prostate phantom developed from a combination of hydrogel casting and three-dimensional printing,22,23 which is often used in surgeon training. Each phantom comprises a prostate (50 g) with peripelvic fat, all made of polyvinyl alcohol. A porcine bladder and urethra were introduced and attached to the model to better simulate organic tissue for resection and suturing. 24 The porcine bladder and hydrogel phantom can be seen in Figure 2(A and B). Each phantom is inserted into a benchtop station, shown in Figure 2(C), which enables controlled continuous irrigation during the experiments.
Resection
Transurethral radical prostatectomy was performed via a laser enucleation in a standard manual laser enucleation of the prostate (“LEP”) procedure by a single surgeon in all cases. The surgeon is a board-certified endourologist who performs robotic and LEP procedures. Both the Lumenis 120H dual-pedal laser unit in 7 cases, as well as the Karl Storz: MultiLASE Thulium Fiber Laser in 4 cases. Prostatic resection was performed with a 26F resectoscope with the continuous irrigation of normal saline. Resection was performed via en bloc technique with an early apical release. The apical prostatic urethra was circumferentially incised until peripelvic fat was identified. Circumferential incision of the surrounding periprostatic tissue was performed proximally toward the bladder neck. A top–down incision of the bladder neck was performed to incise the bladder neck mucosa completely and free the prostate from the surrounding attachments. The prostate specimen was then removed. The result after removal is shown in Figure 3(A–C).
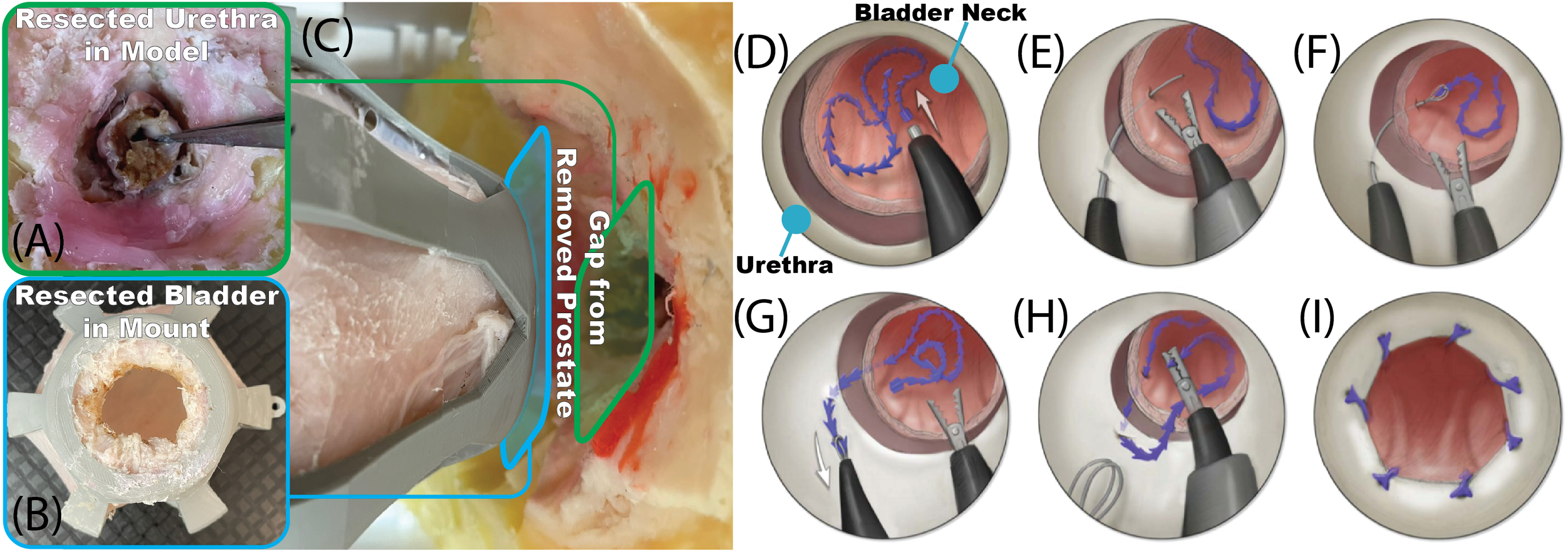
After the prostate was removed, the resected bladder and urethra were sutured back together.
Suturing method
In all phantoms, at the start of suturing, the needle arm initially exited the left side of the endoscope, and the gripper arm exited the right. Barbed suture (V-Loc 180 3–0, Covidien, Dublin, Ireland) was used to eliminate the need for knot-tying, as the barbs maintain tension after closure. The anastomosis was divided into thirds circumferentially, and a running suture was run in each segment in parallel, aiding in suture length and control and resulting in tissue apposition.
Prior to insertion of the robot transurethrally into the model, the suture was preloaded into the grasper of the gripper arm, and the outer sheath of the endoscope was pulled over to cover the tools to prevent harm to them and the urethra during insertion. Once the robot was inserted through the urethra, the surgeon visually inspected the surgical workspace, identifying both the edge of the urethra and the bladder neck. The suture was then advanced and placed into the bladder. The surgeon then identified a desired location for the first suture placement and aligned the needle arm to pierce the bladder neck. Once the bladder neck was pierced, the surgeon extended the needle arm’s inner tube, deploying the 3-pronged grasper. The surgeon then manipulated the suture into the 3-prong grasper using the gripper arm and retracted the needle arm to pull the suture through the bladder. The surgeon then handed the suture back to the gripper arm to prepare to pass the suture through the urethral wall. The robot was pulled back into the urethra such that the needle arm, when extended, could pierce the side of the urethra about 5 mm from the transected edge. The surgeon repeated the same piercing, hand off, and retraction steps as before to pull the suture through the urethra and complete a full suture throw.
Three sutures were used in parallel, each covering one-third of the circumference of the bladder neck and urethra with a running suture of 3–4 suture throws. Early in the procedure, when the bladder and urethra were far from one another, they were pierced sequentially. After a pass was made with each suture, they are lightly and evenly tensioned. As more suture passes were added and each was iteratively tensioned, the bladder and urethra eventually came into contact. At this point, both could be pierced with one needle pass. Once all the sutures were placed, they were tensioned one final time as much as possible. The suture line was examined for any separation, and the robot was then removed. An overview of the suturing method is shown in Figure 3(D–I).
Outcome metrics
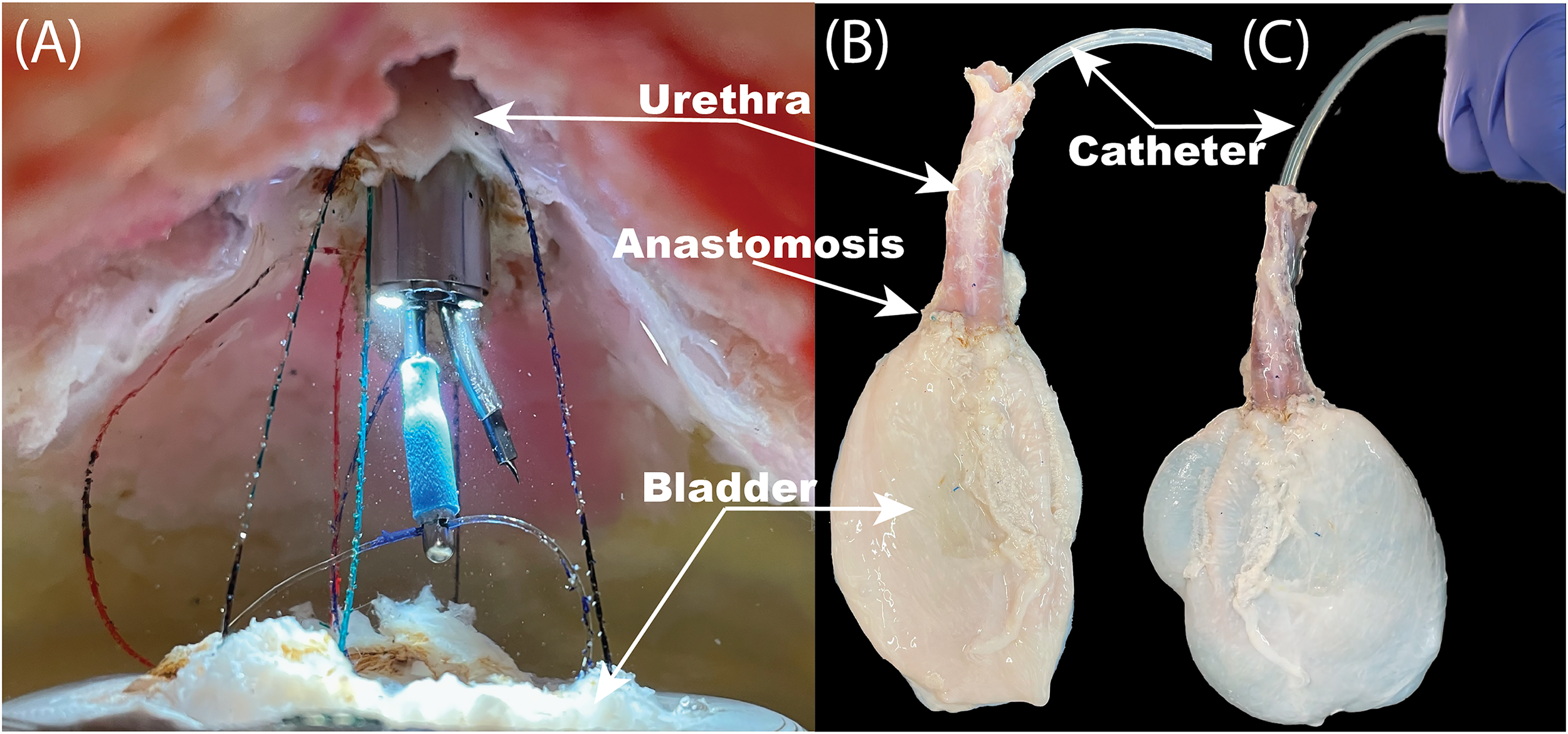
After completion of the vesicourethral anastomosis, a 16F Foley catheter was inserted, and the balloon was inflated with 10ccs of fluid. Anastomosis success was evaluated using a leak test, which is performed by injecting 100ccs of saline through a catheter after completion of the anastomosis. We defined a successful anastomosis as one that exhibited no identifiable leak. Additionally, we evaluate the surgical time of prostate resection and the suturing time of vesicourethral anastomosis.
Results
We performed transurethral radical prostatectomy in 11 phantoms. See Video for an example of suturing and result. The median time of resection was 19 minutes (IQR: 18–21 minutes) and the median suturing time was 103 minutes (IQR: 91–115 minutes). A successful anastomosis was performed in 10 out of 11 (91%) experiments. To demonstrate technical feasibility and justify future translational developments, we sought to perform 10 successful phantom cases. As we had one failure, we performed an additional experiment to achieve 10 successful cases as part of the project, resulting in 11 cases in total. In the case of the failure in the 7th trial, insertion of the catheter led to disruption of the anastomosis because of the catheter catching on tissue and being forced through the interface between the tissue. Figure 4 shows an example of the suturing mid-procedure, as well as the final anastomosis seal both before and during the leak test.
Discussion
In this study, we demonstrated the feasibility of performing an intraluminal vesicourethral anastomosis with a CTR in a validated phantom model. This study revisits a line of investigation that previously showed promise but was limited by the inability to reliably perform the vesicourethral anastomosis transurethrally. By providing highly dexterous, needle-sized manipulators capable of bending and elongating within a standard endoscope, our system overcomes the primary technical barrier identified in earlier work.
The robotic arms comprise pre-curved tubes of nitinol that telescope in and out of one another and can rotate axially with respect to each other. Additionally, they are manipulated by two 3D Spacemouse Controllers and can move independently of the endoscope, enabling dexterous manipulation within the endoscopic field of view. This configuration allows passage sutures circumferentially surround the urethra and bladder neck, enabling a complete intraluminal vesicourethral anastomosis.
To address instrumentation challenges, our novel robotic system enables intraluminal suturing via needle-size robot arms that bend and elongate and are delivered through an endoscope. Though we focused on vesicourethral anastomosis in this study, the prostatectomy itself is also possible using this robot. In that case, the concentric tube arms can be adapted to incorporate laser energy for resection. By limiting tissue disruption, it is possible that transurethral prostatectomy could minimize retraction and cutting-related stress on neurovascular bundles, which run along the outside surface of the prostate, similar to Retzius sparing approaches. 25 A completely natural orifice approach could also improve cosmetic and pain outcomes. Additionally, the bladder neck and urethra will remain supported by the natural tissues that are currently resected during prostate and bladder mobilization. Because of this, the postoperative loading on the anastomosis at the interface of the bladder neck and urethra will be substantially reduced, potentially enabling better tissue-to-tissue contact and healing.
In the current study, our CTR paradigm consists of two manipulators delivered simultaneously through the 5 mm port included in a standard 26 F (8.6 mm diameter) clinical endoscope. Adapting CTRs for use through other endoscopes is straightforward, since Nitinol tubes are available off-the-shelf at diameters down to 0.2 mm. The use of concentric tube manipulators, therefore, enabled us to break traditional size barriers and provide dexterity in spaces inaccessible to existing surgical robots. For example, we have developed similar robotic systems to use during rigid bronchoscopy to address central airway obstruction for patients with large tumors. Similarly, we have applied a similar design for hysteroscopy and polypectomy.14,18
Our study does have several limitations. First, the experiments were performed by a single surgeon in a series of phantoms, limiting generalization. Second, we defined a successful anastomosis by the lack of leakage with the instillation of 100ccs of saline through a catheter at the end of each experiment. Though this leak test is similar to what is done in clinical practice to ensure successful completion of a vesicourethral anastomosis in patients, it is subjective as we did not measure fluid pressure or standardize instillation rates. Additionally, our median suturing time was 103 minutes, which is longer than current robotic approaches. However, the primary aim of this work was to demonstrate feasibility, as this represents the first fully intraluminal anastomosis performed with a CTR. Additionally, we anticipate that these times will improve with further practice and refinement of the hardware. Despite these limitations, we demonstrate a CTR that enables intraluminal suturing and can be used to perform a vesicourethral anastomosis after radical prostatectomy. We plan on future experiments on animals and cadavers to further validate the system towards ultimate clinical use.
Conclusion
We developed a CTR to perform a transurethral vesicourethral anastomosis in a series of validated phantoms. The robot enables the surgeon to manipulate the robotic arms independently of endoscopic angulation. One arm acts as a needle, and the other as an endoscopic forceps. Our CTR overcomes the main barrier for providing a natural-orifice approach to radical prostatectomy by enabling intraluminal completion of the vesicourethral anastomosis.
Authors’ Disclosure Statement
No competing financial interests exist.
Authors’ Contributions
J.F.D.: Designed the robotic system conducted the experiments, and led the writing of the manuscript. K.A.M. and T.E.E.: Contributed to robot testing and experimental design. E.W.: Contributed to hardware design and assisted with experiments. J.A.S.: Contributed to software development. L.S. and A.E.G.: Contributed the model used in the experiments. R.J.W. and S.D.H.: Conceptualized the research, provided guidance on all aspects throughout, and edited the manuscript. N.L.K.: Contributed to experimental design, provided clinical and technical feedback, and performed the experiments.
Footnotes
Funding Information
This material is based upon work supported by the National Institutes of Health, under grant No. R01 EB026901, and by the National Science Foundation Graduate Research Fellowship Program under Grant No. 1937963 \& 2444112. Any opinion, findings, and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of neither the National Institutes of Health nor the National Science Foundation.
