Abstract
Introduction:
Covered metal ureteral stents (CMUSs) have shown promising results for ureteral stricture. However, 3-year outcomes of CMUSs are lacking. This study aimed to evaluate the 3-year efficacy and safety of CMUSs in the maintenance treatment of refractory ureteral stricture (RUS) and explore risk factors for CMUSs-related complications.
Methods:
Between August 2018 and July 2021, 321 patients (366 renal ureter units [RUUs]) with RUS who underwent CMUSs implantation surgery were prospectively recruited in this study. A regular 3-year follow-up protocol including serum biochemistry test, abdominal imaging, and ureteral stent syndrome questionnaire was established to analyze ureteral patency and patient’s quality of life (QoL). The primary ureteral patency was defined as patent ureters without CMUSs-related complications, while secondary ureteral patency also included those that restored patent after endoscopic interventions.
Results:
The study included 105 (32.7%) patients with stone-related benign ureteral stricture (BUS), 90 (28.0%) with nonstone-related BUS, 76 (23.7%) with radiation-induced ureteral stricture (RIUS), and 50 (15.6%) with malignant ureteral stricture. There existed significant differences in mean serum creatinine levels and renal pelvis widths before and after CMUSs implantation (all p < 0.001). Patients’ QoL significantly improved at postoperative 12 and 36 months (both p < 0.001). CMUSs-related complications occurred in 95 (26.0%) RUUs, including 24 (6.6%) with end-stent ureteral restenosis, 28 (7.7%) with stent migration, 31 (8.5%) with stent encrustation, 7 (1.9%) with infection, and 5 (1.4%) with ureteral fistula. The 3-year primary and secondary ureteral patency rate was 74.0% and 90.4%, respectively. However, among patients with RIUS, the primary and secondary ureteral patency rates were only 54.7% and 77.4%, respectively.
Conclusion:
CMUSs are effective and safe in the 3-year maintenance treatment of RUS. Patients with CMUSs experience long-term improved QoL. However, patients who underwent radiotherapy are susceptible to developing CMUSs-related complications.
Introduction
Refractory ureteral stricture (RUS) is characterized by the resistance to conventional curative therapies or the ineligibility for such treatments and represents a persistent clinical challenge for urologists. RUS can occur after various factors, including radiation therapy for pelvic malignancies, impacted stones or endoscopic interventions, scar formation following ureteroplasty, and the compressive effects of tumor masses.1–4 The timely management of RUS is crucial, as untreated or inadequately addressed cases can progressively impair ureteral patency and renal function, leading to a decline in the quality of life (QoL) of patients and exacerbating financial strain. 5
The ureteral reconstruction surgery is a golden standard treatment to addressing ureteral stricture disease and succeeds in over 90% of patients for a long term. 6 However, its application in the management of RUS is often limited because of the severe fibrous adhesion around the affected ureter and the prolonged recovery associated with surgical trauma or complications. The endoscopic ureterotomy and balloon dilatation, while less invasive, have shown relatively lower long-term success rate of 50%–60% for patients with RUS. 7 In addition, percutaneous nephrostomy tubes and double-J ureteral stents in the maintenance treatment of RUS require frequent exchanges and cause several complications that seriously impair patients’ QoL.8,9 The advent of metal ureteral stents provides an alternative maintenance treatment for alleviating RUS, but variability in success rates and patient tolerability highlight the need for further refinement.10–12
Covered metal ureteral stents (CMUSs) (Allium, Israel) have emerged as a promising maintenance treatment for ureteral stricture. Moskovitz et al. first reported that 70% of patients maintained stent patency for up to 17 months, and no re-obstruction was detected in 20% of cases after stent removal. 13 In a comparative study including three types of metal ureteral stents, Khoo et al. found that CMUSs succeed in only 52.8% of cases. 14 These retrospective studies with a small sample size have exhibited unstable success rates with various stenting indications. Gao and colleagues in prospective studies reported that the 1-year and 2-year success rates of CMUSs for the management of ureteral stricture were 73.2% and 71.7%, respectively.15,16 However, there was no study to date illustrating the 3-year outcomes of CMUSs and their impact on patients’ QoL. Therefore, this prospective and multi-center cohort study aimed to investigate the long-term clinical efficacy and safety profile of CMUSs in the maintenance treatment of RUS, evaluate patients’ QoL, and explore risk factors for stents-related complications.
Patients and Methods
Patients enrollment
Patients from 11 medical institutions across China who had been clinically diagnosed with RUS and subsequently underwent CMUSs implantation surgery from August 2018 to July 2021 were prospectively recruited into this study. The inclusion criteria were as follows: (1) 18–80 years old; (2) presence of one of the following stenting indications: stone or nonstone-related benign ureteral stricture (BUS), radiation-induced ureteral stricture (RIUS), and malignant ureteral stricture (MUS); (3) history of failed ureteral reconstruction surgery or endoscopic surgery; (4) frequent exchange of double-J ureteral stents (at least three procedures, and the last stent’s function time <3 months), or intolerable stent-related symptoms; and (5) patients refused to have ureteral reconstruction surgery. The exclusion criteria were as follows: (1) bladder capacity <50 mL or severe bladder outlet obstruction; (2) active urinary tract infection or pyonephrosis; (3) untreated coagulopathy; (4) patients lost to follow-up or missing data; and (5) CMUSs removed without stent-related complications and indwelled for <3 years. All eligible patients provided written informed consent for the anonymized evaluation and publication of their data. This study received approval from the Ethics Committee of Peking University People’s Hospital (approval number: 2018PHB024-01).
Initial evaluation
A systematical protocol was set for the initial evaluation in all the eligible patients. Serum creatinine levels were examined to assess patient’s renal function. The maximal transverse diameter of the renal pelvis opening on the cross-sectional imaging was measured to quantify the degree of hydronephrosis. Contrast-enhanced computed tomography (CT) or dynamic renography was performed for assess ureteral patency. Ureteral stent symptoms questionnaire (USSQ) was used to measure patient’s QoL, with lower USSQ scores indicating better QoL.
CMUSs implantation surgery
Patients were placed in the lithotomy position on a fluoroscopy table and administered either lumbar or general anesthesia. First, a hydrophilic guidewire (COOK, USA) was inserted into the affected ureteral orifice to reach the renal pelvis. Following the contrasting agent injected into the renal pelvis with a ureteral catheter (5 Fr, Urovision, Germany), retrograde pyelography was performed to evaluate the exact location and length of ureteral stricture. Subsequently, a ureteral balloon dilatation catheter (21 Fr, BARD, USA) was inserted into the stricture location along the guidewire and the balloon was pressurized to 25 atm for 3 minutes. The 18 Fr balloon was selected for patients with RIUS to prevent injury to the iliac vessel. After retracting the balloon dilatation catheter, CMUSs (30 Fr, 12 cm, Allium, Israel) were inserted along the guidewire until both ends of the stent exceeded the narrow margins and were slowly released. For long-segment stricture, sequential balloon dilatation was performed across the entire stricture location from top to bottom, and then two or three tandem CMUSs were inserted. Both ends of stents were required to extend at least 2 cm beyond the stricture margins. Finally, retrograde pyelography was performed to confirm the patency of the entire length of the affected ureter (Figure 1).
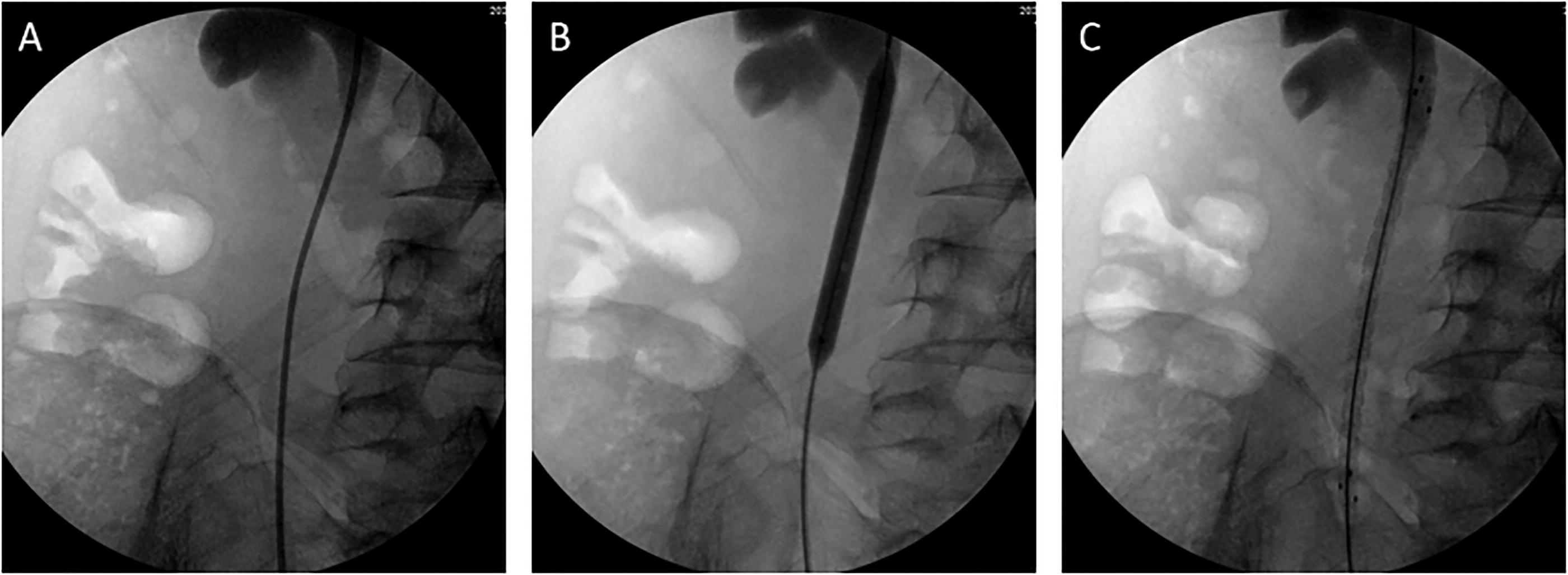
CMUSs implantation surgery.
When the guidewire failed to pass through the stricture location retrogradely, the anterograde surgery was performed, with patients shifted to the oblique supine position. The target calices was punctured under ultrasonography guidance, and a 16 Fr percutaneous renal access was established using a fascia dilator. After the guidewire anterogradely passed though the stricture location, the balloon dilation and CMUSs insertion were completed. For patients with severe stricture, endoscopic combined surgery was performed to recanalize the stenosis segment.
Patients follow-up
A scheduled follow-up protocol was established for evaluating patients’ outcomes every 6 months after CMUSs implantation. Combination of serum creatinine levels and abdominal imaging was used to assess patients’ renal function and ureteral patency. USSQ scores were measured to assess patients’ QoL. CMUSs-related complications were identified by abdominal imaging and endoscopy, including stent migration, encrustation, end-stent restenosis, and ureteral fistula.
Definition of patency outcomes
Primary ureteral patency was defined as cases maintaining unobstructed urinary drainage throughout the 3-year follow-up period without developing any CMUSs-related complications. Secondary ureteral patency also included cases where patency was successfully restored following endoscopic management, including stent adjustment, stent exchange, and pneumatic lithotripsy. Stent failure was characterized by mandatory stent removal due to severe complications, including bacterial biofilm formation, ureteral fistula, and large-volume stent encrustation.
Statistical analyses
The categorical variable was presented as “frequency (percentage)”. The continuous variable was presented as “mean ± standard deviation” or “median (minimum–maximum)” according to a normality test. Paired t-tests, rank-sum tests, or chi-square tests were used to compare the differences in clinical characteristics before and after CMUSs implantation surgery. Kaplan–Meier curves were used to graphically depict primary and secondary ureteral patency survival rates. Univariate and multivariate Cox regression analyses were used to predict the risk factors for CMUSs-related complications. All statistical analyses were performed using SPSS 22.0, with p < 0.05 considered statistically significant.
Results
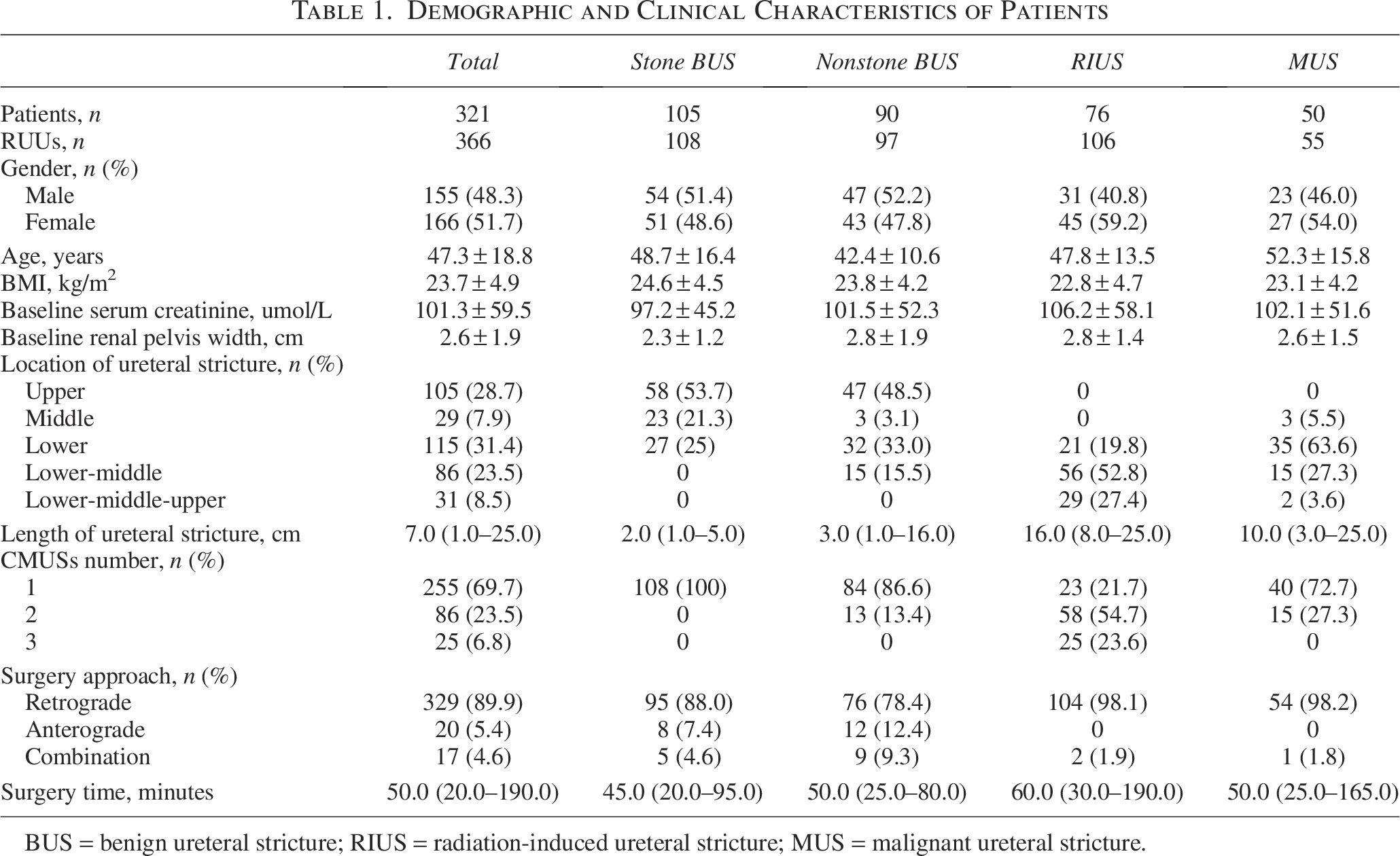
Of the 344 patients initially recruited into this study, 9 patients with MUS passed away unrelated to CMUSs, and 14 patients had their stents removed without CMUSs-related complications with indwelling time of <3 years. Consequently, 321 patients with 366 renal ureter units (RUUs) were included in the final analysis (Figure 2). The mean age of all patients was 47.3 ± 18.8 years, and the mean body mass index (BMI) was 23.7 ± 4.9 kg/m2. There were 155 (48.3%) male patients and 166 (51.7%) female patients. CMUSs implantation indications included stone-related BUS (105, 32.7%), nonstone-related BUS (90, 28.0%), RIUS (76, 23.7%), and MUS (50, 15.6%). The demographic characteristics are presented in Table 1.
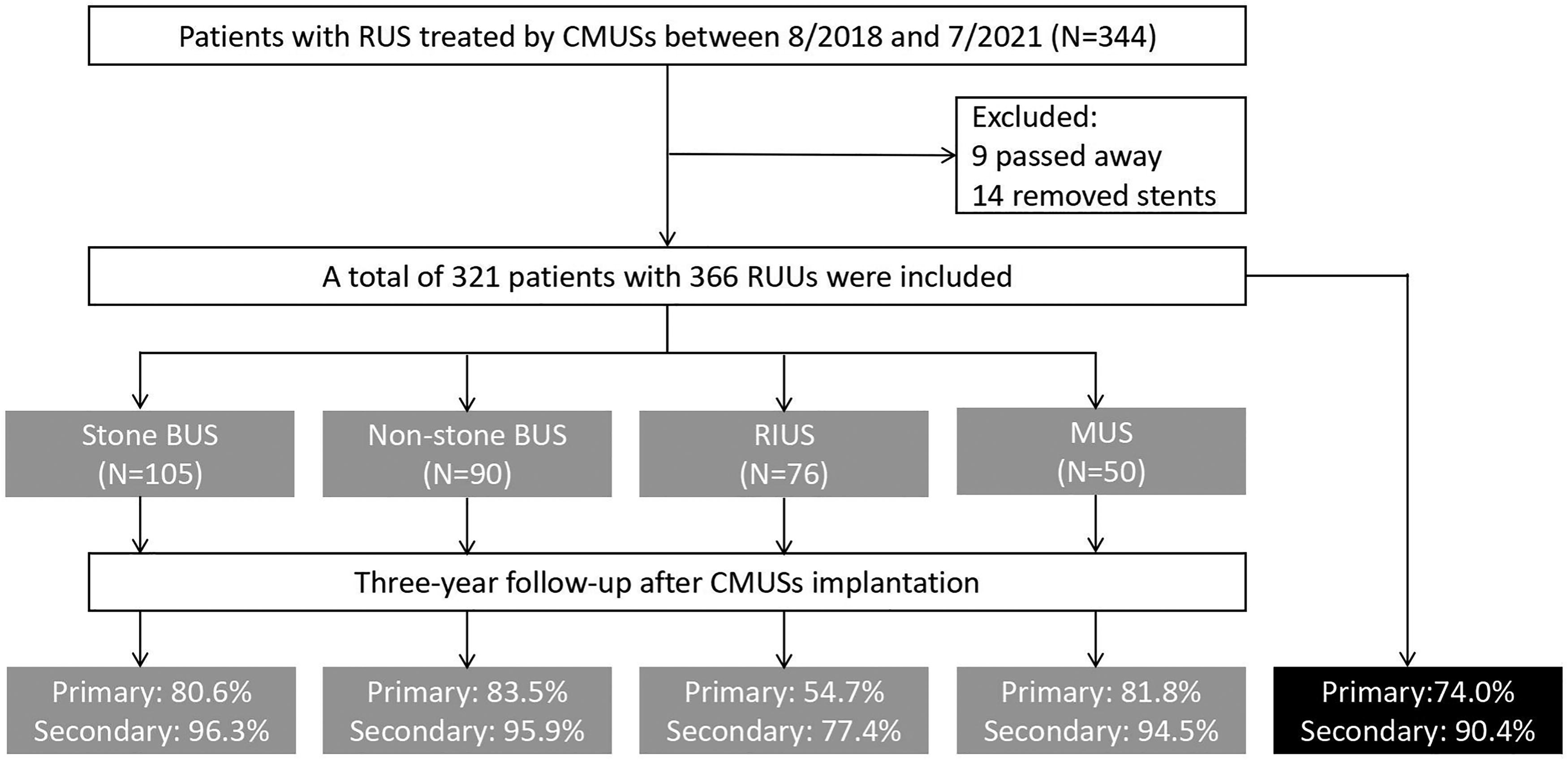
Flowchart of patient enrollment and follow-up.
Demographic and Clinical Characteristics of Patients
BUS = benign ureteral stricture; RIUS = radiation-induced ureteral stricture; MUS = malignant ureteral stricture.
Ureteral obstruction was located at the upper ureter in 105 (28.7%) cases, the lower ureter in 29 (7.9%), the middle ureter in 115 (31.4%), the lower-middle ureter in 86 (23.5%), and the entire ureter in 31 (8.5%) (Table 1). The median length of ureteral stricture was 7.0 (1.0–25.0) cm, and patients with RIUS had the longest ureteral stricture of 16.0 (8.0–25.0) cm. The median surgery time was 50.0 (20.0–190.0) minutes, and surgical technical success rate was 100%.
The mean serum creatinine levels of all patients significantly decreased from preoperative 101.2 ± 50.5 μmol/L to 95.2 ± 47.1 μmol/L at postoperative 12 months, 91.8 ± 52.3 μmol/L at postoperative 24 months, and 94.5 ± 53.9 μmol/L at postoperative 36 months (all p < 0.001 before and after surgery) (Figure 3). At the same time points, the mean renal pelvis widths also significantly reduced from 2.6 ± 1.6 cm to 2.1 ± 1.2 cm, 1.5 ± 0.8 cm, and 1.5 ± 0.9 cm, respectively (all p < 0.001) (Figure 4). However, among patients with RIUS, the significant increase in serum creatinine levels and renal pelvis widths were observed from postoperative 24 months to 36 months (95.3 ± 52.1 μmol/L vs. 99.5 ± 55.6 μmol/L, p = 0.001; 2.2 ± 1.5 cm vs. 2.5 ± 1.5 cm, p < 0.001).
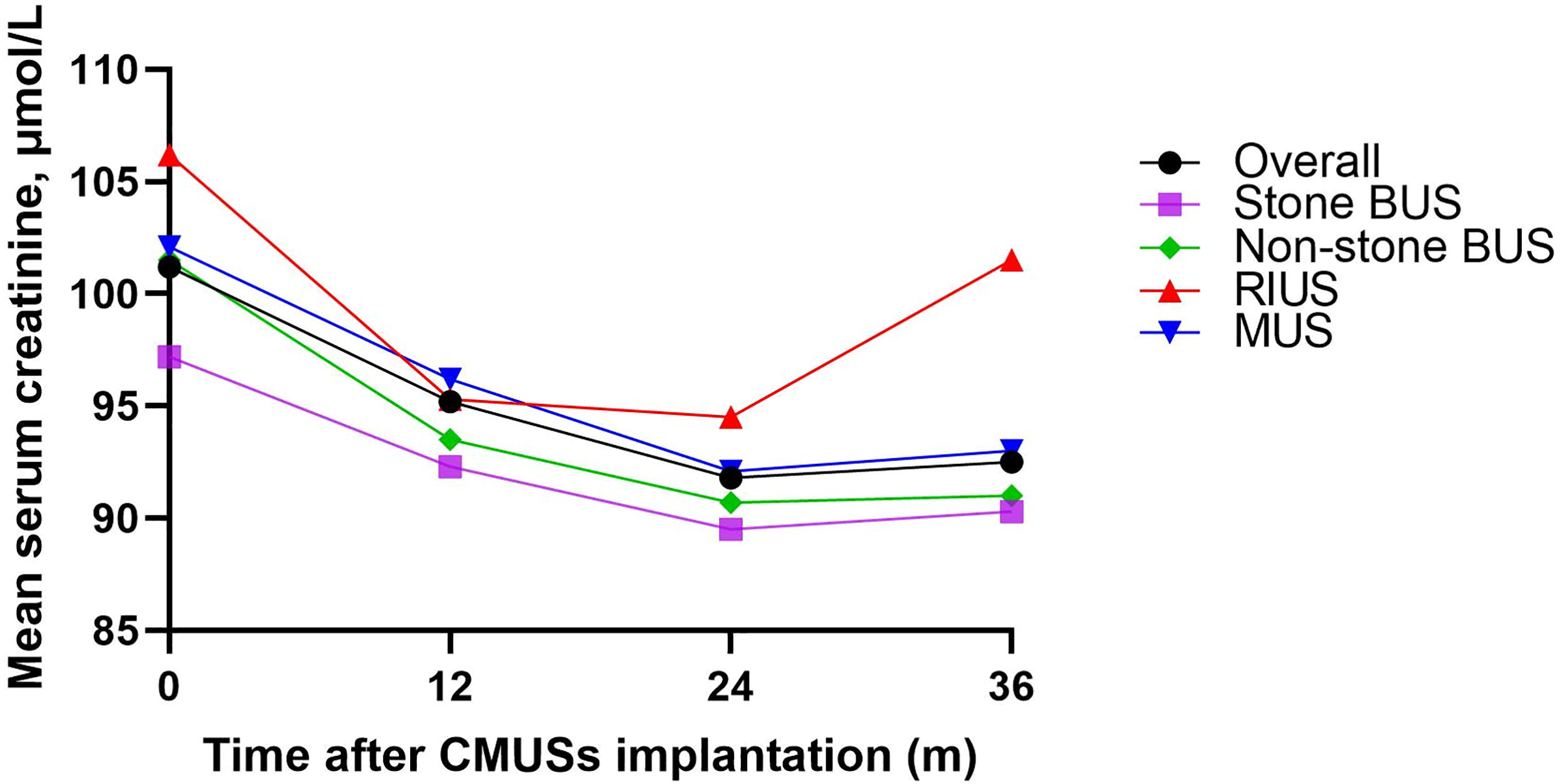
Changes in serum creatinine levels.
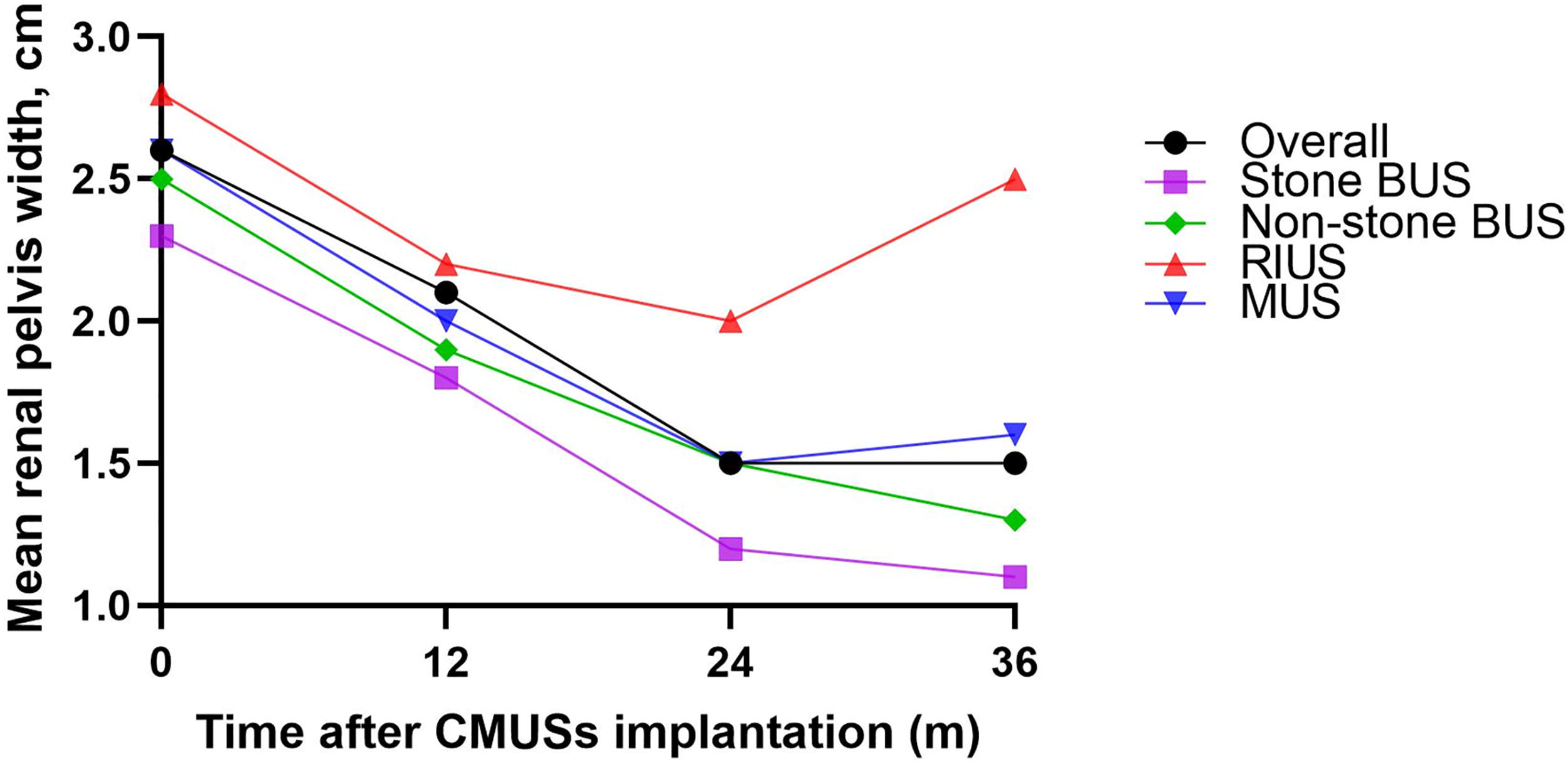
Changes in renal pelvis width.
Compared with the preoperative USSQ total scores with double-J ureteral stents, the USSQ total scores at both postoperative 12 and 36 months with CMUSs showed statistically significant decrease (98.5 ± 7.0 vs. 82.6 ± 5.1 and 82.1 ± 4.0, respectively, all p < 0.001), which implied improved patients’ QoL (Table 2). Although there was no significant difference in the USSQ total scores between postoperative 12-month and 36-month assessments, the subdomain analysis revealed that both body pain and general health scores had further improved (13.8 ± 1.5 vs. 12.9 ± 1.3, p = 0.001; 14.7 ± 1.9 vs. 13.5 ± 1.2, p = 0.015, respectively).
Patient Quality of Life
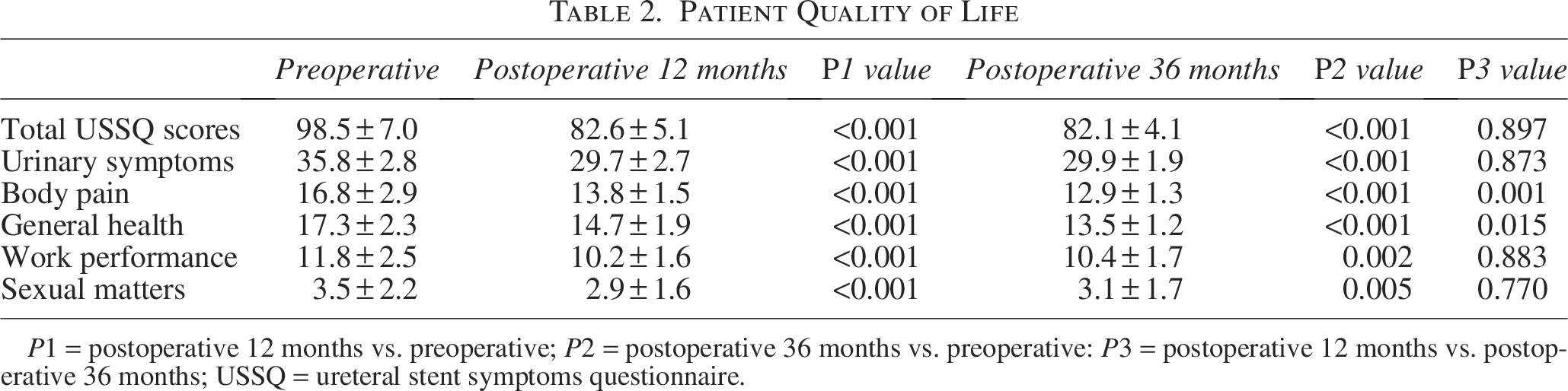
P1 = postoperative 12 months vs. preoperative; P2 = postoperative 36 months vs. preoperative: P3 = postoperative 12 months vs. postoperative 36 months; USSQ = ureteral stent symptoms questionnaire.
There were 95 (26.0%) RUUs with CMUSs-related complications, including 24 (6.6%) with end-stent ureteral restenosis, 28 (7.7%) with stent migration, 31 (8.5%) with stent encrustation, 7 (1.9%) with serious urinary tract infection, and 5 (1.4%) with ureteral fistula (Figure 5). Patients with RIUS experienced the highest complication rate (n = 48, 45.3%). Among the 95 cases with CMUSs-related complications, 60 cases were successfully treated with secondary endoscopic surgery, while the remaining 35 cases underwent CMUSs removal. Consequently, the primary ureteral patency rate was 74.0% (271 out of 366 RUUs), and the secondary ureteral patency rate was 90.4% (331 out of 366 RUUs) (Figure 6). Nevertheless, patients with RIUS had the lowest primary and secondary ureteral patency rates of 54.7% and 77.4%, respectively (Figure 2). Additionally, the subgroup analysis based on stricture length demonstrated a positive correlation between stricture length and CMUSs-related complication rates (Table 3).
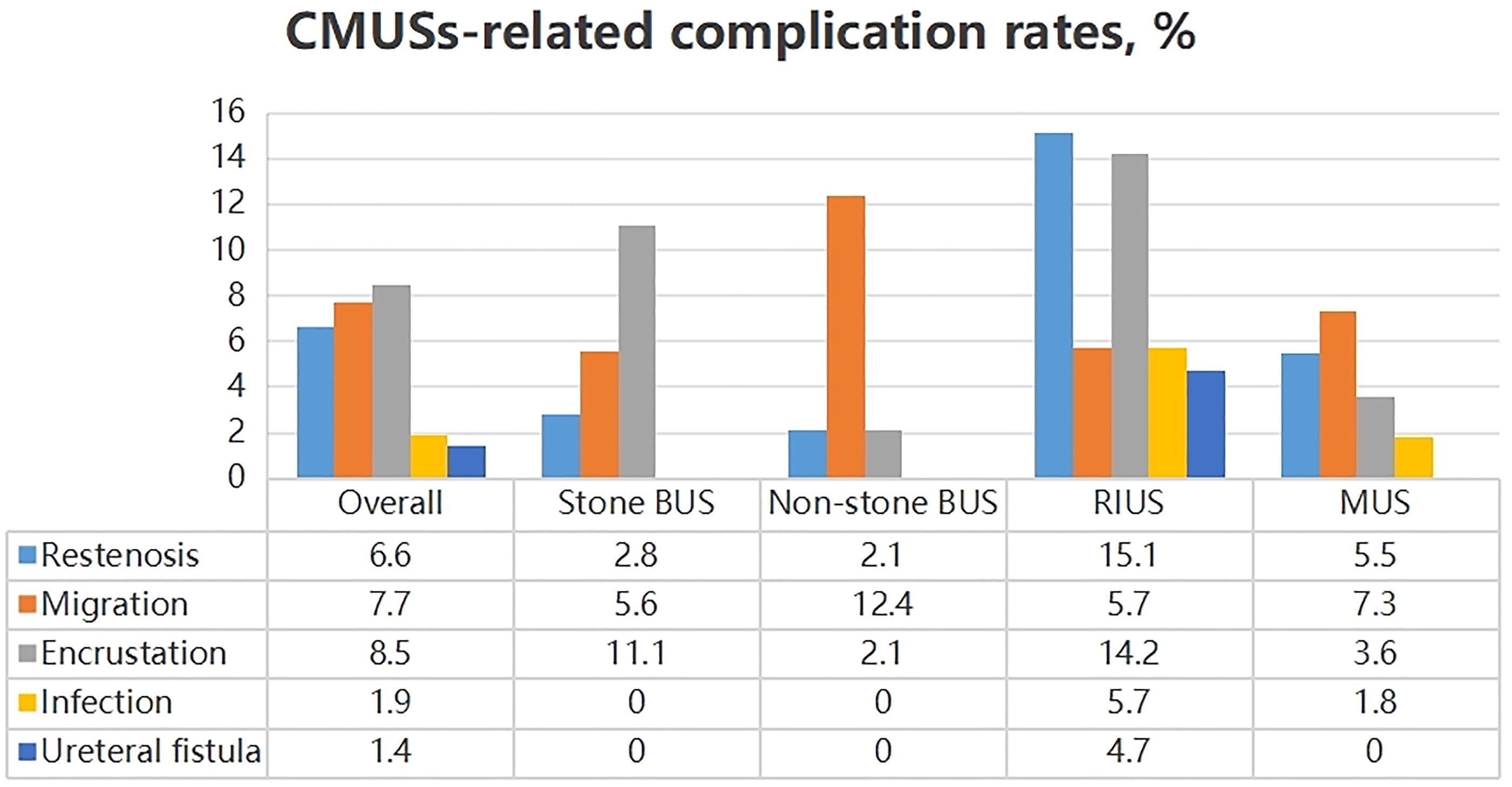
CMUSs-related complication rates.
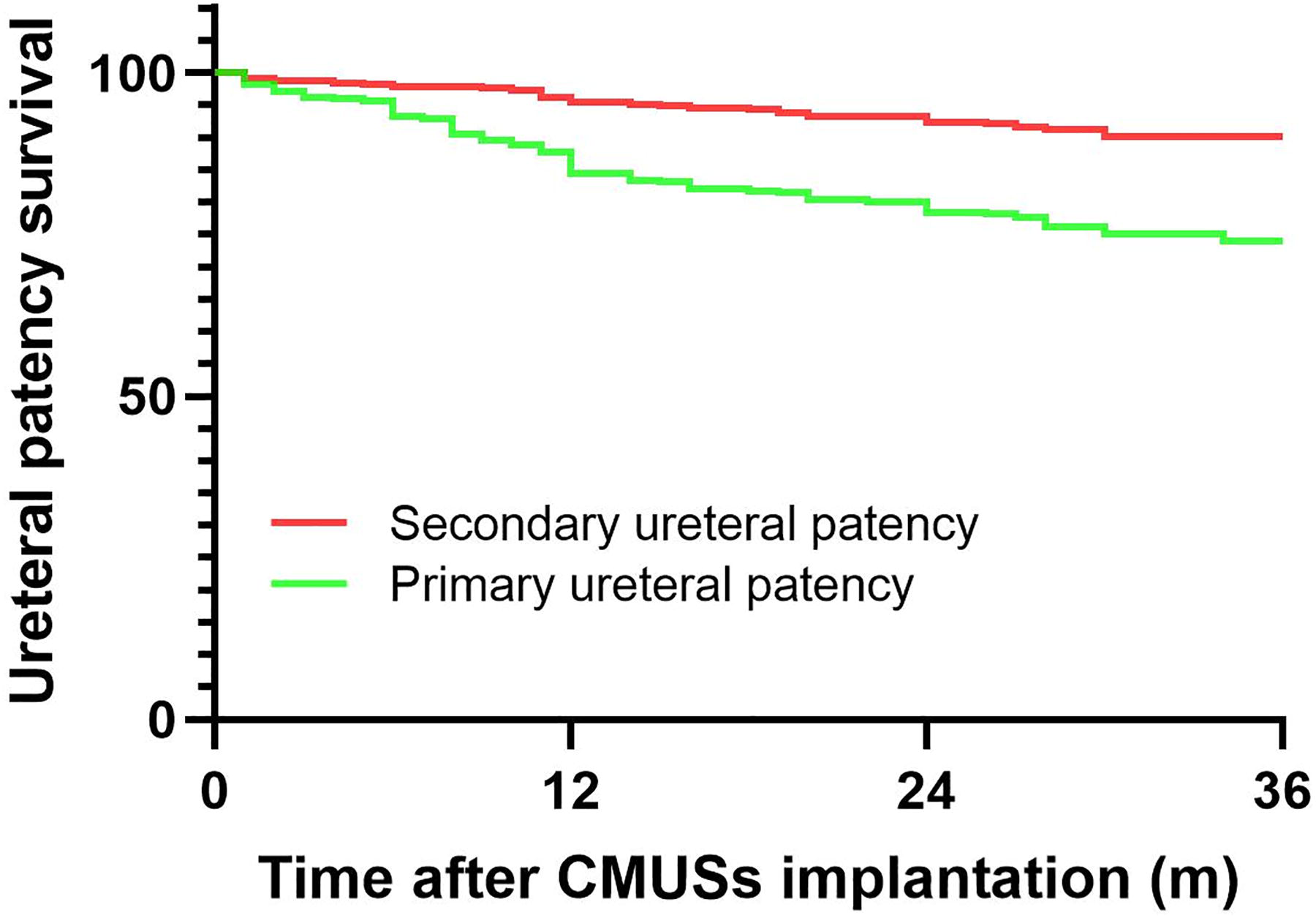
Overall ureteral patency survival.
CMUSs-Related Complications Based on Stricture Length

RUUs = renal ureter units.
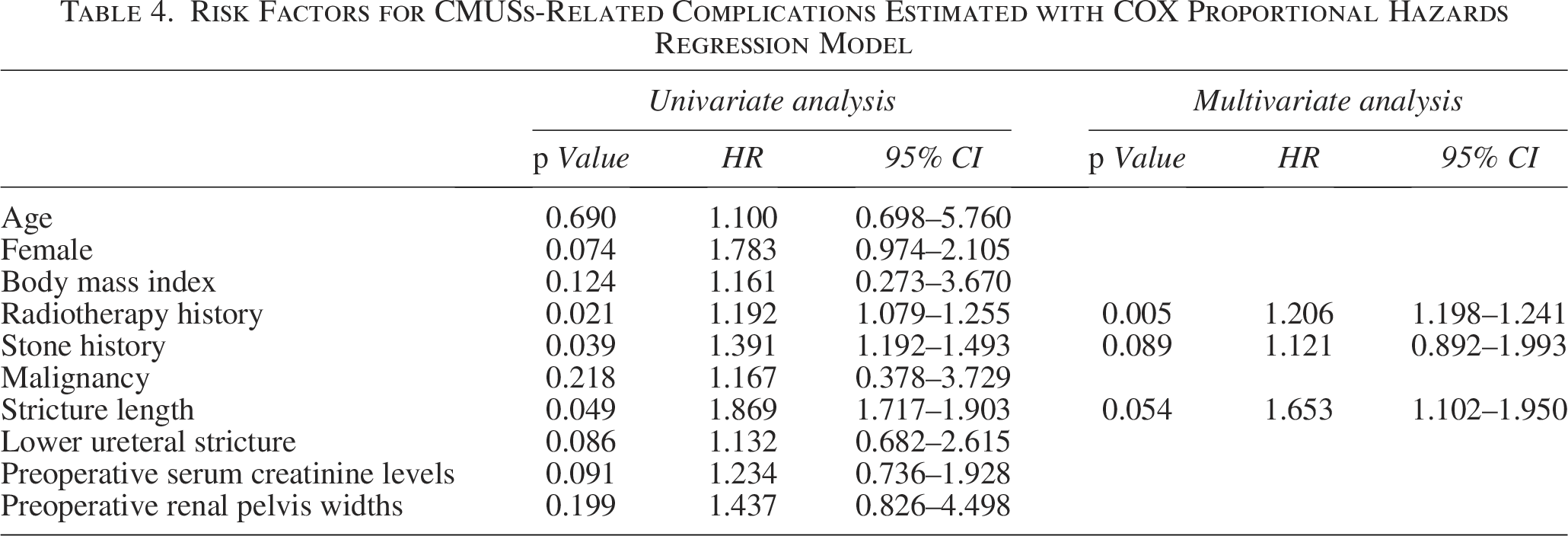
Cox univariate proportional hazards regression analysis demonstrated that CMUSs-related complications were significantly associated with stone history (Hazards ratio (HR) = 1.391, p = 0.039), radiotherapy history (HR = 1.192, p = 0.021), and the length of ureteral stricture (HR = 1.869, p = 0.049). Cox multivariate analyses showed that the radiotherapy history (HR = 1.206, p = 0.005) was an independent predictor for CMUSs-related complications (Table 4).
Risk Factors for CMUSs-Related Complications Estimated with COX Proportional Hazards Regression Model
Discussion
Based on a comprehensive review of the existing literature to date, this study represents the largest prospective and multi-center cohort study, investigating the 3-year clinical efficacy and safety of CMUSs in the maintenance treatment of RUS. Notably, this study illustrated that patients with RIUS might not be suitable for the long-term indwelling of CMUSs.
Serum creatinine levels and renal pelvis widths are established markers of ureteral patency and renal function. 17 Our study demonstrated that serum creatinine levels and renal pelvis widths decreased 6%–9% and 19%–42% at 12–36 months after CMUSs implantation, respectively. However, Khoo et al. reported that serum creatinine levels rose 21%–47% from the baseline. 14 Further analysis found that Khoo et al. study included more patients (87%) with MUS or retroperitoneal fibrosis who usually had severely impaired renal function, which might answer the reason of increased serum creatinine levels. Furthermore, the present study showed that the 3-year primary and secondary ureteral patency rates of CMUSs were 74.0% and 90.4%, respectively. These 3-year outcomes were consistent with the 1-year and 2-year success rates of 73.2% and 71.7%, respectively, as reported by Gao et al.15,16 Following the immediate mechanical effect of balloon dilation on stricture segment, large-diameter CMUSs could provide sustained tissue remodeling and supporting, thereby improving long-term ureteral patency. Therefore, these results were indicative of the long-term efficacy of CMUSs in maintaining adequate renal urine drainage and restoring kidney function.
Ureteral stricture is a chronic and often symptomatic condition that reduces patients’ QoL due to its associated complications and the presence of double-J ureteral stents. 9 The pain or discomfort experienced by patients with double-J ureteral stents is associated with irritation on ureteral tissue and irregular peristaltic activity of ureters.18,19 In addition, the incorrect location of the stent’s distal ends in the bladder may also cause poor QoL. 20 Taguchi et al. reported that patients with loop-tail ureteral stents experienced better QoL, which minimized the irritation on bladder mucosa. 21 This longitudinal cohort study demonstrated a notable and sustained improvement in patients’ QoL following the placement of CMUSs compared with preoperative double-J ureteral stents, especially in terms of body pain and general health. The segmental design, soft-end structure, and polymer covering might contribute to the reduction in tissue irritation. 22 Consequently, the long-term asymptomatic indwelling of ureteral stents might be another key advantage of CMUSs in the maintenance treatment of RUS.
Stent migration of segmental ureteral stents remains a prevalent challenge. 16 This study revealed that the incidence rate of CMUSs migration was 7.7%. Supporting our findings, Gao et al. reported that 12.5% of patients with recurrent ureteral stricture following ureteroplasty experienced stent migration. 23 This observation underscored the potential influence of anatomical factors on stent stability, such as the goblet collecting system. Despite the large caliber and robust metal mesh structure of CMUSs, which were designed to enhance anchoring, the ability to firmly embed at the site of short stricture was limited because of the reduced surface friction between CMUSs and ureteral wall. Additionally, Gao et al. suggested that tandem CMUSs could help reduce stent migration rates. 15 However, contrary to their findings, our study revealed that the junction between two stents remained prone to dislocation, leading to the exposure of primary stricture and recurrence of ureteral obstruction. Therefore, personalized stent selection based on individual patient anatomy, such as the shape and size of the collecting system, could offer a more tailored approach to limit stent migration.
The process leading to ureteral stent encrustation is intricate, involving multiple contributing factors. Upon insertion, a newly implanted stent quickly accumulates a conditioning film of glycoproteins, which can serve as a substrate for the subsequent development of bacterial biofilm or, more problematically, for encrustation. 24 Our study revealed that 8.5% of CMUSs exhibited salt deposition, predominantly at the distal end extending into the bladder, and along the stent inner wall. This finding is corroborated by an earlier study that reported an encrustation rate of 13.3%, which was slightly higher than our results. 14 Furthermore, patients with a history of radiotherapy had an increased risk of CMUS encrustation, which was observed in 14.2% of this cohort. It is imperative to consider stent removal when recurrent salt deposition is detected to mitigate the risk of serious infection and ensure the ongoing efficacy and safety of the stent.
The phenomenon of end-stent restenosis of CMUSs is similar to the use of covered metal mesh stents in the cardiovascular system. 25 The recurrence of ureteral stricture at stent’s ends is attributed to the urothelial follicular hyperplasia, which is associated with the increased shear stress between the stent’s ends and ureteral mucosa. 26 Our study is the first to illustrate that the incidence of end-stent restenosis was 6.6%. However, among patients with RIUS, 15.1% of cases were affected. Patients who have a history of radiotherapy exhibit severe fibrosis of the ureter and surrounding tissues, which may aggravate the urothelial hyperplasia at the ends of CMUSs. The insights derived from our findings suggest that specific surgical techniques, such as positioning each end of the CMUSs at a distance far away from the stricture margins, could help alleviate urothelial hyperplasia and subsequent obstruction, thereby improving the long-term patency of CMUSs.
Tissue fibrosis after pelvic radiation therapy can also lead to fixation of the ureter to surrounding tissues, ischemic necrosis of the ureter wall, and potentially formation of ureteral fistulas, especially in the presence of indwelling ureteral stents.27,28 The current study demonstrated that 1.4% of the whole cohort developed ureteral fistula after the indwelling of CMUSs, all of whom had a history of radiotherapy. It is shocking that Kim et al. reported that 28.0% of patients with chronic ureteral obstruction treated with Uventa ureteral stents developed ureteral fistula, which was obviously higher than our results. 29 These findings highlight the importance of carefully evaluating the pelvic condition of patients who have undergone radiation therapy, with particular attention given to the relationship between the ureter and adjacent tissues. Additionally, when ureteroarterial fistula is suspected, iliac artery angiography should be performed to confirm the diagnosis and guide appropriate management.
This study demonstrated that radiotherapy is an independent risk factor for CMUSs-related complications. Among patients with RIUS, serum creatinine levels and renal pelvis widths from postoperative 24 to 36 months appeared an upward trend. Moreover, the 3-year primary and secondary ureteral patency rates in patients with RIUS were only 54.7% and 77.4%, respectively. Considering these results, we suggest that long-term indwelling of CMUSs may not be suitable for patients with RIUS. Enhanced follow-up and timely management of CMUSs-related complications are necessary. Special attention should be paid to the occurrence of ureteral fistula and stent encrustation, which may lead to major bleeding or urosepsis.
This study also has some limitations. First, the lack of comparative analysis with other types of metal ureteral stents may limit the evidentiary power of this study. Future studies need to complete head-to-head comparisons with other stent types, including Memokath ureteral stents, to better contextualize segmental stent outcomes. Additionally, the economic burden on patients was not systematically collected. The reduced exchange frequency might neutralize the higher single medical expense of CMUSs. Furthermore, because of the financial constraint, the study did not include renal dynamic imaging, which are highly sensitive tools for assessing unilateral renal function. Instead, alternative diagnostic methods were utilized, such as serum biochemistry test and abdominal ultrasonography or CT. Despite these limitations, the present study remains the first to prospectively report the 3-year outcomes of CMUSs in the maintenance treatment of RUS.
Conclusions
CMUSs are effective and safe in the 3-year maintenance treatment of RUS. Patients with CMUSs experience long-term improved QoL. However, patients who underwent radiotherapy are susceptible to developing CMUSs-related complications. Timely diagnosis and intervention are essential for effectively addressing any complications.
Authors’ Contributions
M.W. and R.Z. conducted the literature search and wrote the article. S.L., H.H., Y.Z., W.D., Yi.Z., X.D., W.Y., F.C., K.L., H.L., X.C., W.Q., and K.X. obtained informed consent and collected data. G.X., T.X., and Ha.H. revised the article. All authors have read and approved the final article.
Footnotes
Author Disclosure Statement
The authors have no conflicts of interest to disclose.
Funding Information
This study was supported by Capital’s Funds for Health Improvement and Research (2020-2Z-40810).
Ethical Approval
This study was approved by the Ethics Committee of Peking University People’s Hospital (2018PHB024-01).
